Abstract
Manganese (III) meso-tetrakis(N-ethylpyridinium-2-yl)porphyrin (MnTE-2-PyP or BMX-010; CASRN 219818-60-7) is a manganese porphyrin compound developed as a potential drug substance for use as a radioprotective and for the ex vivo treatment of cells, tissues, and organs intended for transplantation. In preparation for an investigational new drug filing, a full good laboratory practice nonclinical safety assessment was conducted in order to evaluate the safety of MnTE-2-PyP and included the performance of in vitro genotoxicity studies, local tissue tolerance evaluation, safety pharmacology core battery studies, and single- and repeat-dose intravenous (iv) toxicity studies in mice and monkeys. The MnTE-2-PyP was determined not to be genotoxic or hemolytic, did not demonstrate flocculation or elicit adverse pharmacologic effects on respiration, the central nervous system (CNS), and had limited transitory effects on the cardiovascular system only at levels well above the therapeutic target dose. The intended iv clinical solution did not cause venous irritation in rabbits. The no observed adverse effect level (NOAEL) in mice was determined to be 10 mg/kg/day after 18 consecutive days of bolus iv dosing once daily in the morning. The NOAEL in monkeys after 14 days of bolus iv dosing in the morning was determined to be 5 mg/kg/day. At doses relevant to clinical use in humans, neither study revealed any indication of any specific target organ toxicity, including the classic heme porphyrin kidney, liver, CNS, or cardiac toxicities, or manganese toxicity. Mortality seen shortly after dosing in individual animals at higher doses was not accompanied by any organ or clinical pathology indications, suggesting a functional pharmacological-mediated effect. Based on the results of these studies, a conservative safe initial starting clinical dose of 5.0 mg (0.083 mg/kg in a 60 kg adult) was proposed for the initiation of human trials. Because of patent life issues, use of MnTE-2-PyP as a transplantation aid or radioprotective agent is not currently being pursued past the preclinical stages. It serves as a model for the clinical development of this class of drugs.
Introduction
Metalloporphyrines are potent antioxidants and have been demonstrated as effective in ameliorating inflammation and injury in a large number of animal models of human disease such as reducing the pneumotoxic effects of paraquat. 1 Manganese (III) meso-tetrakis(N-ethylpyridinium-2-yl)porphyrin (known as MnTE-2-PyP or BMX-010; CASRN 219818-60-7) is a manganese porphyrin drug candidate intended for use as a protective agent against radiation exposure in terrorist events or over the course of radiotherapy 2 –6 or for the ex vivo treatment of cells and tissue intended for transplantation. 7,8 The structure is shown in Figure 1. Generally speaking, it was believed to be an oxidoreductase that functions as a highly potent catalytic antioxidant. 7 One possible clinical indication identified in animal studies and not linked to an antioxidant effect was for the protection of islet cells during isolation and after transplantation. For pancreatic islet cells, oxidative stress occurs during organ harvesting, during the isolation process, and during storage and transport. Isolation insults result in osmotic, mechanic, ischemic, and oxidative stress for the islet cells. These stressful events result in an increase in the production of proinflammatory cytokines and free radicals by pancreatic acinar cells, passenger leukocytes, and islet cells themselves, leading ultimately to β cell dysfunction and death. Therefore, the ability to reduce these early nonspecific proinflammatory events (ie, free radical generation and cytokine production) during islet cell isolation should afford increased β cell mass as well as improved and more stable metabolic function. 9
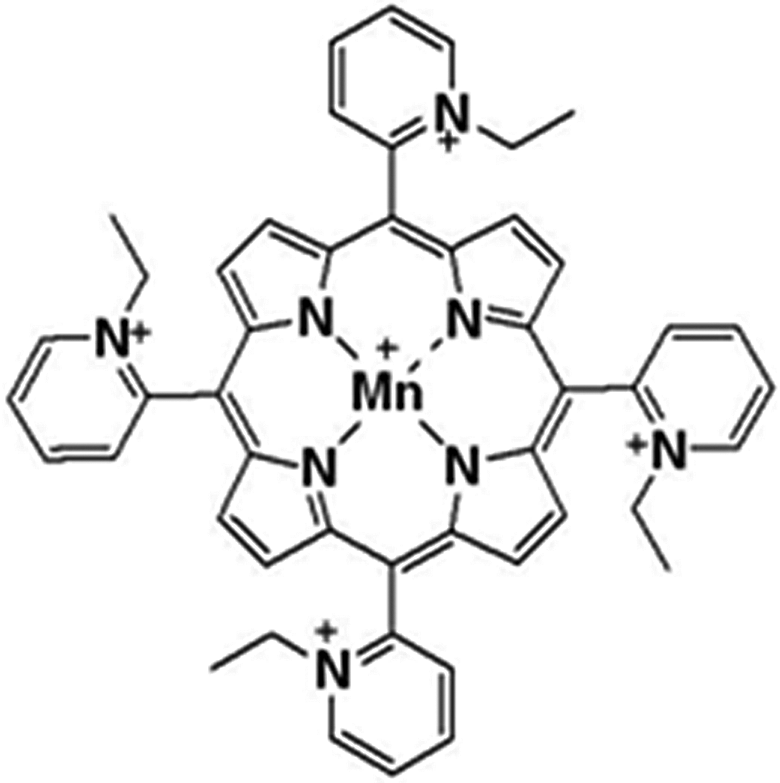
The MnTE-2-PyP structure (CASRN 219818-60-7). Molecular weight: 787.8696.
In order to evaluate toxicity of the MnTE-2-PyP drug substance, a series of studies were conducted, based on the International Conferences of Harmonisation (ICH) M3(R2) good laboratory practice (GLP) compliant safety assessment program. 10 The relevant studies include in vitro genotoxicity studies, hemolysis and flocculation study, safety pharmacology core battery studies (in vitro human ether-à-go-go related gene [hERG], respiratory and central nervous system (CNS) in mice, and cardiovascular in monkeys), a venous irritation study in rabbits, and the performance of intravenous (iv) toxicity studies in mice (single dose and once daily repeat dose for 5 and 18 days) and monkeys (once daily repeat dose for 5 and 14 days).
Materials and Methods
Manufacturing and Characterization of MnTE-2-PyP
The GLP (21 CFR 58) 11 and good manufacturing practices (ICH Q7) 12 compliant MnTE-2-PyP compound was synthesized by Ricerca Biosciences (Concord, Ohio) and supplied as a dark brown solid with 87% purity and 5 specifically identified porphyrin impurities (individual masses ranged from >73.29-836.30). The compound was packaged in vials, each containing 51 mg of prepared MnTE-2-PyP compound in a sterile glass vial with a self-sealing stopper through which compound was able to be reconstituted and removed. Vials containing compound requiring reconstitution were vented to reduce the internal pressure with a commonly used venting device utilizing a 26-gauge needle, or other appropriate procedure, to reduce the probability of spraying, spillage, and loss of the test article. One milliliter of sterile H2O, or normal saline, was delivered to the sterile vial, using a sterile syringe fitted with a 23-gauge needle. The compound was easily soluble in 100% H2O and saline. However, to ensure complete reconstitution, the contents of the vial were swirled around, and the vial was mildly shaken via inversion and rotation over a period of several minutes. After reconstitution, the compound concentration was 44.4 mg/mL (with less than 0.4 µg/mL of free manganese), and a 1500× stock was prepared to reconstitute a working stock of 34 µmol/L and to add to various media as needed.
Genotoxicity
Bacterial reverse mutation assay
The MnTE-2-PyP was evaluated in the bacterial reverse mutation assay using Salmonella typhimurium tester strains TA98, TA100, TA1535, and TA1537 as described by Ames et al 13 and an Escherichia coli tester strain WP2 uvrA as described by Green and Muriel, 14 in the presence and absence of Aroclor-induced rat liver S9 metabolic activation system. The assay was performed in 2 phases, using the plate incorporation method. In the first phase, the initial toxicity mutation assay was used to establish the dose range of the confirmatory mutagenicity assay and to provide a preliminary evaluation of potential mutagenicity. The maximum dose tested in each of the 5 strains (with or without metabolic activation) was 5000 µg/plate; this dose was achieved using a stock concentration of test article of 50 mg/mL and a 100-µL plating aliquot. The dose levels evaluated were 1.5, 5.0, 15, 50, 150, 500, 1500, and 5000 µg/plate. The test article formed cloudy solutions in phosphate-buffered saline (PBS) from 1.5 to 50 mg/mL and clear solutions from 0.015 to 0.50 mg/mL. In the second phase, the confirmatory mutagenicity assay was used to evaluate and confirm the mutagenic potential of the test article. The dose levels tested were 15, 50, 150, 500, 1500, and 5000 µg/plate. For a response to be considered positive, it must cause a dose-related increase in the mean revertants per plate of at least 1 tester strain over a minimum of 2 increasing concentrations of MnTE-2-PyP.
In vitro mammalian chromosome aberration test
The test article MnTE-2-PyP was evaluated in the chromosome aberration assay using Chinese hamster ovary (CHO) cells in both the absence and the presence of S9 according to the standard procedures. 15 A preliminary toxicity test was performed to establish the range of doses for the performance of the chromosome aberration assay. This chromosome aberration assay was used to evaluate the clastogenic potential of the test article.
The PBS was used as a vehicle for these studies based on the solubility of the test article and compatibility with the target cells. The test article was soluble and formed a clear solution in PBS at a concentration of approximately 50 mg/mL, the maximum concentration tested for solubility. Cyclophosphamide and mitomycin C were incorporated into the study protocol as the concurrent positive controls for treatments with and without S9, respectively.
In the preliminary toxicity assay, the dose levels studied ranged from 0.5 to 5000 µg/mL. The CHO cells were treated for 4 and 20 hours in the nonactivated test system and for 4 hours in the S9-activated test system. All the cells were harvested 20 hours after treatment initiation. Substantial toxicity (ie, at least 50% inhibition of cell growth, relative to the vehicle control) was observed at 5000 µg/mL in the nonactivated 4-hour exposure group and at the dose levels of ≥500 µg/mL in the nonactivated 20-hour continuous exposure group. Substantial toxicity was not observed at any dose level in the S9-activated 4-hour exposure group. Based on these findings, the doses chosen for the chromosome aberration assay ranged from 500 to 5000 µg/mL for the nonactivated and the S9-activated 4-hour exposure groups and for the nonactivated 20-hour continuous exposure group ranged from 75 to 750 µg/mL.
Local Tissue Tolerance
Hemolysis induction in human blood
The test article MnTE-2-PyP at the concentrations of 400, 2000, and 4836 ng/mL, the vehicle (PBS), and the positive control (water) were each individually mixed with a dilution of whole blood from a random human donor. Samples were incubated for 1 hour at 37°C ± 2°C. The red cells were removed from the samples by centrifugation, and the amount of hemoglobin in the supernatant was measured spectrophotometrically by determining the optical density (OD) at 540 nm.
A series of dilutions (1:2, 1:3, 1:4, 1:5, and 1:10) of blood from the donor were prepared in saline. When added to sterile water, it was determined that the 1:3 dilution for the donor blood sample resulted in an OD value of 1.179 nm at 540 nm. Therefore, a 1:3 dilution of the blood sample was used for the main study and is referred to as the blood substrate.
Flocculation induction in human plasma and serum
The MnTE-2-PyP at the concentrations of 800, 4000, and 9672 ng/mL and vehicle (PBS) were individually mixed with equal volumes of human plasma and serum from a human donor. The final concentrations of the test article were 400, 2000, and 4836 ng/mL. Samples were then incubated for 30 minutes at room temperature. After incubation, the tubes were examined macroscopically and microscopically for the presence of precipitation or coagulation.
Intravenous and perivenous irritation
The purpose of this study was to assess the potential for the acute irritation of MnTE-2-PyP when injected intravenously and perivenously into rabbits. The hair surrounding the injection sites was clipped, and the skin area was swabbed with 70% ethanol. Rabbits were administered with the test article and vehicle control via either iv or perivenous injection. The dosing regimen is presented in Table 1. All dose sites were marked with an indelible black-ink marker. The area beyond the point of entry of the needle and its progress were evaluated macroscopically and microscopically. Clinical signs were recorded at 1, 4, 24, 48, and/or 72 hours, and irritation was scored at 1, 24, 48, and 72 hours (as appropriate). Grading of irritation was according to the method of Draize. 16
Intravenous and Perivenous Dosing Regimen.
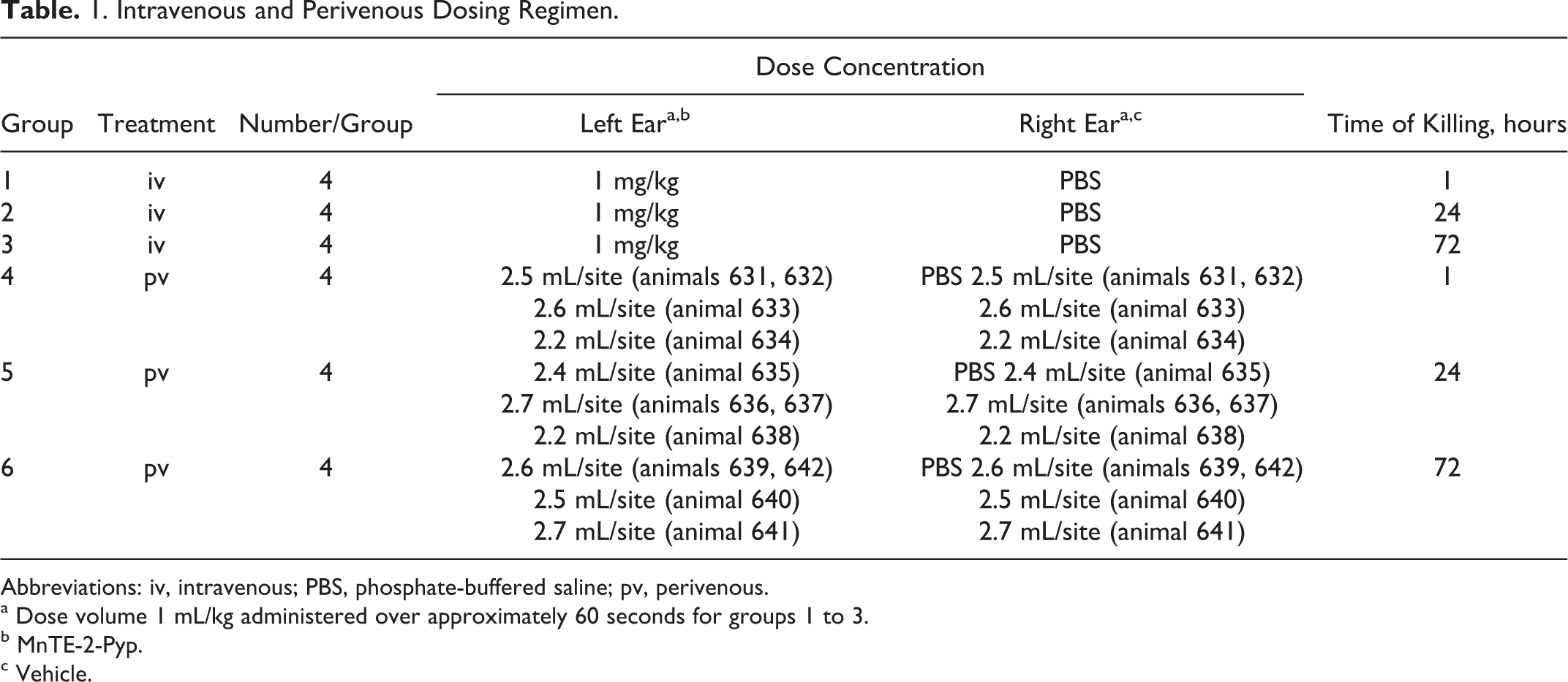
Abbreviations: iv, intravenous; PBS, phosphate-buffered saline; pv, perivenous.
a Dose volume 1 mL/kg administered over approximately 60 seconds for groups 1 to 3.
b MnTE-2-Pyp.
c Vehicle.
Safety Pharmacology Studies
hERG inhibition in HEK 293 cells
The objective of this study was to examine the in vitro effect of MnTE-2-PyP on the hERG channel current (a surrogate for IKr, the rapidly activating delayed rectifier cardiac potassium current) at near-physiological temperature.
Four concentrations of MnTE-2-PyP (10, 30, 100, and 300 μmol/L) were selected to evaluate the concentration–response relationship based on the outcome of the initial concentration range determination. Each concentration was tested in 3 cells (n = 3). Vehicle control solution was applied to 3 cells (n = 3). Duration of application was approximately 7.3 minutes, which was at least as long as the longest test article application in the study.
Cells were transferred to the recording chamber and superfused with the vehicle control solution. The micropipette solution for whole cell patch clamp recordings was composed of: potassium aspartate, 130 mmol/L; MgCl2, 5 mmol/L; ethylene glycol tetraacetic acid, 5 mmol/L; adenosine triphosphate, 4 mmol/L; 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid, 10 mmol/L; and pH adjusted to 7.2 with potassium hydroxide. Micropipette solution was prepared in batches (120604JO027K_MP; expiration date: December 4, 2012), aliquoted, stored frozen, and a fresh aliquot thawed each day. The recording was performed at a temperature of 33°C to 35°C using a combination of in-line solution preheater, chamber heater, and feedback temperature controller. Temperature was measured using a thermistor probe in the recording chamber. Micropipettes for patch clamp recording were made from glass capillary tubing using a P-97 micropipette puller (Sutter Instruments, Novato, California). A commercial patch clamp amplifier was used for whole cell recordings. Before digitization, current records were low-pass filtered at one-fifth of the sampling frequency.
Phase I of the study identified the approximate concentration range, and phase II determined the concentration–response relationship. For phase I, cells stably expressing hERG were held at −80 mV. Onset and steady state inhibition of hERG potassium current due to MnTE-2-PyP were measured using a pulse pattern with fixed amplitudes (conditioning prepulse +20 mV for 1 second; repolarizing test ramp to −80 mV [−0.5 V/s] repeated at 5 seconds intervals). Each recording ended with a final application of a supramaximal concentration of the reference substance (E-4031, 500 nmol/L) to assess the contribution of endogenous currents. The remaining uninhibited current was subtracted off-line digitally from the data to determine the potency of the test substance for hERG inhibition.
The MnTE-2-PyP at a concentration of 300 μmol/L was applied to 3 cells (n = 3). A mean inhibitory effect on hERG potassium current amplitude of 76.6% was observed. Based on this result, additional nominal concentrations (10, 30, and 100 μmol/L) were selected to span the range of approximately 10% to 90% inhibition in half-log increments to evaluate the concentration–response relationship (phase II).
For phase II, 1 test article concentration was applied to each independent cell (n = 3 cells per concentration). Peak current was measured during the test ramp. A steady state was maintained for at least 20 seconds before applying test article or positive control. Peak current was measured until a new steady state was achieved.
The 300 μmol/L formulation in HEPES-buffered physiological saline (HB-PS) was analyzed for stability, homogeneity, and concentration verification. Due to the test method limitations, 10, 30, and 100 μmol/L formulations in HB-PS were not analyzed for stability, homogeneity, and concentration verification. However, these formulations were prepared from the same stock from which the 300 μmol/L formulation was made. The stability under conditions of use and the concentration of the stock solution were determined.
Evaluation of respiratory function of MnTE-2-PyP following single-dose iv administration in CD-1 mice
To evaluate respiratory functions, 24 male mice were acclimated to a head-out plethysmographic chamber for 5 to 15 minutes before the day of the experiment. On the day of dosing, each animal was placed in its plethysmographic chamber, and baseline values were obtained for 5 minutes following an approximately 5-minute stabilization/acclimation period. The mice (6/dose group) were then removed from the chambers and administered 0, 1.0, 3.0, or 10 mg/kg of the test article. Each dose was administered intravenously over a period of 3 minutes. Following dosing, each animal was returned to its previously designated plethysmograph chamber, and respiratory parameters were recorded at 15 minutes and 1, 2, 4, and 6 hours after the administration of the test article. Again, animals were allowed to stabilize in the plethysmographic chamber for at least 5 minutes before each reading was taken. The following parameters were acquired, recorded, and analyzed using DSI Dataquest Open ART (v3.2) and PONEMAH Physiology Platform (v4.20; Data Sciences International (DSI), St. Paul, Minnesota):
Tidal volume
Minute volume
Respiratory rate
Values for the test article-treated group were compared back to the vehicle control and baseline using a 2-way repeated measures analysis of variance (ANOVA) followed by a Bonferroni multiple comparison test (SigmaStat, v2.03; SPSS Inc, Chicago, Illinois). Differences with P ≤ 0.05 were considered statistically significant.
Neuropharmacological profile of MnTE-2-PyP in mice (Irwin screen)
The purpose of this study was to evaluate the potential neuropharmacological effects of MnTE-2-PyP in mice. Three groups of 10 male CD-1 mice were intravenously administered with MnTE-2-PyP at intended (nominal) doses of 1.0, 3.0, or 10 mg/kg in the morning. An additional group of 10 male mice was intravenously administered with the vehicle at 10 mL/kg. Doses were administered over a 3-minute period of time. The mice were observed for the development of apparent neuropharmacological or toxicological signs at 15, 30, 45, and 60 minutes, 2, 3, 4, and 24 hours (±5-15 minutes as appropriate) following dosing. Body temperatures were measured at 30 minutes (±5 minutes) following dose administration. No statistical analysis was performed for behavioral tests, because the data generated were not quantitative enough to allow for meaningful comparison. Body temperature values were presented as mean ± standard error of the mean (SEM), and statistically significant effects were determined by ANOVA with Tukey honestly significant difference multiple comparison test (Systat V. 9.01, Systat Software, Inc, San Jose, California). A P ≤ 0.05 denotes statistical significance.
Evaluation of cardiovascular function following administration of MnTE-2-PyP in conscious telemetered male cynomolgus monkeys
The purpose of this study was to determine the potential acute effects of MnTE-2-PyP on cardiac and hemodynamic functions and the electrocardiograms (ECGs) of conscious radiotelemetered male cynomolgus monkeys. The study consisted of a single treatment group of 4 cynomolgus monkeys, each receiving the vehicle (PBS) and 4 doses of MnTE-2-PyP (1, 3, and 10 mg/kg per day) in the morning. The dose volume administered was 5 mL/kg. Animals were observed predose, 2 to 4 hours, and 22 hours postdose administration. The control and test articles were administered according to a Latin-square design. A minimum washout period of 72 hours was allowed between the doses. One-minute means of hemodynamic parameters as well as ECG parameters were measured for a period of 22 hours following each dose. A board-certified veterinary cardiologist examined the 1-minute tracings of the ECGs at 15 minutes prior to dosing and at 30 minutes, 1, 2, 4, 8, 12, and 22 hours following dose administration. The means for the treated groups were compared to the vehicle control means as well as to baseline using a 2-way repeated measures ANOVA followed by a Bonferroni multiple comparison test (SigmaStat, v2.03). Differences with P < 0.05 were considered statistically significant.
Systemic Toxicity
Escalating Dose Range Finding and 5-Day Repeat-Dose Study by iv Injection With MnTE-2-PyP in Mice
The maximum tolerated dose (MTD) level of MnTE-2-PyP was evaluated in both male and female CD-1 mice following iv dose administration in the morning. The toxicity of the test article was further evaluated by dosing once daily in the morning for 5 consecutive days.
For phase I, MnTE-2-PyP was administered in the morning by iv slow bolus (administered over 3 minutes at a dose volume of 10 mL/kg) to 3 to 4 naive mice per sex (3 males/4 females) at 20 mg/kg/dose. After a minimum period of approximately 48 hours, an escalated dose level of 40 mg/kg/dose was administered to an additional 4 naive mice (3 males/1 female). A lower dose level of 30 mg/kg/dose was administered to 6 naive mice (3/sex) using the same dosing scheme. Mortality/morbidity was evaluated twice daily. Clinical observations were recorded prior to each dose and immediately postdose, 1 hour postdose, and 4 to 6 hours postdose, and once daily on nondosing days. Body weights were recorded at the time of randomization/selection and prior to the administration of each dose. Food consumption was assessed daily. All the surviving animals were killed at least 2 days after dose administration. A gross necropsy was performed, and tissues were evaluated and discarded.
For phase II, the test article was administered in the morning by iv slow bolus to 4 groups of naive mice (6/sex/group) once daily for 5 days at 0, 10, 20, or 30 mg/kg/dose. Mortality and morbidity were evaluated twice daily. Clinical observations were recorded prior to each dose, immediately postdose, and approximately 1-hour postdose. Body weights were recorded prior to dose administration on days 1, 3, and 5. A fasted body weight was recorded prior to the terminal killing on day 6. Food consumption was assessed daily. Blood was collected prior to terminal killing on day 6 for the evaluation of hematology and clinical chemistry. Selected tissues were harvested at necropsy, targeted organs were weighed, and the selected tissues were saved for possible future histopathological evaluation.
A 18-Day iv Toxicity Study With MnTE-2-PyP in Mice With a 17-Day Recovery
A total of 339 experimentally naive CD-1 mice (170 males and 169 females), approximately 6 weeks old, and weighing 24 to 34 g for the males and 18 to 28 g for the females at the outset (day 1) of the study were administered with the test article of 0, 4, 10, and 20 mg/kg/day by iv slow bolus injection (administered over 3 minutes at a dose volume of 10 mL/kg) in the morning. Vehicle control and high-dose groups comprised n = 40 animals/group (20 males and 20 females), whereas both low- and mid-dose groups comprised n = 20 animals/group (10 males and 10 females). On day 18, a previously specifically identified 10 animals/sex/group were euthanized. The remaining animals in the control and high-dose groups remained on study, untreated, until their scheduled killing on day 36 (recovery animals).
The dosing phase was shortened from 28 to 18 days, followed by a 17-day drug-free recovery period, due to the development of bruising and swelling at the tail injection sites, which was observed to occur as early as day 3 in females dosed at 4, 10, and 20 mg/kg to as late as day 16 in females dosed at 0 mg/kg. Because of this difficulty in finding the tail vein, several animals were not dosed consecutively for 18 days.
Mortality and morbidity observations were evaluated daily. Clinical observations were recorded prior to dose administration and approximately 1 to 2 hours postdose on dosing days and once daily on nondosing days. Body weight and food consumption values were recorded weekly. Blood samples were collected from a targeted 5 animals/sex/group for evaluation of hematology parameters and from a separate targeted 5 animals/sex/group for evaluation of clinical chemistry parameters on day 36 (recovery animals). For pharmacokinetic evaluation, blood was collected from animals in the specifically identified pharmacokinetic groups at selected 0 (immediate predose), 0.5, 2, 4, 8, and 24 hours postdose on days 1 and 18. All surviving animals in the toxicology groups were killed on day 18 or 36. Selected tissues were harvested at necropsy; targeted organs were weighed as per the protocol, and selected tissues from the control and high-dose groups were collected and subjected to microscopic evaluation.
A 5-Day iv Toxicity Study With MnTE-2-PyP in Monkeys
The purpose of this study was to evaluate the toxicity of the test article MnTE-2-PyP, when administered once daily by iv slow bolus to cynomolgus monkeys for a minimum of 5 consecutive days.
Four experimentally naive cynomolgus monkeys (2 males and 2 females), approximately 2 to 3 years old and exhibiting weight ranges of 2.4 to 2.6 kg for males and 2.3 to 2.6 kg for females at the outset of the study, were administered with 20 mg/kg/day of MnTE-2-PyP in the morning by iv slow bolus over a 2-minute period of time for 5 consecutive days at 1.0 mL/kg. Mortality and morbidity were evaluated twice daily. Clinical observations were recorded prior to each dose, immediately postdose, 1 hour postdose, and 4 to 6 hours postdose, and once daily on nondosing days. Body weights were recorded daily prior to dose administration on days 1 to 5. A fasted body weight was recorded on day 6. Food consumption was assessed daily. Electrocardiograms and blood samples were obtained from all animals prior to treatment initiation and prior to terminal killing on day 6 for evaluation of serum chemistry, hematology, and coagulation parameters. All surviving animals were killed on day 6. Selected tissues were harvested at necropsy, and the targeted organs were collected and weighed for possible future histopathological evaluation.
A 14-Day iv Toxicity Study With MnTE-2-PyP in Cynomolgus Monkeys With a 14-Day Recovery
The purpose of this study was to evaluate the toxicity of the test article MnTE-2-PyP when administered once daily in the morning by iv slow bolus over a 3-minute period to cynomolgus monkeys for a minimum of 14 consecutive days followed by a 14-day recovery period. A total of 32 experimentally naive cynomolgus monkeys (16 males and 16 females; 4/sex/group), approximately 2 to 3 years old and weights ranging from 2.2 to 2.9 kg for males and 2.1 to 2.6 kg for females at the outset (day 1) of the study, were administered with 0, 2.0, 5.0, or 15 mg/kg/day of MnTE-2-PyP once daily in the morning for 14 consecutive days. Mortality and morbidity were evaluated daily. Clinical observations were recorded prior to the administration of the dose, immediately postdose and approximately 1 to 2 hours postdose on dosing day, and once daily on nondosing days. Body weights were recorded daily prior to each dose administration on days 1 to 14. Animals in the designated recovery group were weighed weekly during the 2-week recovery period. Fasted body weights were recorded prior to scheduled killing on days 15 and 29 (recovery animals). Food consumption values were recorded daily. Ophthalmology examinations were performed prior to treatment initiation and during the last week of treatment. Electrocardiograms and blood samples were obtained from all animals prior to treatment initiation and prior to terminal killing on days 15 and 29 (recovery animals) for evaluation of serum chemistry, hematology and coagulation parameters. For pharmacokinetic evaluation, blood samples were collected from all animals at selected time points on days 1 and 14. Selected tissues were harvested at necropsy, targeted organs were collected and weighed, and histopathological evaluation was performed on all the tissues for all the animals killed on days 15 and 29 (recovery animals).
For all of the repeat-dose studies, statistical evaluations were performed for in-life, clinical pathology, and organ weight numerical data. For in-life and clinical pathology parameters, statistical significance was determined by following a decision tree. First, the homogeneity of the data was determined by Bartlett test. If the data was homogenous, a 1-way ANOVA was performed to assess the statistical significance. If the statistically significant differences between the means were found, the Dunnett test was used to determine the degree of significance from the control means. If the data was nonhomogeneous, the Kruskal-Wallis nonparametric analysis was performed to assess the statistical significance. If the statistically significant differences between the means were found, the Mann-Whitney U test was used to determine the degree of significance from the control means. For necropsy organ weight data, the evaluation of the equality of means was made by a 1-way ANOVA using the F distribution to assess statistical significance. If statistically significant differences between the means were found, Dunnett test was used to determine the degree of significance from the control means. 17
Results
Genotoxicity
Bacterial reverse mutation assay
In the initial toxicity-mutation assay, no positive mutagenic responses were observed with any of the tester strains in the presence or absence of S9 activation. No presence of precipitate was observed. Cell toxicity was observed beginning at 500 µg/plate with all the test conditions except tester strain TA98 in the presence of S9 activation. Based on the findings of the initial toxicity mutation assay, the maximum dose plated in the confirmatory mutagenicity assay was 5000 µg/plate.
In the confirmatory mutagenicity assay, no positive mutagenic responses were observed with tester strains TA98, TA1535, and WP2 uvrA in the absence of S9 activation and tester strains TA98, TA100, TA1535, and WP2 uvrA in the presence of S9 activation, as shown in Table 2. A slight increase in revertant counts (2.3-fold maximum increase) was observed with tester strain TA100 at 500 µg/plate in the absence of S9 activation. No presence of any precipitate was observed. Cytotoxicity was observed at ≥1500 µg/plate with all Salmonella tester strains. Due to presence of unacceptable vehicle control values, tester strain TA1537 was not evaluated for mutagenicity but instead was retested. In the absence of S9 activation, the tester strain TA100 was also retested to clarify the response that was observed.
Confirmatory Mutagenicity for MnTE-2-PyP.
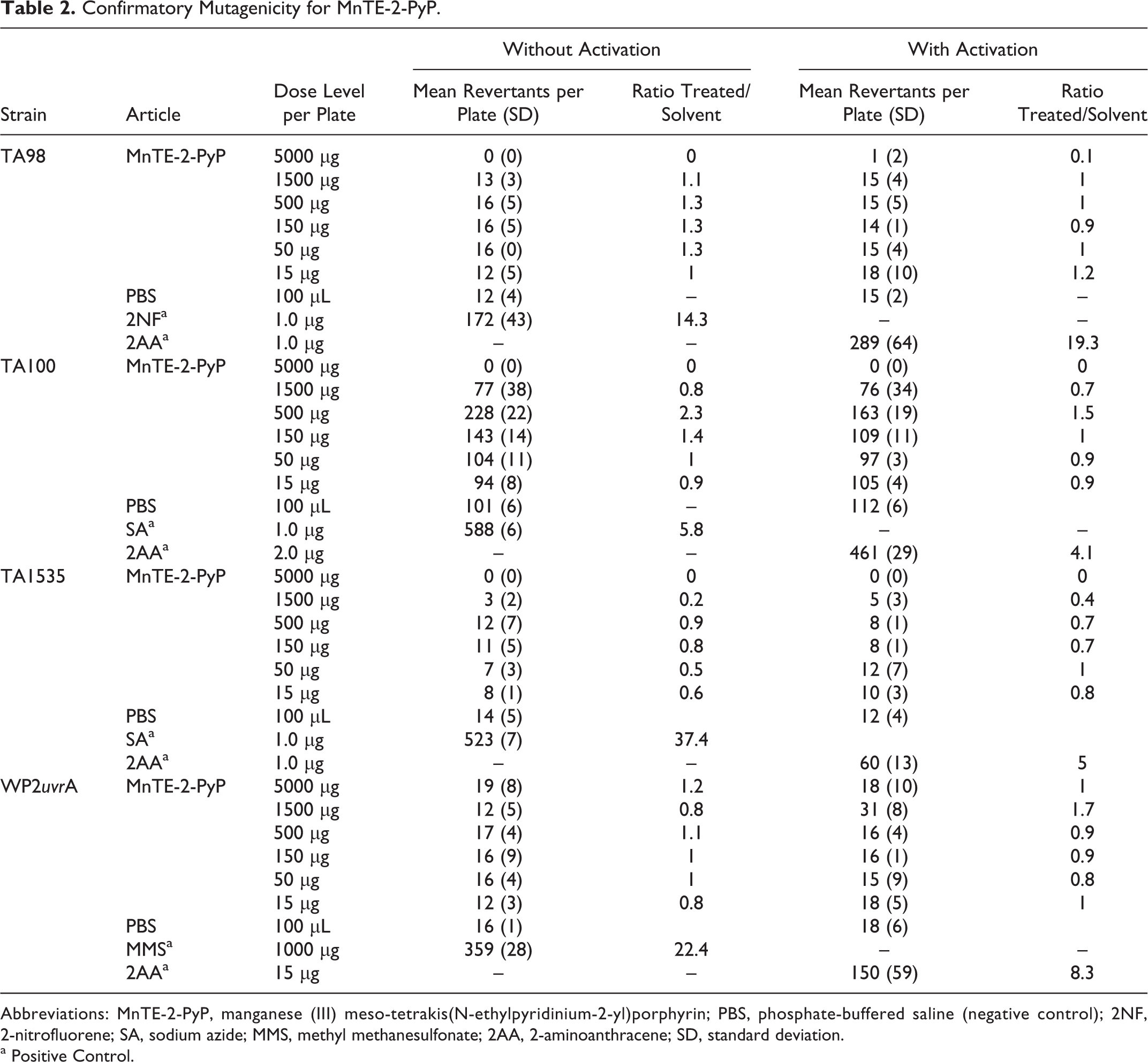
Abbreviations: MnTE-2-PyP, manganese (III) meso-tetrakis(N-ethylpyridinium-2-yl)porphyrin; PBS, phosphate-buffered saline (negative control); 2NF, 2-nitrofluorene; SA, sodium azide; MMS, methyl methanesulfonate; 2AA, 2-aminoanthracene; SD, standard deviation.
a Positive Control.
In the retest of the confirmatory mutagenicity assay, no positive mutagenic responses were observed with tester strain TA100 in the absence of S9 activation and tester strain TA1537 in the presence or absence of S9 activation, as shown in Table 3. The dose levels tested were 15, 50, 150, 250, 500, 600, 1500, and 5000 µg/plate. No precipitate was observed. Cytotoxicity was observed at ≥1500 µg/plate. Under the conditions of this study, MnTE-2-PyP was concluded to be negative in the bacterial reverse mutation assay with all the test conditions
Retest of Confirmatory Mutagenicity for MnTE-2-Pyp.
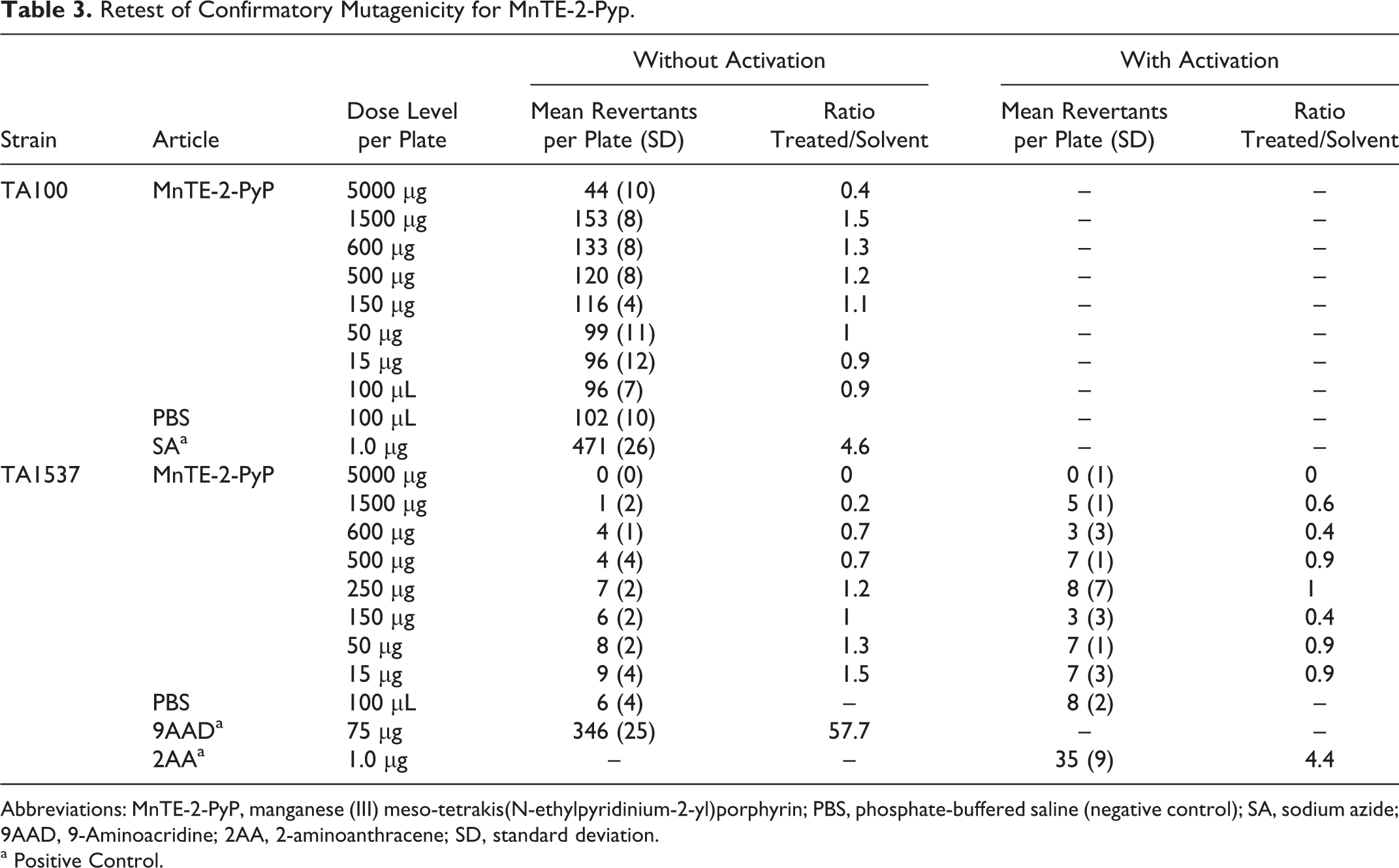
Abbreviations: MnTE-2-PyP, manganese (III) meso-tetrakis(N-ethylpyridinium-2-yl)porphyrin; PBS, phosphate-buffered saline (negative control); SA, sodium azide; 9AAD, 9-Aminoacridine; 2AA, 2-aminoanthracene; SD, standard deviation.
a Positive Control.
In vitro mammalian chromosome aberration test
No significant or dose-dependent increases in aberrant metaphases, or polyploid or endoreduplicated cells, were observed in the treatment groups with or without S9 (P > 0.05; Fisher exact test). All vehicle control values were within historical ranges, and the positive controls induced significant increases in the percentage of aberrant metaphases (P ≤ 0.01). Thus, all criteria for a valid study were met. These results indicate that MnTE-2-PyP was negative in the in vitro chromosome aberration assay in CHO cells under the conditions and according to the criteria of the study protocol.
Local Tissue Tolerance
Hemolysis induction in human blood
Incubation of the human blood substrate with MnTE-2-PyP at 400, 2000, and 4836 ng/mL did not cause the development of any significant level of hemolysis of the human blood. The percentages of hemolysis were calculated to be −0.54%, −0.54%, and −0.36%, respectively. Based on the results of this study, MnTE-2-PyP at 400, 2000, and 4836 was found to be compatible with human blood.
Flocculation induction in human plasma and serum
There was no presence of precipitation or coagulation observed either macroscopically or microscopically in the human plasma or serum when mixed with the MnTE-2-PyP to result in final concentrations of 400, 2000, or 4836 ng/mL in PBS. When these samples were centrifuged, no pellets were observed to be present. Based on the results of this study, MnTE-2-PyP is determined to be compatible with human plasma and serum at a concentration of up to 4836 ng/mL.
Intravenous and perivenous irritation
In general, the iv or perivenous injection of test article exhibited similar reactions when compared with the iv or perivenous injection of PBS. The development of clinical signs of a very slight to moderate degree of erythema and edema was observed at 1 hour in the areas treated with test article and PBS, respectively, and the lesion appeared to ameliorate over the course of the ensuing 72 hours.
Microscopic examinations also revealed that the test article, when injected intravenously or perivenously, elicited a very similar tissue response when compared with the injection of PBS alone. The tissue response was characterized by the development of signs of a minimal to moderate degree of perivascular edema and/or hemorrhage, and the development of a minimal to slight degree of perivascular-mixed inflammatory infiltration, which was composed predominantly of heterophils. Overall, the tissue response was comparable between sites injected with PBS and the sites injected with test article at all time points (1, 24, and 72 hours).
Macroscopically, the iv administration of MnTE-2-PyP was found to be slightly irritating at 1 hour postexposure, but the irritation appeared to improve by 24 and 48 hours postdose and appeared to be completely resolved by 72 hours after exposure. The perivenous administration of MnTE-2-PyP appeared to be moderately irritating at 1 hour postdose, with the severity of irritation found to be decreasing at 24 and 48 hours after exposure, and the irritation resolving in 2 animals and with irritation persisting in 2 animals at 72 hours after injection. Microscopically, under the conditions of this study, a single iv or perivenous injection of the test article caused a tissue response that is comparable with a single iv or perivenous injection of PBS.
Safety Pharmacology Studies
hERG inhibition in HEK 293 cells
The MnTE-2-PyP inhibited hERG current by (mean ± SEM; n = 3) 2.7% ± 0.8% at 10 μmol/L, 10.9% ± 2.1% at 30 μmol/L, 27.5% ± 0.5% at 100 μmol/L, and 76.6% ± 3.0% at 300 μmol/L versus 1.9% ± 0.5% (n = 3) in control. The hERG inhibition observed at 30, 100, and 300 μmol/L was found to be statistically significant (P ≤ 0.05) when compared with vehicle control values. However, with only 2.7% hERG inhibition at 10 μmol/L, there is no clinically relevant hERG potential at a minimum potential clinical concentration (0.1 µmol/L and below).
The half maximal inhibitory concentration (IC50) for the inhibitory effect of MnTE-2-PyP on hERG potassium current was determined to be 160.6 μmol/L (Hill coefficient = 1.7).
Under similar conditions, the positive control (60 nmol/L terfenadine) inhibited hERG potassium current by (mean ± SD; n = 2) 78.9% ± 4.7%. This result confirms the sensitivity of the test system to hERG inhibition.
Evaluation of respiratory function of MnTE-2-PyP following single-dose iv administration in CD-1 mice
The iv administration of the vehicle, PBS, or MnTE-2-PyP at 1.0 or 3.0 mg/kg did not produce any statistically significant changes in the respiratory rate, tidal volume, or minute volume when compared with baseline values.
Statistically significant (P ≤ 0.05) decreases in respiratory rate and minute volume were observed at 60 minutes following the iv administration of MnTE-2-PyP at 10 mg/kg when compared with the baseline values. However, these changes were not statistically significant when compared with the vehicle control group at 60 minutes following dosing and were within the normal range of variation observed in this model. There were no other effects on respiratory rate or minute volume observed at other time points following dosing of MnTE-2-PyP. Tidal volume was not affected.
Single iv administrations of MnTE-2-PyP at doses up to 10 mg/kg did not produce any definitive effects on respiratory rate, tidal volume, or minute volume in conscious male mice.
Neuropharmacological profile of MnTE-2-PyP in mice
The iv administration of MnTE-2-PyP at 1.0, 3.0, or 10 mg/kg did not produce any neuropharmacological or toxicological signs up to 24 hours postdose, with the exception of an apparent dose-dependent darkened urine color. The darkened urine was most likely a consequence of the dark brown color of the dosing formulations.
No effects were observed upon body temperature at 30 minutes postdose in any mice receiving the vehicle at 10 mL/kg or MnTE-2-PyP at doses of 1.0, 3.0 or 10 mg/kg, administered intravenously. In summary, the iv administration of MnTE-2-PyP at doses up to and including 10 mg/kg did not produce any neuropharmacological signs or effects upon body temperature. Based on the results of this study, the no observed adverse effect level (NOAEL) is 10 mg/kg.
Evaluation of cardiovascular function following administration of MnTE-2-PyP in conscious telemetered male cynomolgus monkeys
There were no test article-related clinical signs of toxicity observed following the administration of MnTE-2-PyP at a dose of 1 mg/kg. Decreased activity and yellow skin tone and/or amber-colored urine were noted for all the animals at about 2 to 4 hours following the administration of MnTE-2-PyP at 3 and 10 mg/kg. Animal #1 that received MnTE-2-PyP at 10 mg/kg also was found to exhibit decreased activity and appetite for up to 22 hours following dosing. The only sign that was exhibited by an animal receiving the vehicle control article (PBS) was decreased appetite (monkey #2, at 22 hours following the second dosing day). This animal was also found to have had decreased appetite on day 12, 22 hours following administration of 3 mg/kg MnTE-2-PyP. Monkey #2, which received a dose of 1 mg/kg of MnTE-2-PyP on day 1, exhibited some food particle emesis during the course of the dosing procedure. Food particle emesis was also noted on day 1 during dosing for animal #3 receiving 3 mg/kg MnTE-2-PyP and at 5 hours postdose for animal #4 receiving 10 mg/kg MnTE-2-PyP. However, review of the data indicates that these animals were inadvertently given their morning feed approximately 2.5 hours prior to ketamine administration, which was the most likely cause of this emesis.
Administration of 1 mg/kg MnTE-2-PyP was not associated with the development of any statistically significant changes in heart rate or arterial blood pressure.
There were no statistically significant changes in heart rate or rhythm following the administration of 3 mg/kg MnTE-2-PyP. A mild decrease in arterial pressure was noted during the 15-minute period following dose administration that was only statistically significant versus the vehicle control for systolic arterial pressure (127 vs 156 bpm, 19% decrease).
Administration of 10 mg/kg MnTE-2-PyP was associated with a statistically significant decrease in arterial pressure from the time of dosing until approximately 45 minutes following dosing. Heart rate was found to be increased from a time approximately 1.5 to 2.5 hours following dose administration. The maximum increase in mean heart rate was observed to occur at approximately 2 hours postdose with a mean heart rate of 154 versus 107 bpm for the vehicle control (44% increase).
Intravenous dosing with MnTE-2-PyP at up to 10 mg/kg was not found to have any toxicologic effects on cardiac rhythm or ECG morphology in monkeys in this study.
There were no definitive test article-related changes in the quantitative ECG values measured. A few statistically significant changes were noted, but these were considered incidental and unrelated to treatment.
Intravenous dosing of MnTE-2-PyP at 1 mg/kg to cynomolgus monkeys was not associated with any changes in heart rate or arterial blood pressure. Intravenous dosing of MnTE-2-PyP was associated with a decrease in arterial pressure just following dosing at 3 and 10 mg/kg. This was followed by the observation of an increase in heart rate from approximately 1.5 to 2.5 hours following the administration of 10 mg/kg MnTE-2-PyP intravenously. There were no changes observed in cardiac rhythm or ECG morphology in vivo at any dose level nor were there any changes in quantitative ECG parameters noted.
Systemic Toxicity
Escalating Dose Range Finding and 5-Day Repeat-Dose Study by Intravenous Injection With MnTE-2-PyP in Mice
The presence of MNTE-2-PyP used in this study was found to cause adverse clinical signs when administered once in an escalating dose paradigm at 20, 30, and 40 mg/kg/dose and when administered daily at amounts of 20 and 30 mg/kg/dose for 5 consecutive days. The test article-related clinical findings included but were not limited to abnormal stance and gait, decreased activity, hunched posture, labored breathing, and body trembling. Isolated individual seizures were also observed when animals were dosed at 30 and 40 mg/kg/dose. Based on these findings, the NOAEL in mice after 5 consecutive days of treatment was determined to be 10 mg/kg/dose.
An 18-Day iv Toxicity Study With MnTE-2-PyP in Mice With a 17-Day Recovery
A total of 21 unscheduled deaths were recorded, 7 of which were from the main study groups, and an additional 14 were from the pharmacokinetic satellite groups. Of the 7 deaths in the main study groups, 2 (1 male/1 female) were from animals dosed at 4 mg/kg/day (group 2, low dose) and 5 (1 male/4 females) were from animals dosed at 20 mg/kg/day (group 4, high dose). The deaths of 2 animals in the low-dose group and at least 1 animal in the high-dose group were not considered to be test article related for the following reasons. The low-dose male did not exhibit any adverse clinical signs during the dosing phase, whereas the low-dose female and a high-dose female died inside the restrainer while being dosed, so their deaths were considered to be accidental. There were no deaths observed in controls or animals dosed daily at 10 mg/kg during the study.
There were clinical findings observed postdose in animals dosed at 4, 10, and 20 mg/kg/day from days 1 to 18. The test article-related clinical signs included but were not limited to body tremors (mild to moderate), abnormal stance and gait, ruffled hair, hunched posture, decreased activity, yellow tail skin color, discolored urine (amber or red), and bruised/swollen tails (mild to moderate to severe), dissipating as 1 moved away from the injection site. In general, yellow tail skin color and discolored urine were also noted predose starting at day 2. The number of animals that exhibited these clinical signs and their frequency of occurrence generally increased with the escalation of the dose level. For animals dosed at 20 mg/kg/day, the manifestation of clinical signs might have been higher if all the animals had been dosed consecutively for 18 days. However, due to swollen and bruised tails at the injection sites of animals in all groups including controls, some animals did not receive their daily dose consecutively for 18 days or were only partially dosed due to the leakage of test article from previous injection sites. Consequently, the frequency of missed doses and the number of animals that missed their daily dose were highest in the high-dose group when compared with the low- and mid-dose groups.
There were no test article-related effects on mean body weights and food consumption parameters during the treatment phase and recovery period. There were no test article-related effects in surviving animals on red blood cell parameters, erythrocyte morphology, or clinical chemistry parameters on day 18 or 36.
Histopathological evaluation of tissues indicated that there were no macroscopic or microscopic findings to account for the observed mortality of the 7 animals that were found dead during the course of the study. At day 18, there were no microscopic observations in systemic organs/tissues that were found to be related to the administration of MnTE-2-PyP. The increased incidence and/or severity of several microscopic findings (perivascular acute inflammation, necrosis of the treatment veins, and perivascular edema) at the injection sites in males and females at 20 mg/kg/day of MnTE-2-PyP were consistent with local irritation and toxicity of the test article.
The MnTE-2-PyP administered intravenously daily to mice at 20 mg/kg over an 18-day treatment period caused mortality and the development of adverse clinical signs including body tremors, abnormal stance and gait, ruffled hair, hunched posture, and decreased levels of activity, but no identifiable target organ toxicity was specifically observed. Animals dosed at 20 mg/kg had a higher frequency and severity of occurrence of the development of adverse clinical signs, and more animals exhibited these signs when compared with controls and those dosed at 4 and 10 mg/kg/day. Although clinical findings were noted at day 1 in 1 male dosed at 4 mg/kg and at day 1 in 1 female dosed at 10 mg/kg, there was no recurrence in all subsequent dosing days and following a 17-day drug-free recovery period. Based on these observations as well as the clinical pathology and histopathology findings, this study supported an assignation of a NOAEL of 10 mg/kg/day in mice (which were more sensitive than monkeys to the toxicity of MnTE-2-PyP by the iv route).
A 5-day iv toxicity study with MnTE-2-PyP in monkeys
All these animals survived until their scheduled time of killing on day 6. There were test article-related clinical findings observed during the course of the study. Immediately after dosing on day 1, all animals exhibited ataxia, and 1 male and 2 females had decreased activity and squinting of the eyes. On days 2 to 5, the most common clinical findings observed immediately after dosing were ataxia, decreased activity, squinting of the eyes, recumbency (lateral or sternal), and labored breathing. Other animals also exhibited vocalization, reddening of the face, and total lack of activity. Overall, gradual recovery was observed in all animals occurring within 30 minutes postdose on day 1 to 5. By 1-hour postdose, all animals appeared normal except on days 2 and 5, where 1 female exhibited persistence in eye squinting. At 4 to 6 hours postdose, almost all animals had dark brown-stained areas (presumptive urine) in their cage pans. All animals appeared normal prior to terminal killing on day 6. There were no test article-related effects on body weights during the course of the study.
There were test article-related effects observed on food consumption values. There was a considerable decrease in the amount of food consumed by all the animals during the course of the study. Out of 100 g of dry food provided daily to each animal, 0 to 56 g and 0 to 67 g were consumed daily by the males and females, respectively, from days 1 to 5. Specifically, on day 1, male 01 did not consume its food, male 02 ate only 5 g, female 03 ate 28 g, and female 04 ate 32 g. On day 2, males 01 and 02 consumed 34 and 49 g of food, respectively, whereas females 03 and 04 consumed 35 and 0 g of food, respectively. A similar pattern of decreased food consumption was observed on days 3 to 5.
There were no test article-related effects on ECG rate or rhythm or morphology. There were no test article-related effects found with group mean hematology parameters. There were no test article-related effects on erythrocyte morphology. The hemogram indicated that with regard to the red blood cell parameters, the profile was normocytic and normochromic on day 6. There were no test article-related effects on group mean coagulation parameters on day 6.
No definitive test article-related effects were observed on any group mean clinical chemistry parameters. However, the mean levels of glucose and creatinine in males and females were slightly outside or above the upper limits of the historical database range for glucose and creatinine levels and were 6.25% and 22.1% higher, respectively, in males and 19% higher in females when compared with the database range (see Table 4). There were no rest article-related gross necropsy findings and no test article-related effects on organ weights.
Clinical Chemistry Data in Male and Female Cynomolgus Monkeys Administered MnTE-2-PyP.
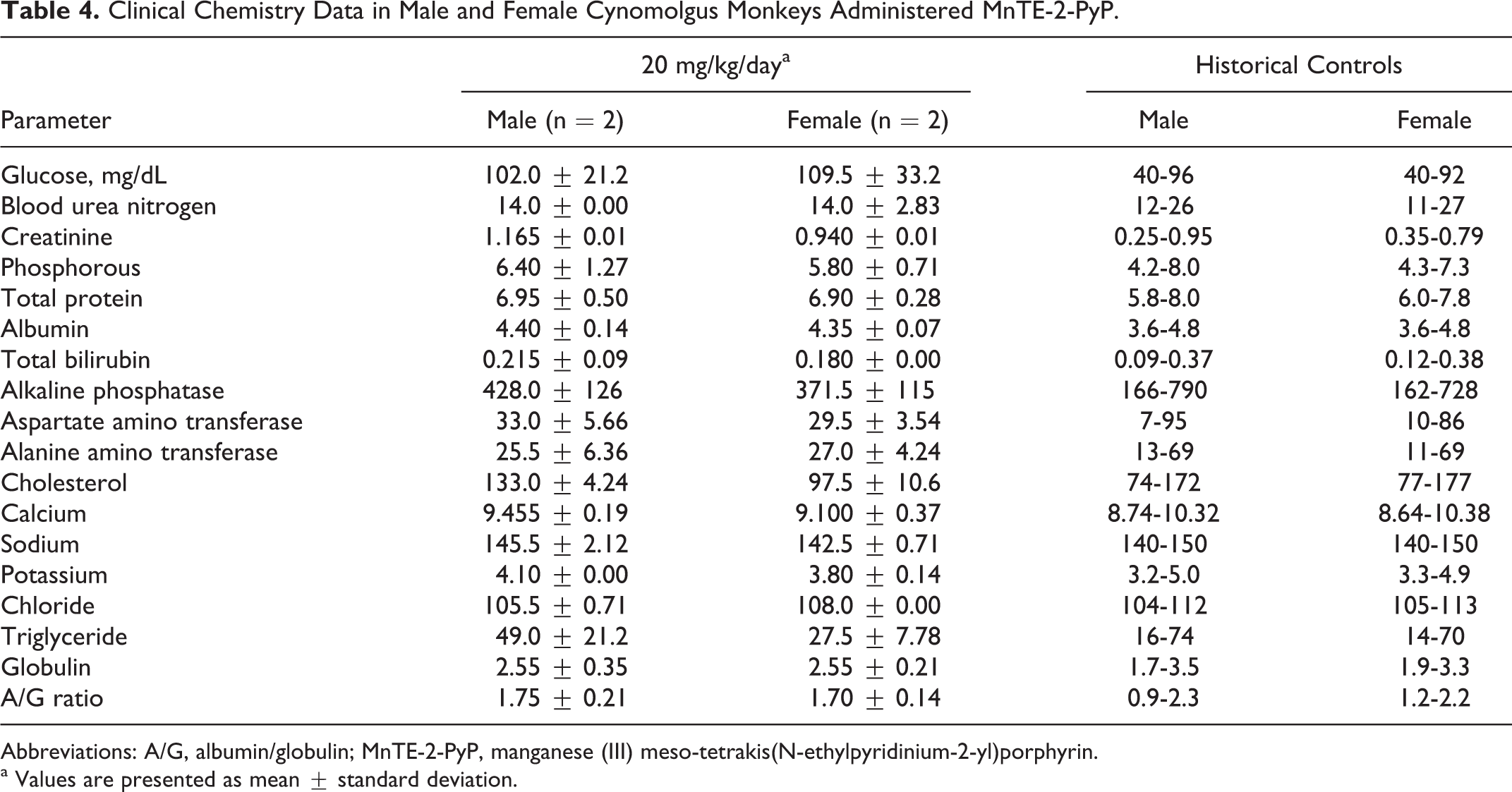
Abbreviations: A/G, albumin/globulin; MnTE-2-PyP, manganese (III) meso-tetrakis(N-ethylpyridinium-2-yl)porphyrin.
a Values are presented as mean ± standard deviation.
The dosing of cynomolgus monkeys with 20 mg/kg of MnTE-2-PyP daily for 5 consecutive days was determined to cause the development of severe clinical signs and a considerable decrease in food consumption values. Clinical signs observed included ataxia, decreased activity, squinting of the eyes, recumbency, labored breathing, vocalization, reddened face, lack of activity, and the presence of a dark brown staining (presumptive urine) in cage pans. It was also observed that there was a strong potential for test article-related increases in the levels of glucose and creatinine.
A 14-day iv toxicity study with MnTE-2-PyPin cynomolgus monkeys with a 14-day recovery
All these animals survived until their scheduled killing times at days 15 and 29 (recovery animals). There were test article-related clinical findings when animals were dosed at 15 mg/kg/day for 14 consecutive days. Postdose findings in the early treatment phase (ie, days 1-4) were mild and included decreased activity (mild), salivation, fully or partially closed eyes, yellow skin color, reddened face and limbs, and amber-colored urine. As dosing continued from days 5 to 14, more adverse findings started to develop, and these included the development of an abnormal stance and gait, lateral recumbency, decreased activity (moderate to severe), and decreased muscle tone. Other adverse findings at 15 mg/kg/day occurred once and included moderate vocalization, labored respiration, emesis and the presence of mild body tremors, and body twitching. Animals dosed at lower than 15 mg/kg/day had only mild and significantly less frequent clinical signs.
Overall, the onset of the development of clinical signs was immediate and lasted no more than 1 hour after dosing, after which, most animals appeared to return to normal except for the presence of the discolored urine and presence of yellow skin. During the drug-free recovery period (days 15-28), amber-colored skin was found to persist in 2 males at days 15 to 17. All other animals appeared normal until killing at day 29. There were no test article-related effects on body weights during the study.
There were test article-related effects on food consumption parameters. During the treatment phase (days 1-14), average food consumption values were found to decrease significantly in males and females dosed at 15 mg/kg/day. However, food consumption values reversed to normal levels by day 15 and remained so until the end of the recovery period at day 28. There were no test article-related ophthalmological findings noted during the course of the study. There were no test article effects on ECG rate or rhythm or morphology. All monkeys maintained normal sinus rhythms, and the presence of no ventricular arrhythmias was noted. There were no test article-related effects observed on group mean hematology, coagulation, clinical chemistry parameters, organ weights, or macroscopic, or microscopic findings at day 15 or 29.
The MnTE-2-PyP administered intravenously to cynomolgus monkeys once daily for 14 consecutive days produced adverse clinical signs at 15 mg/kg/day, which included abnormal stance and gait, lateral recumbency, decreased muscle tone, decreased activity (moderate to severe), moderate vocalization, labored respiration, mild body tremors, and twitching, most of which lasted for less than an hour after dosing. In addition, animals dosed at 15 mg/kg/day were observed to have significantly lower food consumption values throughout the 14-day treatment period. However, this decrease was found to be reversible at the end of the recovery period. Based on these results, this study supported an assignation of a NOAEL of 5 mg/kg/day and a histopathological-based no observed effect level (NOEL) of 15 mg/kg/day in male and female monkeys.
Pharmacokinetic Results
The in vivo pharmacokinetics of MnTE-2-PyP were assessed in the mouse and monkey. Pharmacokinetic parameters for the mouse and monkey are provided in Tables 5 and 6, respectively. For the mouse, plasma levels as characterized by area under the curve (AUC) and C max increased proportionately with dose on days 1 and 22. Levels were lower after the last administration (day 22) than after the first and were generally lower in females after the first and last administrations. However, the differences between genders were less after the last administration than after the first.
Pharmacokinetic Parameters for the Mouse Repeat-Dose Study.
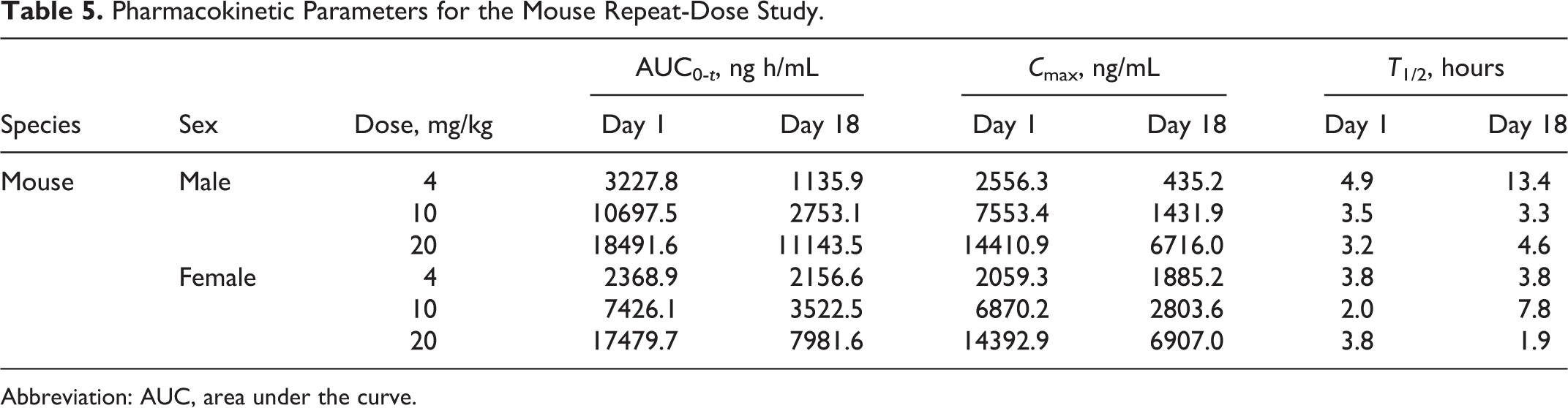
Abbreviation: AUC, area under the curve.
Pharmacokinetic Parameters for the Monkey Repeat-Dose Study.
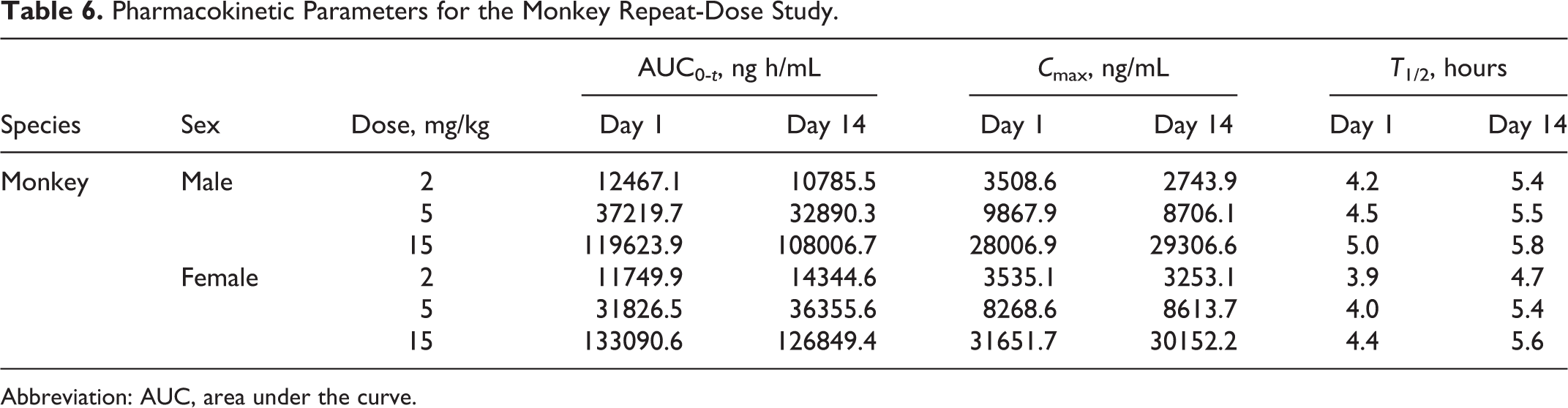
Abbreviation: AUC, area under the curve.
Monkey plasma levels as characterized by AUC and C max increased proportionally with administered dose on days 1 and 14. In males (but not females), plasma levels were lower after the last administration than after the first. Although day 1 plasma levels were lower in females than males, this was not the case after 14 days of dosing. Half-lives were comparable across the range of doses studied after 14 days of administration.
To assess the potential risks from carryover of drug when perfused tissues or organs were transplanted, the measured maximum retained levels of MnTE-2-PyP in perfused primate livers (the tissue in which the levels of retention are highest) were 9236 or 9.24 µg/g of tissue. In a 70-kg human, the liver weighs 1.5 kg. The MnTE-2-PyP levels translated across species would, in the worst case, be 13.86 mg. Therefore, any organ or tissue weighing 541 g (0.54 kg) would have 5 mg or less of retained MnTE-2-PyP in the worst case.
Discussion
Porphyrins are carbon-based compounds made up of 4 pyrrole rings connected by methane bridges. 18 These molecules often form complexes with metals by incorporating them into the nucleus of the porphyrin structure, situated between the interiorly located vertices of the pyrrole rings. In terms of toxicity, the most common and best-studied metalloporphyrin compound is heme, which is an iron–protoporphyrin complex. 19,20 Heme is an essential molecule for living aerobic organisms, because it plays an essential role in various biological reactions including oxygen transport, respiration, drug detoxification, and signal transduction. 21
In some pathological states, including sickle cell disease, ischemia reperfusion, and malaria, high levels of free heme are released. The currently published toxicity data for porphyrins largely involves the toxicity associated with excessive amounts of free heme. Toxic effects typically occur through oxidative stress following the generation of reactive oxygen species. Target organs of heme toxicity include the kidney, liver, CNS, and heart. 19
Manganese is a ubiquitous constituent in the environment, occurring in soil, air, water, and food. As a result, all humans are exposed to manganese. In addition, manganese is a normal component of the human body. Typically, food is the most important route of exposure for people with typical daily intakes of 2.5 to 5 mg/day. 22
The available human toxicity data for manganese are limited to inhalation exposure, usually in the industrial setting. Acute inhalation exposure results in manganese pneumonitis. In workers with chronic inhalation exposure, iron deficiency and liver cirrhosis are commonly observed. Chronic inhalation exposure also affects the CNS, resulting in manganism with clinical characteristics including behavioral changes, Parkinsonian-like symptoms, and dystonia with severe gait disturbances. 22,23 No such signs were seen in any test animal. Maximum daily doses of free manganese for any test animal were less than 0.1 µg/kg/day. In addition, the drug is metabolically quite stable resulting in no measurable amount of free manganese being released by metabolic degradation of the test drug.
Here, we describe the nonclinical safety assessment of a manganese porphyrin, MnTE-2-PyP. The battery of tests performed was based on the ICH M3(R2) standards designed to determine the toxicity of drug substances prior to human use. The tests conducted to evaluate the potential toxicity of MnTE-2-PyP looked at a broad range of end points including genotoxicity, tissue tolerance, neurological toxicity, respiratory function, cardiovascular function, and systemic toxicity. No treatment-related signs of toxicity were seen at dose levels less than or equal to the dose proposed for use in human trials, and no specific target organ toxicity was identified. Clinical signs and mortality seen with iv injection were not seen at similar dose levels for MnTE-2-PyP or a similar manganese porphyrin (MnTnBuOE-2-PyP) administered subcutaneously in mice, although higher plasma levels (expressed as AUCs) were achieved. It is hypothesized that these were direct pharmacologic effects evoked in the vasculature in response to achieving a sufficient triggering local concentration. The MnTnBuOE-2-Pyp is currently being assessed as a drug substance, and data will be provided at a later date.
Conclusion
The MnTE-2-PyP has been evaluated for safety in a full set of GLP compliant nonclinical studies sufficiently appropriate to support the filing of an investigational new drug or clinical trial application. No specific target organ was identified, and observed clinical signs were transitory and mortalities occurred soon after dosing or not at all, although pharmacokinetics showed that the drug stayed present in the body several hours following iv dosing. Based on the results of these studies, if a clinical study was to be initiated a safe starting dose would be based on the mouse NOAEL of 10 mg/kg. When compared with monkey, mouse is a more sensitive species as indicated by toxicity data coupled with the pharmacokinetic data. Allowing for species differences, a scaling factor of 12.1 (for mice) would be used. When an uncertainty factor (UF) of 10 is also applied, the starting dose would be 5 mg in a 60-kg human, as shown below.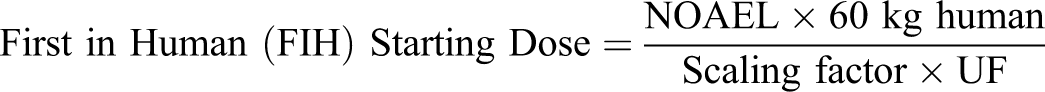
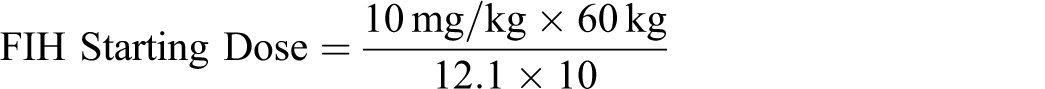

Toxicity and mortality are rapidly evoked at higher iv dose levels, but not at similar (or higher) dose levels administered subcutaneously. The subcutaneous data for this drug, as well as the second-generation manganese porphyrin drug, MnTnBuOE-2-PyP, will be the subject of a subsequent manuscript.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Jewish Health.
