Abstract
Pulpitis is a common oral inflammatory disease in dental pulp commonly associated with bacterial infection. G protein-coupled receptor 55 (GPR55) is a member of the G protein‐coupled receptors family that has been found to regulate inflammatory response. However, its roles in dental pulp inflammation have not been investigated. In this study, we used lipopolysaccharide (LPS) to induce inflammation in human dental pulp cells (hDPCs) to simulate an in vitro model of pulpitis. We found that LPS markedly induced the GPR55 expression in hDPCs. Treatment with the GPR55 antagonist ML-193 ameliorated the LPS-caused decrease in cell viability and increase in lactate dehydrogenase release. The upregulated inflammatory cytokines, interleukin-6 (IL-6) and tumour necrosis factor α, in LPS-challenged hDPCs were also attenuated by ML-193. Treatment with ML-193 ameliorated LPS-induced production of the inflammatory mediators cyclooxygenase-2/prostaglandin E2 (COX-2/PGE2), and inducible nitric oxide synthase/nitric oxide (iNOS/NO) in hDPCs. ML-193 also inhibited the activation of Toll-like receptor 4-myeloid differentiation primary response 88-nuclear factor-κB (TLR4-Myd88-NF-κB) signaling in LPS-induced hDPCs via decreased expressions of TLR4, Myd88, and p-NF-κB 65. Our study provides evidence that the GPR55 antagonist ML-193 exhibited anti-inflammatory activity in LPS-stimulated hDPCs through inhibiting TLR4-Myd88-NF-κB signaling. The results imply that ML-193 might be a novel agent for pulpitis.
Keywords
Introduction
Dental pulp is a unique tissue because it resides in a rigid chamber. 1 Its specific location provides mechanical support and protects dental pulp from the microbial-rich environment in the oral cavity. Pulpitis is a common oral inflammatory disease in dental pulp and is characterized by the accumulation of inflammatory mediators, such as chemokines and cytokines. 2 It is documented that both genetic and epigenetic factors are essential for the inflammatory response of dental pulp. Bacterial infection is the most important etiological factor in the inflammatory process of dental pulp injury through triggering multiple events including methylation and acetylation of histones, DNA methylation, and modifications to small non-coding RNAs. 3 Thus, understanding how to attenuate dental pulp inflammation after bacterial infection could have important clinical ramifications for pulpitis treatment.
G protein‐coupled receptors (GPCRs) constitute a large membrane receptors family responsible for the transduction of external environmental cues. 4 These proteins are widely expressed in brain and peripheral tissues and can be activated by a broad range of ligands, such as metabolites, hormones, neurotransmitters, ions, and sensory signals.5,6 Accordingly, GPCRs participate in a very broad variety of physiological processes. Their dysfunction is involved in many diseases and they are already used as therapeutic targets by 34% of drugs approved by the Food and Drug Administration (FDA). 7
GPR55 is a member of the GPCRs family that has been found to regulate inflammatory response, and innate and adaptive immune systems. 8 Wlodarczyk et al. 9 reported that GPR55 expression in inflamed tissues is statistically significantly higher in Crohn’s disease patients compared to ulcerative colitis patients. Moreover, in Crohn’s disease patients, GPR55 expression in inflamed tissues is higher compared with non-inflamed colonic tissues. 9 GPR55 plays a role in inflammatory and neuropathic pain associated with the regulation of certain cytokines such as IL-4, IL-10, and IFN gamma. 10 The pharmacological blockade of GPR55 using its antagonist CID16020046 reduces intestinal inflammation by regulating the migration and activation of leukocytes and suppressing inflammatory cytokines production in a colitis model. 11 In another experiment, inhibition of GPR55 by CID16020046 protects human aortic endothelial cells (HAECs) against ox-LDL-induced inflammatory injury. 12 These findings suggest a pro-inflammatory role of GPR55 in experimental inflammatory conditions. Thus, GPR55 has become a new pharmaceutical target for the treatment of inflammatory diseases. 8
Although there is a good understanding of GPR55, its roles in dental pulp inflammation have not been investigated. Here, we used LPS to induce inflammation in human dental pulp cells (hDPCs) to simulate an in vitro model of pulpitis and thereby evaluate the role of GPR55.
Materials and methods
Human dental pulp cells isolation and treatment
The hDPCs were isolated from healthy third molars without any carious disease or pathological conditions such as hyperemic pulp tissue as previously described. 13 Briefly, the dental pulp tissues were gently isolated, minced, and digested in collagenase type I for 20 min. Subsequently, the samples were cultured in DMEM (Hyclone, Logan, Utah, USA) supplemented with 20% FBS. Cells from passages two and three were used for the next experiments. The teeth were collected from 12 healthy donors who were subjected to orthodontic reasons at Huashan Hospital North, Fudan University. This study was approved by the Ethical Review Board of Huashan Hospital North, Fudan University, and the donors were informed and have written an informed consent.
For the LPS treatment group, hDPCs were incubated with 2.5, 5, or 10 μM of LPS for 24 h. For the ML-193 intervention group, hDPCs were pretreated with an antagonist of GPR55 (ML-193, 5 or 10 μM) for 1 h before LPS treatment.
Real-time PCR
After indicated induction, total RNAs of hDPCs were extracted using TRIzol reagent (Invitrogen, Waltham, Massachusetts, USA) for the examination of GPR55, IL-6, TNF-α, COX-2, iNOS, and TLR4 mRNA levels. The reverse transcription reaction was performed using PrimeScript RT master mix (TaKaRa, Japan), followed by polymerase chain reaction (PCR) with an SYBR Premix Ex Taq (TaKaRa). The standard 2−ΔΔCT method was used to quantify the relative mRNA level of target genes.
Western blot
Western blot analysis for the determination of GPR55, COX-2, iNOS, TLR4, Myd88, and p-p65 was performed using the isolated cell lysates of hDPCs as previously described. 14 The primary antibodies against GPR55, COX-2, iNOS, TLR4, Myd88 p-p65, and β-actin were obtained from Abcam and diluted at 1: 1000–3000. The secondary antibody (Abcam) was diluted at 1: 3000. The ImageJ software was applied for detecting the intensity of protein bands.
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT)
The hDPCs viability was measured using the MTT (Sigma-Aldrich, St. Louis, Missouri, USA). After incubation with MTT solution (5 mg/mL) for 4 h, the formazan crystals were solubilized in DMSO buffer. The absorbance at 570 nm was measured using a microplate reader (Bio-Tek, Winooski, Vermont, USA).
Enzyme-linked immunosorbent assay
The levels of IL-6, TNF-α, and PGE2 in a culture supernatant of hDPCs were determined using ELISA kits (R&D Systems, Minneapolis, Minnesota, USA). Finally, the absorbance was measured at 450 nm using a microplate reader (Bio-Tek).
3-Amino,4-aminomethyl-2’,7’-difluorescein, diacetate (DAF-FM DA) staining
The nitric oxide (NO) production was examined using a fluorescence probe DAF-FM DA (Molecular Probes, Eugene, Oregon, USA), which can be converted into DAF-FM triazole in the presence of NO. 15 After incubation with 10 μM DAF-FM DA for 30 min, the hDPCs were examined using a fluorescence microscopy and the intensity of fluorescent signaling was measured using image J to reflect the NO level.
Statistical analysis
All data were analyzed by Prism 6.0 software (GraphPad Software, USA) and presented in the form of mean ±standard error of mean (SEM). Differences among the groups were tested by one-way analysis of variance (ANOVA). Differences were considered significant when p < 0.05.
Results
Cytotoxicity of ML-193 in human dental pulp cellss
To investigate the effect of ML-193 in dental pulp cells, we first evaluated its cytotoxicity via LDH assay. After incubation with ML-193 at concentrations of 0.5, 1, 5, and 10 μM for 24 h, the LDH levels and cell viability in hDPCs did not change significantly (Figure 1(a) and (b)). However, treatment with 50 and 100 μM ML-193 caused significant increases in LDH levels (1.25-fold and 1.36-fold) and decreases in cell viability (0.91- and 0.83-fold). Cytotoxicity of the G protein-coupled receptor 55 antagonist ML-193 in human dental pulp cells. Cells were stimulated with ML-193 at concentrations of 0.5, 1, 5, 10, 50, 100 μM for 24 h (a). Lactate dehydrogenase release; (b). Cell viability (*, **, p < 0.05, 0.01 vs Control group).
Lipopolysaccharide increased the expression of GPR55 in human dental pulp cells
Next, we assessed the expression of GPR55 in response to stimulation with different doses of MP-193. In Figure 2(a), the mRNA levels of GPR55 in hDPCs were respectively increased to 1.9-, 2.6, and 3.2-fold after stimulation with LPS (2.5, 5, and 10 μM) for 24 h. Meanwhile, LPS stimulation (2.5, 5, and 10 μM) also led to 1.8-, 2.3-, and 2.7-fold increases in the protein levels of GPR55 in hDPCs (Figure 2(b)). LPS increased the expression of GPR55 in human dental pulp cells. Cells were stimulated with LPS (2.5, 5 and 10 μM) for 24 h (a). mRNA of GPR55; (b). Protein of GPR55 (*, **, ***, p < 0.05, 0.01, 0.005 vs Control group). LPS: Lipopolysaccharide; GPR55: G protein-coupled receptor 55.
ML-193 ameliorated lipopolysaccharide-induced expressions of IL-6 and tumour necrosis factor α in human dental pulp cells
In Figure 3(a), we found that the mRNA levels of IL-6 and TNF-α were increased to 6.3- and 7.8-fold by treatment with LPS (10 μM), whilst ML-193 (5, 10 μM) treatment reduced the mRNA levels of IL-6 (31.8% and 44.4%) and TNF-α (28.2% and 47.4%) in LPS-treated hDPCs. Consistently, the increased protein levels of IL-6 (9.3-fold) and TNF-α (7.6-fold) in LPS-treated hDPCs were also rescued by 5 and 10 μM ML-193 (Figure 3(b)). ML-193 ameliorated LPS-induced expressions of IL-6 and TNF-α in human dental pulp cells. Cells were stimulated with LPS (10 μM) with or without ML-193 (5, 10 μM) for 24 h (a). mRNA of IL-6 and TNF-α; (b). Proteins of IL-6 and TNF-α (***, p < 0.005 vs Vehicle group; #, ##, p < 0.05, 0.01 vs LPS group). LPS: Lipopolysaccharide; TNF-α: tumour necrosis factor α.
ML-193 inhibited the lipopolysaccharide-induced expression of COX-2 and the production of PGE2 in human dental pulp cells
Treatment with LPS (10 μM) alone resulted in higher expression of COX-2 at both mRNA (3.5-fold) and protein levels (2.9-fold) in hDPCs, which could be attenuated by ML-193 (5, 10 μM) (Figure 4(a) and (b)). In addition, LPS increased the production of PGE2 with a 4.4-fold change, which was then reduced by 30.9% and 48.2% ML-193 (5, 10 μM) (Figure 4(c)). ML-193 inhibited the LPS-induced expressions of COX-2 and the production of PGE2 hDPCs. (a). mRNA of COX-2; (b). Protein of COX-2; (c). The production of PGE2 (***, p < 0.005 vs Vehicle group; #, ##, p < 0.05, 0.01 vs LPS group). LPS: Lipopolysaccharide; PGE2: prostaglandin E2; COX-2: cyclooxygenase-2.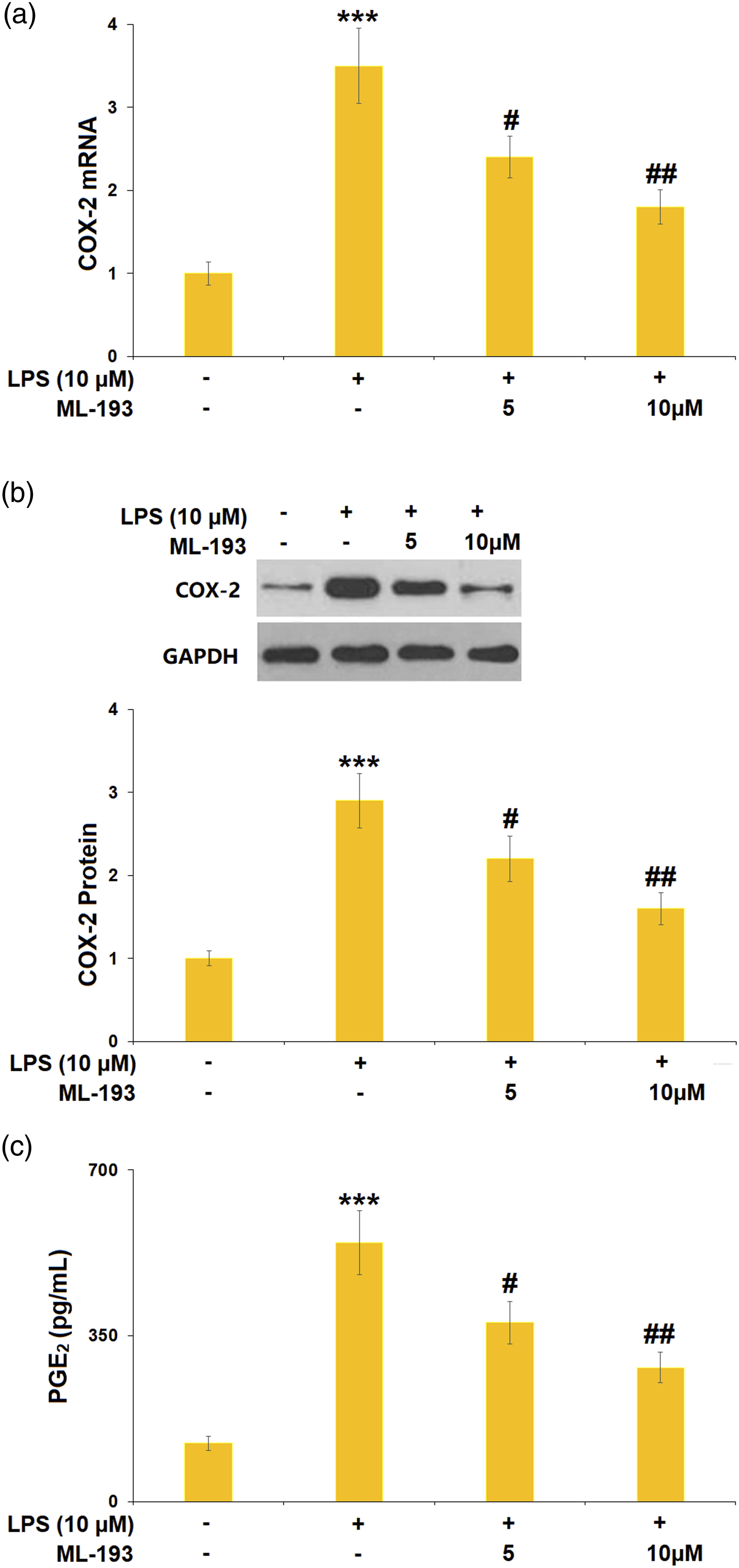
ML-193 ameliorated the lipopolysaccharide-induced expression of inducible nitric oxide synthase and production of nitric oxide in human dental pulp cells
After LPS (10 μM) stimulation, the mRNA and protein levels of iNOS were respectively increased by 3.1- and 2.8-fold in hDPCs (Figure 5(a) and (b)). Treatment with ML-193 (5, 10 μM) caused apparent reductions in both mRNA (25.8% and 45.2%) and protein levels of iNOS (25.0% and 46.4%). The production of NO was markedly increased by 3.0-fold in LPS-treated hDPCs, which could be reduced by ML-193 (5, 10 μM) with 0.7- and 0.5-fold changes, respectively (Figure 5(c)). ML-193 ameliorated the LPS-induced expression of iNOS and the production of NO in hDPCs. (a). mRNA of iNOS; (b). Protein of iNOS; (c). The production of NO (***, p < 0.005 vs Vehicle group; #, ##, p < 0.05, 0.01 vs LPS group). LPS: Lipopolysaccharide; iNOS: inducible nitric oxide synthase; NO: nitric oxide.
ML-193 ameliorated lipopolysaccharide-induced activation of TLR4/Myd88/NF-κB signaling in human dental pulp cells
The TLR4 mRNA in hDPCs was significantly increased by 3.6-fold after LPS stimulation, whereas intervention with 5 or 10 μM of ML-193 reduced the mRNA levels of TLR4 with 0.72- and 0.50-fold changes, respectively (Figure 6(a)). The hDPCs treated with LPS alone exhibited a higher protein level of TLR4 with a 2.7-fold change, which could be decreased by 5 or 10 μM of ML-193 treatment (22.2% and 37.0% reduction) (Figure 6(b)). ML-193 ameliorated LPS-induced expression of TLR4 in human dental pulp cells. (A). mRNA of TLR4; (B). Protein of TLR4 (***, p < 0.005 vs Vehicle group; #, ##, p < 0.05, 0.01 vs LPS group). LPS: Lipopolysaccharide; TLR: Toll-like receptors.
LPS induced the activation of Myd88 and NF-κB in hDPCs, accompanied by a 3.2-fold increase in Myd88 expression and a 2.9-fold increase in p-NF-κB p65 expression (Figure 7). However, 5 or 10 μM of ML-193 treatment caused significant decreases in both Myd88 expressions (25.0% and 40.6% reduction) and p-NF-κB p65 expression (24.1% and 41.4% reduction). ML-193 suppressed LPS-induced activation of Myd88 and NF-κB. The expressions of Myd88 and p-NF-κB p65 were measured by western blot analysis (***, p < 0.005 vs Vehicle group; #, ##, p < 0.05, 0.01 vs LPS group). LPS: Lipopolysaccharide.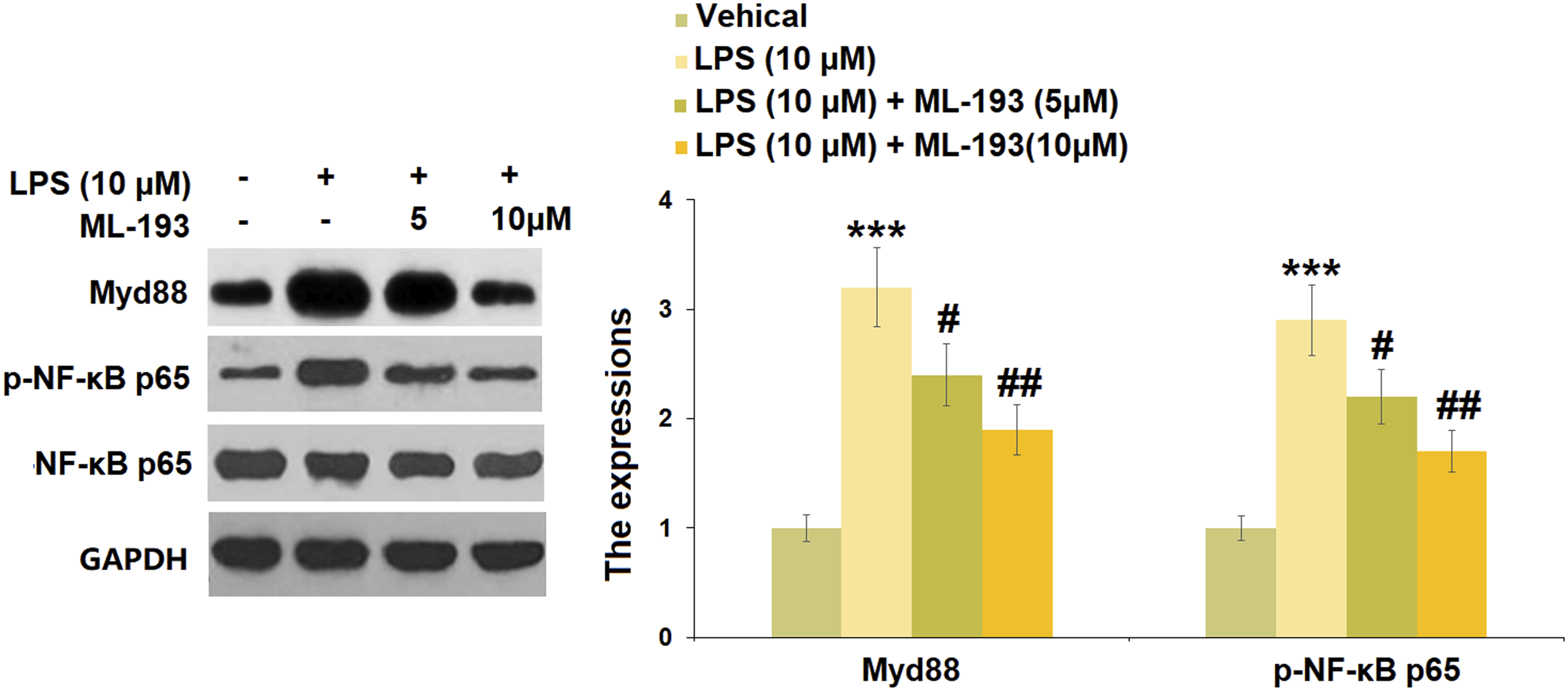
Discussion
As mentioned above, bacterial infection is a significant factor that triggers dental pulp inflammation. LPS is an endotoxin that exists on the cell walls of Gram-negative bacteria and maintains bacterial cell integrity and viability. It helps the bacteria defend against environmental stress. 16 During bacterial infection, LPS exists ubiquitously in the environment and exhibits a potent ability to induce inflammatory responses. Intensive research has led to the identification of LPS in modulating the immune system and susceptibility to plenty of diseases, based on the potency of its pro-inflammatory reaction. 17 Here, we used LPS to induce inflammation in hDPCs to simulate an in vitro model of pulpitis. A group of researchers demonstrated that the increased level of LDH, is a cell toxicity marker also connected with a reduction in cell viability due to the production of pro-inflammatory cytokines. 18 Moreover, the present study shows that LPS successfully induced a potent inflammatory response and cellular injury in hDPCs by decreasing cell viability, increasing the release of LDH followed by upregulation of inflammatory cytokines.
Previous studies have demonstrated that GPR55 participates in the LPS-induced inflammatory response in various cells and organs. GPR55 is involved in intestinal inflammation since its expression is increased in LPS-treated intestines, and its knockout attenuates inflammation in colitis mice. 19 GPR55 antagonists exhibit anti-neuroinflammatory effects in LPS-activated primary microglial cells with decreased PGE2 and COX-2. 20 A putative GPR55 antagonist, cannabidiol, presents a concentration-dependent inhibitory effect on inflammatory cytokines expression in cerebellar granule cells induced by LPS. 21 GPR55 plays a pro-inflammatory role since it can boost IL-12 and TNF-α production, as well as decrease the endocytic activity in LPS-activated monocytes. 22 Here, we found that LPS increased the expression of GPR55 in hDPCs. Treatment with the GPR55 antagonist ML-193 ameliorated LPS-induced expressions of IL-6 and TNF-α, COX-2/PGE2, as well as iNOS/NO in hDPCs, implying its proinflammatory role in LPS-induced hDPCs.
Toll-like receptors (TLRs) are a group of proteins expressed by a variety of immune cells and participate in the inflammatory response by recognizing various pathogen-associated molecules.23,24 TLR4, the most studied TLR, is a critical driver of immune response that has been confirmed as the signaling receptor for LPS.
25
The adaptor molecules activated by TLR4 in response to LPS include Myd88, TIR-containing adapter molecule (TRIF), and TIRF-related adaptor protein (TRAM).
26
The most extensively studied Myd88 plays a critical role in the early response to LPS.
27
One of the important intracellular events following the activation of Myd88 is the late activation of NF-κB, which leads to the induction of proinflammatory cytokines and chemokines, as well as other transcription factors.28,29 Therefore, the TLR4-Myd88-NF-κB signal transduction mediates the inflammatory response following LPS stimulation. It has been found that KIT 10, a novel GPR55 receptor antagonist, reduces neuroinflammatory parameters in microglia by inhibiting the phosphorylation of IκB-α, which is a crucial upstream step of the NF-κB signaling pathway.
30
We found that ML-193 ameliorated LPS-induced expressions of TLR4, Myd88, and p-NF-κB 65 in hDPCs, implying that it inhibited the activation of TLR4-Myd88-NF-κB signaling in LPS-induced hDPCs. The underlying mechanism is illustrated in Figure 8. Diagram of pharmacological functions of ML-193 in against lipopolysaccharide-induced damages in human dental pulp cells.
In conclusion, our study provides evidence for the anti-inflammatory activity of the GPR55 antagonist ML-193 in LPS-induced hDPCs. Mechanistically, TLR4-Myd88-NF-κB signaling was involved in the protective role of ML-193. Our findings suggest the GPR55 antagonist could be further investigated as a potential therapeutic agent for pulpitis.
Supplemental Material
Supplemental Material - Antagonism of G protein-coupled receptor 55 prevents lipopolysaccharide-induced damages in human dental pulp cells
Supplementary Material for Antagonism of G protein-coupled receptor 55 prevents lipopolysaccharide-induced damages in human dental pulp cells by Wei-Jie Li and Jie Shen in Human & Experimental Toxicology.
Footnotes
Acknowledgements
The authors would like to express our appreciation to “Jiading District Central Hospital Affiliated Shanghai University of Medicine & Health Sciences” for their support.
Author contributions
WeiJie Li and Jie Shen contributed to the experimental design and data analysis; WeiJie Li contributed to the investigation; Jie Shen prepared the manuscript. All authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The protocol of this study was approved by the Ethical Committee of Jiading District Central Hospital Affiliated Shanghai University of Medicine & Health Sciences.
Data availability
Requests for data and materials should be addressed to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
