Abstract
Background and Aim
The epithelial cells are the strongest determinants of the physical intestinal barrier. Tight junctions (TJs) hold the epithelial cells together and allow for selective paracellular permeability. Larazotide acetate (LA) is a synthetic octapeptide that reduces TJ permeability by blocking zonulin receptors. In this study, we aimed to investigate the effects of LA, a TJ regulator, on the liver and intestinal histology in the model of acute liver failure (ALF) in rats.
Materials and Methods
The thioacetamide (TAA) group received intraperitoneal (ip) injections of 300 mg/kg TAA for 3 days. The TAA+LA(dw) (drinking water) group received prophylactic 0.01 mg/mL LA orally for 7 days before the first dose of TAA. The LA(dw) group received 0.01 mg/mL LA orally. The TAA + LA(g) (gavage) group received prophylactic 0.01 mg/mL LA via oral gavage for 7 days before the first dose of TAA. The LA(g) group received 0.01 mg/mL LA via oral gavage. While liver tissue was evaluated only with light microscopy, intestinal samples were examined with light and electron microscopy.
Results
Serum ammonia, AST, and ALT levels in the TAA group were significantly higher than in control groups (all p < 0.01). Serum ALT levels in the TAA + LA(dw) group were significantly lower than in the TAA group (p < 0.05). However, serum ammonia and ALT levels did not differ between the TAA and other groups. Serious liver damage in the TAA group was accompanied by marked intestinal damage. There was no significant difference between the TAA and TAA + LA(dw) groups and TAA and TAA + LA(g) groups for liver damage scores. However, intestinal damage scores significantly decreased in the TAA + LA(dw) group compared to the TAA group. In the TAA + LA(dw) group, fusion occurred between the surface epithelial cells of neighboring villi and connecting regions formed as epithelial bridges between the villi.
Conclusion
Our findings suggest that LA reduced intestinal damage by acting on TJs in the TAA-induced ALF model in rats.
Introduction
The gut and liver share essential anatomical and functional connections, and their interactions play a crucial role in the development of many liver diseases such as non-alcoholic fatty liver disease, cirrhosis, and liver failure. Increasing evidence has highlighted the relevance of gut barrier dysfunction in the development of liver damage. 1 As a result of intestinal barrier dysfunction, bacteria and bacterial products in the intestine migrate to extraintestinal sites such as the liver. This process, which is also defined as bacterial translocation, has been shown to play an important role in the pathogenesis of many liver diseases such as alcoholic liver disease 2 and drug-induced liver damage. 3 Augmented pro-inflammatory response to gut-derived products and failure to control invading bacteria cause remote organ injury. 4 In addition, it has been reported that liver diseases are accompanied by many structural and functional changes in the gastrointestinal tract, such as edema of lamina propria, distended intercellular spaces, impaired motility, and changes in microbiota composition.5–8
Normally, the intestine protects the body against harmful substances by creating a complex physical, chemical, functional, and immunological barrier. The epithelial cells are the strongest determinants of the physical intestinal barrier. 9 Tight junctions (TJs) are structures located between epithelial cells. These hold the epithelial cells together and allow for selective paracellular permeability, normally excluding passive movement of large hydrophilic uncharged compounds, such as bacteria and macromolecules (e.g., lipopolysaccharides and peptidoglycans). 10 In liver diseases, intestinal barrier dysfunction develops via many mechanisms, such as intestinal oxidative damage and changes in intestinal microbial composition. 11 Also, qualitative and quantitative changes in TJs have been observed in liver diseases.12,13
Larazotide acetate (LA) (Innovate Biopharmaceuticals, Raleigh, NC, USA) is a synthetic octapeptide that reduces TJ permeability by blocking zonulin receptors. 14 Presently, LA is being studied in Phase 3 clinical trials for the treatment of celiac disease. 15
The main driving force that motivated us to do this study was to determine whether a substance that reduces TJ permeability in the experimental liver failure model could reduce gut barrier dysfunction and consequent liver and intestinal damage. Therefore, in this study, we aimed to investigate the effects of LA, a TJ regulator, on the liver and intestinal histology in the model of acute liver failure (ALF) in rats.
Materials and methods
Animals
Healthy male Wistar rats weighing 300–350 g were obtained from Inonu University Animal Laboratory (Malatya, Turkey). They were housed under strictly controlled humidity conditions, temperature (about 23°C), and illumination (12 h dark: 12 h light cycle) and were given food and water ad libitum. The study protocol was in accordance with the guidelines for animal research and was approved by the Ethics Committee of Animal Experimentation at the Medical Faculty of Inonu University (2018/A-27).
Induction of liver failure
Liver failure was induced by intraperitoneal (ip) injections of 300 mg/kg thioacetamide (TAA; Merck, Darmstadt, Germany) daily for 3 days.16,17 Subcutaneous 5% dextrose (25 mL/kg) and 0.9% NaCl with potassium (20 mEq/l) every 6 h were given as supportive therapy to prevent weight loss, hypoglycemia, and renal failure, as described previously. 18
Experimental design
Forty-eight male rats were divided into six groups (n = 8 rats). (1) The control group received 0.1 mL NaCl intraperitoneally (IP) for 7 days. (2) The TAA group received 300 mg/kg/day thioacetamide (TAA) IP for 3 days at intervals of 24 h. (3) The TAA + LA(dw) (drinking water) group received 300 mg/kg/day TAA IP for 3 days + prophylactic LA orally (autoclaved water supplemented with 0.01 mg/mL LA and bicarbonate 1.5 g/dl to buffer gastric acidity) for 7 days before the first dose of TAA. The dose of LA was determined based on an earlier experimental study related to this substance. 19 (4) The LA(dw) group received autoclaved water supplemented with 0.01 mg/mL LA and bicarbonate 1.5 g/dl to buffer gastric acidity for 7 days (5) The TAA + LA(g)(gavage) group received 300 mg/kg/day TAA IP for 3 days + prophylactic 300 μL of 0.1 mg/mL LA via oral gavage twice daily for 7 days before the first dose of TAA. (6) The LA(g) group received 300 μL of 0.1 mg/mL LA via oral gavage twice daily for 7 days.
Tissue samples
On day 8, we performed laparotomy under anesthesia with 50 mg/kg ketamine (Ketalar; Parke-Davis, Eczacibasi, Turkey) and 10 mg/kg xylazine HCl (Alfazyne 2%; Alfasan, Woerden, Netherlands) under strict sterile conditions. After the skin was sterilized with iodine and shaved, the abdomen was opened wide. The liver and ileum were excised for histopathological examination. Blood was taken from the right ventricle for biochemical determination of ammonia, aspartate transaminase (AST), and alanine transaminase (ALT).
Liver and intestinal histology
Liver histology
Midsections of the liver’s right lobe were processed for light microscopy. Liver samples were divided into pieces of 0.5 × 0.5 × 0.5 cm in size and processed for a routine examination. Specimens were fixed in 10% neutral formol solution, embedded in paraffin, and 5–6 μm-thick sections were cut and stained with hematoxylin and eosin (H&E). The tissue slices were scanned and scored by an experienced histologist unaware of which experimental group the samples were taken. The degree of inflammation and necrosis was expressed as the mean of 10 different fields within each slide, classified on a scale of 0–3 (0, normal; 1, mild; 2, moderate; and 3, severe). 14
Intestinal histology
Terminal ileum samples were removed and fixed for 24 h in 10% neutral formol solution. Samples were divided into pieces of 0.5 cm and processed for a routine examination. The samples from each animal were obtained in separate blocks. Tissue sections were cut at 5–6 μm thickness, mounted on slides, stained with H&E, and examined by a Leica DFC280 light microscope and a Leica Q Win Image Analyses System (Leica Microsystems Imaging Solutions, Cambridge, UK). The tissue slices were scanned and scored by two expert pathologists, unaware of which experimental groups the samples were taken. A scoring system ranging from 0 to 3 (0, normal; 1, mild; 2, moderate; 3, severe) was used for intestinal histopathological evaluations. Mucosal integrity, edema, inflammatory cellular infiltration of the lamina propria, and vessel vasodilation were scored accordingly. 20 Also, we examined intestinal samples by using electron microscopy.
Transmission electron microscopy
For the transmission electron microscopy (TEM) examination, the ileum specimens were fixed in 2.5% glutaraldehyde buffered with 0.2 M NaH2PO4 and Na2HPO4 (pH: 7.2) and postfixed in 1% osmium tetroxide. After dehydration in acetone, the specimens were embedded in Araldite CY212. Ultrathin sections were stained with uranyl acetate and lead citrate and examined in a Zeiss Libra 120 transmission electron microscope (Carl Zeiss, Oberkochen, Germany).
Biochemical analyses
Blood samples of ∼4 mL were taken from each rat. Aliquots (1 mL) from each blood sample were transferred to centrifuge tubes that contained 1 mL 10% trichloroacetic acid for deproteinization and centrifuged at 3000 g for 10 min following mixing. The clear supernatant at the top of the tube was used for the analysis of ammonia. The remaining 3 mL of each blood sample was used to separate serum following transfer into marked centrifuge tubes. These samples were kept at room temperature for 30 min and centrifuged at 3000 g for 10 min. Serum samples were aliquoted for use in biochemical analysis and stored at −20°C until analysis. Blood levels of ammonia were analyzed using the colorimetric Berthelot (indophenol) reaction. 21 Free ammonia in previously deproteinized samples generates a blue-colored indophenol molecule by reacting with sodium nitroprusside in an alkaline environment that contains phenol and hypochlorite ions. The color intensity of the chromogen, spectrophotometrically measured against blank samples at 625 nm, increases in direct proportion to the concentration of ammonia. (NH4)2SO4 was used as a standard. Serum ammonia levels were expressed in micrograms of ammonia nitrogen per deciliter. Serum AST and ALT levels were measured using a kinetic UV method defined by the International Federation of Clinical Chemistry (IFCC) using pyridoxal phosphate and NADH as cofactors. An Olympus autoanalyzer and commercial kits of the same brand were used for two analyses in the routine biochemistry laboratories of Turgut Ozal Medical Center. AST and ALT levels were expressed as units per liter.
Statistical analysis
Descriptive statistics were presented with a mean of values. The numerical values of the six groups were compared with the Kruskal–Wallis test since the sample numbers in the groups were <10 (α = 0.05). In binary comparisons, Bonferroni correction was made by dividing the α value by the number of comparisons. In binary comparisons, since the difference of only the TAA group from other groups was examined, the α value of 0.05 was divided into five (α = 0.01). Statistical analysis was performed with IBM SPSS 25.0 program (IBM, Armonk, NY, USA).
Results
Serum aspartate transaminase, alanine transaminase, and ammonia Levels
Serum levels of ammonia, AST, and ALT.

Results expressed as mean ± SD.
*Ammonia was expressed in μg ammonia nitrogen/dL.
**p < 0.01 compared with the TAA group.
***p < 0.0001 compared with the TAA group.
****p < 0.05 compared with the TAA group.
Liver histology
Hepatic necrosis, hydropic degeneration, and inflammation scores.
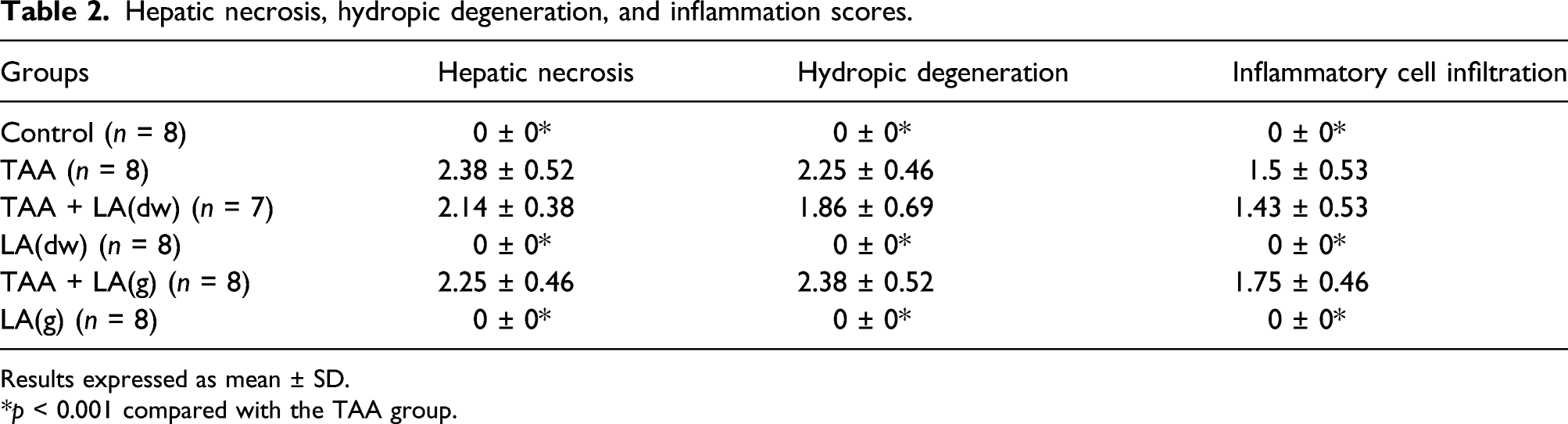
Results expressed as mean ± SD.
*p < 0.001 compared with the TAA group.

Light microscopic images of the liver. a. Control group: Normal liver parenchyma (white aster), central vein (cv), portal triad (arrows). H-E, x20. b. TAA group: Central vein (Cv), portal triad and periportal inflammation and necrosis (black aster), hydropic degeneration of hepatocytes. H-E, x20. c. TAA + LA(dw): Central vein (Cv), portal triad and periportal inflammation and necrosis (black aster). H-E, x20. D. TAA + LA(g). Central vein (Cv), portal triad and periportal inflammation and necrosis (black aster), hydropic degeneration of hepatocytes. H-E, x20.
Intestinal histology
Villous epithelial fusion, villous length, epithelial damage, villous damage, subepithelial edema, and inflammatory cell infiltration in the ileum.

Results expressed as mean ± SD.
*p < 0.001 compared with the TAA group.
**p < 0.01 compared with the TAA group.

Light microscopic images of the ileum. a. Control group: Villous (V), the crypt of Lieberkühn (Lc), tunica muscularis (M). H-E, x10. b. TAA group: Villous (V), the crypt of Lieberkühn (Lc), tunica muscularis (M), subepithelial edema (arrow), villous degeneration (aster). H-E, x10. c. TAA + LA(dw) group: Villous (V), the crypt of Lieberkühn (Lc), tunica muscularis (M), the epithelial fusion between villi (arrow). H-E, x10. D. TAA + LA(dw): Villous (V), the crypt of Lieberkühn (Lc), the epithelial fusion between villous (thin arrow), the surface epithelium (thick arrow), H-E, x20.
Transmission electron microscopy
Microvilli of epithelial cells, intercellular connections, epithelial basement membrane, and epithelial cells showed normal ultrastructural appearance in the ileum in the control group. The ultrastructural appearance of the LA(dw) and LA(g) groups was similar to that of the control group. TAA-induced ultrastructural changes were observed in the ileum of the TAA group. Vacuolization, increased secondary lysosomes, autophagosomes, mitochondrial damage, and degeneration were observed in the cytoplasm of enterocytes. Damage and degeneration were observed in the intercellular connection complexes between epithelial cells. Also, dilatation in intercellular spaces was observed (Figure 3(a)). Epithelial basement membrane damage and large vacuoles were detected in the cell–basal membrane junction region. There was a marked formation of myelin figures in these vacuoles. In the TAA + LA(dw) group, microvilli of enterocytes, intercellular connection complexes, and distances between epithelial cells and epithelial basement membranes were similar to those in control, LA(dw), and LA(g) groups (Figures 3(b) to (d)). Vacuolization, secondary lysosomes, autophagosomes, and epithelial cell mitochondrial damage in the TAA + LA(dw) group decreased significantly compared to the TAA group. The ultrastructural appearance of the TAA + LA(g) group was similar to that of the TAA group. There was no significant improvement in the ultrastructural damage in this group. Electron microscopic images of the ileum. a. TAA group: enterocyte nuclei (N), intraepithelial lymphocyte (Ln), dilatation in the inter-enterocyte space (white arrow), vacuolization (V), myelin figure in the vacuole (arrowhead), damaged basement membrane (black arrow). 8000×, bar: 2 μm. b. TAA + LA(dw) group: microvilli (Mv), junctional complex (rectangle and thick arrow), damaged mitochondrion (thin arrow). Large photograph 6300×, bar: 2 μm. Small photograph 25 000×, bar: 500 nm. c. TAA + LA(dw) group: enterocyte nuclei (N), inter-enterocyte space (arrow), damaged mitochondrion (thin arrow), intracytoplasmic autophagosome (*).6300×, bar: 2 μm. d. TAA + LA(dw) group: enterocyte nuclei (N), damaged mitochondrion (thin arrow), lysosome (arrowhead), basement membrane (thick arrow). 6300×, bar: 2 μm.
Discussion
Liver failure was induced by using TAA as in previous studies.16,17 There was significant liver damage in the TAA group, and AST, ALT, and ammonia levels were significantly higher than in the control group. Serious liver damage in the TAA group was accompanied by marked intestinal damage. Intestinal damage scores significantly decreased in the TAA + LA(dw) group compared to the TAA group. The TEM examination of the intestines confirmed the decrease in intestinal injury observed in the TAA + LA(dw) group. However, we found that TAA-induced liver damage did not improve with the LA treatment. In terms of liver damage scores, there was no significant difference between the TAA + LA(dw) and TAA + LA(g) and TAA groups.
The intestinal damage scores were high in the TAA group, and significant intestinal damage was observed in this group (Figure 2(b)). It has been reported that significant intestinal damage is observed in TAA-induced liver damage models.
18
We observed that intestinal damage scores significantly decreased in the TAA + LA(dw) group compared to the TAA group. In the TAA + LA(dw) group, fusion occurred between the surface epithelial cells of neighboring villi and connecting regions formed as epithelial bridges between the villi (Figures 2(c) and (d)). TEM examination of the TAA + LA(dw) group supported the light microscopy findings (Figure 3). The appearance of intercellular connection complexes and distances between epithelial cells and epithelial basement membranes in the TAA + LA(dw) group were similar to those in control, LA(dw), and LA(g) groups. Maintenance of the intestinal epithelial barrier is an essential function of the intestinal epithelial cells.
22
Another structure involved in the function of the intestinal barrier is TJs located between intestinal epithelial cells. TJs are the apical-most adhesive complexes that largely seal the intercellular space and consist of transmembrane proteins (e.g., claudins), peripheral membrane proteins (e.g., zonula ocludens 1), and regulatory proteins.
9
TJs are connected to the cytoskeleton.
1
TJ function is dynamic and controlled by signaling molecules, including myosin light chain kinases.
11
It has been reported that LA inhibits TJ disassembly and dysfunction caused by endogenous and exogenous stimuli in intestinal epithelial cells.
23
The fusion observed among the intestinal villi in the TAA + LA(dw) group was one of the most remarkable findings in our study. This fusion appearance may be related to the inhibitory effect of LA on the disassembly and dysfunction of TJs, which are dynamic structures connected to the cytoskeleton. If we consider all of these together, our light and electron microscopy findings suggest that LA improved intestinal damage by acting on TJs. Figure 4 visually summarizes the design and results of the study. Intestinal damage and increased intestinal permeability play an important role in the pathophysiology of many liver diseases such as cirrhosis, non-alcoholic fatty liver disease, and alcoholic liver disease, as well as in ALF.
11
Investigating the effects of LA on these models may contribute to a better understanding of the pathophysiology of these diseases and their subsequent treatment. This illustration simply summarizes the design and results of the study visually. TAA: Thioacetamide, LA: Larazotide acetate.
TAA has been used for years because it is useful for inducing experimental liver injury models. TAA causes membrane damage, oxidative stress, and accumulation of lipid droplets in the hepatocyte cytoplasm to enhance inflammation and liver injury. 24 In our study, severe liver injury was observed in the TAA group. When we compared liver damage in the TAA + LA(dw) and TAA + LA(g) groups with that in the TAA group, we found no improvement. Severe liver damage caused by TAA may be one of the reasons for this. Another reason may be the complexity of the underlying pathogenesis of ALF. ALF occurs when the rate of hepatocyte death exceeds the rate of hepatocyte regeneration due to various causes that lead to apoptosis or necrosis. 25 Cell death is mediated by many inter-related factors and signals, including caspases, oxidative stress and antioxidants, transcription factors, cytokines, chemokines, and kinases. In ALF, there is immune dysregulation caused by an imbalance between pro-inflammatory and anti-inflammatory factors. 26 Apart from these, the intestines also play a role in the development of liver damage. Some studies have highlighted the importance of increased intestinal permeability and subsequent bacterial translocation in the development of liver damage. 1 In our study, although intestinal damage was significantly decreased in the TAA + LA(dw) group compared with the TAA group, there was no significant difference in liver damage between the two groups. Our results suggest that factors such as liver cell death, regeneration, and immune dysregulation, which are non-intestinal, are more dominant than the intestines in the development of liver damage in TAA-induced ALF.
Studies on LA in humans have mostly investigated the effect of LA on the treatment of celiac disease. 15 However, experimental animal studies on LA are very limited.19,23 There are no reports in the literature about the effect of LA in experimental liver injury models. Since there is no established method for LA treatment in experimental models, we administered LA treatment in two ways by adding LA to drinking water or by gavage. While improvement in intestinal damage caused by TAA was observed in the TAA + LA(dw) group, we did not see this effect in the TAA + LA(g) group. We cannot fully explain why the administration of LA in gavage or drinking water causes such different effects. However, LA may have been dispersed and homogenized differently in water in the dw group. In addition, long-term consumption of LA-containing drinking water in the TAA + LA(dw) group, unlike the TAA + LA(g) group, may have been another factor affecting the result. As a result, our findings suggest that adding LA to drinking water is superior to gavage to prevent intestinal damage in this model.
There are some limitations of our study. First, there is no established method for the treatment of LA in experimental models. Another limitation is that other parameters that can give an idea about intestinal permeability, such as bacterial translocation, were not analyzed in this study.
In conclusion, our findings suggest that LA reduced intestinal damage by acting on TJs in the TAA-induced ALF model in rats.
Footnotes
Authors contribution
Conceptualization: [Ali Riza Caliskan, Murat Harputluoglu, and Ulvi Demirel].
Methodology: [Ali Riza Caliskan, Baris Otlu, Mehmet Gul, Ilkay Kilicaslan, Semir Gul, Nuray Uremis, and MM Uremis].
Formal analysis and investigation: [Ali Riza Caliskan, Murat Harputluoglu, Ismet Yilmaz, Muhammed Yalcin, Deniz Tikici, Osman Saglam, and Ilkay Kilicaslan].
Writing—original draft preparation: [Ali Riza Caliskan, Murat Harputluoglu, and Ulvi Demirel].
Writing—review and editing: [Ali Riza Caliskan, Murat Harputluoglu, and Ulvi Demirel].
Funding acquisition: [Ali Riza Caliskan and Murat Harputluoglu].
Resources: [Ali Riza Caliskan and Murat Harputluoglu].
Supervision: [Murat Harputluoglu and Ulvi Demirel].
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Inonu University Scientific Research Projects Unit (Project number: TSA-2019-1601).
Ethical approval
Ethics Committee approval was received for this study from İnönü University (Decision Date: 09.04.2018/Decision No.: 2018/A-27).
Informed consent
Written informed consent was obtained from patients who participated in this study.
