Abstract
Introduction
MiR-200c plays a central role in glucose metabolism in cancer cells. However, its upstream regulators in this process are unknown. CircRNA CSPP1 (circCSPP1) was predicted to bind to premature miR-200c, an oncogenic miRNA. Therefore, we explored their interaction in osteosarcoma (OS).
Methods
Differential circCSPP1 and miR-200c expression in OS was analyzed using RT-qPCR. Glucose metabolism was analyzed by glucose uptake assay. Subcellular circCSPP1 location in OS cells was detected using cellular fractionation assay. The direct interaction between circCSPP1 and miR-200c was explored using RNA-RNA pull-down assay. The role of circCSPP1 in miR-200c maturation was investigated by analyzing both mature and premature miR-200c levels in OS cells with circCSPP1 overexpression.
Results
CircCSPP1 and premature miR-200c levels were increased while mature miR-200c level was decreased in OS. CircCSPP1 was detected in both the nuclear and cytoplasm fractions of OS cells. CircCSPP1 directly interacted with premature miR-200c. CircCSPP1 overexpression increased premature miR-200c level, glucose uptake, and cell proliferation, but decreased mature miR-200c level. MiR-200c overexpression suppressed the role of circCSPP1 in OS cells.
Conclusions
CircCSPP1 promotes OS cell proliferation and increases glucose metabolism by suppressing miR-200c maturation.
Introduction
Although osteosarcoma (OS), the most common bone tumor, mostly affects long bones, such as the femur and tibia, it can develop in any bones. 1 In each year, about 1000 new OS cases are diagnosed in the United State. Of these cases, about 50% are children and teenagers.2,3 With the development of novel anti-OS therapies and early diagnostic rate increasing, more than 75% of OS patients with localized tumors can live more than 5 years after diagnosis. 4 However, distant tumor metastasis to lung and other organs is diagnosed in about 15–20% of OS patients.5–7 Once distant tumor metastasis occurs, the overall survival rate will significantly decrease. 7 Therefore, novel therapies are needed to treat metastatic OS patients.
With the advantages of high specific targeting and fewer side effects, targeted molecular therapy has become the next generation of cancer therapy, especially for metastatic cancers. 8 Recent studies on the molecular genetics of OS have significantly increased our understanding of molecular factors involved in this disease.9,10 Some molecular pathways, such as CK1α/CBX4 and PI3K/Akt pathways, are proven to have therapeutic potentials for OS.11,12 CBX4 promotes OS metastasis by increasing CBX4 phosphorylation via CK1α. 11 PI3K/Akt pathway mainly regulates OS cell viability and proliferation. 12 Inhibition of these two pathways reduces tumor metastasis and growth. However, more molecular targets are needed to improve the clinical applicability and feasibility of targeted molecular therapy. With no or limited coding capacity, circRNAs and miRNAs participate in cancers mainly by regulating expression of genes with protein-coding abilities.13–15 It has been well established that miRNAs directly target protein-coding genes to affect disease progression and circRNAs may serve as endogenous competing RNAs for miRNAs to indirectly affect protein synthesis.13–15 The critical functions of circRNAs and miRNAs have also been characterized in OS, suggesting the potential roles of circRNAs and miRNAs as molecular targets to treat OS.13–15 MicroRNA 200c (miR-200c) plays a central role in glucose metabolism in cancer cells. However, its upstream regulators are unclear.16,17 We have predicted the interaction between premature miR-200c and circRNA CSPP1 (circCSPP1), an oncogenic circRNA. 18 It is known that translocation of premature miRNAs from the nucleus to the cytoplasm determines their maturation. Therefore, we hypothesized that circCSPP1 may interact with premature miR-200c, thereby affecting mature miR-200c production and analyzed the interaction between circCSPP1 and premature miR-200c in OS.
Materials and methods
Study design
OS patients and healthy controls were included to study differential gene expression. The potential roles of circCSPP1 and miR-200c in cancer cell behaviors were analyzed using correlation analysis and further validated by in vitro experiments.
Participants
Chi-squared analysis of the associations between circCSPP1 expression in OS tissues and patients’ clinical data.

Cells and transfection
MG-63 cells from a male OS patient and SAOS-2 cells from a female OS patient (ATCC, USA) were used in in vitro experiments to match the gender distribution of patients included in this study. These cells were cultured in DMEM containing 10% FBS, glutamine, 100 mg/mL streptomycin and 100 μL/mL penicillin at 37°C in an incubator with 5% CO2 and 95% humidity.
Both circCSPP1 and miR-200c overexpression were achieved by Lipofectamine 3000 (Invitrogen)-mediated transfections of circCSPP1 vector and/or mimic of miR-200c. In each transfection, 107 cells were incubated with the mixture of Lipofectamine 3000 and 10 mM vector or 100 mM miRNA. Transfections were confirmed by RT-qPCR 48h later.
RNA preparation
Total RNAs were isolated using Monarch Total RNA Miniprep Kit (Cat# T2010, NEB). Prior to the addition of buffers, cells (1x10 7 ) were pelleted and tissue samples (10 mg) were ground in liquid nitrogen. DNA contamination was eliminated by DNase I digestion. RNA concentration and RIN numbers were analyzed using a Bioanalyzer.
Reverse transcription and qPCR
About 1000 ng total RNAs were reverse transcribed into cDNA samples using RevertAid First Strand cDNA Synthesis Kit (Thermo Scientific). All cDNA samples were subjected to PCR amplification of GAPDH to examine RNA quality and showed satisfactory quality. With cDNA samples as templates, the levels of circCSPP1 and miR-200c were determined by qPCRs with 18S rRNA and U6 as internal controls, respectively. Relative gene expression levels were analyzed using the delta delta Ct method.
Pull-down assay for RNA-RNA interaction
CircCSPP1 and negative control (NC) RNA transcripts were prepared by in vitro transcription using HiScribe™ T7 In Vitro Transcription Kit (NEB) and purified using GeneJET RNA Purification Kit (Thermo Fisher Scientific). After DNase I treatment to remove DNA templates, the transcripts were labeled with biotin using Biotin RNA Labeling Mix (Roche Diagnostics). The biotin-labeled transcripts Bio-circCSPP1 and Bio-NC were transfected into cells as mentioned above. Cells were lysed 48h after transfection. Biotin-labeled RNA complexes in the lysates were pulled down using magnetic beads (Invitrogen), purified, and subjected to RT-qPCR to detect miR-200c abundance.
Subcellular localization analysis
The nuclear and cytoplasm fractions of cells were prepared using PARIS Kit (Life Technologies) following the manufacturer’s instructions. Briefly, about 107 cells were collected after low-speed centrifugation and incubated with pre-cold cell fractionation buffer on ice for 10 min. After that, cell lysates were centrifuged at 500g for 10 min and the supernatants were collected as the cytoplasm fractions. The pellets were collected and mixed with cell fractionation buffer. After vortex, samples were incubated on ice for a least 10 min to collect the nuclear fractions. RNAs were extracted from both nuclear and cytoplasm fractions, reverse transcribed into cDNA samples, and subjected to RT-PCR to determine circCSPP1 levels.
Glucose uptake assay
Cells harvested at 48h post-transfection were cultured in serum-free medium supplemented with 10 mM glucose for 48h. After that, intracellular glucose content was determined using Glucose Colorimetric Assay Kit II (K686, BioVision, Inc.).
BrdU cell proliferation assay
Cell proliferation after transfections was analyzed by determining BrdU incorporation. Briefly, cells were washed with ice-cold PBS and incubated in fresh medium containing 10 μM BrdU for 1h. After washed with PBS three times, cells were incubated first with peroxidase-coupled, anti-BrdU antibody for 2h and then with peroxidase substrate tetramethylbenzidine for 2h. After that, OD values at 450 nm were measured to reflect cell proliferation.
Statistical analysis
Statistical power was calculated and reached higher than 85% in all cases. GraphPad Prism 9 was used for normality test (D’Agostino-Pearson). All data were expressed as mean ± standard deviation (SD) and compared using Student’s test (two groups) or ANOVA Tukey’s test (multiple groups). Patients were divided into high and low circCSPP1 level groups (n = 31, cutoff value = median circCSPP1 level in OS tissues), and associations between patients’ clinical data and circCSPP1 expression were analyzed with Chi-squared test. p < 0.05 was considered statistically significant.
Results
Differential circCSPP1 expression in OS and its correlations with patients’ clinical data
CircCSPP1 expression in paired OS and non-tumor tissue samples from 62 OS patients was determined using RT-qPCR. It was observed that circCSPP1 expression level was significantly increased in OS tissue samples (Figure 1(a), p < 0.01). Chi-squared test analysis showed that circCSPP1 expression in OS tissues was not closely correlated with patients’ age, gender, location, tumor metastasis, body weight, and clinical stages but was closely correlated with patients’ tumor size and blood glucose (Table 1). Therefore, circCSPP1 may mainly regulate tumor growth in OS. Differential circCSPP1 expression in OS and non-tumor tissues. RT-qPCR was performed to examine circCSPP1 expression in paired OS and non-tumor tissue samples from 62 OS patients. **, p < 0.01.
MiR-200c expression and its correlations with circCSPP1
Differential expression of both premature and mature miR-200c in paired tissue samples was analyzed with RT-qPCR. It was observed that miR-200c was significantly upregulated in OS at premature level (Figure 2(a), p < 0.01) but was significantly downregulated at mature level (Figure 2(b), p < 0.01). Pearson’s correlation coefficient analysis showed that circCSPP1 was significantly and positively correlated with premature miR-200c (Figure 2(c)) and closely and inversely correlated with mature miR-200c (Figure 2(d)). Therefore, inhibiting miR-200c maturation may participate in OS. In addition, circCSPP1 may regulate miR-200c maturation. MiR-200c expression and its correlations with circCSPP1.
Differential expression of both premature (A) and mature (B) miR-200c in paired tissue samples was analyzed with RT-qPCR. Pearson’s correlation coefficient analysis showed that circCSPP1 was significantly and positively correlated with premature miR-200c (C) and closely and inversely correlated with mature miR-200c (D). **, p < 0.01.
Subcellular location of circCSPP1 in OS cells and its crosstalk with premature miR-200c
Cellular fractionation assay was applied to detect circCSPP1 in the nuclear and cytoplasm fractions of both MG-63 and SAOS-2 cells. It was observed that circCSPP1 could be detected in both nuclear and cytoplasm fractions of both cell lines (Figure 3(a)). IntaRNA2.0 was applied to predict the potential interaction between circCSPP1 and premature miR-200c. It was predicted that circCSPP1 and premature miR-200c could form strong base pairing (Figure 3(b)). RNA-RNA pull-down analysis showed that premature miR-200c levels were significantly higher in Bio-circCSPP1 group than in Bio-NC group, (Figure 3(c), p < 0.01), confirming the direct interaction between circCSPP1 and premature miR-200c. Therefore, circCSPP1 may directly interact with premature miR-200c in the nucleus. Subcellular location of circCSPP1 in OS cells and its crosstalk with premature miR-200c.
Cellular fractionation assay was applied to detect circCSPP1 in the nuclear and cytoplasm fractions of both MG-63 and SAOS-2 cells (A). IntaRNA2.0 was applied to predict the potential interaction between circCSPP1 and premature miR-200c (B). RNA-RNA pull-down analysis was performed to further confirm the interaction between circCSPP1 and premature miR-200c (C). **, p < 0.01.
The role of circCSPP1 in miR-200c maturation
To explore the role of circCSPP1 in miR-200c maturation, both MG-63 and SAOS-2 cells were overexpressed with circCSPP1 or miR-200c. Their overexpression was confirmed every 24h until 96h (Figure 4(a), p < 0.01). It was observed that circCSPP1 overexpression increased premature miR-200c expression (Figure 4(b), p < 0.01) but decreased mature miR-200c level (Figure 4(c), p < 0.01). Therefore, circCSPP1 could suppress miR-200c maturation. The role of circCSPP1 in miR-200c maturation.
Both MG-63 and SAOS-2 cells were overexpressed with circCSPP1 or miR-200c, as confirmed every 24h until 96h (A). The effects of circCSPP1 on premature miR-200c (B) and mature miR-200c (C) levels were analyzed using RT-qPCR. **, p < 0.01.
The roles of circCSPP1 and miR-200c in OS cell proliferation and glucose uptake
Correlation analysis revealed a close association between circCSPP1 and tumor size, suggesting the potential role of circCSPP1 in regulating tumor growth. Therefore, BrdU incorporation and glucose uptake assays were applied to analyze proliferation and glucose uptake of both MG-63 and SAOS-2 cells, respectively. CircCSPP1 overexpression increased proliferation and glucose uptake of both cell lines, while miR-200c overexpression suppressed proliferation (Figure 5(a), p < 0.01) and glucose uptake of both cell lines (Figure 5(b), p < 0.01). Therefore, circCSPP1 may suppress OS cell proliferation by inhibiting miR-200c maturation. The roles of circCSPP1 and miR-200c in OS cell proliferation and glucose uptake.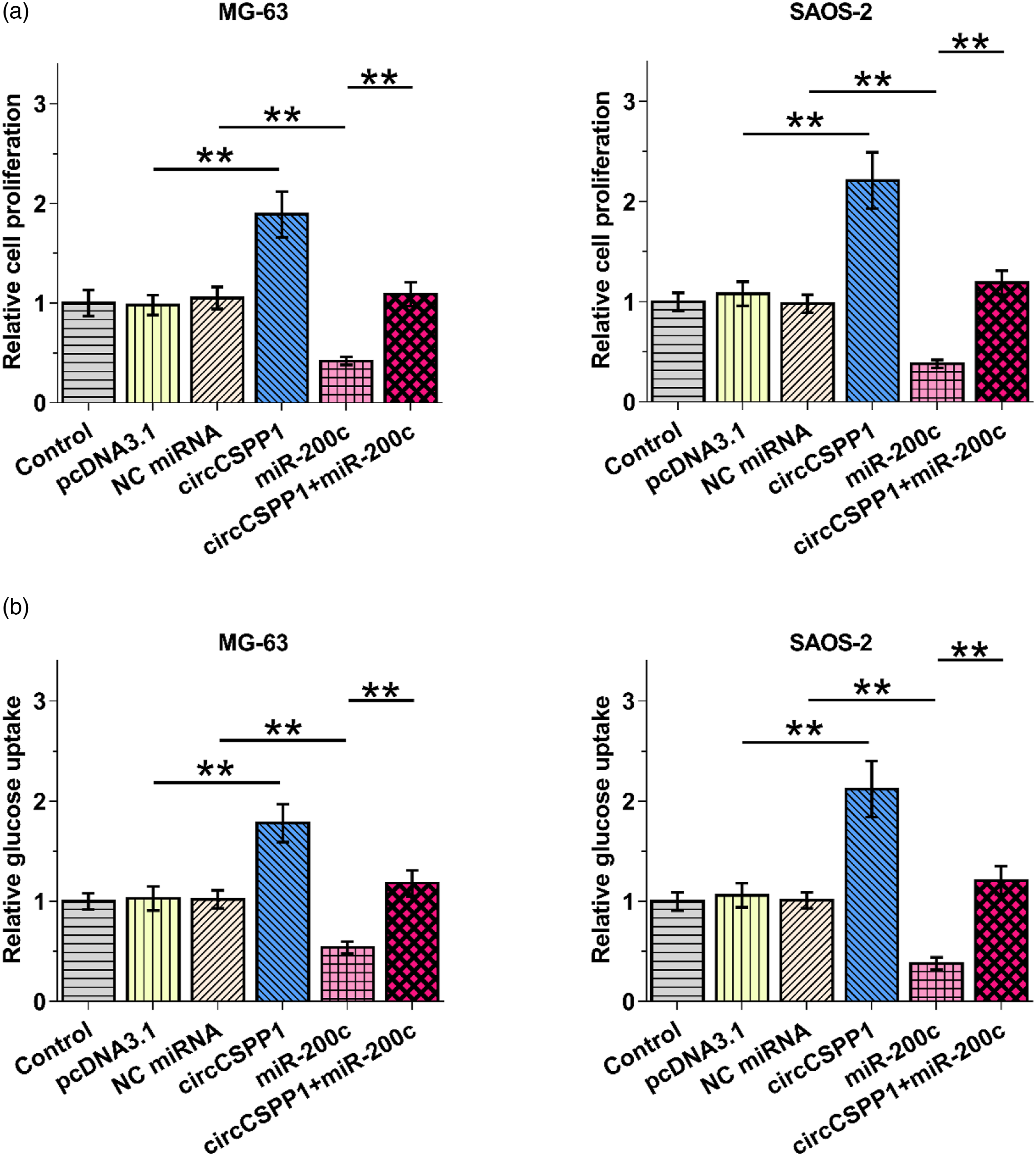
BrdU incorporation and glucose uptake assays were applied to analyze proliferation (A) and glucose uptake (B) of both MG-63 and SAOS-2 cells, respectively. Data of three independent replicates were presented and compared. **, p < 0.01.
Discussion
The present study explored the functions of two ncRNAs, namely circCSPP1 and miR-200c, in OS. We observed altered circCSPP1 expression and miR-200c maturation in OS. Moreover, circCSPP1 may affect miR-200c maturation to regulate glucose uptake and cell proliferation to promote OS.
MiR-200c plays a tumor suppressor role in many types of cancers, including OS.19–21 It has been reported that miR-200c targets ABCB1 and AKT2 to suppress tumor growth and increase chemosensitivity.19,21 The decreased miR-200c expression also predicts poor survival and lung metastasis of OS patients. 20 Increased glucose metabolism provides energy for cancer cell proliferation, suggesting glucose metabolism as a potential target to treat cancers, including OS. 22 MiR-200c targets Notch1 and GLUT1 to regulate glucose uptake.16,17 However, the role of miR-200c in glucose metabolism in OS is unclear. This study showed the inhibitory effects of miR-200c on glucose uptake and proliferation of OS cells. Therefore, miR-200c may target glucose uptake to suppress OS cell proliferation.
As a novel circRNA identified in liver cancer, circCSPP1 sponges miR-1182 to promote cancer development. 18 However, its role in other cancers is unclear. Our study revealed an increased circCSPP1 expression in OS and its enhancing effects on OS cell proliferation and glucose uptake. Therefore, circCSPP1 is an oncogenic circRNA in OS. Interestingly, we observed an inhibitory effect of circCSPP1 on miR-200c maturation and direct interaction between circCSPP1 and premature miR-200c in OS cells. It is well established that premature miRNAs are mainly localized in the nuclear fraction. We detected circCSPP1 in both nuclear and cytoplasm fractions of OS cells. Therefore, circCSPP1 in the nucleus may sponge premature miR-200c to suppress its transportation to the cytoplasm, thereby inhibiting its maturation. It has been well established that circRNAs in the cytoplasm may sponge mature miRNAs to suppress their role.13–15 However, the role of circRNAs in the maturation of miRNAs has not been well studied. This study reported a novel interaction between circRNAs and miRNAs. This novel interaction may be applied to study the function of other circRNAs. Interestingly, circCSPP1 is only closely correlated with tumor size, but not tumor metastasis. In vitro cell proliferation assay also performed the role of circCSPP1 in tumor growth. Therefore, circCSPP1 may only participate in tumor growth, but not tumor metastasis of OS. However, the role of circCSPP1 in tumor growth and metastasis remains to be further analyzed by in vivo experiments. In addition, the sample size of our study is small. The conclusions should be further validated by future studies with a larger sample size.
In conclusion, circCSPP1 is downregulated and miR-200c maturation is inhibited in OS. CircCSPP1 may suppress miR-200c maturation to regulate glucose uptake and cell proliferation to promote OS. The novel circCSPP1/miR-200c pathway may serve as a potential target to treat OS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Yunnan Provincial Bone and Joint Disease Clinical Medicine Center Project (No. ZX2019-03-04).
