Abstract
Background
Human dental pulp stem cells (hDPSCs) possess mesenchymal stem cell properties, originating from migrating neural crest cells. hDPSCs have received extensive attention in the field of tissue engineering and regenerative medicine due to their accessibility and ability to differentiate in several cell phenotypes. In this study, we cultured hDPSCs with Y-27632 to observe their biological behaviors changes.
Methods
The hDPSCs were separately cultured with Y-27632 (0, 0.156, 0.312, 0.625, 1.25, 2.50, 5, 10, 20, 40 μ
Results
The hDPSCs proliferation was significantly enhanced after cultured with 2.50 μ
Conclusions
The biological behaviors of hDPSCs could be changed when they cultured with Y-27632.
Keywords
Introduction
Human dental pulp stem cells (hDPSCs) possess mesenchymal stem cell properties, originating from migrating neural crest cells. 1 hDPSCs have received extensive attention in the field of tissue engineering and regenerative medicine due to their accessibility and ability to differentiate into adipogenic, chrondrogenic, and osteogenic cell.1,2 In the context of regenerative therapies, hDPSCs provide a hope for repairing lost dental pulp and tooth tissue because of their abilities to differentiate into dentin cells and vascular.3,4 However, the environment of receptor regions is generally uncertain, including inflammation and stress state, which could affect the differentiation of hDPSCs.
Y-27632 (R)-(+)-trans-4-(1-Aminoethyl)-N-(4-Pyridyl)cyclohexanecarboxamide), a highly selective Rho kinase inhibitor, could increase the dentin sialophosphoprote in expression and ALP activity, when long-term cultured with human odontoma-derived cell (hODC). 5 Wang et al. have reported that Y-27632 significantly promoted human periodontal ligament stem cells (hPDLSCs) proliferation, chemotaxis, wound healing, fat droplets formation, and pluripotency, while inhibited ALP activity and mineral deposition. 6 Several studies have showed that Y-27632 regulates the cytoskeletal changes associated with cell adhesion and has been used to protect cultured cells during their passaging.7–9 Additionally, Yang and colleagues found Y-27632 transfection in human exfoliated deciduous teeth (SHEDs) enhanced cell viability, colony formation, and neurosphere formation. 10 These evidences suggest that Y-27632 is a potential reagent that is able to enhance the proliferation of different stem cells types.
In this study, we assessed the effects of Y-27632 on biological behaviors of hDPSCs, including the proliferation, apoptosis, migration, wound-healing ability, and differentiation capacities, at the same time, complemented with expression of stem cell self-renewal, osteogenic, and adipogenic marker genes.
Methods
Cells isolation and primary culture
Primary human dental pulp stem cells (hDPSCs) were isolated by as previous described 11 and cultured in α-MEM medium (12571063, GibcoTM) contained with 10% fetal bovine serum (FBS) at 37°C with 5% CO2. The medium was replaced every 3 days. When they reached 85%–90% confluence, the cells were used for experiments. The procedure for obtaining human dental pulp stem cells from extracted teeth without any personal identity information was approved by the Medical Ethical Committee of the Shengli Oilfield Central Hospital of Shandong province (No. Q/ZNN-ZY-YWB-LL202118). Early passage (passage2 or 3) dental pulp stem cells were used for all experiments.
Separation method refers to and improves the Virginia method as followings: 1. Rinsed and disinfected after the tooth was completely extracted, split the tooth under sterile conditions, and removed the pulp 2. Disinfection solution: 70% ethanol, 2× antibiotics PBS: 1% penicillin (100 U/mL) and streptomycin (100 mg/L) in PBS 3. Enzyme mixture: 2.5 mg/mL Dispase and 2.5 mg/mL type I collagenase in DMEM 4. Neutralization solution: 10% FBS in DMEM with 1% penicillin/streptomycin 5. Inoculation medium (The culture system was consisted of α-MEM, 10% FBS and 100 U/mL penicillin, 100 μg/mL streptomycin; The culture conditions were 100% relative humidity, 5% C02, 37°C) 6. Routine passage when cell growth was close to fusion, amplification culture, inverted microscopy to observe cell morphology. 7. Materials for tissue processing: Forceps, autoclaved scalpel holder, sterile blades, 50 mL falcon tube, 100 μm cell strainers, and 100 mm tissue culture dishes.
Cell Counting Kit-8(CCK-8 assay)
The cells suspension (100 μL, 5 × 103 cell/well) was cultured for 24 h at 37°C with 5% CO2. Then, 10 μL different concentrations (0, 0.156, 0.312, 0.625, 1.25, 2.50, 5, 10, 20, 40 μ
Cells groups
According to the results of CCK-8, the suitable culture condition of Y-27632 was concentration 2.50 μ
EdU (5-Ethynyl-2’-deoxyuridine) assay
After trypsin digestion, 100 μL cell suspensions (1 × 103 cell/well) were cultured in 5% CO2 incubator at 37°C. The hDPSCs were incubated with preheated 20 μM equal volume of BeyoClick TM EdU solution (C0088s, Beyotime, China) for 2 h at 24 h and 96 h, separately. Then, the cultured medium was removed and the hDPSCs were fixed with 1 mL fixed solution for 15 min at room temperature. Removing the fixed solution, the cells were washed with 1 mL PBS three times, each time for 5 min. The hDPSCs were incubated at room temperature for 10–15 min. After washing, the cells were cultured with endogenous peroxidase blocking solution for 20 min at room temperature. Then, adding 50 Click reactions into each well, the hDPSCs were cultured for 30 min at room temperature. After washing, the cells were cultured with Streptavidin-HRP solution for 30 min at room temperature. After washing, the cells were cultured with 0.1 mL TMB solution for 20 min. The reaction was terminated by adding 25 μL H2SO4 (2
Plate cloning
After digestion with 0.25% trypsin (BL006A, Biomiky), the cell suspension (5000 cell/well) was inoculated in 6-well plate, three multiple wells in each group. The inoculated hDPSCs were cultured for 8 days at 37°C with 5% CO2, and the medium was changed every 3 days. Before the termination of the experiment, the cell clones were photographed under the fluorescence microscope (MF53, Mshot). After washing with PBS, the cells were fixed with 4% paraformaldehyde (1 mL) for 45 min. After washing with PBS, the cells were stained with 1000 μL crystal violet staining solution (C0775, Sigma) for 15 min. The cells were photographed with camera after washing with ddH2O and drying in the air.
Apoptosis was detected by flow cytometry
After digestion with 0.25% trypsin, the cell suspension (1000 cell/well) was collected into 5 mL centrifuge tube. After centrifugation at 1300 rmp for 5 min, the supernatant was discarded and the cell precipitates were washed with precooled D-Hanks (pH=7.2–7.4). The cells were suspended with 1×binding buffer, then centrifuged at 1300 rmp for 3 min. Suspending with 1×binding buffer (200 μL), the hDPSCs were cultured with 2 μL Annexin V-FITC and PI (22837, AAT Bioquest) for 45 min at room temperature without light. The results were measured using a flow cytometry (BD FACSCanto).
Transwell assay
The serum-free cell suspension (5 × 104 cell/well) was inoculated in 24-well plate. Removing the medium from the upper chamber, hDPSCs suspension (100 μL) was added into the upper chamber. In the lower chamber, 30% FBS (600 μL) was added. The plates were cultured at 37°C for 48 h. After removing the non-transferred cells with a cotton swab, the transferred cells were stained with crystal violet staining solution (C0121, Beyotime, China) for 15 min. Washing and drying the chamber, the results were observed under a light microscope with 100 magnifications.
Scratch test
Two horizontal lines were drawn on the back of 12 wells. About 2 × 105 cells were added to reach 90% convergence degree on the next day. On the second day, the cells were cultured with low concentration serum medium and scratched with a 10 μL gun head. The cells were observed after cultured for 8 h, 24 h at 37°C, respectively.
Alkaline phosphatase (ALP) activity of hDPSCs was detected by ELISA
The hDPSCs supernatant was collected after centrifuging 1300 rpm for 20 min. The cells suspension was diluted with PBS (pH = 7.2–7.4). According to the instructions of ALP ELISA kit (P0321S, Beyotime, China), the absorbance (OD) of each well was measured at 450 nm. The determination should be carried out within 15 min after adding the termination solution.
Alizarin red staining
The hDPSCs were digested with 0.25% Trypsin-0.04% EDTA, and seeded into 24 well plates with 0.1% gelatin. In each well, 1 mL complete medium (HUXDP-90011, OriCell) was added. When the degree of cell fusion reached 60%–70%, removed the complete medium, and added 2 mL complete medium for osteogenic differentiation (HUXDP-90021,OriCell). The complete medium for osteogenic differentiation was changed every 3 days. After 21 days, removed the medium and washed with PBS. In each well, 4% neutral formaldehyde was added for 30 min. After washing with PBS, 2% alizarin red S solution (C0138, Beyotime, China) was added for 3–5 min. After washing, the cells were observed under a light microscope.
Oil red O staining
The hDPSCs were digested with 0.25%Trypsin-0.04% EDTA, and seeded into 24 well plates with 0.1% gelatin. In each well, 2 mL complete medium was added. When the degree of cell fusion reached 100%, removed the complete medium, and added 1 mL adipogenic differentiation medium A (HUXDP-90031,OriCell). After 3 days, removing the medium A, and added the adipogenic differentiation medium B (HUXDP-90031,OriCell) for 24 h. Then, replaced medium B with medium A and cultured for 3 days. Alternately cultured with medium A and medium B for 14 days, the cells were cultured with medium B for 7 days. After washing with PBS, the cells were fixed with 4% neutral formaldehyde for 30 min, and stained with oil red O staining (C0158
Quantitative RT-PCR (qRT-PCR) assay
PCR Primers used for qRT-PCR.

Statistical analysis
The SPSS19.0 (IBM, USA) was used for statistical analysis. The samples used in each experiment were independent, and the same samples were not used in different tests. All data in the experimental results were repeated independently for 3 times and represented as mean ± standard deviation. Between two groups, the t test was used to compare the difference (p < .05).
Results
Suitable conditions of Y-27632 cultured with hDPSCs
The effects of different concentrations of Y-27632 (0, 0.156, 0.312, 0.625, 1.25, 2.50, 5, 10, 20, 40 μ CCK-8 assay was used to analyze the proliferation of human dental pulp stem cells (hDPSCs) treated with different concentration of Y-27632 (0, 0.156, 0.312, 0.625, 1.25, 2.50, 5, 10, 20, 40 μ
Effects of Y-27632 on the proliferation of hDPSCs
The proliferation of hDPSCs was analyzed by EdU (Figure 2(a)) and plate cloning (Figure 2(b)), and the results showed that the proliferation of hDPSCs was significantly increased after treatment with 2.50 μ EdU (a) and plate cloning (b) experiments were used to analyze the proliferation ability of hDPSCs. Compared with control group (n = 3), *p < 0.05.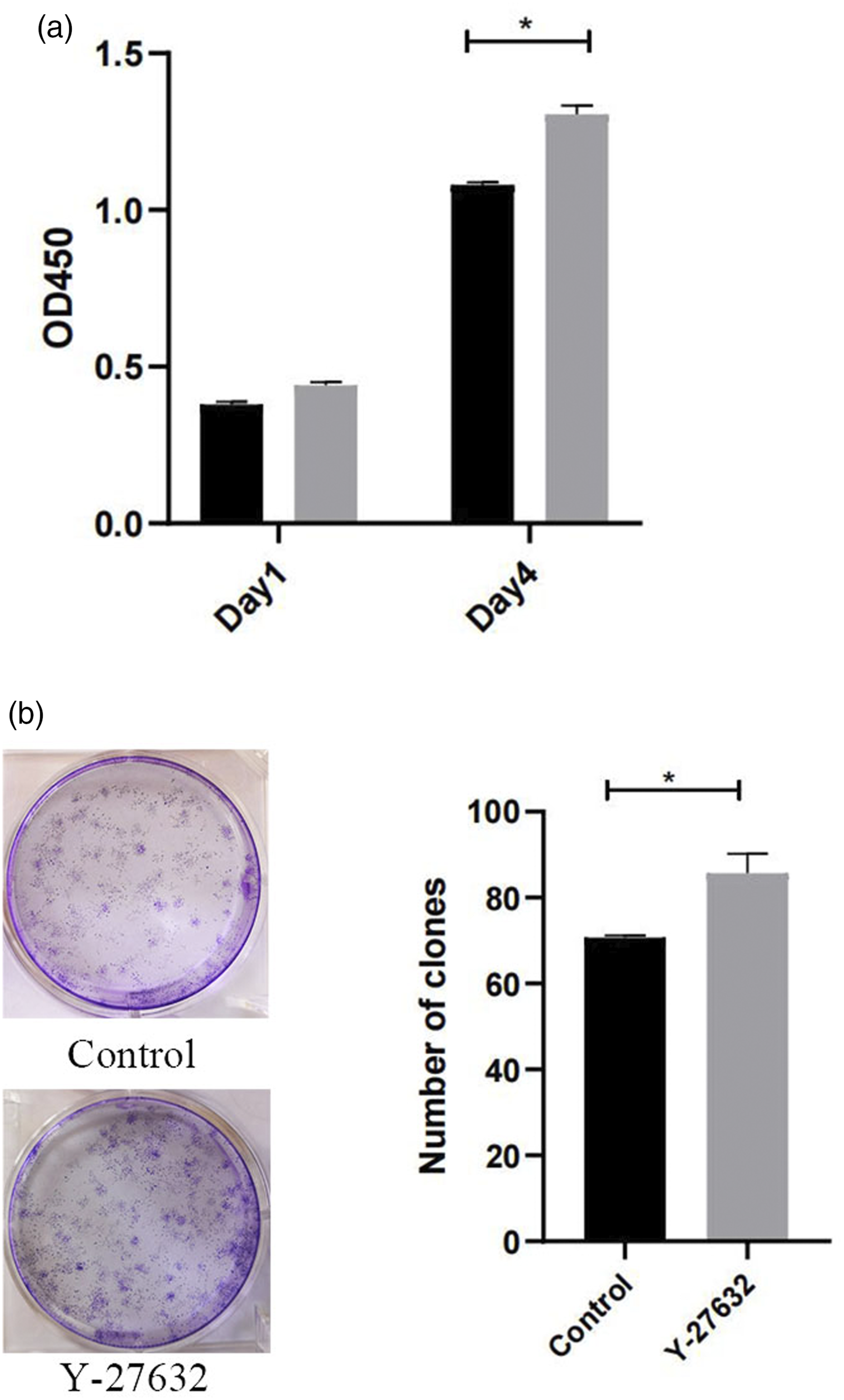
Effects of Y-27632 on the migration ability of hDPSCs
The migration ability of hDPSCs was analyzed using transwell (Figure 3(a)) and scratch (Figure 3(b)) experiments. Transwell results showed that there was no significant difference between control group and Y-27632 group (p = .419); Scratch results also showed that there was no significant difference between control group and Y-27632 group at 8 h (p = 0.25) and 24 h (p = 0.71). The migration ability of hDPSCs was analyzed using transwell (Figure 3(a)) and scratch (Figure 3(b)) experiments. In Figure 3(a), the pictures were observed under 100 magnifications; In figure 3(b), the images were observed under 40 magnifications (n = 3).
Effects of Y-27632 on the apoptosis of hDPSCs
The apoptosis of hDPSCs was analyzed using Annexin V FITC/PI kit (Figure 4), and the data showed that treatment with 2.50 μ Effects of Y-27632 on the apoptosis of hDPSCs were analyzed using Annexin V FITC/PI apoptosis kit (n = 3).
Effects of Y-27632 on ALP of hDPSCs
The effect of Y-27632 on ALP activity of hDPSCs was measured by the ALP ELISA kit (Figure 5). The results showed that 2.50μ The effect of Y-27632 on ALP activity of hDPSCs was measured by the ALP ELISA kit. Compared with control group (n = 3), **p < .01.
Effects of Y-27632 on osteogenic calcium nodules and lipid droplets of hDPSCs
Alizarin Red S staining was performed to identify extracellular calcium deposits of cells differentiated into osteoblasts (Figure 6(a)), and oil red O staining was performed to identify lipids in cells differentiated into adipocytes (Figure 6(b)). Compared with the control group, the number of mineralized nodules in Y-27632 group clearly decreased (p = 0.00), but the amount and size of lipid droplets in Y-27632 group were increased. The proportion of average area of lipid droplets in Y-27632 group was significantly larger than that in control group (p = .047). Alizarin Red S staining was performed to identify extracellular calcium deposits of cells differentiated into osteoblasts (Figure 6(a)), and oil red O staining was performed to identify lipids in cells differentiated into adipocytes (Figure 6(b)). In Figure 6, the pictures were observed under 100 magnifications. Compared with control group (n = 3), *p < .05, **p < .01.
Effects of Y-27632 on genes expression of hDPSCs
The effects of Y-27632 on genes expression related to osteogenesis (Figure 7(a)), adipogenesis (Figure 7(b)), stemness maintenance (Figure 7(c)), and inflammation (Figure 7(d)) of hDPSCs were analyzed by qRT-PCR. The results showed that the Runx2 and ALP genes expression in the Y-27632 group were significantly lower than those in the control group (p = .00, Figure 7(a)). Compared with the control group, AP2 and PPARγ-2 genes in the Y-27632 group were significantly increased (p = 0.00, Figure 7(b)). Compared with the control group, CD73, CD90, CD105, and CD166 genes in the Y-27632 group were significantly decreased (p = .00, Figure 7(c)), while there was no significant difference between groups for STRO-1 gene (p = .573). After treatment of Y-27632, IL-8 gene was significantly increased (p = 0.00), while TLR4 and NF-κB genes were significantly decreased (p = .00, Figure 7(d)). The effects of Y-27632 on genes expression related to osteogenesis (Figure 7(a)), adipogenesis (Figure 7(b)), stemness maintenance (Figure 7(c)) and inflammation (Figure 7(d)) of hDPSCs were analyzed by Quantitative RT-PCR (qRT-PCR). Compared with control group (n = 3), **p < .01.
Discussion
Dental caries and periodontal disease, such as periodontitis, fractures, genetic defects, or aging, can lead to tooth damage and loss, thereby reducing the quality of life. 12 In current dental clinical practice, the therapeutic methods are major replacing missing teeth using osseointegrated dental implants, 13 or removing the infected hard tissue part and replacement with composite resins. 14 When the bacterial infection reaches the pulp, root canal therapy is the first choice. 15 In recent years, regenerative dentistry has become a hot topic in the research of prosthodontics, such as, injection of stem cells and/or growth factors.16–18 Various studies have demonstrated that the potential of hDPSCs for dental pulp regeneration.19,20 However, there is still little information for the effects of Y-27632 on the hDPSCs properties and differentiation.
Many studies have showed that supplementary Y-27632 could promote the proliferation and suppress the apoptosis of pluripotent stem cells, including human embryonic stem cells
21
and hPDLSCs.
6
In this study, we selected 2.5 μ
ALP activity plays an important role in the mineralization process, which is usually used as a marker for osteogenic differentiation. 22 Runx2 is the main gene controlling the differentiation of osteoblasts and plays a key role in the differentiation of mesenchymal stem cells (MSCs) into osteoblasts. 23 Adipocyte protein 2 (AP2) and peroxisome proliferator activated receptor γ-2 (PPARγ-2) are mainly located in adipocytes, participating in the transportation of fatty acids in cells and promoting the metabolism of fat in the body. 24 In our study, Y-27632 co-culture notably inhibited ALP activity, decreased the Runx2 and ALP genes expression, indicating that Y-27632 decreased the ability of hDPSCs to produce mineralized nodules. In turn, Y-27632 increased the ability of hDPSCs to produce fat droplets through promoting the AP2 and PPARγ-2 genes expression. The results of alizarin red staining and oil red O staining also indicated that Y-27632 enhanced the potential of adipogenic differentiation, and suppressed the potential of osteogenic differentiation.
MSCs-associated markers, namely CD73, CD90, CD105, and CD166, 25 were clearly decreased when hDPSCs co-cultured with Y-27632. But the STRO-1 gene express was not affected by Y-27632, which is also an essential MSCs-associated marker. 26 These data suggested that the cell type specific regulation of hDPSCs during Y-27632 treatment. Additionally, we measured the genes expression of interleukin (IL)-8, TLR4 and Nuclear Factor-κB (NF-κB) p65 in cells. The IL-8 expression significantly increased, but TLR4 and NF-κB p65 expression clearly decreased when hDPSCs co-cultured with Y-27632. IL-8 has been shown to promote homing of bone marrow-derived cells to injured sites, and enhances the angiogenic potential of human bone marrow mesenchymal stem cells by increasing vascular endothelial growth factor. 27 TLR4/NF-κB p65 signaling is an important inflammatory response pathway. The activation of TLR4 promotes the phosphorylation of NF-κB, thereby promoting inflammatory effects. 28 These data provided an enlightenment that the regulatory mechanism of Y-27632 may be related with IL-8, TLR4/NF-κB p65 signaling.
Conclusions
In conclusion, we demonstrated that Y-27632 could enhance the proliferation of hDPSCs, but have no significant effects on the migration, wound-healing ability, and apoptosis. At the same time, Y-27632 could enhance the potential of adipogenic differentiation, and suppress the potential of osteogenic differentiation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Shandong Province Medicine and Health Science and Technology Development Project (Grant No. 2018WS163, 2019WS045).
