Abstract
Objective
Bone marrow mesenchymal stem cells (BMMSCs) exert protective effects against myocardial infarction (MI). Here, we focused on the function and mechanism of miR-455-3p from BMMSCs-derived exosomes (BMMSCs-Exo) in myocardial infarction.
Materials and methods
BMMSCs were isolated from rat bone marrow, and the exosomes from the culture medium of BMMSCs were separated, and administered to H9C2 cells under hypoxia-reperfusion (H/R) stimulation. MTT and TUNEL staining analyzed cell viability and apoptosis, respectively. RT-qPCR determined miR-455-3p expression. Apoptosis-related proteins, autophagy-associated proteins, and the MEKK1-MKK4-JNK signaling pathway were detected. The interaction between miR-455-3p and MEKK1 was confirmed through dual luciferase activity and RIP assay. An in vivo ischemia reperfusion (I/R) model was established in rats. 2, 3, 5 triphenyltetrazolium chloride (TTC) staining, hematoxylin-eosin (H&E) staining, Masson staining, and TUNEL staining evaluated the infarct volume and histopathological changes.
Results
miR-455-3p′s expression was down-regulated in BMMSCs-derived exosomes, I/R myocardial tissues, and H/R myocardial cells. miR-455-3p enriched by BMMSC exosomes reduced H/R-mediated cardiomyocyte damage and death-related autophagy. miR-455-3p upregulation suppressed MEKK1-MKK4-JNK. MEKK1 overexpression notably mitigated cell apoptosis, cramped cell viability, suppressed autophagy expansion, and attenuated Exo-miR-455-3p′s protection on H/R myocardial cells. In-vivo trials reflected that BMMSC exosomes enriched with miR-455-3p repressed ischemia reperfusion-induced myocardial damage and myocardial cell function.
Conclusion
miR-455-3p, shuttled by exosomes from MSCs, targets the MEKK1-MKK4-JNK signaling pathway to guard against myocardial ischemia-reperfusion damage.
Introduction
Myocardial infarction (MI), also called heart attack, features acute myocardial injury resulting from limited blood supply. A decrease or complete stop in the blood flowing towards the myocardium will lead up to such a phenomenon and, in severe cases, contribute to cardiogenic shock and heart arrest.1–2 As the most common coronary heart disease, it is as well the primary contributor to worldwide mortality and incidence. 3 Myocardial infarction has been confirmed to be inextricably correlated with a slew of risk factors, including high cholesterol, smoking, psychological stress, type 2 diabetes, hypertension, and obesity. 4 Moreover, non-alcoholic fatty liver disease also results in myocardial infarction and other cardiovascular disorders due to regular flavor-enhancing high-lipid diets.5–6 At present, we still need to develop a better understanding of the disease’s pathological process and investigate new therapies, for all preventive and therapeutic solutions utilized clinically, such as blood cholesterol level and blood pressure control, regular workout, the application of aspirin, nitroglycerin and opioids, smoking cessation, and angioplasty.7–8 Reportedly, myocardial cell apoptosis mediates the occurrence of acute myocardial injury subsequent to myocardial infarction, 9 but autophagy exerts a protective effect against cardiac damage following myocardial infarction. 10 However, the cellular pathogenesis of myocardial infarction remains obscure, and a new approach to dampening myocardial cell apoptosis needs to be further clarified.
Mesenchymal stem cells (MSCs) are multipotent cells that can differentiate into myocytes, osteoblasts, chondrocytes, adipocytes and many other types of cells. 11 MSC application has become a strategy for treating myocardial infarction and other cardiovascular diseases. 12 Exosomes have been demonstrated as a promising acellular remedy for cardiovascular diseases due to their better circulatory stability, less toxicity, controllable transplantation dose, and other merits. 13 MSCs can secrete soluble exosomes to modulate the therapeutic effect. 14 For instance, exosomes originating from mesenchymal stem cells can incur myocardial cell autophagy via the AMPK/mTOR and Akt/mTOR pathways to alleviate myocardial ischemia-reperfusion damage. 15 miR-150-5p, an exosome derived from BMMSCs, targets Bax to lessen myocardial cell apoptosis and enhance the cardiac function of MI mice. 16 These findings signify that multiple exosomes stemming from mesenchymal stem cells can exert an outstanding protective effect against MI and myocardial cell damage.
Exosomes, marked by vesicles containing microRNAs, are derived from umpteen cell types, such as stem cells, and are extensively present in blood and urine. They are often discovered in cell culture mediums. 17 With anti-apoptotic, anti-fibrosis, and pro-angiogenesis functions, exosomes are closely linked to heart repair and regeneration. 18 Recent studies have indicated that various exosome miRNAs stemming from MSCs also participate in myocardial injury progression. For instance, exosome miR-143-3p, derived from MSCs, modulates the CHK2-Beclin2 pathway to bolster H/R-caused autophagy and dampen myocardial ischemia-reperfusion damage. 19 Exosome miR-30e, originating from BMMSCs, targets LOX1 to down-regulate the activity of the NF-κB p65/Caspase-9 pathway and hamper the apoptosis and fibrosis of myocardial cells, thus ameliorating heart failure in MI rats. 20 Fu DL et al. have discovered that MSC-derived exosome miR-338 can modulate the MAP3K2/JNK signaling pathway to suppress myocardial cell apoptosis and thus enhance MI rats’ cardiac function. 21 miR-455-3p is another significant member of the miRNA family. It has been demonstrated that lncRNA SNHG7 silencing regulates the miR-455-3p/PTAFR axis to exert its protective function in the angiotensin II-caused apoptosis, fibrosis, and inflammation of cardiac fibroblasts, offering an underlying molecular target for MI treatment. 22 Shao M et al. have confirmed that exosome miR-455-3p stemming from human umbilical cord mesenchymal stem cells can mitigate macrophage infiltration and local liver injury and attenuate the levels of serum inflammatory factors, hence relieving acute liver injury elicited by IL-6. 23 Notwithstanding, whether MSC-derived exosome miR-455-3p functions in MI remains poorly understood.
The C-Jun NH2-Terminal kinase (JNK) pathway, also referred to as stress-activated protein kinase (SAPK), is a crucial member of the MAPK superfamily, mainly activated by inflammatory cytokines and environmental stress. 24 Reportedly, JNK activity is modulated by dual phosphorylation on certain threonine and tyrosine residues through MKK4 (also known as SEK1). 25 MKK4 is initiated by the upstream protein kinase MEKK1. 26 MEKK1-MKK-JNK-AP1 signaling pathway activation exerts a strong inhibitory function in modulating the Rgs4 transcription of rabbit colonic smooth muscle cells. 27 PLCγ2 gene expression inhibition can restrain the MEKK1/MKK4/JNK1/2/c-Jun signaling pathway to induce caspase-dependent cell death. 28 Nevertheless, the function of the pathway in cardiovascular diseases has not hitherto been reported.
The research concentrates on studying the influence of BMMSCs-derived exosome miR-455-3p on MI by establishing an MI animal model in vivo and a myocardial ischemia and hypoxia cell model ex vivo. The underlying mechanism of such an interplay can provide us with a new understanding of exosomes’ function in cardiovascular disease treatment.
Materials and methods
Animal and exosome treatment
Thirty Sprague Dawley (SD) rats (three weeks old, 80–100 g in weight) were supplied by the Animal Center of Hubei University of Medicine, were kept in a cage at 22–24°C with a 12-hour light/dark cycle, fed by standard food particles and water. The animals were randomized to four groups: the sham group (n = 5), the Exo-miR-455-3p group (n = 5), the ischemia-reperfusion (I/R) group (n = 10), and the I/R+Exo-miR-455-3p group (n = 10). Twenty male rats went through left anterior descending (LAD) coronary artery ligation and reperfusion to induce myocardial ischemia/reperfusion (I/R) model. 29 Briefly, the rats were anaesthetized with sodium pentobarbital (50 mg/kg i.p.) prior to surgery. Thoracotomy was conducted between the second and third rib, with a 7–0 Prolene suture utilized to ligate the left anterior descending coronary artery for 30 min. Following 24-hour reperfusion, the scarred area was evaluated. All in all, only rats in the I/R and I/R+Exo-miR-455-3p groups were surgically ligated at the left anterior descending coronary artery. The rats in the Sham group and Exo-miR-455-3p group went through the same surgery, but didn’t receive coronary artery ligation and reperfusion. Exosomes (10 μg) separated from the rats’ bone marrow mesenchymal stem cells were intramyocardially transfused into the left ventricular wall of rats in the I/R+Exo-miR-455-3p group. Twenty-four hours after reperfusion, thoracotomy was performed on the furry animals again to build the aforementioned rat myocardial ischemia-reperfusion model. The left anterior descending coronary arteries were ligated once again, and the separated hearts was dyed with TTC and then harvested for analysis. All surgical operations were implemented in line with the Guidelines for The Care and Use of Laboratory Animals of Hubei University of Medicine in this research which had received the approval from the Medical Ethics Committee of Sinopharm Dongfeng General Hospital and was implemented as per the revised version Guidelines for the Care and Use of Laboratory Animals published by the National Institute of Health (NIH) (Publication No. 85–23, (1996)).
Isolation and culture of rat bone marrow mesenchymal stem cells
BMMSCs were separated from the rats (4 weeks-old, 90–100 g body weight) and then rinsed in α-minimal essential medium (α-MEM) (Thermo Fisher Scientific, Inc.) supplemented with 15% FBS (Thermo Fisher Scientific, Inc.), 1% penicillin and streptomycin (Beyotime, Shanghai, China). 30 Subsequent to filtration and centrifugation, particles with bone marrow mesenchymal stem cells were cultivated for the following analysis. BMMSCs were grown in a culture dish in a 37°C-humidification chamber with 5% CO2 for 7 days. The medium was changed three times a week. Cellular immunofluorescence was done to identify the separated MSCs and then validated by the positive profiles of cell surface antigens CD29 and CD44 instead of CD31 and CD34. Proteintech company supplied the primary antibodies Anti-CD29 (1:100; Catalog number: 12594-1-AP), Anti-CD44 (1:100; Catalog number: 60224-1-Ig), Anti-CD31 (1:100; Cat No. 28083-1-AP), and Anti-CD34 (1:100; Cat No. 60180-1-Ig) employed in cellular immunofluorescence.
Exosome isolation and electron microscopy
Predicated on the method used by Khan M et al., 31 we obtained exosomes from the rats’ bone marrow mesenchymal stem cells. Separated MSCs (passages 4–6) were cultivated for more than 40 h, and exosomes in the medium were gathered through 70 min’ ultracentrifugation at 4°C and 100000 × g. Purified exosomes originating from MSCs were resuspended and kept in saline for the following trials. Exosomes separated from BMMSCs cultivated with normoxia and hypoxia/reoxygenation (O2 <1%, hypoxia 48 h, reoxygenation 24 h) were shortened as Nor-Exo and Hypo-Exo. The exosome suspension was conflated with 4% paraformaldehyde at room temperature for 15 min and applied to a template-painted carbon grid which was pre-coated with 0.01% polylysine at room temperature for a minute and dyed with 1% phosphotungstic acid at the same temperature for 2 min. The FEI Tecnai Spirit G2 transmission electron microscope (Thermo Fisher Scientific, Inc.), with a magnification of 120-kV voltage and ×49,000, was operated to monitor the morphological features of the isolated exosomes.
Histological analysis of rat myocardial tissues
Pentobarbital sodium (135 mg/kg) was adopted to sacrifice the rats. The rats’ hearts were harvested following I/R model construction and certain treatments. The pathogenic characteristics of rats’ myocardial tissues were assessed through histological analysis. After being fixed with 4% paraformaldehyde for 24 h at room temperature, the heart tissues went through dehydration for 2 min with 70% ethanol, 2 min with 90% ethanol, and 2 min with 95% ethanol, respectively. The tissues were embedded in paraffin and sliced up into consecutive sections 4 µm thick. The tissue slices were dyed with hematoxylin at room temperature for 5 min and then with eosin at the same temperature for 2 min. A microscope with an ×400 magnification (TE200, Nikon Corporation) was taken for observation in at least six fields. For TUNEL staining, the slices were incubated with Protease K solution at 37°C for 30 min and then with the mixture of TdT and dUTP (2:29) at 37°C for 2 h, blocked by endogenous peroxidase supplemented with 3% hydrogen peroxide solution in methanol in darkness for 15 min, and then incubated with converter-POD at 37°C for 30 min. A reversed fluorescence microscope (TE2000-E; Nikon Corporation) was applied to observe the sections mounted with an Antifade Mounting medium (cat. no. P0126; Beyotime of Biotechnology). Prior to 2, 3, 5-triphenyltetra-zolium chloride (TTC) staining, the sections were immersed in 1% TTC at room temperature for 15 min. For Masson staining, the slices were dyed with Ponceau S acid fuchsin for 5–10 min, incubated with molybdophosphoric acid solution for 3 min, stained by aniline blue for 5 min, all at room temperature, and ultimately observed using a camera (EOS 80D; Canon, Inc.).
The cellular hypoxia-reoxygenation model
H9C2 cells in the rat heart were adopted to construct a cell H/R model in this work. H9C2 cells, supplied by American Type Culture, were grown in a DMEM medium (Thermo Fishier Scientific, Inc.) containing high glucose, 10% FBS (Thermo Fishier Scientific, Inc.), penicillin, and streptomycin at 37°C with 5% CO2. As described previously, 27 the cells were cultivated in DMEM containing low glucose but no FBS with 5% CO2 in a hypoxic environment for 4 h and then cultivated under normal oxygen for 24 h to set up the cellular H/R model. To figure out the function of exosomes, we treated H9C2 cells with 2 μg of the exosomes at 37°C for 48 h.
Cell transfection
H9C2 cells or BMMSCs in the logarithmic growth stage were digested, passed, and seeded into 6-well plates with a density of 5 × 106/well. The cells were transfected when they achieved stable growth. MEKK1 overexpression plasmid, or miR-455-3p mimics, the miR-455-3p inhibitor, and corresponding negative control fragments were transfected into H9C2 cells or BMMSCs, as guided by the instructions of FuGENEHD Transfection Reagent (Roche, Shanghai, China). The cells were cultivated in an incubator at 37°C with 5% CO2 and then transfected for 24 h in preparation for the following procedures.
MTT assay
H9C2 myocardial cells were inoculated into 96-well plates with a density of 5 × 103 cells/well. Then, 10 μL of MTT solution (Sigma-Aldrich, St. Louis, MO, USA) in PBS (5 mg/mL) was given to each well for 3 h’ incubation in an incubator. With the supernatant removed, 100 μL of dimethyl sulfoxide (DMSO, Sigma-Aldrich, St. Louis, MO, USA) was administered to dissolve the formazan crystals. The optical density was gauged at 450 nm to confirm the cells’ viability in each well.
RNA isolation and quantitative PCR
Primer sequences of each gene.

Western blot
For western blot, total protein was extracted from exosomes, rat tissues, and cultivated cells with the use of RIPA lysis and extraction buffer solution (Thermo Fisher Scientific, Inc.). The protein concentration was confirmed by the Thermo Bicinchoninic Acid Protein Assay kit (cat. no. 23227; Thermo Fisher Scientific, Inc.). Next, 20 μg of the proteins were isolated with 10% SDS-PAGE gel and then moved onto PVDF membranes. After being sealed with 5% skimmed milk at room temperature for 2 h, the membranes incorporating targeted proteins were incubated with primary antibodies for 2 h and with secondary antibodies for 2 h, both at room temperature, successively. A fortified chemiluminescence detection reagent (cat. no. WB KLS0500; EMD Millipore) was administered to develop blots, with the Image-Pro Plus 6.0 software (Media Cybernetics, Inc.) introduced to analyze the band intensity. GAPDH was taken as the internal inference for the examination of the relative protein abundance. The antibodies below were utilized in this study (concentration: 1: 1000): Anti-Alix antibody (ab275377), Anti-CD9 antibody (ab92726), Anti-CD63 antibody (ab108950), Anti-Bad antibody (ab220116), Anti-Bak antibody (ab104124), Anti-Bcl2 antibody (ab182858), Anti-LC3B antibody (ab221794), Anti-P62 antibody (ab109012), Anti-Beclin-1 antibody (ab207612), Anti-MEKK1 antibody (ab138662), Anti-p-MKK4 antibody (ab52958), Anti-MKK4 antibody (ab242052), Anti-p-JNK antibody (ab124956), Anti-JNK antibody (ab199380), and Anti-GAPDH antibody (ab9485) (overnight at 4°C); horseradish peroxidase (HRP) goat anti-Rabbit IgG (cat. no. BA1054; 1:20,000; Wuhan Boster Biological Technology) and HRP goat anti-mouse IgG (cat. no. BA1051; 1:20,000; Wuhan Boster Biological Technology).
Dual luciferase reporter assay
Luciferase reporter gene detection was done through the dual-luciferase reporter assay system (Promega, Madison, WI, USA). Wild-type and mutant-type MEKK1 target fragments were established and consolidated into the pGL3 vector (Promega, Madison, WI, USA) to establish pGL3-ACSL4-wild type (MEKK1-WT) and pGL3-ACSL4-mutant (MEKK1-MT) reporter vectors. MEKK1-WT or MEKK1-MT and miR-455-3p or its negative control were transfected together into H9C2 cells. Forty-eight hours subsequent to the transfection, the luciferase activity was examined in keeping with the supplier’s recommendations. All the procedures were conducted in triplicate and repeated three times.
RNA binding protein immunoprecipitation assay
The EZ-Magna RIP kit (Millipore, MA, USA) was utilized for RIP. In brief, about 2 × 107 H9C2 cells were pelleted and lysed with 100 μL of RIP lysis buffer supplemented with protease and RNase inhibitors. Cell lysates were incubated with the IgG or Ago2 antibody, with IgG as the negative control. Following proteinase K treatment, the immunoprecipitated RNAs were extracted. RT-qPCR was implemented to check miR-455-3p′s abundance.
Statistical analysis
The analysis was implemented with the SPSS17.0 statistical software (SPSS Inc., Chicago, IL, USA). T-test was adopted for comparing two different groups, while One-way ANOVA was taken for comparison among several groups. p < 0.05 was deemed to be statistically meaningful.
Results
The separation of BMMSCs-derived exosomes and determination of miR-455-3p′s expression
To delve into the underlying function of exosomes originating from rat BMMSCs in myocardial infarction, we isolated MSCs from the bone marrow tissues of the SD rats. The harvested MSCs were identified through cellular immunofluorescence. The positive profiles of cell surface genes CD29 and CD44 and the lower profiles of CD31 and CD34 were employed as biomarkers to validate the separation of MSCs (Figure 1(a)). Exosomes were then extracted from the medium culturing the separated rat BMMSCs. A transmission electron microscope was operated to observe the regular shape of the separated MSC-derived exosomes (Figure 1(b)). Western blot was conducted to further analyze the profiles of exosome marker genes CD63, CD9, and Alix. It turned out that these proteins’ expressions were notably higher in exosomes than in MSCs (p < 0.05, Figure 1(c)). Subsequent to BMMSCs’ H/R culture, exosomes were separated to confirm the profile of miR-455-3p. Surprisingly, the results revealed that miR-455-3p was down-regulated after H/R treatment (p < 0.05, Figure 1(d)), and its expression was substantially down-regulated in I/R myocardial tissues and H/R myocardial cells (p < 0.05, Figures 1(e) and (f)). In a nutshell, these findings corroborated the success in isolating MSCs from the rats’ bone marrow tissues and in enriching the exosomes stemming from MSCs for the following analysis. The separation of BMMSCs-derived exosomes and examination of mir-455-3p′s expression. a): Cellular immunofluorescence was adopted to examine rat BMMSCs which were validated by the positive profiles of CD29 and CD44 and the absence of CD31 and CD34 expressions. b): The TEM images (magnification, ×49,000) of the morphological characteristics of rat MSC-originated exosomes. The arrow shows the represented image of isolated exosomes. c): BMMSCs were treated under normoxia and hypoxia/regeneration (H/R) circumstances. Exosomes were separated. Western blot was conducted for detecting exosome markers, including Alix, CD63, and CD9. The gray value was calculated as fold change of BMMSC group. (d–f): RT-qPCR measured the profile of miR-455-3p in BMMSCs (d), I/R myocardial tissues (e), and H/R myocardial cells (f). ***p < 0.001. N = 3.
miR-455-3p overexpression in BMMSC exosomes weakened H/R-elicited myocardial cell apoptosis and autophagy damage
To interrogate the function of exosomal miR-455-3p in myocardial damage induced by H/R, we transfected BMMSCs with miR-455-3p mimics. The exosomes were isolated and added to H9C2 cells induced by H/R. RT-PCR was conducted to check miR-455-3p′s expression. It was demonstrated that in contrast with the control group, the profile of miR-455-3p was considerably lowered in the H/R group, while it was substantially heightened after Exo-miR-455-3p treatment (in comparison with the H/R+Exo-miR-NC group) (p < 0.05, Figure 2(a)). MTT and TUNEL staining indicated that in contrast with the control group, H9C2 cells’ viability was remarkably attenuated, and apoptosis was prominently boosted in the H/R group. However, exosome miR-455-3p mimics’ transfection contributed to a substantial increase in the viability of the cells and a distinct reduction in the apoptosis (by contrast to the H/R+Exo-miR-NC group) (p < 0.05, Figures 2(b) to (d)). The profiles of apoptosis-concerned proteins Bad, Bak, and Bcl2 were examined through western blot. H/R resulted in an increase in the profiles of Bad and Bak and restrained the profile of Bcl2, but the H/R+Exo-miR-455-3p group exhibited the opposite outcome (in contrast with the H/R+Exo-miR-NC group) (p < 0.05, Figure 2(e)). By contrast to the control group, the H/R group had a decline in the profiles of LC3Ⅱ/Ⅰand Beclin-1 (autophagy-associated proteins) as well as an increase in P62 expression, whereas the H/R+Exo-miR-455-3p group reversed the phenomenon (vis-a-vis the H/R+Exo-miR-NC group), as denoted by western blot (p < 0.05, Figure 2(f)). These outcomes signified that miR-455-3p enriched by BMMSC exosomes mitigated H/R-induced apoptosis and autophagy damage in myocardial cells. miR-455-3p-enriched by BMMSC exosomes ameliorated H/R myocardial cell apoptosis and autophagy injury. miR-455-3p mimics or miR-NC were transfected into BMMSCs, and the exosomes were isolated (named as Exo-miR-455-3p and Exo-miR-NC, respectively). H9C2 cells were treated with Exo-miR-455-3p or Exo-miR-NC after they were induced by H/R. a): RT-PCR determined miR-455-3p expression in H9C2 cells. (b–d): MTT and TUNEL staining examined H9C2 cell viability and apoptosis, respectively. (e–f): Western blot ascertained the profiles of apoptosis-concerned proteins Bad, Bak, and Bcl2 and autophagy-associated proteins LC3Ⅱ/Ⅰ, Beclin-1, and P62. *p < 0.05, **p < 0.01, ***p < 0.001. N = 3.
MEKK1 was directly targeted by miR-455-3p in H9C2 cells
The Starbase database (http://starbase.sysu.edu.cn/) confirmed a targeted binding correlation between MEKK1 and miR-455-3p (Figure 3(a)). To validate the targeted relationship, we performed dual luciferase activity assay in H9C2 cells and discovered that miR-455-3p vigorously suppressed the luciferase activity of MEKK1-WT (p < 0.05, Figure 3(b)) but exerted no evident impact on MEKK1-MUT (p > 0.05, Figure 3(b)). Figure 3(c) displayed that the profiles of miR-455-3p and MEKK1 mRNA were both considerably enriched in Ago2 complexes, reflecting the direct interplay between miR-455-3p and MEKK1 (p < 0.05, Figure 3(c)). RT-qPCR and western blot were done in H9C2 cells transfected with miR-455-3p mimics, respectively, to verify the mRNA and protein profiles of MEKK1, denoting that miR-455-3p overexpression conspicuously restrained those expressions of MEKK1 (p < 0.05, Figures 3(d) and (e)). These discoveries revealed that miR-455-3p negatively modulated MEKK1 in H9C2 cells. miR-455-3p directly targeted MEKK1 in H9C2 cells. a): The Starbase database (http://starbase.sysu.edu.cn/) predicted the binding target of MEKK1 and miR-455-3p. (b–c): Dual luciferase activity and RIP assays validated the targeted correlation between them. (d–e): The mRNA and protein profiles of MEKK1 were examined in H9C2 cells transfected with miR-455-3p mimics. NS p > 0.05, ***p < 0.001. N = 3.
The regulatory function of miR-455-3p in the MEKK1-MKK4-JNK pathway
To investigate the exact mechanism of exosome miR-455-3p affecting H9C2 myocardial cell apoptosis and autophagy, we transfected miR-455-3p mimics or inhibitors into H9C2 cells and implemented western blot to examine the protein profile of the MEKK1-MKK4-JNK axis. In contrast with the control group, transfected miR-455-3p mimics led up to a substantial decrease in the protein profile of the pathway (p < 0.05, Figure 4(a)), which was remarkably elevated after miR-455-3p inhibitors were transfected (p < 0.05, Figure 4(b)). This reflected that miR-455-3p overexpression dampened MEKK1-MKK4-JNK axis activation in H9C2 cells. The regulatory effect of miR-455-3p on the MEKK1-MKK4-JNK pathway. miR-455-3p mimics or inhibitors were transfected into H9C2 cells. (a–b): Western blot confirmed the protein profile of the MEKK1-MKK4-JNK signaling pathway. ***p < 0.001. N = 3.
MEKK1 overexpression weakened the protective function of Exo-miR-455-3p in H/R myocardial cells
To verify the mechanism of Exo-miR-455-3p′s function in H9C2 cells, we established MEKK1 overexpression plasmid in the cells. RT-qPCR determined MEKK1’s expression, indicating that by contrast to the control group, the profile of MEKK1 was notably heightened after it was overexpressed (p < 0.05, Figure 5(a)). This demonstrated the success in constructing the MEKK1 overexpression model. Exo-miR-455-3p mimics were transfected into H9C2 cells impaired by H/R to construct MEKK1 overexpression plasmid. In comparison with the H/R group, H9C2 cells’ viability was evidently enhanced, and their apoptosis was conspicuously attenuated following the transfection of Exo-miR-455-3p mimics, whereas MEKK1 overexpression produced the opposite outcome, as displayed by MTT and TUNEL staining (p < 0.05, Figures 5(b) and (c)). Western blot manifested that in contrast with the H/R group, Exo-miR-455-3p mimics’ transfection caused a decline in the profiles of pro-apoptotic proteins Bad and Bak and an increase in the profile of the anti-apoptotic protein Bcl2, while H/R+Exo-miR-455-3p+MEKK1 presented the reverse phenomenon (in comparison with the H/R+Exo-miR-455-3p group) (p < 0.05, Figure 5(d)). As per the outcomes of western blot, when compared to the H/R group, the profiles of LC3Ⅱ/Ⅰ and Beclin-1 were up-regulated, P62’s expression was reduced, and the protein profile of the MEKK1-MKK4-JNK signaling pathway was also apparently attenuated in the H/R+Exo-miR-455-3p group, but such an effect was inverted in the H/R+Exo-miR-455-3p+MEKK1 group (vis-a-vis the H/R+Exo-miR-455-3p group) (p < 0.05, Figures 5(e) and (f)). These discoveries denoted that exosome miR-455-3p defended myocardial cells from H/R-induced damage, whereas MEKK1 overexpression abated its protective function. MEKK1 overexpression weakened the protective function of Exo-miR-455-3p in H/R myocardial cells. Exo-miR-455-3p mimics were added to H9C2 cells transfected with MEKK1 overexpression plasmid. H9C2 cells were injured by H/R. a): RT-qPCR checked the profile of MEKK1. (b–c): MTT and TUNEL staining examined H9C2 cell viability and apoptosis. (d–f): Western blot was done to confirm the protein profiles of Bad, Bak, Bcl2, LC3Ⅱ/Ⅰ, Beclin-1, P62, and the MEKK1-MKK4-JNK signaling pathway. Ns p > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001. N = 3.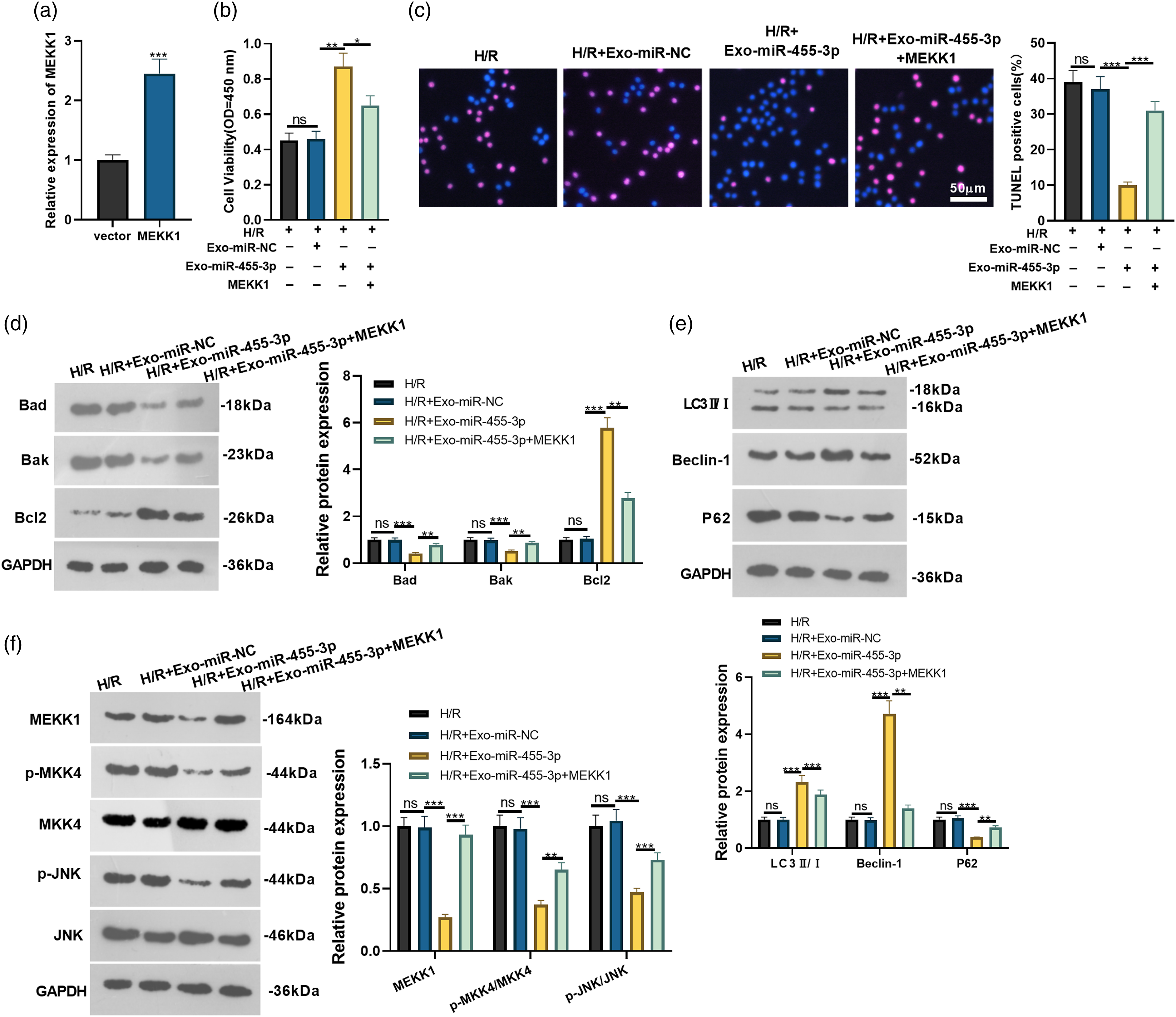
BMMSCs-derived exosome miR-455-3p repressed myocardial damage
To analyze the protective function of BMMSCs-derived exosome miR-455-3p against myocardial infarction, we constructed a myocardial ischemia-reperfusion model via left anterior descending coronary artery ligation and treated the model with the exosome. TTC staining revealed that by contrast to the sham group, the I/R model rats exhibited much more severe myocardial injury, while the administration of Exo-miR-455-3p dampened I/R-caused myocardial infarct (p < 0.05, Figure 6(a)). The histological characteristics of the rats’ myocardial tissues were further examined through H&E and Masson staining. It transpired that the morphology and arrangement of the myocardial tissues manifested irregular alterations after the induction of myocardial infarction. The transfusion of the exosome alleviated the aberrant changes in the cells (p < 0.05, Figure 6(b)). TUNEL staining measured rat myocardial cell apoptosis, indicating a huge increase in the apoptotic cells of I/R rats. Exosome miR-455-3p treatment suppressed myocardial cell apoptosis in rats that went through the ligation of the left anterior descending coronary artery (p < 0.05, Figure 6(c)). Given the outcomes of western blot, in comparison with the sham group, there was a notable increase in the profiles of Bad and Bad and a reduction in the profile of Bcl2 in the I/R group. When compared to the I/R group, the profiles of Bad and Bak were evidently lowered, and Bacl2 expression was considerably enhanced in the I/R+Exo-miR-455-3p group (p < 0.05, Figure 6(d)). In contrast with the sham group, the I/R group had a notable reduction in the profiles of LC3Ⅱ/Ⅰ and Beclin-1 and an increase in P62 expression, whereas the I/R+Exo-miR-455-3p group produced the opposite outcome vis-a-vis the I/R group (p < 0.05, Figure 6(e)). Western blot also reflected that in comparison with the sham group, MEKK1-MKK4-JNK expression was remarkably augmented in the I/R group. On the contrary, it was substantially leveled down in the I/R+Exo-miR-455-3p group in comparison with the I/R group (p < 0.05, Figure 6(f)). In conclusion, the histological and molecular discoveries signified that BMMSCs-derived exosome miR-455-3p repressed MI-induced myocardial tissue injury effectively. BMMSCs-derived exosome miR-455-3p dampened myocardial damage. A myocardial ischemia reperfusion model was built via left anterior descending coronary artery ligation and treated with Exo-miR-455-3p. a): TTC staining was conducted for evaluating infarct volume. b). H&E staining and Masson staining were performed for evaluating the histopathological changes of heart tissues. Red circles indicate myocardial fibrosis. c). TUNEL staining was performed for detecting apoptosis in the tissues. TUNEL-positive cells were shown by arrows. (d–f): Western blot determined the protein profiles of Bad, Bak, bcl2, LC3Ⅱ/Ⅰ, Beclin-1, P62, and the MEKK1-MKK4-JNK signaling pathway. Ns p > 0.05, **p < 0.01, ***p < 0.001. N = 5.
Discussion
WHO has expected myocardial infarction and stroke burden to increase by 11% by 2030. 32 Conventional therapies for myocardial infarction include thrombolysis, percutaneous coronary intervention, coronary artery bypass grafting, and myoblast ischemia relief. However, none of the remedies can repair necrotic heart tissues. 33 That is why a probe into new therapies for myocardial infarction is high on the agenda.
Exosomes were characterized in mammalian reticulocytes as a significant factor in the removal of plasma membrane proteins during erythrocyte formation. 34 An increasing amount of evidence has indicated that exosomes can partake in intercellular communication by transferring proteins, lipids, or miRNAs. 35 Exosomes originating from pluripotent stem cells, in particular, have been confirmed to be of great potential in repairing damage and treating various human diseases. 36 Reportedly, exosomes derived from BMMSCs can modulate the renin-angiotensin-aldosterone system to enhance cardiac remodeling and protect rats from MI. 37 Zou L et al. have also discovered that BMMSCs-derived exosomes can hamper MI by bolstering autophagy. 38 These findings mean that BMMSCs-derived exosomes are of great value in MI development.39,40 Here, we chose an extensively applied method to separate BMMSCs from rat bone marrow tissues. The isolated BMMSCs were identified through flow cytometry to investigate the profiles of CD29 and CD44 on the cell surface, which were regarded as specific biomarkers for exosomes derived from MSCs before. 41 CD31 and CD34 were taken as negative controls. The identification of other biomarkers, such as CD90 and CD45, further stepped up the characterization specificity. Exosomes separated from the medium of the rats’ BMMSCs were further validated by a transmission electron microscope through which the profiles of a good few exosome-specific proteins were also corroborated. The isolation and confirmation of rat BMMSCs and secretory exosomes laid a solid foundation for the following analysis.
miRNAs are critical to cardiovascular pathophysiology and cardiovascular diseases. 42 It has been demonstrated that lncRNASOX2-OT modulates the miR-455-3p/TRAF6 axis to boost inflammation in ischemic heart failure. 43 miR-455-3p expression is notably abated in acute myocardial infarction patients, and miR-455-3p can act as an underlying biomarker for acute myocardial infarction diagnosis in an early stage, 44 which signifies that miR-455-3p takes part in cardiovascular disease progression. Reportedly, exosome miR-455-3p stemming from BMMSCs also exerts a strong function in acute live injury. 23 Nevertheless, whether it functions in MI has not been hitherto reported. However, many other exosome miRNAs originating from BMMSCs have been demonstrated to exert a prominent function in MI. For instance, exosome miR-185 derived from BMMSCs can restrain SOCS2 to dampen MI mice’s ventricular remodeling. 45 BMMSCs-derived exosome miR-25-3p targets pro-apoptotic genes FASL and EZH2 to exert its cardioprotective function in MI. 46 Predicated on the studies above, we conjectured that BMMSCs-derived exosome miR-455-3p exhibited conspicuous value in MI. Fortunately, in this research, miR-455-3p was discovered to be down-regulated in BMMSCs-derived exosomes, I/R myocardial tissues, and H/R myocardial cells. Moreover, BMMSCs-derived exosome miR-455-3p overexpression defended H9C2 cells against H/R-elicited damage by reducing cell apoptosis, augmenting viability, and elevating autophagy. As indicated by in vivo experiments, BMMSCs-derived exosome miR-455-3p repressed ischemia reperfusion-incurred myocardial injury. These outcomes demonstrated that exosome miR-455-3p exerted a protective effect against myocardial damage. These findings could provide a new way of thinking for myocardial infarction treatment.
MEKK1 (also referred to as MAP3K1) is a pivotal kinase activator for cellular stress. Myocardial infarction contributes to the activation of the caspase and MAP3K1/MAP3K10 pathways in aging mice, which may boost resistance generated in them to suppress apoptosis, thus augmenting myocardial cell apoptosis. 47 MKK4/JNK shows a function in myocardial ischemia reperfusion as a downstream MEKK1 pathway whose enhanced activity will lead to greater mitochondrial and myocardial injury in reperfusion following coronary artery occlusion. 48 Therefore, we further probed the exact mechanism of the myocardial protection mediated by exosome miR-455-3p. Bioinformatics analysis confirmed a targeted binding correlation between miR-455-3p and MEKK1, which was also validated by dual luciferase and RIP assays. miR-455-3p overexpression vigorously restrained the profile of MEKK1 in myocardial damage caused by H/R, which implied that miR-455-3p targeted and negatively modulated MEKK1 expression in H9C2 cells. Western blot manifested that miR-455-3p overexpression obviously repressed the protein profile of the MEKK1/MKK4/JNK signaling axis, whereas miR-455-3p knockdown produced the opposite situation. The rescue experiment denoted that in H9C2 cell damage elicited by H/R, myocardial cell viability substantially declined, apoptosis considerably increased, and myocardial cell autophagy weakened after MEKK1 overexpression plasmid was further transfected following Exo-miR-455-3p intervention. This revealed that MEKK1 overexpression weakened the protective function of exosome miR-455-3p, originating from BMMSCs, against H/R-induced damage in myocardial cells.
To conclude, our work has demonstrated that miR-455-3p is down-regulated in BMMSCs-derived Exo, H/R myocardial cells, and I/R myocardial tissues, and miR-455-3p overexpression weakens H/R myocardial cell apoptosis and autophagy injury, which may pertain to the MEKK1/MKK4/JNK signaling pathway mediated by miR-455-3p (Figure 7). Our findings underpin the development of a new myocardial infarction therapy based on exosomes. Nonetheless, whether other signaling pathways are implicated in such a process needs to be further investigated. The mechanism diagram: Exosome miR-455-3p from BMMSCs showed a protective effect against myocardial hypoxia-reoxygenation (H/R). miR-455-3p targeted MEKK1-MKK4-JNK and relieved H/R-caused damage to cardiomyocytes by repressing apoptosis and death-concerned autophagy.
Footnotes
Author contributions
Conceived and designed the experiments: Yusi Shen;
Performed the experiments: Yue Wang;
Statistical analysis: Yue Wang, Yusi Shen;
Wrote the paper: Yue Wang, Yusi Shen;
All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data sets used and analyzed during the current study are available from the corresponding author on reasonable request.
Ethics statement
Our study was approved by the Medical Ethics Committee of Sinopharm Dongfeng General Hospital.
