Abstract
The therapeutic efficacy of cisplatin (CIS) is limited owing to its hepatotoxic side effects. The current study aimed to investigate the protective impact of ferulic acid (FA) and low-doses of γ-irradiation (LDR) against CIS-prompted hepatotoxicity in rats. Adult male Swiss albino rats were divided into eight groups: untreated group; FA, LDR, and CIS treated groups; and combinations of one or more of the above treatments. Post-treatment analyses included measuring redox markers like SOD and CAT activity, NO free radical content, and lipid peroxidation in liver tissue. Serum aminotransferase activities were also determined. Additionally, gene transcript levels of liver NF-ҡB-P65, caspase-1, COX-2, and IL-1β were quantified. Moreover, immunohistochemistry for caspase-3 and histopathological examinations were estimated in liver tissue. Our findings revealed increased levels of oxidative stress along with a significant reduction in anti-oxidative responses and a significant increase in serum aminotransferase activities in the CIS-intoxicated group. A similar increase was also observed in COX-2 and IL-1β transcript levels and caspase-3 enzyme activity, besides a decrease in transcript levels of NF-ҡB-p65 and caspase-1, indicating an overall inflammatory trend and an increase in the apoptotic shift. The co-administration of FA and/or treatment with LDR has ameliorated the hepatotoxic effect induced by CIS. The histopathological investigation of liver tissues confirmed this ameliorating action of these adjuvant therapies against CIS toxicity. In conclusion, it is plausible to suggest that the hepatoprotective effects of co-administration of FA and/or LDR against CIS-induced hepatotoxicity are attributed to the possession of anti-oxidative, anti-inflammatory, and anti-apoptotic capabilities.
Introduction
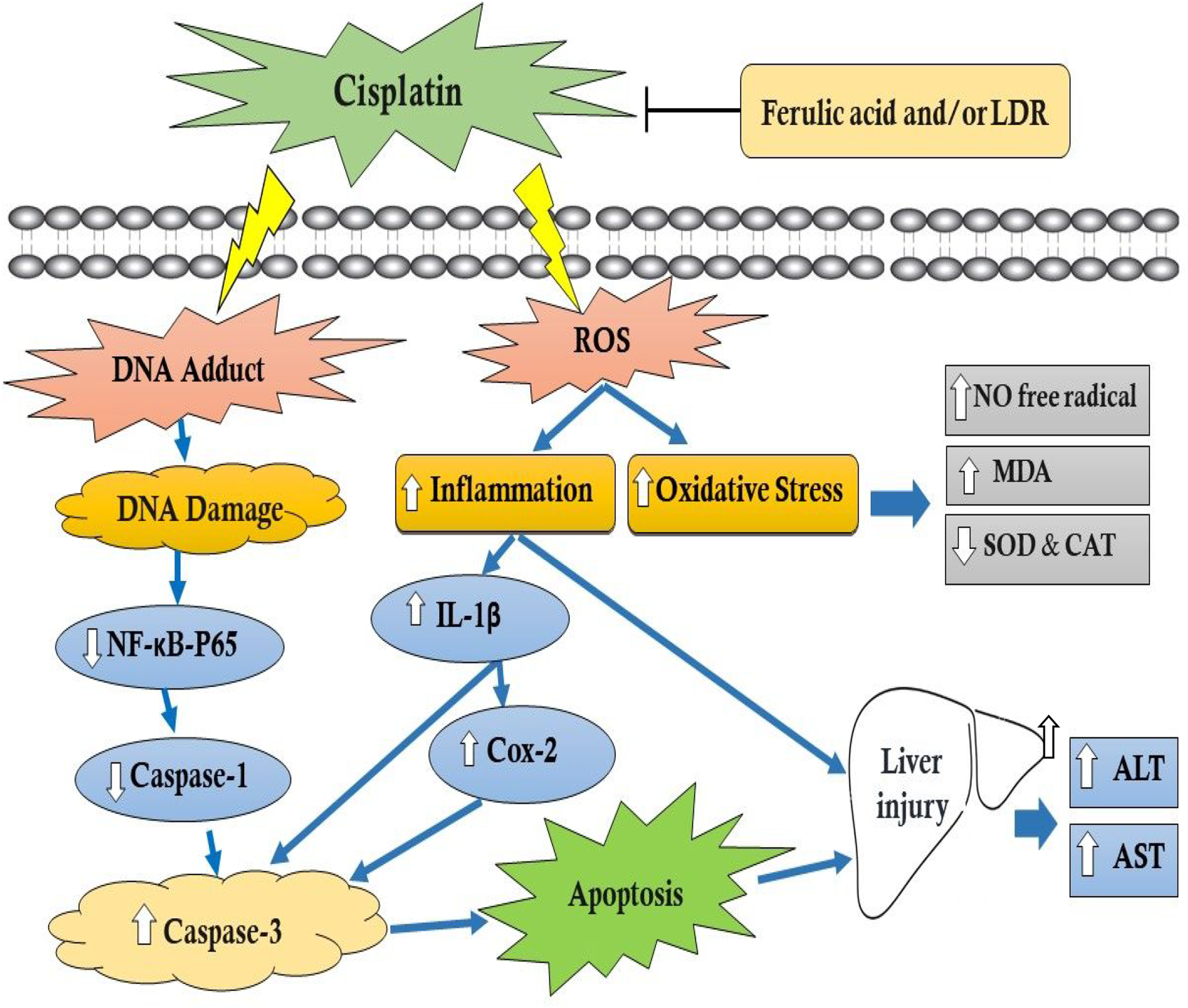
Hepatotoxicity is one of the most serious side effects of cancer treatment, especially when platinum-based medicines like cisplatin are used. 1 Cisplatin (Pt (NH3)2Cl2, CIS) has been widely used due to its potent penetration and antineoplastic spectrum to treat a variety of tumors, including testicular, ovarian, bladder, and lung. Despite its wide therapeutic uses, its use is restricted due to severe ototoxicity, nephrotoxicity, and hepatotoxicity. 2 The primary target of CIS is the genomic DNA, where CIS becomes activated and induces direct breaks in DNA strands, resulting in the production of reactive oxygen species (ROS) and cell damage. 2 Moreover, long-term accumulation of high doses of CIS has been shown to cause hepatotoxicity by causing oxidative stress, inflammation, and hepatocellular apoptosis. 3 The combination approach has been confirmed as an excellent and effective strategy against carcinogenesis in the recent past. 4 It basically includes the combined use of two or more molecules (drugs/phytochemicals/chemo-preventive agents) to enhance the efficacy of the treatment. Therefore, in the present study, we study the hepatoprotective agents or adjuvant therapies that could reduce the adverse effects of CIS. Of these approaches, combining conventional chemotherapeutics, especially CIS, with natural compounds provides synergistic antitumor efficacy and reduces its cytotoxic effects.
Among these natural products, ferulic acid, also known as 4-hydroxy-3-methoxycinnamic acid (C10H10O4, FA), is a phenolic chemical found in cereals, fruits, and vegetables. FA is a natural antioxidant with anti-oxidative, anti-inflammatory, anti-apoptotic, hepatoprotective, anti-carcinogenic, and radioprotective characteristics that have been demonstrated to have therapeutic effects in several disease types.5–9 It possesses unique pharmacokinetic features, such as the ability to stay in the bloodstream for several hours and the ability to be absorbed in the intact form in mouse models. 5
Various studies adopted different strategies to manage liver injury induced by CIS toxicity. One of these strategies is irradiation with low doses of ionizing radiation (LDR) (less than 1 Gy), which has been shown to elicit a hormetic effect by increasing cellular metabolic functions such as protein synthesis and DNA repair. 10 Furthermore, it was noticed that exposure to LDR in different disease models exerts an anti-oxidative, anti-apoptotic, anti-inflammatory, hepatoprotective, and anti-carcinogenic effects.11,12 Thus, LDR has the potential to improve the efficacy of cancer therapy while also lowering the toxicity of anti-cancer drugs. 13 The combination approach is now an established strategy against cancer. Therefore, the current investigation examined potential hepatoprotective mechanisms behind the synergistic effects of ferulic acid and/or low dose γ-irradiation against cisplatin-induced liver damage in rats.
Materials and methods
Materials
Chemicals and drug
Cisplatin (50 mg/50 mL) vial was purchased from Mylan Company, France. Ferulic acid was obtained from Santa Cruz Biotechnology, Inc., Heidelberg, Germany as a powder that reconstituted into a crystal-like suspension in distilled water. RNA extraction kit was purchased from QIAGEN (QIAGEN, USA), SensiFAST™ SYBR® No-ROX One-Step Kit was obtained from (Bioline Inc., USA). Serum aminotransferase and redox markers kits were purchased from Bio-Diagnostics (Egypt). The other chemicals and reagents used in this study are the products of Sigma-Aldrich Chemical Co USA.
Animals
The animal breeding unit of the National Center for Radiation Research and Technology (NCRRT) in Cairo, Egypt, provided male albino rats weighing 120–150 g. Throughout the experiment, the animals were housed in standard humidity and temperature (22–24°ϲ) conditions and fed standard pellets of concentrated food containing all the necessary nutritive elements (23% protein, 4.68% lipids, and 2.6% fibers), with free access to water. This study was conducted in accordance with the recommendations in the Guide for the Use and Care of Laboratory Animals of the National Institute of Health (NIH no. 85:23, revised 1996) and in compliance with the regulations of Ethical Committee (REC) of NCRRT, Atomic Energy Authority, Cairo, Egypt (Approval Number: 9 A/21).
Irradiation process
Rat whole-body gamma irradiation was performed at the NCRRT, Atomic Energy Authority (Cairo, Egypt), using the Cesium-137 biological irradiator source (gamma-cell-40), Atomic Energy of Canada Limited (Chalk River, ON, Canada) with a dose rate of 0.46 Gy/min during the time of exposure. Rats were exposed to two doses of 0.25 Gy with a total dose of 0.5 Gy. 14
Experimental design
The animals were randomly divided into eight groups of eight rats each, which were left untreated (Normal Control, NC), and treated with FA at the dose of 40 mg/kg b. w.t., 15 with minor modification for 19 successive days using the gastric tube, exposed to two low doses of γ-irradiation (0.25 Gy/each) 14 on day 14 and day 18 from the zero time of the experiment, and a combination of both treatments in the absence (Group 1–4, respectively), or in the presence of CIS at a dose of 2 mg/kg b. w.t., intraperitoneally, for 5 successive days, starting from day 15 to day 19 from the zero time of the experiment) 16 (Group 5–8, respectively).
On the 20th day from the zero time of the experiment and after rats fasted overnight, they were anesthetized and sacrificed. Blood samples were taken from all animals via heart puncture. Sera were separated, aliquoted, and used for the determination of the activity of liver enzymes. Liver tissues were excised and were used in histopathological and immunohistochemistry examinations; electron spin resonance (ESR) measurements of nitric oxide (NO) free radicals; gene expression analysis by RT-qPCR; and the estimation of antioxidant parameters; SOD, CAT, and MDA.
Methods
Determination of nitric oxide free radical
Liver tissues (1 gm each) were rapidly taken from rats, gently lyophilized, and vacuum-dried to dryness. All the samples were dissolved in a small amount of toluene and placed in a circular ESR cell. Before ESR spectroscopy, the cells were closed with a rubber septum and fully deoxygenated by nitrogen bubbling. A Bruker EPR ER-200D spectrometer was used to record ESR spectra at room temperature, and a Bruker ER-140 (ASPECT 2000) data system was used for spectrum accumulation. The power of the microwave was 2 mW, the modulation amplitude was 1 G, and the receiver gain was 1 E4. With a field-sweeping rate of 100 G/42 S, the response time was constant at 10 ms. The powder sample inside the quartz tube had a height of roughly 10 mm. A computer simulation software was used to carry out the ESR spectrum analysis. 17
Biochemical investigation
Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities in the serum were detected 18 and liver tissue lipid peroxidation level (MDA), superoxide dismutase (SOD), and catalase (CAT) were measured as oxidative stress markers.19–21 All the colorimetric kits were supplied by Biodiagnostic, Cairo, Egypt.
Histopathological examinations
After sacrifice, specimens were autopsied from the liver tissues of distinct groups and preserved in neutral buffered formalin (10%) for 24 h, washed properly under running water, dehydrated in ascending grades of alcohol, cleaned in xylene, and fixed in paraffin wax. For examination under the light microscope, the embedded specimens were sectioned by microtome (4–5 m thick), deparaffinized, and stained with hematoxylin and eosin (H&E). 22 Another set of sections was used for the immunohistochemical staining of the caspase-3 antibody.
Immunohistochemical assay for active Caspase-3 enzyme
In rehydrated paraffin-embedded sections, endogenous peroxidase activity was stopped by incubating them in 3% hydrogen peroxide (H2O2) in methanol for 15 min at 23°C, then microwaved for 9 min in citrate buffer (PH 6.0). Polyclonal rabbit anti-active caspase-3 (1:100) in phosphate buffer saline (PBS) was applied for 60 min at 23°C before the secondary biotinylated goat anti-rabbit antibody was added. Slides were then incubated and developed with diaminobenzidine (DAB) and counterstained with hematoxylin for examination under a light microscope. 23
Molecular investigation
Sequence of the primers, programs and PCR products of NF-ҡB-p65, IL1β, caspase-1, COX-2 and β-Actin.

Statistical analyses
Mean and standard error values were determined for all the parameters and the results were expressed as Mean ± SE. All data were analyzed with SPSS version 22 employing One-way ANOVA followed by the Tukey post hoc test. Differences between groups was considered significant when p < 0.05.
Results
Hepatic NO free radical content
Figure 1 Illustrates that CIS exhibited a significant elevation in liver tissue NO free radical content with respect to the untreated group. On the other hand, supplementation with FA or exposing the rat’s whole body to LDR together with CIS injection significantly ameliorated liver NO free radical content, and the co-treatment of FA and LDR prior to and during CIS injection resulted in a marked enhancement in the hepatic NO free radical content as compared to the CIS group, which indicated the fact that co-treatment gave the best results. Impact of CIS, FA, LDR and their combination on liver NO free radical concentration.
Biochemical studies
Effect of CIS, FA and/or LDR on the activities of serum ALT, AST and liver antioxidant enzymes as well as liver lipid peroxidation level among the different studied groups.

The values represent mean ± SE from 8 samples in each group. All data represent one out of three independent experiments.
ais significant difference versus untreated control group at p < 0.05.
bis significant difference versus CIS treated group at p < 0.05.
Molecular studies
The data in Figure 2(a) and (b) revealed that, when compared to the control group, exposure of normal rats to LDR or FA treatment produced a marked elevation in the expression level of the studied genes, except for the Caspase-1 gene, which was down-regulated after FA administration. The CIS-treated group exhibited a significant down-regulation (p < 0.05) in the transcription level of NF-ҡB-p65 and Caspase-1 genes, with a significant up-regulation (p < 0.05) in the transcription level of IL-1β and COX-2 genes relative to the control group. Prior to and during CIS injection, rats were given FA and/or fractionated LDR, which resulted in a significant improvement (p < 0.05) in the previously described parameters compared to the CIS group. (a) Effect of CIS, FA and/or LDR on the expression level of NF-ҡB-P65 and Caspase-1 genes in the hepatic tissues of the different studied groups. The values represent mean ± SE from 8 samples in each group. ais significant difference versus untreated control group at p < 0.05. bis significant difference versus CIS treated group at p < 0.05. (b) Effect of CIS, FA and/or LDR on the expression level of IL-1β and COX-2 genes in the hepatic tissues of the different studied groups. The values represent mean ± SE from 8 samples in each group. ais significant difference versus untreated control group at p < 0.05. bis significant difference versus CIS treated group at p < 0.05.
Histopathological findings
Untreated rats' liver tissues show a normal histological structure of the hepatic lobule, which includes a central vein and concentrically distributed hepatocytes. Figure 3(a). Rat groups receiving FA Figure 3(b), exposed to LDR Figure 3(c), and treated with FA+LDR Figure 3(d) showed mild histopathological changes in their hepatic tissues. On the opposite side, liver tissues of the CIS-treated group are characterized by histological alterations consisting of a dilated central vein (CV) which is surrounded by cords of hepatocytes exhibiting severe steatosis and others with pyknotic nuclei with the appearance of inflammation areas, Figure 3(e). Pretreatment of CIS-intoxicated rats with FA and/or LDR ameliorated the hepatic changes and inflammatory process induced by CIS. The combination treatment of (FA+LDR) gave the best protective effect against CIS hepatic toxicity, Figure 3(f-h). Rat liver sections from (a) Normal group showing the normal histological structure of hepatic lobule where normal central vein (black arrow) is surrounded by normal hepatocytes (blue arrow); (b) rats exposed to LDR have a mildly dilated central vein (black arrow) surrounded by hepatocyte cords, some of which are normal (blue arrow) and others have mild steatosis (red arrow); (c) FA treated rats showing a normal sized central vein (black arrow) surrounded by cords of hepatocytes exhibiting mild steatosis (red arrow); (d) FA + LDR treated rats exhibit Kupffer cell activation, with a mildly dilated central vein (black arrow) surrounded by cords of hepatocytes, the majority of which are normal and a few exhibiting mild steatosis (red arrow). (e) CIS-treated rats showing central vein (black arrow) surrounded by cords of hepatocytes exhibiting severe steatosis with others with pyknotic nuclei (red arrow) and areas of inflammation (green arrow); (f) F, CIS+ LDR treated rats have a mildly dilated central vein surrounded by a few inflammatory cells (black arrow) and hepatocyte cords, some of which are normal (blue arrow) and some of which have mild steatosis (red arrow); (g) CIS + FA treated rats showing a mild dilated central vein (black arrow) surrounded by cords of normal sized hepatocytes (blue arrow); (h) CIS + FA + LDR treated rats showing a normal sized central vein (black arrow) surrounded by cords of normal sized hepatocytes (blue arrow). (H&E, scale bar 50μm, X 100).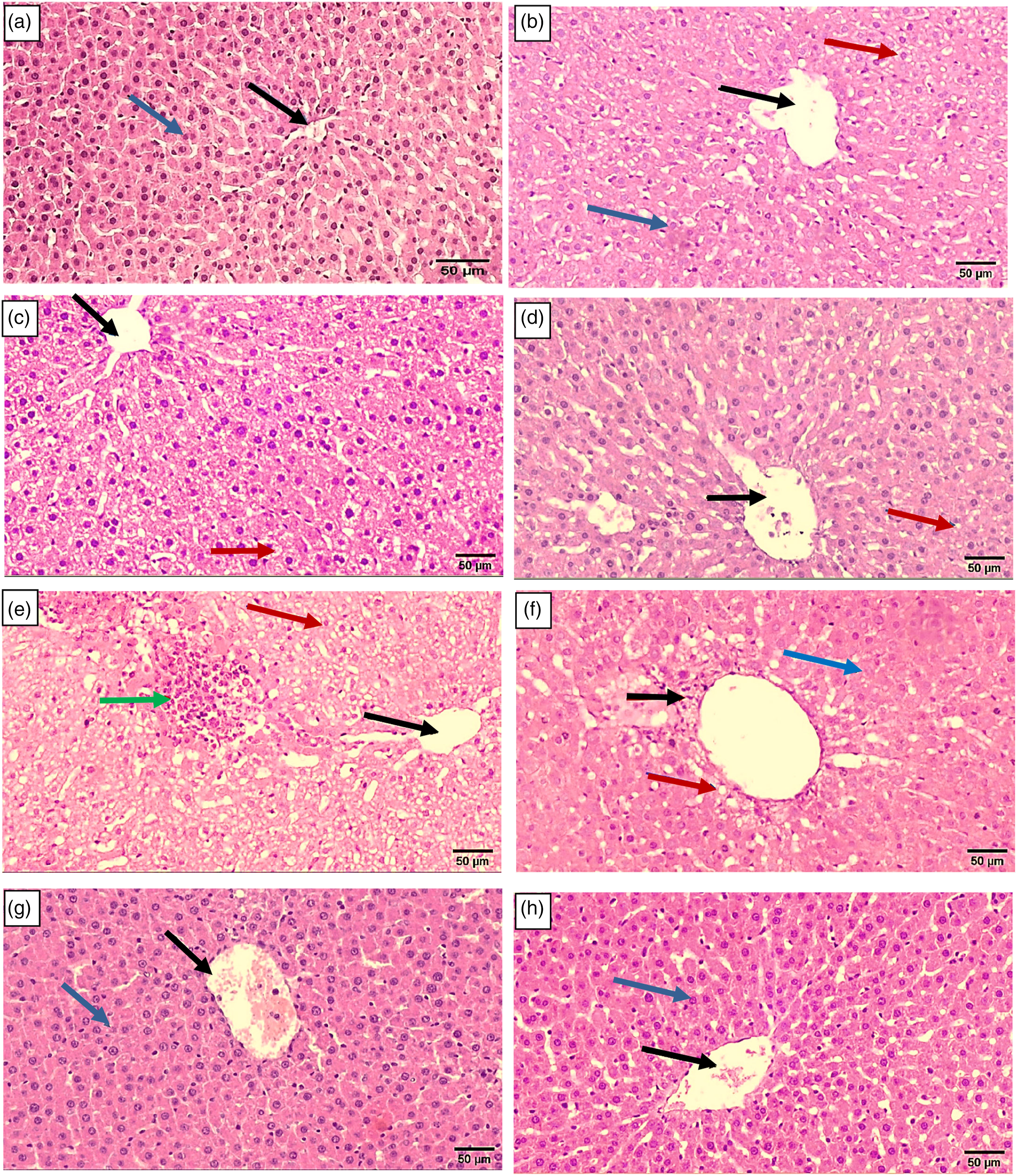
Immunohistochemical expression of caspase-3
Photomicrographs of the untreated group’s liver tissues showed negative expression of caspase-3 (Figure 4 (a)), while animals treated with either FA or LDR showed a few numbers of caspase-3 positive cells in their liver tissues (Figure 4 (b) and (c)), respectively. Furthermore, the FA+LDR treated group showed mild expression of caspase-3 (Figure 4 (d)). On the other hand, the CIS-intoxicated group (CIS group) was characterized by high expression of hepatic caspase-3, the highly brown stain reveals marked apoptosis of the hepatocytes (Figure 4 (e)). However, pretreatment of CIS-treated rats with LDR (CIS+LDR group) or with FA (CIS +FA) relieved the apoptotic effect of CIS on liver cells, which was indicated by the moderate expression of caspase-3 in these two groups (Figure 4 (f) and (g)). Furthermore, the combination treatment of CIS-injected rats with FA and LDR (CIS + FA + LDR group) ameliorated the toxic effect of CIS on the hepatocytes, this was observed by the mild expression of active caspase-3 in the liver tissue of this group (Figure 4 (h)). Immunostaining for cleaved caspase-3 protein in liver sections of rats, (100X). (a) Normal control group showing no cleaved caspase-3 immune-reactive cells. (b) Treated with LDR, showing a very low expression of caspase-3. (c) Treated with FA, showing negative expression of caspase-3. (d) Co-treated with FA+LDR, showing a mild expression of caspase-3. (e) Treated with CIS showing strong positive immune reactive hepatocytes. (f) Co-treated with FA+CIS, showing moderate positive immune expression. (g) Co-treated with LDR+CIS, showing moderate positive immune expression. (h) Co-treated with FA+LDR+CIS, showing a mild positive cleaved caspase-3 reaction. (i) Immunostaining area (%) of cleaved caspase-3 expression. Data shown as mean±SE; error bars show the variations of determinations in terms of standard errors, mean values with unlike superscript letters were significantly different (p ≤ 0.05). Figure 4(i) revealed the evaluation of immunostaining expression of cleaved caspase-3 in rats from different experimental groups.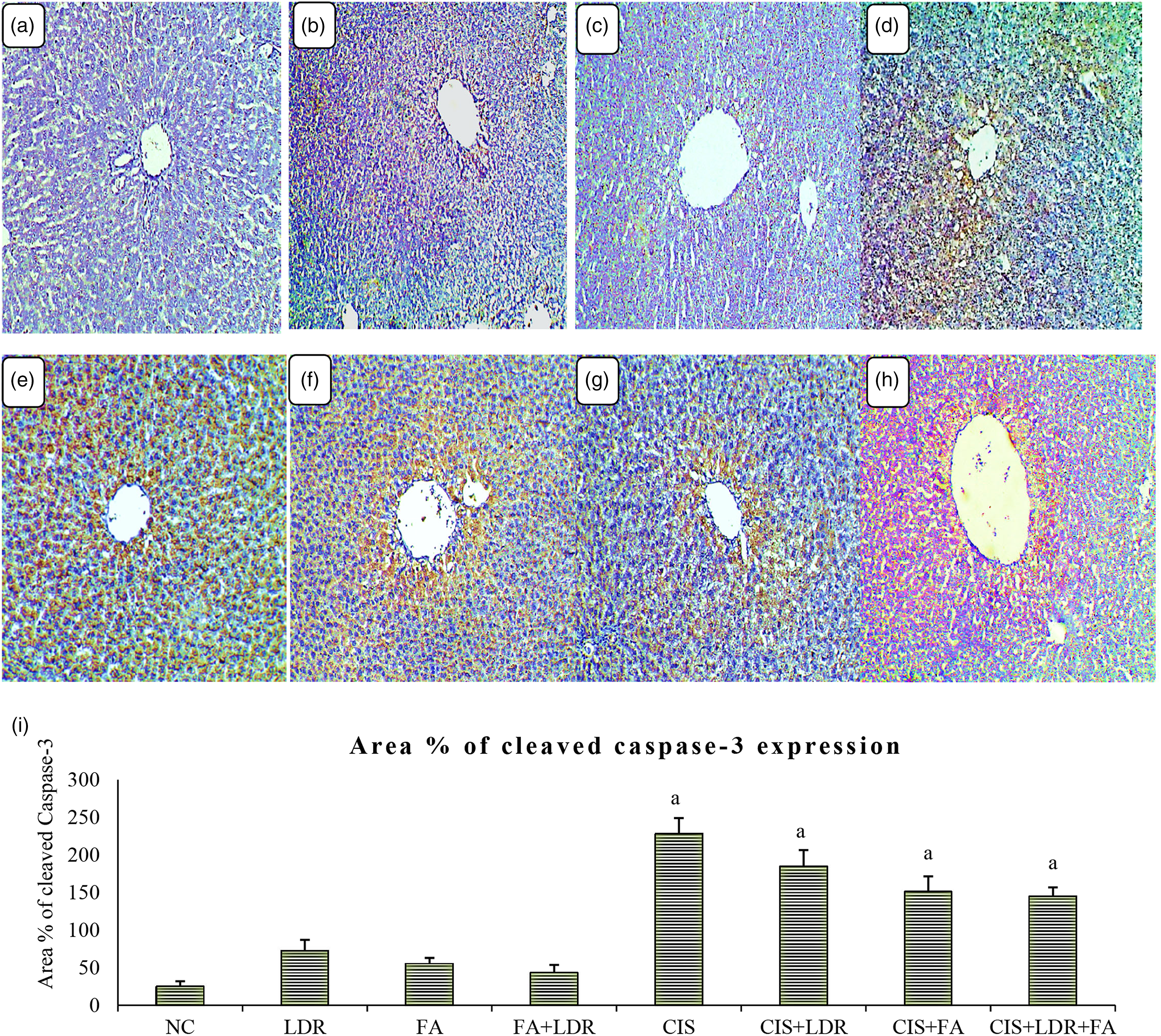
Discussion
Hepatic toxicity is a severe drawback of giving the chemotherapeutic drug CIS to cancer patients. 29 The etiology of hepatotoxicity is still unknown, although it is thought to be caused by a variety of factors; it is a complicated and multifaceted process. Contributory factors have been identified, including oxidative stress, inflammation, DNA adducts, and apoptosis. 30 Therefore, during CIS treatment, it is critical to apply particular hepatoprotective approaches, such as antioxidants, free radical scavengers, and natural herbal extracts. The combination of therapies with different compounds is likely to increase the effectiveness of drug treatment as well as reduce adverse outcomes. Thus, the goal of this investigation was to study the possible mitigating role of a combination of FA and/or LDR against CIS-evoked hepatotoxicity in rats. FA is a well-known hepatoprotective natural phenolic agent that exhibits a strong free radical scavenging activity. It has been described as having antioxidant, anti-inflammatory, and anti-apoptotic properties.5,31 LDR improves the efficacy of cancer therapies and lowers their harmful side effects. 13 Also, the stimulating effects of gamma irradiation were recorded to raise flavonoid, alkaloid, phenolic compound, and antioxidant activity. 32 Moreover, it has an anti-toxic hepatoprotective function through its antioxidant, anti-inflammatory, and anti-apoptotic actions.11,12
Reactive oxygen and nitrogen species (ROS/RNS) such as nitric oxide (NO) and peroxynitrite are thought to be one of the key mediators in the pathophysiology of CIS-induced hepatotoxicity.33,34 In the current study, CIS injection has drastically increased NO radical production in rats' liver tissue compared to control animals, which could be explained by the fact that CIS increases inducible nitric oxide synthase 2 (iNOS2) enzyme activity in the liver, which in turn produces a high level of NO radical that interacts with oxygen radicals to generate peroxynitrite, a powerful oxidative and nitrosative agent, causing damage to liver tissue. 35 On the other hand, the administration of FA and/or exposure to LDR the CIS-deleterious effects noticeably. This could be attributed to their antioxidant capabilities, thus preventing the harmful effect of NO generated during CIS injection on liver cells, which aligns with prior report. 36
In the current investigation, increases in serum ALT and AST levels were observed after CIS injection, indicating liver injury. These findings are consistent with earlier research1,37 indicating liver dysfunction due to CIS-induced oxidative stress, which leads to liver damage and subsequent enzyme leakage from hepatocytes into the systemic circulation. The results obtained showed that changes in serum ALT and AST levels were mitigated by the administration of FA and/or LDR exposure, presumably by maintaining hepatocyte membrane integrity. This is evidence of FA’s hepatoprotective properties. 38 Low-dose radiation (LDR) has been shown to trigger a variety of bio-effects, particularly anti-oxidative ones. 39
In the current study, the CIS-treated group showed a disturbance in the cell redox tone manifested by an increase in MDA as well as a decrease in the antioxidant enzymes CAT and SOD in the liver tissue as compared to their respective controls; these findings are in line with other studies.40,41 and verify that CIS-induced oxidative stress plays a major role in liver disease.2,42 The increase of MDA may be attributed to the excess of ROS’s toxicity that breaks down cell membrane unsaturated fatty acids. 40 Moreover, the persistent and immense production of free radicals like superoxide and hydroxyl free radicals alter the antioxidant system and decrease the activity of the antioxidant enzymes pool. 42 The antioxidant system, including anti-oxidative enzymes such as CAT and SOD is responsible primarily for the defense against the production of ROS in cells and their potential toxicity-related effects. 43 Also, the loss of zinc and copper, both of which are essential for enzyme activity, could explain the decrease in SOD activity after CIS treatment. 36 Interestingly, in the current study, treating CIS-intoxicated animals with FA, LDR and their combination has ameliorated oxidative stress by wiping out the ROS, reducing lipid peroxidation levels, and restoring the activity of the protective antioxidant enzymes, demonstrating a defensive effect against ROS. It is suggested that FA and LDR may have a synergetic protective effect due to the exhibition of potent anti-oxidative efficacy to reduce the hepatic oxidative damage induced by CIS.
In the current approach, a slight elevation in the tissue NO free radical content and MDA levels, and serum ALT and AST with a slight decline in hepatic SOD and CAT activities were detected in FA-treated animals versus the control group, this could be attributed to FA’s low toxicity under normal rat metabolic circumstances. 5 On the opposite side, CIS-intoxicated rats supplemented with FA reduced CIS accumulation in liver tissue and protected against CIS-induced damage via modulating the oxidative insult. Nonetheless, LDR resulted in a dose-dependent improvement in anti-oxidative properties, which are critical in protecting cells from oxidative stress caused by ROS. 44 Conversely, Temme and Bauer. 45 LDR promotes superoxide anion generation by non-irradiated cells via TGF-1-dependent bystander signaling. This explains the elevation in the hepatic NO free radical content, serum ALT and AST, as well as the hepatic level of MDA and the reduction in hepatic CAT and SOD activities after exposure of whole bodies of rats to LDR alone in the present work.
Our histopathological findings demonstrated that CIS administration caused various degenerative alterations in the liver cells of normal rats, which supports the biochemical evidence of oxidative stress. 46 Hepatocytes are known to accumulate substantial amounts of CIS, so the toxicity in hepatocytes is attributed to CIS storage. 47 These changes were reduced when FA, LDR, or both were applied with CIS. The ameliorative effects of FA and LDR on structural changes might be attributed to their roles in the modulation of essential cellular activities, including cell growth and differentiation, as well as their potent antioxidant properties and ability to scavenge free radicals.
Excessive ROS production by CIS damages endogenous macromolecules such as DNA, lipids, and proteins, causing them to be modified and resulting in cellular malfunction and apoptosis. 48 In the same manner, our findings revealed severe disturbance in inflammatory and apoptotic markers in normal rats' liver tissues after CIS administration.
Nuclear factor-kappa B (NF-κB), is a leading aspect of the cell death/survival balance. While NF-ҡB can be either pro-or anti-apoptotic, it serves as a cell survival stimulant in most circumstances, 49 and the decline in its activity results in apoptosis. 50 The most prevalent NF-κB dimer is the heterodimer containing the p50 and p65 subunits. 51 P65 is the specified subunit in our study.
Our findings revealed a pronounced decline in NF-ҡB-P65 expression level after injecting normal rats with CIS compared to the untreated controls, which verifies that CIS plays a crucial role in the induction of apoptosis or necrosis through NF-κB down-regulation.52,53 This decline of NF-ҡB gene expression function is mediated by a decrease in its ability to bind to ҡB site(s) via CIS-DNA adducts, which distort DNA conformation. 54 This in turn enhances the activity of several oxidative stress-sensitive transcription factors, which regulate apoptotic gene expression and lead to programmed cell death. 54 Further, CIS-induced sustained oxidative stress results in inhibiting the expression of the NF-ҡB gene by impeding the degradation of I-κB and reducing NF-ҡB-DNA’s binding activity. 51 Furthermore, divalent metals, such as CIS, with high affinity to sulfhydryl groups have been proven to cause apoptosis in cells by blocking the binding of NF-ҡB to its consensus response elements at the DNA-ҡB site (s) in the cell nucleus. 55 Therefore, the repression of NF-ҡB and its downstream pathway appears to be a common underlying mechanism of apoptosis by CIS. According to the results of this study, treating CIS-injected rats with FA or LDR caused an up-regulation of the NF-ҡB-p65 gene expression versus the CIS-treated group. Also, the combined effect was more pronounced than using either treatment individually. This indicates the synergetic hepatoprotective effect of both FA and LDR because of their antioxidant, anti-inflammatory, and anti-apoptotic capabilities, which protect cells from oxidative stress and apoptosis.
Caspases are enzymes that are cysteine-dependent, mediate apoptosis and inflammation, and are stimulated by oxidative stress. 56 Caspase-3 cleaves a variety of substrates, causing DNA to break down and resulting in apoptotic cell death. 57 The results obtained showed that CIS increased active caspase-3 expression, indicating apoptosis in hepatocytes. These findings are in line with preceding reports 58,59 and substantiate that the down-regulation of NF-ҡB-p65 is associated with apoptosis in the hepatocytes induced by CIS toxicity. Our finding showed that Pre-treatments with FA or/and LDR protect liver cells against CIS-induced apoptosis by inhibiting the cleavage of caspase-3 when compared to CIS-treated rats, implying that FA and LDR may have potential anti-apoptotic hepatoprotective properties against CIS-prompted liver injury.
Caspase-1, also called IL-1 converting enzyme (ICE), is an inflammatory caspase that is essential for pro-IL-1 processing and IL-1 secretion and is activated by auto processing within a macromolecular complex called the inflammasome. 60 According to the results of gene expression obtained from qPCR, caspase-1 gene expression was down-regulated in the animal group treated with CIS. Similar observations were obtained by other investigators.61,62 A possible mechanism explained that NF-ҡB is a prime candidate for caspase-1 activation. 63 In addition, Lee et al. 64 provided evidence that NF-κB-P65 is the direct upstream transcriptional activator for caspase-1 gene expression. NF-κB is a key regulator pathway that regulates the expression of the NLRP3 (nucleotide-binding domain, leucine-rich-containing family, pyrin domain-containing-3) in hepatocytes. 65 Upon stimulation of NF-κB, IκB-α is phosphorylated, which in turn leads to the phosphorylation of the p65 subunit at the Ser536 position. The activated subunit then translocates to the nucleus to up-regulate NLRP3 mRNA expression. 66 NLRP3 binds with the adaptor molecule and apoptosis-associated speck-like protein (ASC), which possesses a caspase recruitment domain that can be used to recruit and activate caspase-1. 67 Kong et al. 65 stated that CIS markedly inhibits IκB-α degradation and p65 nuclear translocation in macrophages, hence suppressing NF-κB activation, which is consistent with our observation. Hence, suppressing the expression of the NF-κB gene by CIS prevents NLRP3 gene expression in liver cells and thus decreases caspase-1 expression. Also, Meng et al. 68 referred that NF-κB is activated by caspase-1 independent of its enzymatic activity. It was observed that only the LDR-treated group revealed a significant up-regulation of the expression level of the caspase-1 gene. Inflammasome activation is sustained in most major immune cell subsets after radiation exposure. The caspase-1 enzyme is activated as a result of the formation of the inflammasome. 69
IL-1β is widely seen as an inflammatory biomarker and is released as a result of inflammasome activation and an increase in ROS. Together, they can inflict pyroptosis and apoptosis in cells. 70 In our study, intraperitoneal injection of normal rats with CIS resulted in a significant elevation of IL-1β gene transcript level compared to untreated rats which are consistent with previous reports.58,71 The free radicals generated as a result of CIS administration stimulate T-helper cells, which are important at the start of an immune response. Stimulated T-helper cells may secrete inflammatory cytokines like IL-1β, TNF-α, IL-2, etc. 70 Furthermore, in neutrophils, IL-1β production is mediated by multiple different serine proteases and is not dependent on caspase-1, including, proteinase 3 (PR3), neutrophil elastase (NE), chymase, cathepsin G (CG), and chymotrypsin, along with certain matrix metalloproteinases, that have been proven to be capable of cleaving pro- IL-1β at crucial regions involved in the bioactive molecule’s production and release. 72 This may explain our observations that the increase in IL-1β mRNA level in the CIS-treated group is mediated by other pathways independent of NF-ҡB or caspase-1 gene expression activation. Although the activation of genes that control inflammation and apoptosis via the NF-κB pathway is implicated in a variety of cellular responses. Surprisingly, rats with down-regulated NF-ҡB gene expression are more susceptible to hepatotoxicity with CIS due to the elevation of plasma and tissue IL-1β levels after processing.
In the current investigation, exposing the whole body of rats to LDR induced an increase in the level of IL-1β gene expression compared to the untreated control group. LDR is believed to induce slight damage at the cellular level by elevating the proliferative reactive response of T cells to mitogenic stimulation, enhancing immune cell function, and altering cytokine release. 73 In addition, it was observed that in the LDR-treated group, in an NF-ҡB-dependent mechanism, monocytes, macrophages, and dendritic cells (DCs) produced the majority of IL-1β. 74 In our results, treatment with FA and/or LDR in CIS-intoxicated rats reduced the expression level of the IL-1β gene relative to CIS-treated animals; this could be related to FA and LDR’s abilities to protect against CIS-evoked liver damage by lowering CIS accumulation in the liver and by controlling oxidative insult and inflammation.
The inducible type of cyclooxygenase, cyclooxygenase-2 (COX-2), is stimulated by growth factors and cytokines and over-expressed during inflammation to promote the transformation of arachidonic acid to prostaglandins. 75 COX-2 activity in cells is linked to the formation of ROS and inflammatory markers, both of which are critical in mediating CIS-induced hepatotoxicity. 30 Our results showed that in animals treated with CIS, over-expression of COX-2 was observed within the hepatocytes. These results are consistent with previous findings75,76 which showed that COX-2 is overexpressed in CIS-injured tissue due to its induction of pro-inflammatory cytokines, for example, TNF-α, IL-1β, and endotoxins, which trigger prostaglandin formation. According to a prior study, IL-1β is the major cytokine implicated in the up-regulation of COX-2 in inflammatory processes. 77 Through interactions with two membrane-bound receptors, IL-1 receptor type I (IL-1RI) and IL-1 receptor accessory protein, mature IL-1β leaves the cell and activates target cells, controlling the production of a wide range of genes involved in the regulation of inflammation, such as COX-2. 78 Otherwise, CIS-intoxicated rats were treated with FA or/and LDR, which revealed down-regulation of COX-2 gene expression levels compared to CIS-treated rats, indicating their pivotal role in relieving inflammation.
Conclusion
Finally, our findings in this investigation demonstrated that the combination treatment with FA and LDR reduced CIS-induced liver injury via attenuating oxidative stress, lowering caspase-3 activity, and inhibiting inflammation routes. This might be an optimistic new approach for cancer patients who are receiving a CIS regimen.
Footnotes
Acknowledgements
The authors are thankful to Dr. Yomna Abdelmonem Zamzam Osman, Assistant Professor of Pathology, Faculty of Medicine, Tanta University, for her assistance in examining and interpreting the pathological study aspects of this work.
Author’s note
This data is taken from PhD Thesis of Marwa A. Esmat.
Author contributions
All authors conceived and designed the experiments. Marwa A. Esmat performed the experiments. Marwa A. Esmat and Rasha E. Hassan analyzed and interpreted the data. Marwa A. Esmat, S.A. Hagag and T.K. El-maghraby, contributed reagents/materials/analysis tools. Marwa A. Esmat and Rasha E. Hassan, wrote the paper. Ahmed Osman, Rasha E. Hassan and T.K. El-maghraby, revised the final manuscript. All authors reviewed the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
