Abstract
Purpose: Doxorubicin (DOX) is a widely used chemotherapeutic agent complicated with cardiotoxic adverse effects. Up till now, there are no researches discussing the role of vinpocetine (VIN) in DOX cardiotoxicity. Thus, the aim of our work was to study this effect and explore the different involved mechanisms.
Methods:
50 male Wistar albino rats were subjected to DOX toxicity via administration of single i.p. Dose (15 mg/kg) on the 4th day with or without co-administration of VIN (10, 20, 30 mg/kg/day) orally for 5 days.
Results:
Our data revealed that VIN succeeded in protecting the heart against DOX induced damage as manifested by significant decrease of cardiac enzymes, hypoxia inducible factor alpha (HIF-1α), vascular endothelial growth factor-A (VEGF-A), tissue malondialdehyde (MDA), tumor necrosis factor alpha (TNF-α) and caspase3 levels. Furthermore, VIN given group showed marked improvement of the histopathological changes of cardiac injury, total antioxidant capacity (TAC), elevation of reduced glutathione (GSH), cyclic guanosine monophosphate (cGMP), cyclic adenosine monophosphate (cAMP) and sirtuin-1 (SIRT-1).
Conclusion:
We concluded that VIN could ameliorate DOX induced cardiac damage and this effect may be attributed to modulation of HIF/VEGF signaling pathway, up-regulation of cGMP/cAMP/SIRT pathway, inhibition of phosphodiesterase enzyme, besides its anti-apoptotic, anti-inflammatory, and anti-oxidant properties.
Introduction
Doxorubicin (DOX) is an anthracycline quinone chemotherapeutic agent that is commonly used in treating different malignant disorders such as solid tumors, breast and ovarian cancers. However, its clinical use is associated with serious cardiotoxic effects including cardiomyopathy and heart failure that restrict its administration. 1 There is a great interest and several trials were performed in studying DOX cardiotoxicity, but the precise mechanisms have not been entirely elucidated. Different factors play an essential role in mediating DOX cardiac injury such as synthesis of free radicals accompanied by oxidative stress, release of inflammatory mediators including pro-inflammatory cytokines as tumor necrosis factor alpha (TNF-α), lipid peroxidation, DNA damage, calcium overloading, deterioration of mitochondrial function and cardiomyocyte apoptosis2,3
Vinpocetine (VIN) is an alkaloid derivative that could inhibit phosphodiesterase 1 (PDE1), a superfamily of destructive enzymes that catalyze cyclic adenosine monophosphate (cAMP) and/or cyclic guanosine monophosphate (cGMP) degradation. 4 Thus, usage of VIN is accompanied with increasing cAMP level and stimulation of the cGMP/cAMP/SIRT signaling cascade that is effective in modulating inflammation, apoptosis and oxidative stress. Recently, the beneficial cardioprotective role of cAMP and its enhancers in different disorders were detected as decreasing hyperlipidemia and atherosclerosis. This pathway has a great role in controlling DOX cardiotoxicity.5,6 In addition, other mechanisms of VIN include its ability to antagonize vascular damage, thrombosis, atherosclerosis and cardiac remodeling. These novel pharmacological and physiological effects and high safety of VIN may facilitate its repositioning in preventing or treating relevant disorders in humans.7,8
In addition, VIN could improve and maintain well blood flow with anti-inflammatory, anti-oxidant, anti-platelet aggregation effects and it is used in treatment of cerebral arteriosclerosis, seizures, Alzheimer’s disease, dementia, stroke, and chronic cerebral insufficiency. Furthermore, VIN is currently found to be beneficial in management of brain, liver, and retinal ischemic injuries via modulating immune responses and Na+ channels, scavenging hydroxyl radicals and decreasing oxidative stress.9,10
Hypoxic state stimulates hypoxia inducible factor 1α (HIF-1α) that is responsible for regulating the transcription process of other essential molecules; vascular endothelial growth factor (VEGF) then sirtuin-1 (SIRT-1). VEGF is considered the most potent and effective angiogenic factor that has a great role during different physiological and pathological neovascularization processes.10,11 Moreover, VEGF plays a critical role in angiogenesis process, formation of new blood vessels and improves the vasculature during DOX induced toxicity and other hypoxic states. Thus, up-regulation of HIF/VEGF signal transduction pathway is so essential in controlling many vascular disorders and preserves the vasculature during ischemic conditions.12,13 Up till now, there are no studies that have evaluated the role of VIN in DOX induced cardiotoxicity in spite of its usefulness in other cardiac injuries. This directed us to investigate the possible protective properties of VIN in such model with deep studying of the different involved pathways including HIF/VEGF signaling cascade and cGMP/cAMP/SIRT transcription pathway.
Materials and methods
Ethics
We followed all guidelines of experimental animals' care that were approved by the Institutional Ethical Committee, Faculty of Medicine, Minia University, Egypt in accordance with EU Directive 2010/63/EU for animal experiments under approval No. 6582021
Kits and Chemicals
VIN was obtained from El-Amyria Co., Cairo, Egypt. DOX was purchased from Khandelwal laboratories Co. India. Total antioxidant capacity (TAC) colorimetric kit was from Bio-diagnostic, Egypt. Cleaved caspase3 and SIRT-1 antibodies were obtained from Abcam, Cambridge, UK. Lactate dehydrogenase (LDH), troponin I, creatine kinase-MB (CK-MB), HIF-1α, cAMP and cGMP Elisa kits were purchased from MyBiosource (California, USA). The polyclonal rabbit/anti-rat TNF-α and VEGF-A antibodies and immunostaining detection kits were from Thermo Fisher Scientific (Fermont, CA, USA).
Animals
Fifty 8 weeks male Wistar albino rats of (200–220 g) body weight were obtained from the National Center of Research, Giza, Egypt. The duration of acclimatization was 1 week (12 h lighting cycle, 45 ± 5% humidity, 25 ± 2°C temperature, and free access to water with the available standard chow diet (El-Nile Company, Egypt). Rats were kept in suitable stainless steel cages for each labeled rat.
Experimental design
The rodents were assorted into 5 groups (10 rats/group). VIN was prepared and dissolved in saline immediately before injection
Collecting the samples
At the end of our experiment, blood was obtained from the left ventricles, allowed to coagulate at room temperature and centrifuged for 15 min at 5000 r/min (JanetzkiT30 centrifuge, Germany). We separated the sera and kept them at −80°C for biochemical analysis. The heart tissues were weighed, excised, washed and divided. For histopathological and immunohistochemical evaluation, parts of the left ventricles were kept in 10% formalin. Other parts were homogenized in ice-cold phosphate buffer (pH 7.4; 20% w/v) then allowed to centrifuge for 15 min at 5000 r/min. The supernatant was separated and stored at −80°C for biochemical measurements which were started immediately following obtaining the samples.
Measurements of cardiac enzymes and TAC
The serum levels of TAC and cardiac enzymes (CK-MB, Tropinin I, LDH) were detected according to the manufacturer’s instructions.
Evaluation of MDA, GSH, HIF-1α, cAMP, cGMP
Malondialdehyde (MDA) level is considered as the most reliable index of lipid peroxidation according to the described method of Buege. 16 HIF-1α, cGMP and cAMP were evaluated according to manufacturer’s instructions of the available Elisa kits. Reduced glutathione (GSH) levels were determined calorimetrically based on that Ellman’s reagent that could reduce the thiol group of GSH with production of a yellow color detected spectrophotometrically at 412 nm. 17
Histopathological procedures
Cardiac tissue was fixed in 10% formalin solution then processed for preparation of paraffin blocks. Five μm sections were deparaffinized, rehydrated in graded alcohol for hematoxylin and eosin (H&E) staining. The pathologist conducted the evaluation in a blind fashion using light microscopy (Olympus microscope, Japan).
Histopathological changes as vascular congestion, hemorrhage, hydropic change, loss of muscle striations, and necrosis were graded semiquantitatively as follows: 0 = no change, 1 = mild change, 2=moderate change and 3 = severe change. 18
Immunohistochemistry
Briefly, five μm sections were de-paraffinized with xylene, rehydrated through ethanol and treated with 3% hydrogen peroxide for 30 min. Slides were washed in phosphate-buffered saline (PBS) solution, then boiled in 0.01 M citrate buffer (pH 6.0) by microwave for antigen retrieval for 15 min (three 5-min cycles). Afterwards, rinse in PBS, primary antibodies were incubated overnight in a humidity chamber using TNF-α (1:100), and VEGF-A antibody (Ready to use) overnight. Slides were washed with PBS before treatment with biotinylated secondary antibody for 30 min 3, 3-diaminobenzidinetetra hydrochloride (DAB) was applied for 5 min, washed in distilled water, counterstained with hematoxylin.
TNF-α cytoplasmic immunoreactivity was scored depending on the percentage of positive cells as follow: 0, negative staining; 1, ≤25% positive cells; 2, >25% and ≤50% positive cells; 3, >50% and ≤75% positive cells; and 4, >75% positive cells. 19 Concerning VEGF-A immunoexpression was considered positive when ≥10% of the cells showed cytoplasmic expression . 20
Western blotting technique for measurement of caspase3 and SIRT-1 levels
Cleaved caspase3 and SIRT-1 protein expressions were measured in cardiac specimens using western blotting technique. 21 Briefly, the following steps were performed; tissue was homogenized then applied to sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS–PAGE). The produced protein bands were transferred to a nitrocellulose membrane with a semi-dry blotter (Bio-Rad). These blots were blocked using TBS-T buffer containing 5% skim milk powder at room temperature for 1 h. Anti-cleaved caspase3, SIRT-1 or mouse monoclonal anti-β-actin antibodies were applied overnight at 4°C then secondary antibody for 1 h at room temperature. Analysis of blots were done using BCIP/NBT (5-bromo-4-chloro-3’-indolyphosphate and nitro-blue tetrazolium) colorimetric method. Protein bands were analyzed using Image-J and GraphPad Prism-5 software.
Statistics
Mean ± standard error of the mean (SEM) was used to express the revealed data. Tukey–Kramer post-analysis test of one-way analysis of variance (ANOVA) was used to analyze the results. p-value ≤ .05 was considered significance. GraphPad Prism version 5.01 for Windows, Graph pad Software, San Diego California USA was used.
Results
Effect of VIN on cardiac enzymes (troponin I, CK-MB, LDH)
Effect of VIN in different doses on cardiac biomarkers.

Results represent the mean ± SEM (10 rats/group).
aSignificant (p ≤ .05) difference from control group.
bSignificant (p ≤ .05) difference from DOX group.
cSignificant (p ≤ .05) difference from high dose vinpocetine (30 mg/kg). [VIN = vinpocetine; DOX = doxorubicin; CK-MB: creatine kinase-MB; LDH: lactate dehydrogenase].
Effect of VIN on oxidative stress parameters (MDA, GSH, TAC)
Effect of VIN on oxidative stress parameters (MDA, GSH, TAC).

Results represent the mean ± SEM (10 rats/group).
aSignificant (p ≤ .05) difference from control group.
bSignificant (p ≤ .05) difference from DOX group.
cSignificant (p ≤ .05) difference from high dose vinpocetine (30 mg/kg). [VIN = vinpocetine; DOX = doxorubicin; MDA = malondialdehyde; GSH = reduced glutathione; TAC = total antioxidant capacity].
Effect of VIN on heart weights, HIF-1α, cAMP and cGMP
Effect of VIN on heart weight changes, HIF-1α, cGMP, cAMP.

Results represent the mean ± SEM (10 rats/group).
aSignificant (p ≤ .05) difference from control group.
bSignificant (p ≤ .05) difference from DOX group.
cSignificant (p ≤ .05) difference from high dose vinpocetine (30 mg/kg). [VIN = vinpocetine; DOX = doxorubicin; cGMP = cyclic guanosine monophosphate; cAMP = cyclic adenosine monophosphate; HIF = hypoxia inducible factor].
Histopathological evaluation results
Examination of control group slides revealed preserved cardiac muscles architecture with muscle striations and vesicular central nuclei (Figure 1(a)). DOX group and VIN-low dose groups showed extensive necrosis and loss of muscular striations with vascular congestion Figure 1(b), (c) respectively. VIN-median dose given group exhibited mild necrosis and vascular congestion (Figure 1(d)).VIN- high dose group showed marked improvement of the histopathological abnormalities and unremarkable necrosis (Figure 1(e)). Histopathological evaluation of cardiac tissue Examination of control group revealed preserved cardiac muscles integrity with muscle striations and vesicular central nuclei (green arrow) Figure. 1(a). Figures 1(b) and (c) DOX and VIN-low dose groups showed hydropic change (blue arrow) extensive necrosis (black arrow) and loss of muscular striations with vascular congestion (red arrow). VIN-median dose group exhibited mild necrosis and vascular congestion Figure. 1(d). VIN-high dose group showed marked improvement in histopathological features with unremarkable necrosis Figure.1(e). (X200). (Scale bar = 100 µm).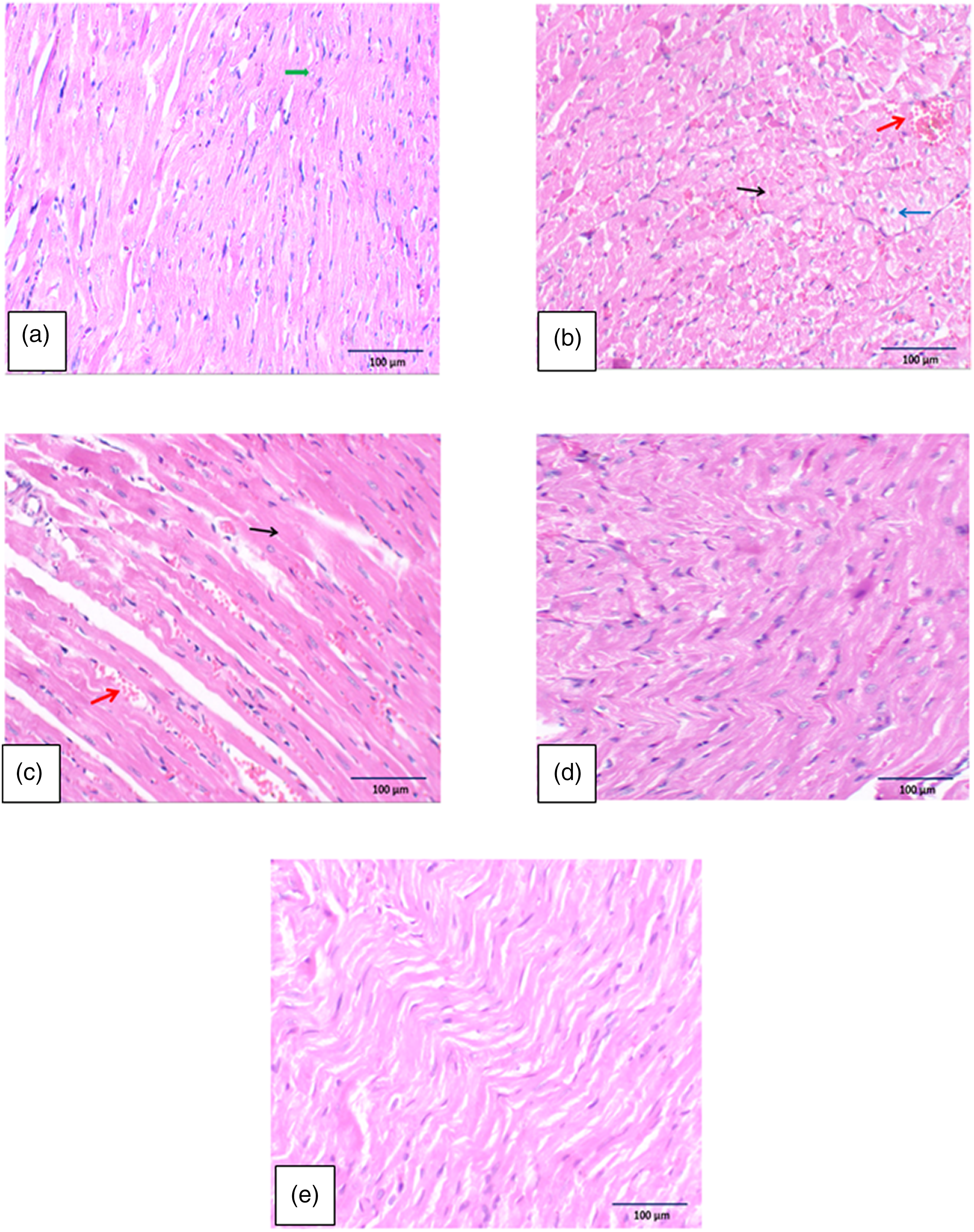
Grading of the histopathological findings
Control group was graded and scored as 0, DOX and DOX+VIN-low dose given group were given grade 3. DOX+VIN-median dose given group was graded as grade 2. DOX+VIN- high dose given group was scored and given grade 1.
Evaluation of TNF-α immunoexpression
There was grade 1immunoexpression in VIN-high dose treated group (Figure 2E ). Meanwhile, DOX group revealed grade 4 immunoexpression and VIN-low dose treated group was given grade 4 (Figures 2(b) and (c)). Grade 2 positivity was observed in VIN- median dose given group (Figure 2(d)). Immunoexpression of TNF-α in cardiac tissue. There was grade 1immunoexpression in control and VIN-high dose groups (Figure 2 A, E). Meanwhile, DOX group revealed grade 4 immunoexpression (Figure 2(b)). VIN-low dose group was given grade 4 (Figure 2(c)). Grade 2 positivity was observed in VIN-median dose group (Figure 2(d)) (X200). (Scale bar = 100 µm).
Semiquantitative analysis of cardiac sections showed that TNF-α expression was significantly higher in DOX group compared to the control group and VIN-high dose treated group. However, co-administration of VIN could markedly decrease TNF-α immunoexpression compared to DOX group (Figure 2(f)).
Evaluation of VEGF-A immunoexpression
Negative expression was observed in control and VIN-high dose given groups (Figures 3(a) and (e)). On the other hand, DOX and DOX+VIN-low dose groups showed positive VEGF-A immunoexpression (Figures 3(b) and (c)). Immunoexpression of VEGF-A in cardiac tissue. Negative expression was observed in control and VIN-high dose groups (Figures 3(a) and (e)). On the other hand, the DOX and VIN-low dose groups showed positive VEGF-A immunoexpression (Figures 3(b) and (c)). Weak positivity was observed in VIN-median dose group (Figure 3(d)) (X200). (Scale bar = 100 µm). Results represent the mean ± SEM (10 rats/group). a Significant (p ≤ .05) difference from control group. b Significant (p ≤ .05) difference from DOX group. c Significant (p ≤ .05) difference from high dose vinpocetine (30 mg/kg). (Figures 2 and 3(f)).
Semiquantitative analysis showed that VEGF-A expression was significantly higher in DOX group compared to control group and VIN-high dose given group. On the other side, VIN treated groups showed significant decrease of VEGF-A immunoexpression in comparison to DOX group (Figure 3(f)).
Changes of cleaved caspase3 and SIRT-1 levels using western blotting
DOX group had a significant elevation of cleaved caspase3 level but a significant decrease of SIRT-1 level compared to control group. However; VIN co-administration plus DOX showed a significant decrease of cleaved caspase 3 level but a significant increase of SIRT-1 level in comparison to DOX untreated group Detection of cleaved caspase3 and SIRT-1 using western blotting. Data revealed significant elevation of cleaved caspase3, but decrease of SIRT-1 level in DOX given group compared to control group. However, there is a significant decrease of cleaved caspase3 but increase of SIRT-1 level in vinpocetine treated groups compared to DOX untreated group.
Discussion
Cardiotoxicity is a very common complication during clinical administration of DOX that restricts its use. Therefore, searching for more effective drugs to reduce DOX induced myocardial damage provides more beneficial outcomes for patients receiving it. 22 Our model aimed to explore the ability of the phosphodiesterase inhibitor; VIN to reduce DOX induced cardiac injuries and study the different pathophysiological pathways mediating this effect. Results revealed that DOX group had a significant increase in serum cardiac enzymes, MDA, TNF-α, caspase3 level with histopathological features of cardiac injuries and up-regulation of HIF/VEGF pathway, but down-regulation of cGMP/cAMP/SIRT signaling cascade. Moreover, there is marked decrease in GSH, TAC and heart weights. Interestingly, co-administration of VIN showed significant cardioprotective effects mainly via down-regulation of HIF/VEGF pathway, ROS scavenging, elevation of cAMP and SIRT-1 levels, and amelioration of inflammation, oxidative stress and apoptosis.
Different factors were found to be responsible for DOX injury such as formation of harmful ROS with further releasing of different inflammatory mediators as TNF-α and interleukins that is responsible for stimulation of other inflammatory/apoptotic pathways involving caspase family mediated apoptosis. 23 Caspase3 is one of this family which was activated by previous upstream of caspase-8 and caspase-9. Unfortunately, cardiomyocyte is a non-renewable tissue and cell apoptosis will terminate with DNA damage and cell death.24, 25 This was evaluated in our study that detected significant increase of the active caspase3 and TNF-α levels in DOX given group but VIN treated groups showed significant decrease of the measured inflammatory and apoptotic agents that could be supported by other studies.8,23,26
Despite extensive research on the mechanisms of DOX cardiotoxicity, the pathogenesis is still unclearly understood. The major causes of cell damage include release of ROS followed by lipid peroxidation, DNA damage, protein denaturation and mitochondria damage. 27 Current model detected marked enhancement of oxidative stress and reduction of anti-oxidants in form of increasing MDA, the most reliable index for evaluating membrane lipid peroxidation via oxidative injury but decrease of GSH, and TAC. Moreover, there is marked elevation of the serum Troponin I, CK-MB and LDH in DOX given group due to the released free radicals accompanied with membrane damage and leakage of cardiac enzymes to the serum. This is in accordance with different previous studies.25,28
Based on the critical role of cAMP/cGMP/SIRT pathway, PDE inhibition is mandatory to keep the myocardium, protect blood vessels and preserve the tissue. It plays an essential role in regulating apoptosis, resisting oxidative stress, alleviating inflammation and protecting the mitochondrial function.29–31 In the heart, sirtuins family protein (SIRT) 1, 2, 3, and 6 activate the autophagic process; protect the heart from atherosclerosis, diabetic cardiomyopathy, ischemia-reperfusion injury, myocardial hypertrophy, and cardiac hypertrophy. Unfortunately, DOX administration down regulates SIRT-1 expression, enhances ROS production leading to excess cellular damage, mitochondrial dysfunction, oxidative stress, apoptosis and cell death. 22
We observed in current study downregulation of such cascade in DOX induced damage. However, co-administration of VIN showed marked increase in cAMP due to its inhibitory effect on phosphodiestrase enzyme with up-regulation of cGMP/cAMP/SIRT signaling pathway reflecting a logical explanation of the associated cardiovascular preserving effect of VIN. Previous studies demonstrated the anti-apoptotic effect of such pathway and its ability to regulate gene transcription, cytokine signaling and cellular proliferation. Modulation of these pathways and suppression of apoptosis are essential to guard against DOX induced damage of the myocardium.29–32 Moreover, DOX plus VIN groups could reduce an important indicator of oxidative stress (MDA), increase the antioxidants (GSH) with elevation of TAC and suppress apoptosis (active caspase 3 apoptotic marker). In addition, VIN showed a significant decrease of troponin I, CK-MB and LDH indicating the preservation of cell membrane against harmfully released ROS, cell injury and lipid peroxidation. These findings are in line with other different previous models.8,9,33–36
Recently, VIN cardioprotective effect was detected in a rat model of induced MI using isoproterenol. VIN pretreatment could ameliorate these changes via elevating cAMP level and suppression of apoptosis, 35 oxidative stress and inflammation. In addition, Wu et al. 37 showed that VIN preserved the heart against angiotensin II induced cardiac remodeling. Furthermore, VIN suppressed fibronectin release, TGFβ-induced fibroblast activation and type I collagen.
One of the earliest adaptive cardiac tissue responses during DOX is releasing of HIF-1α 38 that could stimulate gene transcription of another protective agent; VEGF. The latter has an important role in preserving the vasculature and protecting the tissue against the associated ischemic damage.39,40 Furthermore, the stimulation of HIF-1α induces reprogramming of the metabolic processes to protect the cardiac cell against oxygen deprivation. Up-regulation of HIF-1α/VEGF is able to transform the oxidative phosphorylation processes into glycolysis which could suppress the formation of ROS.39,41–43
In our study, the expression of VEGF as a downstream of HIF-1α was evaluated. We found significant increase of HIF/VEGF in DOX administered group to guard against the harmful effects of DOX. However, co-administration of VIN in different doses could decrease HIF/VEGF level explaining the associated preservation of the myocardium against DOX induced biochemical and pathological changes and this role of VIN was supported with others.15,44–47 In our experiment, we found that VIN is highly safe with variable pharmacological and physiological properties and it may be the future treatment of various cardiac disorders.
Conclusion
Vinpocetine in different doses markedly ameliorated doxorubicin induced cardiotoxicity most probably via phosphodiesterase enzyme inhibition, increasing cAMP, anti-oxidant, anti-inflammatory and anti-apoptotic properties with regulation of HIF/VEGF and cGMP/cAMP/SIRT pathways. Further researches are in critical need to evaluate its role in patients receiving DOX.
Footnotes
Acknowledgements
We’d like to express our appreciation and great thanks to El-Amyria Co., Cairo, Egypt for generous support of our research and Dr. Mohamed El.Sayed, Faculty of medicine El-Fayoum University.
Authors’ contribution
Authors declare that all data were generated in-house and no paper mill was used. Marwa M.M. Refaie detected the research point, shared in the experimental part and writing the manuscript. Sayed Shehata, Elshymaa A. Abdel-Hakeem and Eman Shaaban shared in the experimental part, writing the manuscript and sending it for publication. Maram El-Hussieny performed and wrote the histopathology and immunohistochemistry parts. Michael Atef Fawzy performed and wrote the part of western blot analysis. All authors revised the final version of the manuscript and agreed for publishing it.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Animal handling, medications, and animal sacrifice were carried out according to the guidelines for the care of experimental animals and approved by the Institutional Ethical Committee, Faculty of Medicine, Minia University, Egypt according to the NIH Guide for taking care and use of laboratory animals. Approval No. 6582021.
