Abstract
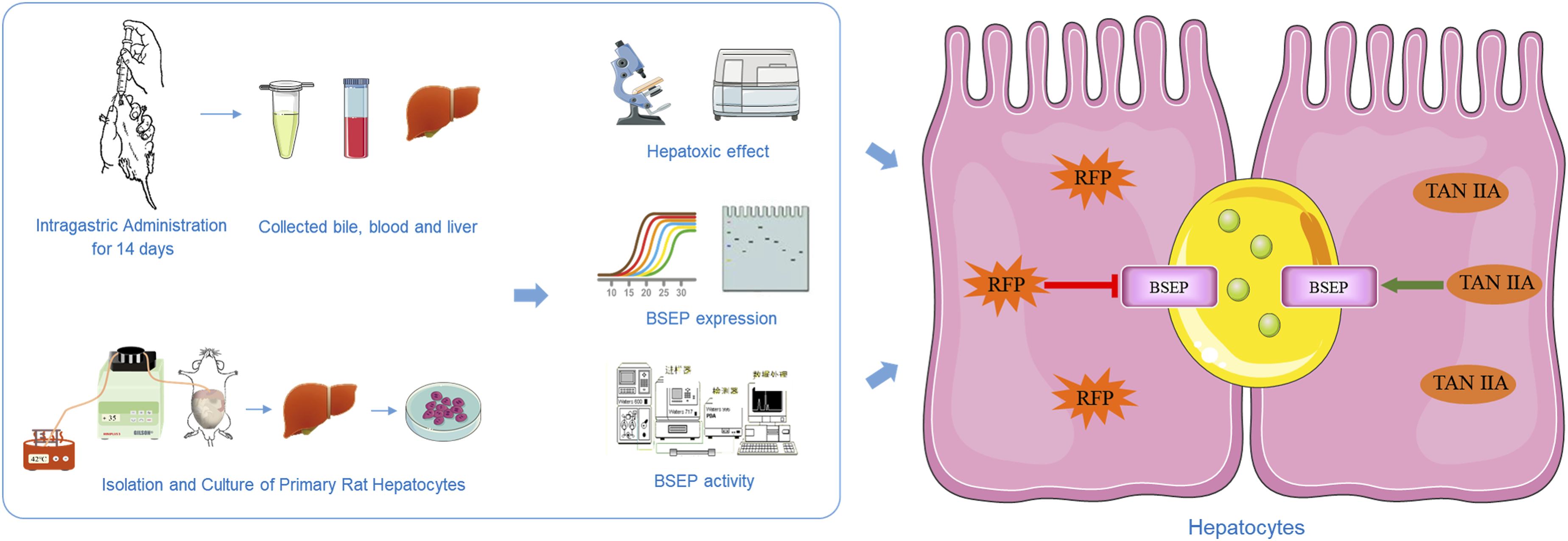
Introduction
Rifampicin (RFP) is one of the first-line drugs for tuberculosis that is clinically administered for 6 months. 1 Due to the long medication cycle, cholestasis liver injury frequently occurs, eventually resulting in cirrhosis and hepatic failure without appropriate treatment. 2 Food and Drug Administration (FDA) suggests that liver function must be monitored and evaluated before therapy and continuously every 2 to 4 weeks during RFP therapy. If signs of liver injury occur or worsen, discontinue RFP is recommended. 3 Therefore, once cholestasis occurred during RFP treatment, it would finally limit the efficacy of anti-tuberculosis regimen. However, there are still no medications available to prevent cholestasis caused by RFP. It is urgent to find a effective drug that may interfere with the cholestasis caused by the RFP.
Hepatic transporters play critical roles in the vectorial transport of bile acids. Bile acids in the hepatocytes are excreted into bile canaliculi predominantly via bile salt export pump (BSEP). 4 A large number of evidences suggest that many drugs can cause cholestasis by inhibiting BSEP. 5 Therefore, BSEP is an important target for treating cholestasis.
Tanshinone IIA (TAN IIA) is one of the main lipophilic active constituents isolated from Salvia Miltiorrhiza Bunge. 6 Previous studies have demonstrated that TAN IIA had hepatoprotective properties. 7 Sodium TAN IIA sulfonate, a derivative of TAN IIA, has been reported to regulate inflammatory cytokine secretion and protect the liver in rabbits. 8 Moreover, TAN IIA has been suggested to suppress cholesterol accumulation in human macrophages. 9 In addition, there is evidence that TAN IIA has potential for treating hepatic fibrosis. 10 However, a comprehensive understanding of RFP-induced liver injury and TAN IIA liver protection relative to BSEP expression and activity is limited. Based on previous research results, we raised a hypothesis that TAN IIA may relieve cholestasis by regulating BSEP, thereby protecting RFP-mediated liver injury.
In this study, we successfully developed RFP-induced rat liver injury model in vivo and the sandwich-cultured rat hepatocytes (SCRH) in vitro. Furthermore, the effects of RFP and TAN IIA on BSEP expression and BSEP transporter activity were characterized to explore the mechanism of RFP-induced liver injury and the hepatoprotective effect of TAN IIA.
Materials and methods
Materials
RFP (No. S01023YA14, purity 99%) and TAN IIA (No. Y20J8C49264, purity 90%) were obtained from Yuanye Bio-Technology Co, Ltd. (Shanghai, China). Sodium taurocholate and glibenclamide (No. A0225A, purity 99%) were purchased from Meilune (Dalian, China). Collagenase (type II) was obtained from Gibco (Grand Island, NY). 5(6)-carboxy-2′,7′-dichlorofluorescein diacetate (CFDA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Hank’s balanced salt solution (HBSS, with Ca2+ and Mg2+) and D-Hank’s balanced salt solution (HBSS, without Ca2+ and Mg2+) were provided by Solarbio (Beijing, China). All other chemicals and reagents were of analytical grade and were readily available from commercial sources.
Animals and treatment
Male SD rats (220 ± 20 g) were obtained from Da Shuo (Sichuan, China) and were fed at room temperature 25°C, 12 h-12 h light-dark environment with appropriate humidity and allowed food and water ad libitum for at least 1 week before treatment.
In vivo experiments, TAN IIA (10, 20 and 30 mg/kg, i.g.) and RFP (100, 150, 200 mg/kg, i.g.) were dissolved in 0.5% sodium carboxymethyl cellulose (CMC-Na). Briefly, 42 rats were randomly divided into seven groups, which were blank group, low (100 mg/kg), medium (150 mg/kg) and high (200 mg/kg) dose groups of RFP, as well as RFP (200 mg/kg) combined with low (10 mg/kg), medium (20 mg/kg) and high (30 mg/kg) dose groups of TAN IIA, respectively. Then they were administered intragastrically for 14 consecutive days. Remarkably, the rats were pretreated with TAN IIA prior to the administration of RFP in combination regimen, and the time interval of two drugs was 4 h. On day 15, rats were anesthetized with isoflurane and bile was collected from 0-10 min, 10–20 min, 20–30 min, 30–40 min, 40–50 min, and 50–60 min, respectively. Serum and liver tissues were harvested. Liver tissue fixed in 10% neutral buffered formaldehyde solution for evaluation of histological damage. The rest of liver was cryopreserved at −80°C. The experimental procedures were implemented in accordance with the Guide for the Care and Use of Laboratory Animals, and approved by the Animal Care of the West China School of Pharmacy, Sichuan University (Approval number: KS2020405).
Isolation and culture of primary rat hepatocytes
Sandwich-cultured hepatocytes (SCHs) are produced by cultivating primary hepatocytes between two layers of gelled collagen. This technique has a favorable influence on the development of canalicular networks and the maintenance of the hepatic transport protein expression and function. 11 Therefore, SCHs are widely utilized to evaluate hepatic drug disposition and drug-induced hepatotoxicity in vitro. 12 In this study, we systematically characterized the effects of RFP and TAN IIA on the expression and function of Bsep by sandwich-cultured rat hepatocytes (SCRH) in vitro.
Briefly, rat hepatocytes were freshly isolated by the collagenase perfusion technique as Seglen described, 13 with modifications. The protocol was shown in our previous experimental method. 14 Morphology of cultured hepatocytes over time were presented in Supplementary Materials.
Estimate of SCRH
The biliary efflux study was conducted only when the bile canaliculi had been formed by the rat hepatocytes. The formation of extensive canalicular networks and the recovery of cell polarity were presented after hepatocytes were cultivated in SCRH for 3–5 days according to previous research.15,16 The bile compartments were analysed with 5(6)-carboxy-2′,7′-dichlorofluorescein (CDF), a fluorogenic reagent that could be excreted into bile canaliculi if the networks were complete.
After rat primary hepatocytes were incubated for 4 days in SCRH, the plates were treated with Hanks or D-Hanks for 15 min. Then, 10 μM 5(6)-carboxy-2′,7′-dichlorofluorescein diacetate (CFDA), which could be hydrolysed to CDF, was added to the wells for 15 min. A fluorescence microscope was used to observe the cells and obtain images. The bile canaliculi were indicated by tubular CDF fluorescence. And the fluorescence images were shown in Supplementary Materials.
MTT assay
The cell viability was determined using the MTT assay. Hepatocytes were incubated until the bile canalicular networks developed, then they were treated with different concentrations of RFP or TAN IIA combined with RFP (50 μM), glibenclamide in a 96-well plate. The medium was carefully removed and replaced with MTT solution. After 4 h, we used DMSO to solubilize the formazan that was generated during incubation. The optical density (OD) was measured at 492 nm, the results were demonstrated in Supplementary Material.
RNA isolation and quantitative real-time PCR
Primer sequences for quantitative real-time PCR analysis.

Western blotting
Following homogenized in RIPA Lysis Buffer (Beyotime, Shanghai, China), the protein concentrations were determined using a BCA protein assay kit (Wanleibio, Shenyang, China). Then, the protein samples were separated by 8% SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and electro-transferred to PVDF membranes. Subsequently, the membranes were blocked with 5% non-fat milk powder in TBST and incubated with primary antibodies (anti-BSEP, Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA; anti-GAPDH, Wanleibio) overnight at 4°C. After incubated with a secondary antibody (goat anti-rabbit IgG-HRP, ZenBio, Chengdu, China) for 1 h at 37°C, the intensity of bands was quantified by ImageJ software.
Biliary efflux study in SCRH
After the bile canalicular networks were formed, the cells were used for a biliary efflux study. In brief, plated cells were treated with RFP or co-administration of TAN ⅡA for 12 h, 24 h and 48 h, respectively. Then, the cells were washed with Hanks or D-Hanks three times and allowed to equilibrate for 15 min at 37°C with the same Hanks. Subsequently, dosing solutions containing 1 μM taurocholic acid (TCA) were added at 37°C. 15 min later, the cells were washed three times with cold Hanks to terminate the biliary efflux of TCA. The cell samples were freeze-thawed three times. Then, the cell lysates were stored at −80°C.
HPLC-MS/MS analysis
TCA analysis was performed with a HPLC-MS/MS system. The method for TCA concentration determination was established and validated. Losartan was utilized as an internal standard. The analyte was separated on a C18 column (50×2.00 mm, 3 μm). The mobile phase was 5 mM ammonium acetate-dd H2O containing 0.1% formic acid (mobile phase A) and acetonitrile (mobile phase B) with a flow rate of 0.4 mL/min. The elution gradient conditions of primary hepatocyte samples were as follows: 0–0.5 min, 5% B; 0.5–1.5 min, 5%-60% B; 1.5–2.5 min, 60% B; 2.5–3 min, 5% B and 3–5 min re-equilibration. While the bile samples were 0–0.5 min, 5% B; 0.5–1.5 min, 5%-45% B; 1.5–2.5 min, 45% B and 2.5–3 min, 45% B and 3–5 min re-equilibration. MRM was used to quantify the concentrations of TCA in the negative ion mode using the mass parameters listed in the Supplementary Data.
After thawing at room temperature, 100 μL of cell lysates or bile samples were mixed with 10 μL of losartan (internal standard, 4500 ng/mL in cell lysates or 100 μg/mL in bile samples) and were vortexed for 1 min. Subsequently, 300 μL of acetonitrile was added to deplete the protein. After centrifugation at 12,000 rpm for 10 min, 80 μL of the supernatant was transferred into auto-sampler vials.
Data analysis
The biliary excretion index (BEI; %) was calculated using Excel 2016 and GraphPad Prism according to the following equations
Results
TAN ⅡA protected against RFP-induced liver injury in vivo
Regarding the histological structure, hepatocytes were arranged in a single line centering on the central vein in the blank control group (Figure 1(a)). The severity of liver damage induced by RFP was dose-dependent and reached the peak at high-dose RFP (200 mg/kg) group indicated by the larger areas of hepatocyte necrosis, feather-like steatosis and the higher levels of serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total bilirubin (TBIL) and total bile acid (TBA) in serum were determined (Figure 1(c)). According to the classification criteria of drug-induced liver injury,
17
the liver injury caused by the high-dose RFP (200 mg/kg) was cholestasis. Rifampicin induced Cholestatic liver injury. Rats were treated with different concentrations of RFP (100 mg/kg, 150 mg/kg or 200 mg/kg) for 14 days. The histologic, liver weight ratio and serum biochemistry were evaluated. (a) H&E staining in rats undergoing different treatments. Scale bar = 50 μm. Areas of featherlike necrosis, fatty degeneration and inflammatory infiltrates are marked by arrows, star and triangle respectively. (b) Liver weight ratio and (c) Serum ALT, AST, ALP, and TBIL, as well as serum TBA in rats. Data are expressed as mean±SD (n = 6 replicates/treatment). *p < 0.05, **p < 0.01, ***p < 0.005, ****p < 0.001, against Blank. ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; TBIL: total bilirubin; TBA: total BAs; RFP: rifampicin.
Thus, the rats were treated with 200 mg/kg RFP combined with different doses of TAN ⅡA for 2 weeks to investigate whether TAN ⅡA had a protective effect on RFP-induced liver injury in further research. As Figure 2 shown, TAN ⅡA attenuated RFP-induced hepatocellular injury compared with the RFP group, including hepatocyte necrosis, inflammatory cell infiltration and steatosis. Furthermore, all liver function indexes were also decreased in the co-administration group, and the downward trend was the most obvious in the high-dose TAN ⅡA group, with statistical significance (p < 0.05). It was suggested that TAN ⅡA had a protective effect on RFP-induced liver injury. TAN IIA prevented cholestatic liver injury caused by RFP. Rats were co-treated with different concentrations of TAN IIA (10 mg/kg, 20 mg/kg, or 30 mg/kg) and RFP (200 mg/kg) for 2 weeks. The histologic, liver weight ratio and serum biochemistry were evaluated. (a) H&E staining in rats undergoing different treatments. Scale bar = 50 μm. Areas of feather-like necrosis, fatty degeneration and inflammatory infiltrates are marked by arrows, star and triangle respectively. (b) Liver weight ratio and (c) Serum ALT, AST, ALP, and TBIL, as well as serum TBA in rats. Data are expressed as mean±SD (n = 6 replicates/treatment). *p < 0.05, **p < 0.01, ***p < 0.005, ****p < 0.001, against Blank; #p < 0.05, ##p < 0.01, ###p < 0.005, ####p < 0.001, against 200 mg/kg RFP. ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; TBIL: total bilirubin; TBA: total BAs; RFP: rifampicin; TAN IIA: tanshinone IIA.
TAN IIA reversed RFP-induced BSEP expressive inhibition
BSEP is a crucial efflux transporter responsible for transferring substances from hepatocytes to the bile canaliculi. 18 To investigate if RFP and TAN IIA regulated liver function by BSEP, we systematically demonstrated the effects of RFP or RFP combined with TAN IIA on BSEP expression both in vivo and in vitro.
In vivo, rats were treated with RFP or co-treated with TAN IIA for 14 consecutive days. Then liver tissues were harvested and BSEP expression were determined by qRT-PCR and western blotting, respectively. As shown in Figure 3, RFP decreased the mRNA and protein expression of BSEP relative to the blank control group, and 200 mg/kg RFP showed the strongest decrease trend (p < 0.01). While TAN IIA combined with RFP reversed the inhibition of RFP on BSEP expression. Compared with RFP group, co-administration of TAN IIA (30 mg/kg) increased RFP-induced down-regulation of the mRNA expression by 3.3-fold, the protein expression by 3.1-fold (p < 0.05). TAN IIA reversed the inhibition of BSEP expression by RFP in vivo. Rats were treated with different concentrations of RFP (100 mg/kg, 150 mg/kg or 200 mg/kg) or co-treated with different concentrations of TAN IIA (10 mg/kg, 20 mg/kg, or 30 mg/kg) and RFP (200 mg/kg) for 2 weeks. The relative mRNA expression levels were determined by qRT-PCR (a, b). The protein expression of BSEP in SCRH was determined by western blotting (c, d). Gapdh was selected as the reference for both mRNA and protein expression. The data were presented as the means ± SD (n = 6). *p < 0.05, **p < 0.01, ***p < 0.005 against the control group. #p < 0.05, ####p < 0.001 vs the RFP-200 group. RFP: rifampicin; TAN IIA: tanshinone IIA; BSEP: bile salt export pump.
Furthermore, similar results were presented in vitro experiments. As shown in Figure 4, RFP decreased the mRNA and protein level of BSEP in a concentration-dependent and time-dependent manner. 50 μM RFP significantly reduced BSEP mRNA level by 64% and BSEP protein level by 31% after 48 h (p < 0.005). Instead, the combination groups showed the significant increases in the BSEP expression levels. As the concentration of TAN IIA increased, the upregulation was more obvious (Figure 5). Rifampicin inhibited the mRNA and protein expression of BSEP in SCRH. Hepatocytes were incubated with different concentrations of RFP (10 μM, 25 μM, or 50 μM) for 12 h, 24 h, 48 h. The relative mRNA expression levels were determined by qRT-PCR (a–c). The protein expression of BSEP in SCRH was determined by western blotting (d–f). Gapdh was selected as the reference for both mRNA and protein expression. The data were presented as the means ± SD (n = 3). *p < 0.05, **p < 0.01, ***p < 0.005 vs the control group. RFP: rifampicin; TAN IIA: tanshinone IIA; BSEP: bile salt export pump; SCRH: sandwich-cultured rat hepatocytes. TAN IIA reversed the inhibition of BSEP expression by RFP in SCRH. Hepatocytes were co-treated with different concentrations of TAN IIA (5 μM, 10 μM, or 20 μM) and RFP (50 μM) for 12 h, 24 h, 48 h. The relative mRNA expression levels were determined by qRT-PCR (a–c). The protein expression of BSEP in SCRH was determined by western blotting (d–f). Gapdh was selected as the reference for both mRNA and protein expression. The data were presented as the means ± SD (n = 3). *p < 0.05, **p < 0.01, ***p < 0.005, ***p < 0.001 vs the control group. #p < 0.05, ##p < 0.01, ###p < 0.005, ####p < 0.001 vs the RFP 50 group. RFP: rifampicin; TAN IIA: tanshinone IIA; BSEP: bile salt export pump; SCRH: sandwich-cultured rat hepatocytes.

In summary, TAN IIA reversed the inhibition of the mRNA and protein expression of BSEP caused by RFP.
TAN IIA alleviated RFP-induced BSEP dysfunction
TCA is one of the substrates for BSEP.
19
It can be excreted into bile ducts. In this study, we used TCA to evaluate the effects of RFP and TAN IIA on BSEP function. Glibenclamide, a selective inhibitor of BSEP,
20
was chosen as the positive control. As demonstrated in Figure 6, the results of in vivo experiments suggested that treatment of 200 mg/kg RFP significantly decreased the content of TCA in bile duct to 50.2% over the blank control group (p < 0.01). While co-administration of TAN IIA mitigated the reduction of TCA in bile caused by RFP. Specifically, 30 mg/kg TAN IIA caused the steepest upward trend, which increased the accumulation of TCA in bile by 1.8-fold than 200 mg/kg RFP group (p < 0.005). TAN IIA reversed inhibition of BSEP function by RFP in vivo. Rats was treated with different concentrations of RFP (100 mg/kg, 150 mg/kg or 200 mg/kg) or co-treated with different concentrations of TAN IIA (10 mg/kg, 20 mg/kg, or 30 mg/kg) and RFP (200 mg/kg) for 14 days. Subsequently, the bile was collected from 0-10 min, 10–20 min, 20–30 min, 30–40 min, 40–50 min, and 50–60 min, respectively. And the accumulation of TCA in bile was determined by HPLC-MS/MS. The effects of RFP on the accumulation of TCA were shown in (a) and the effects of TAN IIA and RFP were presented in (b). The data were presented as the means ± SD (n = 6). **p < 0.01 against the control group. ###p < 0.005 vs the RFP-200 group. RFP: rifampicin; TAN IIA: tanshinone IIA; BSEP: bile salt export pump.
Then, the effects of RFP and TAN IIA on BSEP function were further investigated by SCRH. As illustrated in Figure 7, both the accumulation of TCA in the bile canaliculi and the BEI of TCA decreased in a concentration-dependent manner after treatment with RFP. 50 μM RFP reduced TCA accumulation in bile canaliculi to 6.9% and decreased the BEI of TCA to 14.3% over the blank control group at 48 h (p < 0.005). Moreover, TCA accumulation in bile canaliculi and TCA BEI were gradually increased by co-treatment with TAN IIA. Significantly, co-administration of 30 μM TAN IIA increased TCA accumulation in bile canaliculi by 13.5-fold and elevated the BEI of TCA by 7.8-fold over the 50 μM RFP group at 48 h (p < 0.05). TAN IIA reversed inhibition of BSEP function by RFP in SCRH. The accumulation of TCA in bile canaliculi was determined after treatment of RFP and TAN IIA in SCRH. Different concentrations of RFP (10 μM, 25 μM, or 50 μM) or different concentrations of TAN IIA (5 μM, 10 μM, or 20 μM) and RFP (50 μM) were incubated with cells for 12 h, 24 h, 48 h. Then, 1 μM TCA was administered. Subsequently, the accumulation of TCA was determined by HPLC-MS/MS. The accumulation in bile canaliculi are represent in (a–c). The BEI of TCA were shown in (d–f). The data were expressed as the mean ± SD (n = 3). *p < 0.05, **p < 0.01, ***p < 0.005, ****p < 0.001 vs the control group; #p < 0.05, ##p < 0.01, ###p < 0.005, ####p < 0.001 vs the RFP 50 group. RFP: rifampicin; TAN IIA: tanshinone IIA; TCA: taurocholic acid; BSEP: bile salt export pump; SCRH: sandwich-cultured rat hepatocytes.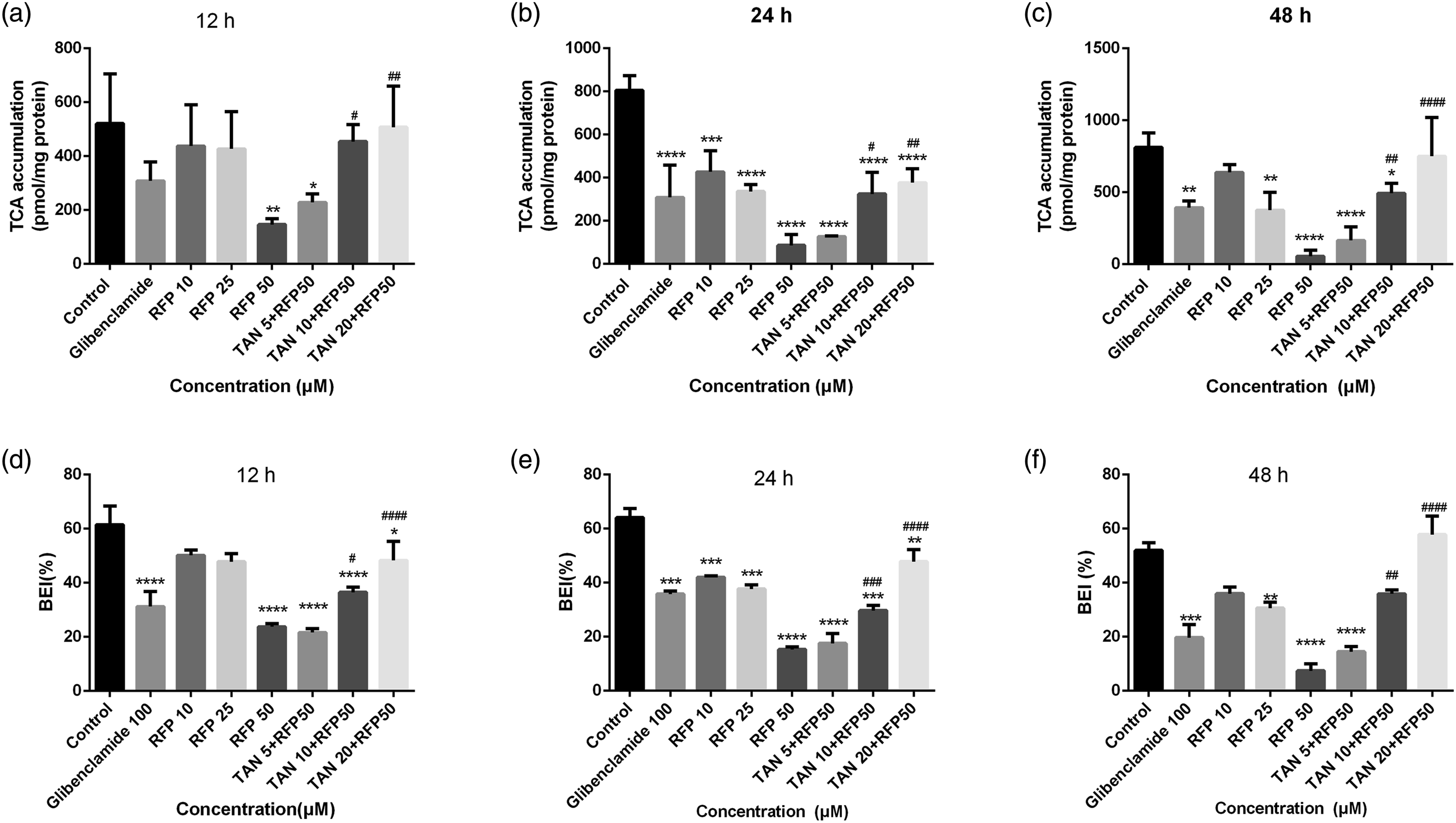
In short, these results indicated that RFP could inhibit BSEP function while TAN IIA could facilitate TCA from hepatocytes to the bile duct.
Discussion
Drug-induced liver injury (DILI) is not only an important cause of liver transplantation due to acute liver failure but also a leading reason for withdrawal of approved drugs. 21 Based on the R-value, which is defined as serum alanine aminotransferase/upper limit of normal (ULN) to serum alkaline phosphatase/ULN, DILI was classified as hepatocellular DILI (R≥5), cholestatic DILI (R<2) or “mixed” DILI (2<R<5). 17 A number of studies show that RFP can cause cholestatic DILI, 22 which is consistent with the results of this study. Hepatic bile acid levels are regulated through basolateral uptake and efflux and canalicular output, during which many transporters are involved to mediate the bile acid transfer, such as OATP, NTCP and MRP2. Meanwhile, RFP could inhibit OATP1B-mediated uptake. 23 And RFP has inhibition effects on NTCP, which we have found before. 14 Similarly, RFP inhibits MRP2 function and reduces the excretion of bilirubin glucuronic acid and bile acids. 24 Besides, BSEP is the vital transporter to mediate the excretion of monovalent bile acids into the canaliculi. 25 BSEP gene mutation induces a variety of types of cholestasis, such as benign recurrent intrahepatic cholestasis type 2 (BRIC2) and progressive familial intrahepatic cholestasis type 2 (PFIC2).26,27 Therefore, to further clarify the mechanism of RFP induced cholestatic DILI, we would comprehensively explore the effects of RFP on both BSEP expression and its transport activity in vitro and in vivo. The results showed that RFP not only inhibited the expression of BSEP mRNA and protein levels but also the function of BSEP. It might inhibit bile acids efflux mediated by BSEP and disrupt the steady-state of bile acids circulation. The above findings indicate that RFP-induced cholestasis might be related to the inhibition of BSEP. Thus, our work provides a clue that Bsep could be a priming target to reduce RFP cholestasis.
Clinically, there is no effective treatment for RFP-induced cholestasis. The general therapy is to use hepatoprotective drugs or strengthen supportive therapy to accelerate drug excretion for liver self-recovery gradually. 28 Currently, there are many kinds of hepatoprotective drugs, which can be divided into anti-inflammation, detoxification, promoting liver cell recovery etc. 29 However, there are no potent drugs to cholestasis. As a treasure of Chinese culture, traditional medicine has a long history of thousand years and provides abundant bioactive compounds. In this paper, we tried to screen a traditional medicine that could prevent RFP-induced cholestasis. Based on our knowledge, although TAN IIA has mainly been used for treating cardiovascular diseases, it also has hepatoprotective properties. A previous report suggested that TAN IIA pre-treatment could protect the liver from APAP-induced hepatic injury by activating the nuclear factor erythroid 2-related factor 2 (Nrf2) pathway. 30 TAN IIA exerts protective effects in LCA-induced cholestasis by upregulating pregnane X receptor (PXR). 31 In addition, our previous study showed that TAN IIA increased BSEP expression via epigenetic activation of NRF2. 32 Therefore, we suspected that TAN IIA could prevent RFP-induced liver injury by regulating BSEP. In this study, we found that co-administration of TAN IIA and RFP could increase the mRNA expression, protein expression and function of BSEP compared with RFP alone. Our data suggested that TAN IIA could reverse the inhibitory effect of RFP on BSEP, which had a positive effect on restoring the steady-state of bile acid circulation. Therefore, TAN IIA had the potential to prevent RFP-induced cholestasis by regulating BSEP expression and function.
In conclusion, our study reveals that RFP-induced cholestasis is associated with decrease of BSEP expression and function. TAN IIA has the potential to prevent RFP cholestasis by increasing BSEP expression and function. Essentially, this work will contribute to a better understanding of RFP-induced cholestatic liver injury and enrich the significance of BSEP in the prevention and treatment of bile acid-related liver injury. In addition, this work also provides the theoretical basis for TAN IIA in preventing cholestasis caused by RFP.
Supplemental Material
Supplemental Material - Tanshinone IIA alleviate rifampicin-induced cholestasis by regulating the expression and function of bile salt export pump
Supplemental Material for Tanshinone IIA alleviate rifampicin-induced cholestasis by regulating the expression and function of bile salt export pump by L Liu, Y Yang, W Li, Y Li, X Jiang and L Wang in Human & Experimental Toxicology
Footnotes
Acknowledgements
The authors are thankful to the subjects who were involved in this study, the research staff.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Natural Science Foundation of China (No. 8157141040).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
