Abstract
The Na+-taurocholate cotransporting polypeptide (NTCP) acts as the major hepatic basolateral uptake system, and plays a key role in balancing bile flow. The anti-tuberculosis drugs rifampin (RFP) can affect bile flow causing liver injury, while tanshinone IIA (TAN IIA) has the effect of protecting liver. This study aimed to investigate the effects of RFP and TAN IIA on the NTCP expression and activity, and explore the potential connections. Herein, we established sandwich-cultured primary rat hepatocytes, and quantified mRNA and protein levels of NRF2 and NTCP after treatment with RFP (10, 25, or 50 μM) or co-treatment with TAN IIA (5, 10, or 20 μM) for 12, 24, 48 h (n = 3). NTCP activity was assessed by measuring the initial uptake rates of known substrates taurocholate (TCA) (n = 3) after treatment with different concentrations of RFP, TAN ⅡA for 12, 24 and 48 h. We found that RFP had inhibition effects on NRF2, NTCP mRNA and protein expression, and co-administration of TAN IIA could reverse RFP inhibition. TCA cellular accumulation was significantly decreased by RFP (39.1%), and TAN IIA could significantly induce TCA uptake of NTCP (2.9-fold at 48 h). The TCA uptake activity was correlated with the NTCP mRNA expression, confirming the role of RFP or TAN IIA on NTCP expression and activity is synchronous, and we can predict NTCP activity by detecting its mRNA expression. In conclusion, our work will enrich the significance of NTCP in the liver protection, and provide theoretical basis for TAN IIA to prevent RFP induced cholestatic liver injury.

Introduction
Hepatic drug transporters contribute significantly to bile acid transport and enterohepatic circulation. Most bile acids are reabsorbed back to the liver through hepatic basolateral uptake systems at the end of enterohepatic circulation, which can prevent excessive bile acids retaining in the circulation system and damaging other tissues. 1 NTCP acts as the major hepatic basolateral uptake system, and plays a key role in reabsorbing conjugated bile acids from the plasma into the hepatocyte. 2 When bile flow is impeded in enterohepatic circulation, toxic bile acids accumulate in the liver or penetrate into the blood, leading to liver injury. 3 Drugs can affect bile flow and bile acid reabsorption, 4 causing drug-induced liver injury (DILI).
In recent years, anti-tuberculosis drug caused liver injury has ranked in first place in DILI. A large number of studies have reported that long-term use of RFP can cause abnormal liver function, including a significant increase in serum bile acids and bilirubin levels, 5 and its pathogenesis is related to enterohepatic circulation disorder. 6 Inhibiting NTCP can lead to increased bile acid levels in plasma. 7 We speculated that the enterohepatic circulation disorder caused by RFP was related to NTCP transport inhibition. 8 Salvia miltiorrhiza Bunge (Dan shen in Chinese) is one of the most prestigious traditional Chinese medicines, and its main fat-soluble component, TAN IIA, has the effect of protecting liver and promoting bile flow. 9 Classical nuclear factor, farnesoid X receptor (FXR) and nuclear factor erythroid 2-related factor 2 (NRF2) play an important role in regulating bile acids efflux and uptake. 10,11 Our previous studies have shown that TAN IIA could induce the expression of NTCP by regulating NRF2. 11 However, the changes in NTCP functional activity after treatment of RFP or TAN IIA were not examined, and the relationship between NTCP expression and activity under this state is still unknown.
A variety of in vitro cell models have been used to study drug transporters expression and activities, 12 such as primary hepatocytes and individual or multiple transporters overexpressing cells. Primary hepatocytes cultured in a 2D sandwich systems have been shown to promote hepatocyte polarization for extended culture periods and to maintain their function for better transporters and metabolizing enzymes activities than in monolayer cultures. 13 Sandwich-cultured hepatocytes are extensively used to investigate the hepatic metabolism and transport, as well as drug-drug interactions and hepatotoxicity based on drug-metabolizing enzymes and transporters. 14 Although the expression and activities of bile acid transporters has been investigated in primary cultured hepatocytes to some extent, 15 a comprehensive understanding of RFP-induced liver injury and TAN IIA liver protection relative to NTCP expression and activity is limited. Thus, we investigated that the effects of RFP and TAN IIA on NRF2, NTCP expression and NTCP transporter activity in rat primary hepatocytes, as well as the relationship between NTCP expression and activity. To clarify the effects of RFP and TAN IIA on NTCP not only will enrich the significance of NTCP changes to bile acid-related liver injury transformation, but it will also show the potential of TAN IIA to prevent the RFP-induced liver injury.
Materials and methods
Chemicals and reagents
Rifampin (No. S01023YA14, purity 99%) was purchased from Yuanye Biotechnology (Shanghai, China). TAN IIA (purity 98%) were purchased from SiHuan Biotechnology (Xi’an, China). Anti-NRF2 monoclonal antibody (catalog no. 62352) and anti-NTCP polyclonal antibody (catalog no. YT5372) were purchased from Abcam (Cambridge, UK) and ImmunoWay (TX, USA), respectively. Goat anti-rabbit IgG (H + L) secondary antibody was purchased from Thermo Fisher Scientific (MA, USA). Taurocholic acid (TCA) (specific activity, 4.6 Ci/mmol) was purchased from PerkinElmer Life Sciences (Boston, MA). Cyclosporine A (CsA) was purchased from PerkinElmer Life Sciences (Boston, MA). Dulbecco’s modified eagle medium (DMEM), Collagenase (type Ⅱ) and ITS-X (Insulin-Transferrin-Selenium-X Supplement 100X) were purchased from GIBCO (Grand Island, NY). Matrigel Basement Membrane Matrix and Collagen Ⅰ were purchased from Corning (Bedford, MA). Insulin from bovine pancreas, L-glutamine (100X), dexamethasone, sodium taurocholate, gentamicin sulfate and gliebenclamide were purchased from Meilune (Dalian, China).
Animals
Male Sprague Dawley (SD) rats (250 ± 20 g) were purchased from Da Shuo (Sichuan, China). Rats were housed in a 12 h light/dark cycle and allowed food and water ad libitum for at least 1 week before hepatocytes isolation. The experimental procedures were implemented in accordance with the Guide for the Care and Use of Laboratory Animals, and approved by the of Animal Care of the West China School of Pharmacy, Sichuan University (Chengdu, China).
Primary rat hepatocytes isolation and culture
Primary Rat Hepatocytes were isolated using a two-step collagenase perfusion, as described previously. 16 After isolation, hepatocytes were counted using a hemocytometer, and cell viability was determined using trypan blue. Hepatocytes, in DMEM medium supplemented with 0.1 μM dexamethasone, 0.1 mg/mL gentamicin, 4 μg/mL insulin, 100 U/mL penicillin G, 100 μg/mL streptomycin, 1% MEM nonessential amino acids, 1% μ-glutamine and 5% bovine calf serum, were seeded onto collagen I coated plates with a density of 5 × 105 cell/mL, and cultured at 37°C in a humidified atmosphere containing 5% CO2. Short-term cultures were assessed 4 h after seeding. Unattached hepatocytes were discarded along with the medium, and hepatocytes were washed with 1× PBS and cultured in serum-free medium supplemented with 0.25 mg/mL Matrigel, 0.1 μM dexamethasone, 0.1 mg/mL gentamicin, 4 μg/mL insulin, 100 U/mL penicillin G, 100 μg/mL streptomycin, 1% MEM nonessential amino acids and 1% μ-glutamine overnight. After an overnight incubation with a Matrigel overlay, hepatocytes were treated with RFP or co-treated with TAN IIA, and the administration doses were based on the previous experimental results (to be published) and existing references. 17
MTT assay
The appropriate experimental concentration range was evaluated using the 1-(4, 5-dimethylthiazol-2-yl)-3, 5-diphenyl-formazan (MTT) assay. After overnight incubation with a Matrigel overlay, the different concentrations of RFP or RFP (50 μM) and TAN IIA were plated in a total volume of 200 µL in 96-well plates, respectively. Following 48 h incubation, 100 µL of the MTT stock solution (0.5 mg/mL) was added to each well. After 4 h of incubation, the absorbance was measured by Multiskan MK3 Reader (Thermo, USA) at 490 nm in 150 µL dimethylsulfoxide (DMSO).
RNA isolation and quantitative real-time PCR
Total RNA was performed using TRIzol reagent (TaKaRa, Japan) according to the Manufacturer’s instructions, and then reverse transcribed to cDNA using PrimeScript™ RT reagent Kit with gDNA Eraser (Perfect Real Time) (TaKaRa, Japan). Quantitative real time PCR was performed with Hieff™ qPCR SYBR®Green Master Mix (Yeasen Biological Technology Co., Ltd. Shanghai, China) using specific primers listed in Table 1. The relative gene expression levels were normalized against the expression of GAPDH.
Primer sequences for quantitative real-time PCR analysis.

Western blot analysis
Following homogenized in RIPA Lysis Buffer, the protein content was determined by a BCATM protein assay Kit (Biyuntian Co. Ltd, China). Total cellular protein was resolved by 10% SDS-PAGE and transferred onto a polyvinylidene difluoride membrane. The membrane was blocked with 5% non-fat milk and was incubated with primary antibody overnight at 4°C, followed by incubation with secondary antibody for 1 h. All antibodies were used at dilutions recommended by the manufacturers. The densities of protein bands were determined by ImageJ software.
Cell uptake experiment
Activity of NTCP transporter was determined by cellular accumulation of TCA, as described previously. 18 In brief, after treatment of 10 μM CsA or different concentrations of RFP, TAN ⅡA for 12, 24 and 48 h, plated cells were washed with standard Hanks’ balanced salt solution and allowed to equilibrate for 15 min at 37°C. CsA, as a known inhibitor of NTCP, was used as a positive control. Subsequently, plated cells were treated with standard HBSS containing 1 μM TCA for 15 min at 37°. The standard buffers were discarded and the cells were washed with the cold HBSS and were collected to determinate the cellular accumulation of TCA. The cell samples were freeze-thawing three times to lyse cells. The protein content of the hepatocytes in each well was measured with the BCA method. The cell lysates were stored at −70° until quantified by HPLC-MS/MS analysis.
HPLC-MS/MS analysis
A high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) system, consisted of an API3000 mass spectrometer (AB scienx, USA) coupled with LC10A HPLC system (Shimadzu, Japan), has been developed and validated for quantification of intracellular TCA. 18 Losartan was utilized as an internal standard substance. Quantification was achieved using multiple reaction monitoring in positive mode, and chromatography was performed on C18 column (50 × 2.00 mm 3 micron) using mobile phase A (5 mM ammonium acetate solution, containing 0.1% formic acid) and B (acetonitrile, containing 0.1% formic acid) at a flow rate of 0.4 mL/min. The binary gradient conditions were as follows: 0–0.5 min, 5% B; 0.5–1.5 min, 5%–60% B; 1.5–2.5 min, 60% B; 2.5–3 min, returned to 5% B; 3–5 min, 5% B.
Before analysis, the cell lysates were thawed at room temperature. An aliquot of 100 µL cell lysates were mixed with 10 µL of acetonitrile (or standard or QC solution) and 10 µL of losartan solutions (4.5 μg/mL). After vortex-mixing for 30 sec, 200 µL of acetonitrile were added to deplete protein. After centrifuged at 12,000 rpm for 10 min, the supernatant (20 μL) was injected into the HPLC system for analysis.
Statistical analysis
Statistical analyses were performed using GraphPad Prism version 6.02. For analysis of differences between the groups, an unpaired two-tailed Student’s t test was used. If the data could not meet homogeneity of variance, the Welch’s correction was used to correct the unpaired t test. Differences were considered significant at the p < 0.05 level. Spearman rank correlation analysis was also performed in GraphPad Prism version 6.02 to measure the strength of association between TCA uptake activity and NTCP expression.
Results
TAN IIA reverses inhibition of NRF2 and NTCP mRNA levels by RFP in primary hepatocytes
Firstly, we measured the gene expression of nuclear receptor NRF2 and BA transporter NTCP in primary hepatocytes using qRT-PCR. As demonstrated in Figure 1(a) to (c), RFP decreased NRF2 and NTCP mRNA in a dose-dependent form but there was no statistical difference at 12 and 24 h. Whereas, NRF2 mRNA was significantly reduced at 48 h by RFP, which decreased the mRNA level of NRF2 and NTCP by approximately 52.6% and 41.4%, respectively. Co-treatment of TAN IIA had a strong induction effect on NRF2 and NTCP mRNA expression. NRF2 and NTCP mRNA expression were significantly increased by TAN IIA-high dose group at 12 and 24 h (5.1- and 3.1-fold at 12 h, 4.9- and 5.4-fold at 24 h over the 50 μM RFP group). However, with the increase of treatment time, the induction of NRF2 and NTCP mRNA expression was stronger in TAN IIA-medium dose at 48 h (9.0- and 7.0-fold over the 50 μM RFP group). In general, co-treatment of TAN IIA could continuously up-regulate the NRF2 and NTCP mRNA levels, and reversed inhibition by RFP in primary hepatocytes.
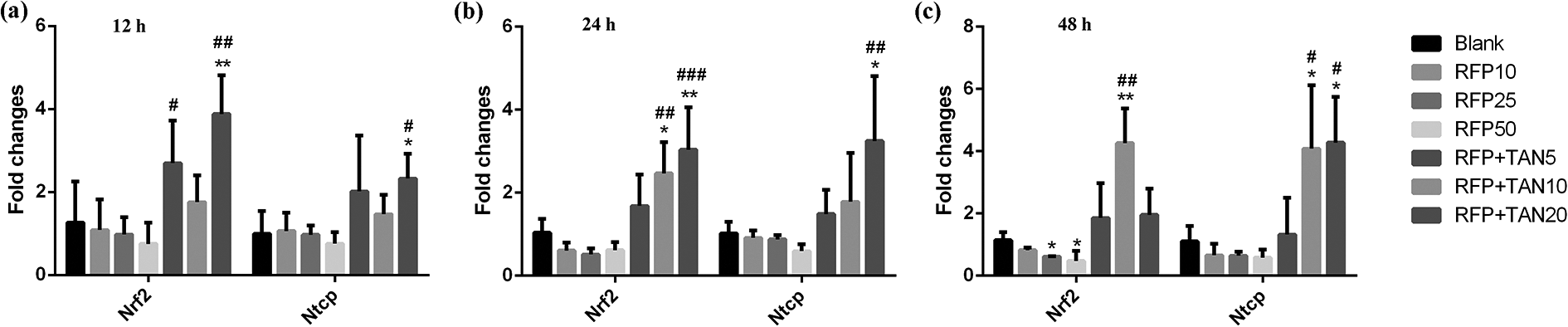
TAN IIA reverses inhibition of NRF2 and NTCP mRNA levels by RFP. qRT-PCR analysis of NRF2 and NTCP in sandwich-cultured rat hepatocytes after treatment with 50 μM RFP or co-treatment with 5, 10, or 20 μM TAN IIA for 12 h (a), 24 h (b) or 48 h (c). NRF2 and NTCP mRNA expression levels were normalized to GAPDH. Data are expressed as the mean ± s.d. (n = 3 replicates/treatment). *P < 0.05, **P < 0.01, ***P < 0.001, against Blank; #P < 0.05, ##P < 0.01, ###P < 0.001, against 50 μM RFP.
TAN IIA reverses inhibition of NRF2 and NTCP protein expression by RFP in primary hepatocytes
To confirm the qRT-PCR results, we further detected their protein levels using western blot analysis. As shown in Figure 2(a) to (c), treatment of RFP had the most significant effect on NRF2 and NTCP protein expression at 12 h, which were reduced by 33.4% and 68.4%, respectively. Meanwhile, co-treatment of TAN IIA-high dose significantly increased NRF2 and NTCP protein expression at 48 h (1.6- and 2.9-fold over the 50 μM RFP group, respectively). In sum, TAN IIA reversed RFP inhibition of NRF2 and NTCP at both the mRNA and protein levels in hepatocytes.

TAN IIA reverses inhibition of NRF2 and NTCP protein expression by RFP. Western blot analysis of NRF2 and NTCP in sandwich-cultured rat hepatocytes after treatment with 50 μM RFP or co-treatment with 5, 10, or 20 μM TAN IIA for 12 h (a), 24 h (b) or 48 h (c). GAPDH was used as a loading control. Controls were set as 1. Data are expressed as the mean ± s.d. (n = 3 replicates/treatment). *P < 0.05, **P < 0.01, ***P < 0.001, against Blank; #P < 0.05, ##P < 0.01, ###P < 0.001, against 50 μM RFP.
TAN IIA reverses inhibition of NTCP function by RFP in primary hepatocytes
TCA is one of the probe substrates of NTCP, which can be ingested into hepatocytes by NTCP. In this study, we used TCA to evaluate the effects of RFP and TAN IIA on NTCP function. CsA, a selective inhibitor of NTCP, was chosen as the positive control for RFP. As described in Figure 3(a) to (c), TCA cellular accumulation was significantly decreased by CsA and RFP. With increasing concentrations of RFP, TCA uptake was gradually reduced. Specifically, 50 μM RFP caused the steepest downward trends, which reduced the accumulation of TCA in hepatocytes by 42.1%, 51.2% and 24.0% at 12, 24 and 48 h, respectively. Meanwhile, 50 μM RFP had a more notable downward trend than CsA at 48 h. Besides, co-administration of TAN IIA-high dose markedly increased TCA accumulation in hepatocytes at 24 and 48 h (1.5- and 2.9-fold over the 50 μM RFP group, respectively), which indicated that the induction ability of TAN IIA increased with the increasing treatment time. In brief, TAN IIA had a reversal effect on RFP inhibition of NTCP function.

TAN IIA reverses inhibition of NTCP function by RFP. TCA cellular accumulation in sandwich-cultured rat hepatocytes after treatment with 50 μM RFP or co-treatment with 5, 10, or 20 μM TAN IIA for 12 h (a), 24 h (b) or 48 h (c). CsA, a selective inhibitor of NTCP, was chosen as the positive control. The protein content of the hepatocytes in each well was used as a loading control. Data are expressed as the mean ± s.d. (n = 3 replicates/treatment). *P < 0.05, **P < 0.01, ***P < 0.001, against Blank; #P < 0.05, ##P < 0.01, ###P < 0.001, against 50 μM RFP.
Correlation between functional activity and expression of NTCP after treatment of RFP or TAN IIA
Since NTCP functional activity showed similar changes with NTCP expression after treatment of RFP or TAN IIA, we used Pearson r correlation coefficients (Significant alpha = 0.05) to measure the strength of association between NTCP activity and expression under this state, and drew the linear regression of data. As showed in Figure 4, NTCP transporter activity showed a good correlation with NTCP mRNA expression (r = 0.8988), but had a poor correlation with NTCP protein expression (r = 0.3475). It confirmed the role of RFP or TAN IIA on NTCP mRNA expression and activity was synchronous, and NTCP functional activity is greatly affected by its expression. This suggested that we may be able to predict NTCP functional activity by detecting NTCP mRNA expression.

Pearson correlation between functional activity and expression of NTCP. Pearson r correlation coefficients was used to measure the strength of association between NTCP activity and mRNA levels (a) or protein expression (b) after treatment of RFP or TAN IIA. TCA uptake and NTCP expression data were normalized to Blank. Data are expressed as linear regression (significant alpha = 0.05).
Discussion
To clarify the mechanism of RFP induced cholestatic liver injury and TAN IIA liver protection, the alterations in factors of NTCP expression and transport activity were examined using qPCR, western blot analysis, and cell uptake experiment. The present study showed RFP decreased the NTCP mRNA level in a dose-dependent form, and nuclear transcription factor NRF2 showed a similar inhibition trend by RFP. Consistent with predictions, co-treatment of TAN IIA had a strong induction effect on NRF2, NTCP mRNA and protein expression. These results are consistent with the mechanism of NRF2 regulating NTCP, which we have found before. 11 With the prolongation of in vitro culture time of primary hepatocytes, the expression of NTCP gradually decreased and reduced more than 10-fold over time (by day 4) relative to day 0. 19 Thus, TAN IIA showed a stronger induction effect on mRNA and protein of NTCP at the late stage of culture (48 h).
We have also determined the functional activity of the hepatic uptake transporter NTCP by determining uptake clearance of probe substrate TCA in suspended rat hepatocytes. The results indicated that TCA cellular accumulation was significantly decreased by CsA and RFP, besides TCA uptake was reduced more with the concentration increases of RFP. It is noteworthy that 50 μM RFP had a more notable downward trend than CsA at 48 h, that is the inhibition ability of RFP was nearly twice that of CSA. Co-administration of TAN IIA increased TCA accumulation in a dose and time dependent manner. Only high dose TAN IIA could significantly induce the functional activity of NTCP at 24 and 48 h. However, the above cell functional results need to be further verified in the whole animal. What’s more, we analyzed the correlation of effects of RFP or TAN IIA on NTCP expression and activity. Furthermore, NTCP transporter activities showed a good correlation with NTCP mRNA expression.
Our data is supported by the observation of the physiological cholestasis in neonates, which is related to NTCP activity immediately declined after birth. 20 Moreover, Miura et al. found that the sustained repression and translocation of Ntcp seem to be responsible for the high blood bile acids levels after massive hepatectomy, which is a marker of cholestasis. 21 So, the inhibition of NTCP expression and transport activity by RFP is probably one of the causes of cholestatic liver injury. The NTCP plays an important role in the hepatic reabsorbing of a number of conjugated bile acids from the plasma into the hepatocyte and uptaking clinically important drugs. 22 Development of NTCP elimination capacity might alter the incidence of cholestasis liver injury. For example, the therapeutic effect of Yin-Zhi-Huang to cholestasis could be attributed to the upregulation of NTCP, including significant decrease in the serum total bile acids (TBA) and direct bilirubin (DBIL). 23 New Therapeutic concepts in bile acid transporters and signaling for cholestasis liver injury will open a new chapter. 24
Conclusion
Our data indicate that RFP has inhibition effects on NTCP expression and transport activity, and co-treatment of TAN IIA can reverse RFP inhibition in hepatocytes. Ultimately, this work will contribute to a better understanding of RFP induced cholestatic liver injury and TAN IIA liver protection, and provide theoretical basis for TAN IIA preventing RFP induced cholestatic liver injury. In addition, our work will enrich the significance of NTCP changes to bile acid-related liver injury, and provide a new research direction for bile acid-related diseases.
Footnotes
Authors’ note
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgment
The authors thank the participants in this study.
Author contributions
YY, XJ and LW conceived and designed the experiments; YY and LL performed research and conducted the data analysis; YY wrote the first-draft manuscript and LL revised the manuscript. YY and LL contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Natural Science Foundation of China [Grant No. 8157141040] and Scientific Research Project of Sichuan Health Commission [Grant No. 20PJ205].
