Abstract
Psoriasis is a chronic autoimmune disease in which peripheral blood mononuclear cells (PBMCs) are involved in the pathological process. Transient receptor potential (TRP) channels expressed in immune cells have been shown to be associated with inflammatory diseases. We aimed to evaluate mRNA expression levels of TRP channels in PBMCs of patients with psoriasis. 30 patients with plaque psoriasis and 30 healthy age- and gender-matched control subjects were included in this study. mRNA expression levels of TRP channels in psoriasis patients were determined by Real-time polymerase chain reaction. A decreased TRPM4, TRPM7, TRPV3, TRPV4, and TRPC6 genes expression levels were found in the patient group compared to controls, respectively (p = 0.045, p = 0.000, p = 0.000, p = 0.045, p = 0.009), whereas, an increased expression level was found in TRPM2 and TRPV1 genes in the patient group compared to controls (p = 0.001 and p = 0.028). This is the first study showing the TRP channel mRNA expressions in PBMCs of psoriasis patients. Different expression patterns of TRP channels may have a role in pathogenesis of psoriasis.
Introduction
Psoriasis is a chronic, immune-mediated, polygenic, inflammatory dermatosis affecting 1–3% of the population. Psoriatic lesions are located in areas such as the face, scalp, intertriginous or palmoplantar areas. 1 It is characterized by skin scaling, an inflammatory cell-rich cutaneous microenvironment, cutaneous pain and chronic itch. 2 Although psoriasis is one of the most studied diseases, the pathogenesis of the disease is not known yet. Interactions between keratinocytes and the immune system cells such as neutrophils, dendritic cells, T-lymphocytes and mast cells (MCs) appear to be effective in the development of psoriasis. 3
Transient receptor potential (TRP) channel superfamily is broadly divided into groups 1 and 2, which are based on sequence and topological differences. TRP channel superfamily is classified into six related subfamilies including (Group1) (TRPC) canonical, (TRPV) vanilloid, (TRPM) melastatin, (TRPA) ankyrin, (Group2) (TRPML) mucolipin, and (TRPP) polycystin, with about 28 protein subfamily members, most have splice variants. All TRP channels have six transmembrane domains with the N- and C-terminal regions located inside the cell. 4 These channels are expressed in a wide number of tissues and the immune system cells such as lymphocytes, dendritic cells, polymorphonuclear neutrophil granulocytes (PMNs), monocytes/macrophages and mast cells. According to the amino acid sequence and structural similarity, members of the TRP superfamily are classified into subfamilies of TRPC, TRPV, TRPM, TRPP, TRPML, and TRPA. 5 These channels have a role in ion entry, responding to a variety of neural signaling processes implicated in the sensation of temperature, pressure, osmolarity, and pH, as well as smell, taste, vision, and pain perception. 6
Changes in intracellular calcium concentrations [Ca2+] is one of the main mechanisms in the control of inflammatory and immune cell functions. Since TRP channels are Ca2+ permeable cation channels, it can be considered that TRP channels may contribute to immune and inflammatory responses in relation to other important molecular pathways. 5 TRPM1, TRPM2, TRPM3, TRPM6/7, TRPM8 channels are Ca2+ permeable non-selective cation channels, while TRPM4, TRPM5 are cation channels activated by the increase in [Ca2+] level inside the cell. 7 TRPV1–TRPV4 channels are temperature-sensitive Ca2+ permeable non-selective ion channels that are activated at different temperatures. 8 TRPV1 and TRPA1 channels are located in peripheral nerve fibers that act as pain sensors. 9 Mutations related to TRPC channels, which have been shown to exist in many different tissues, are associated with many diseases. This shows that TRPC channels are associated with many physiological processes. 10 TRP channels play a role in many pathological processes such as itch, cancers, genetic disorders, and pain. 11,12 There is no study in the literature showing the role of TRP channels expressions in psoriasis. Therefore, this study will shed light on the role of TRP channels in psoriasis.
The current study aimed to investigate mRNA expression levels of group 1 TRP channels in peripheral blood mononuclear cells (PBMCs) of patients with psoriasis compared with healthy controls by Real-Time quantitative PCR (qPCR).
Experimental procedures
Study subjects
30 patients (13 female and 17 males) who were diagnosed clinically with plaque psoriasis and 30 age-and gender-matched healthy control (15 female and 15 males) subjects were enrolled in the study. All patients have received only topical therapies. All of the patients were evaluated according to their duration of disease, Psoriasis Area Severity Index (PASI) and the affected body surface area (BSA). Subjects who also had another systemic disorder were not included in the study. Patients with erythrodermic psoriasis, pregnant and breastfeeding women, patients undergoing topical or systemic therapies for psoriasis, congenital abnormalities, alcohol users, metabolic disease (diabetes mellitus, hypertension), acute-chronic infection, cardiac-thyroid-lung diseases, collagen tissue disease, renal failure, liver diseases, patients below the age of 18 years and those with a known history of cancer or lymphoma were excluded from the study. The study was approved by the ethical committee (Decision number: 2017-KAEK-189_2020.02.12_15), and written informed consents from the subjects were also obtained for all cases.
Peripheral blood mononuclear cell isolation
Human peripheral blood mononuclear cells were isolated using Lymphocytes Separation Media (Capricorn Scientific, Ebsdorfergrund, Germany) by standard density gradient centrifugation and then carefully transferred into an RNase free tube for extraction of RNA.
Preparation of RNA, cDNA synthesis, and qPCR
Total RNA was processed through TRIzol reagent in accordance with the manufacturer’s instructions (Tri Pure Isolation Reagent, Roche Diagnostics, Mannheim, Germany) for PBMCs. The concentration of total RNA was assessed by Nanodrop 2000 (Thermo Scientific, Waltham, MA). RNA was reversely transcribed using a commercial kit (Transcriptor First Strand cDNA Synthesis Kit, Roche Diagnostics, Mannheim, Germany). The individual cDNA samples were stored at −80°C until further gene expressions made by quantitative real-time (qRT-PCR). 13 qRT-PCR analyses were performed using a LightCycler 480 SYBR Green I Master (Roche Diagnostics, Mannheim, Germany) on the LightCycler 480 platform (Roche Diagnostics). Primers were designed using NCBI primer-BLAST. All primers product sizes were less than 200 bp. PCR primers for TRP channel and internal reference gene (β-actin) is presented in Table 1. The expression of each gene was normalized using the housekeeping β-actin gene as a control, and the final results were obtained with LightCycler 480 software. All samples were run in triplicate. The expression of each gene was calculated using the ΔΔCT method and compared with the expression in the control group. A difference was considered significant at p < 0.05.
Primers used for real-time gene expression analysis (5′→3′).

Statistical analysis
Statistical analysis was performed using SPSS 18 package program (SPSS, Chicago, IL). Statistical significance was determined using The Mann–Whitney U test and differences were considered significant at a P-value of <0.05. Spearman’s correlation analysis was used for the correlation analysis of the data not showing normal distribution. All data for graphs are expressed as the mean ± SEM.
Results
Comparisons of clinical characteristics of psoriasis patients
All of the clinical characteristics are presented in Table 2. Gender, PASI, disease duration, and BSA were recorded in the patient group. There was no significant difference between the groups for gender or age, respectively (p < 0.612, p < 0.280). The mean age of the study group was 39.53 ± 10.957 and 36.97 ± 6.749 in the control group. The mean duration of the disease was 10.83 ± 8.53 years, the mean PASI was 10.367 ± 6.24, and the mean affected BSA was 11.03 ± 8.53 in the study group.
Baseline description and disease characteristics of psoriasis patients.

PBMCs mRNA expression of the TRP channel gene s
Significant differences in TRPM2, TRPM4, TRPM7, TRPV1, TRPV3, TRPV4, and TRPC6 genes expression level were observed between the patient and control groups. There was no significant difference in TRPA1, TRPV2, TRPM1, TRPM5, TRPC1, TRPC2, TRPC3, and TRPC5 genes between the patient and control groups (p > 0.05). Although it is not statistically significant, TRPA1 gene expression was increased while the others were decreased in the patient group (p > 0.05) (data not shown). There is no mRNA expression for TRPM6 and TRPC4 both in PBMCs of patient and control groups. Since, TRPM3, TRPM8, TRPV5, TRPV6, and TRPC7 expression were very low or none in PBMCs, these gene expressions were not studied. 14,15
A decreased TRPM4, TRPM7, TRPV3, TRPV4, and TRPC6 genes expression levels were found in the patient group compared to controls, respectively (p = 0.045, p = 0.000, p = 0.000, p = 0.045, p = 0.009), whereas, an increased expression level was found in TRPM2 and TRPV1 genes in the patient group compared to controls (p = 0.001 and p = 0.028) (Figure 1).
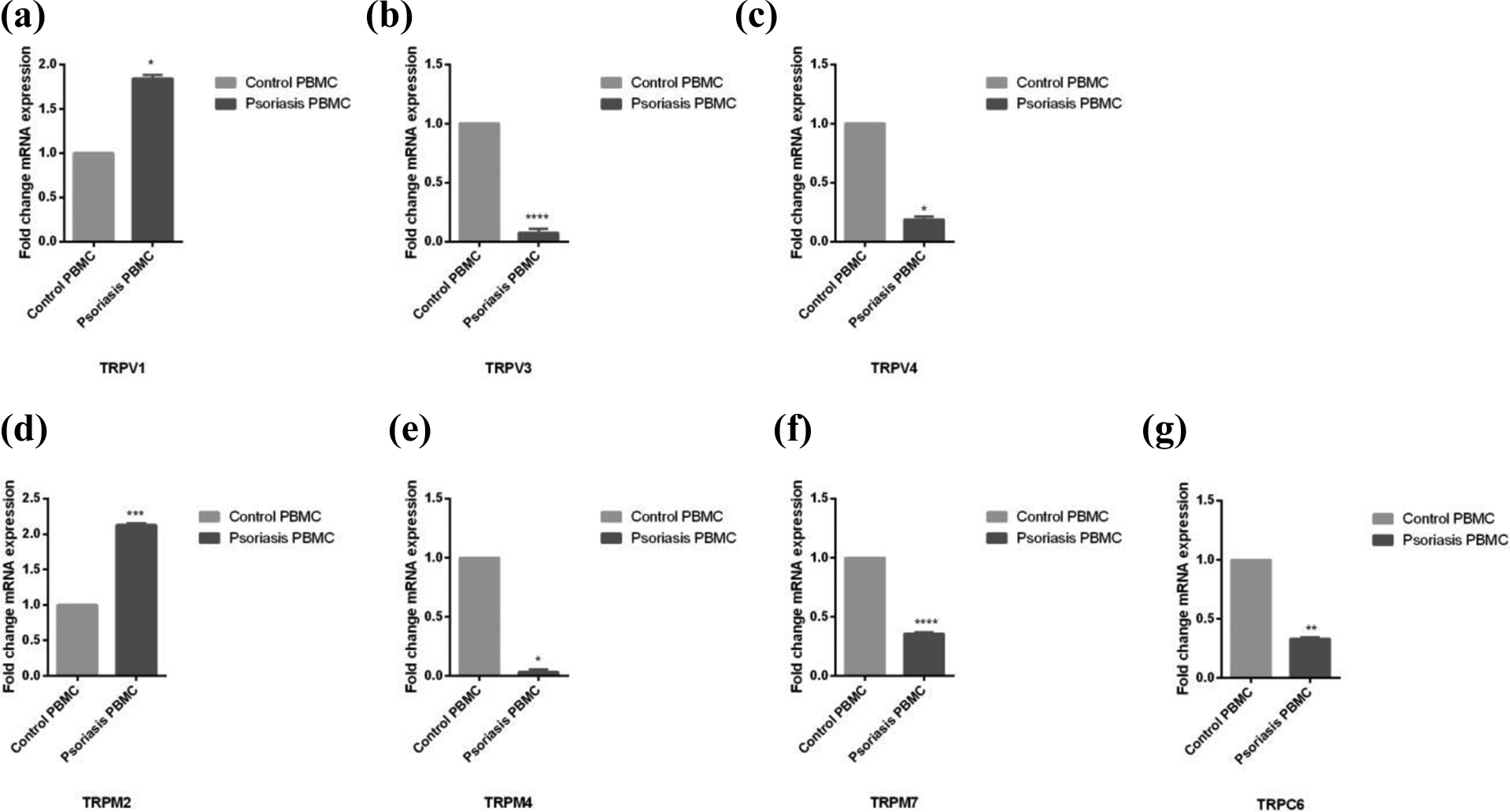
Relative mRNA expression of TRP channels in PBMCs of psoriasis patients and controls. (a) TRPV1 mRNA levels (fold change), (b) TRPV3 mRNA levels (fold change), (c) TRPV4 mRNA levels (fold change), (d) TRPM2 mRNA levels (fold change), (e) TRPM4 mRNA levels (fold change), (f) TRPM7 mRNA levels (fold change), and (g) TRPC6 mRNA levels (fold change). Each value was represented as the mean fold of mRNA expression compared with the controls (n = 30 for control and patient group). The bars represent means ± SEM. The data were analyzed by the Mann–Whitney U test and significant main effects were indicated as follows: *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001, and ****p ≤ 0.0001.
Furthermore, in the current study there is a significant negative correlation between TRPV3 and TRPA1 mRNA expression and PASI value, respectively (p = 0.021, r = −0.418 and p = 0.015, r = −0.440). We also found significant negative correlation between TRPC6 mRNA expression and PASI value and BSA, respectively (p = 0.004, r = −0.514, p = 0.02, r = −0.551).
Discussion
Here, we report that PBMCs TRPM2 and TRPV1 mRNA expression levels in psoriasis patients are significantly higher than control subjects. However, a significant decreased TRPM4, TRPM7, TRPV3, TRPV4, and TRPC6 mRNA expression levels were found in the patient group compared to controls.
TRPM2 is a Ca2+ permeable, non-selective cation channel. When the cellular reactive oxygen species (ROS) increase in the cell, TRPM2 serves as a sensor for ROS. 16 Recent studies have shown that increased ROS and reactive nitrogen species (RNS) aggravate the psoriasis pathogenesis. The antioxidant defense system developed against this condition results in the secretion of inflammatory cytokines which attributed to skin inflammation. 17 Moreover, there is evidence for the role of this channel in inflammatory processes. Immune-mediated skin diseases such as psoriasis are mediated primarily by T cells. 18 In a study, Melzer et al. reported that mouse primary CD4+ T cells have TRPM2 expression. They have demonstrated that expression of TRPM2 contributes to T-cell proliferation and production of pro-inflammatory cytokines after T-cell receptor (TCR) stimulation. 19 Considering all of these studies, the increase in TRPM2 expression in PBMCs of psoriasis patients is consistent with the literature.
TRPM4 channels which are activated by increased [Ca2+], regulate the function of immune cells. Launay et al. reported that in an in vitro study Jurkat T cells (an immortalized cell line of human T cells), silencing of TRPM4 channels resulted in enhanced Ca2+ signaling and increased (interleukin-2) IL-2 production 20 Recent study showed that increased IL-2 level correlated with psoriasis. 21 This implies that decreased TRPM4 mRNA expression might have a role in psoriasis by stimulating the cytokine increase. The kinase-coupled TRPM7 channel is expressed in immune cells, such as lymphocytes, mast cells, neutrophils, and macrophages. 22 TRPM7 has also been related to the activation and proliferation of monocytes and macrophages. 23
In the current study, we demonstrated that decreased mRNA expression of TRPV2, TRPV3 TRPV4 in the psoriasis group whereas increased mRNA expression of TRPV1. TRPV1-4 are non-selective cation channels. 24 In a study, researchers showed that the activation of TRPV1 in human keratinocytes by anandamide, a prototypic endocannabinoid, decreased the proliferation and triggered apoptosis dose-dependently and they reported that targeting these channels may be useful for the treatment of hyperproliferative skin disorders. 25 Moreover, in a disrupted epidermal barrier, stimulation of TRPV1 gen decelerates barrier recovery. 26 However, it was reported that up-regulation of cutaneous TRPV1 transcripts in sensitive skin. 27 TRPV1 is expressed by the primary human T cells and Jurkat cells as well as the primary mouse splenic T cells. TRPV1 leads to T-cell receptor (TCR)-mediated Ca2+ influx and T-cell activation. Moreover, TRPV1 blockers inhibit T-cell activation and production of inflammatory cytokines. 28 Sulk et al. showed that, increased gene expression levels for TRPV1 mRNA in all rosacea subtypes. 29 In addition this studies there are researches regarding TRPV1 gene expressions in several diseases. Rizopoulos et al. reported that decreased expression of TRPV1 in the colonic epithelium of ulcerative colitis patients whereas TRPV4 expression was significantly increased. 30 Moreover, Yoshioka et al. claim that epidermal TRPV3 has an impact on dermatitis and is associated with the development of pruritus. 31 The TRPV1-4 cation channels are moderately permeable to Ca2+. 32 Alterations in intracellular calcium concentrations represent a fundamental mechanism in the control of inflammatory and immune cell functions. Future investigations would clarify the role of TRPV channels in the pathogenesis of psoriasis.
Psoriasis is characterized by hyperproliferative keratinocytes. Relationship between keratinocytes and immune cells have an important role in psoriasis pathogenesis. 3 The maturation of the keratinocyte may vary depending on the gradient of the calcium in the cell. Our findings have demonstrated that TRPC6 mRNA expression in PBMCs of psoriasis patients was decreased and downregulation of this gene has a negative correlation between PASI and BSA values. Similarly, Leuner at al. have shown that defects in this system may cause differentiation and proliferation in psoriatic keratinocytes. Especially, they reported that downregulation of TRPC6 channels are correlated with reduced keratinocyte differentiation and TRPC activators might be useful for psoriasis treatment. 33
Previous studies show that TRPA1 is also expressed on keratinocytes and its specific physiological activation leads to the release of inflammatory mediators. 34,35 In addition to the immunological effects of TRP channels in the development of psoriasis, these channels contribute to symptoms of dermatological diseases. In a recent study reported that expression of genes for TRPV2, TRPA1, protease-activated receptor (PAR2), (PAR4), and (interleukin) IL-10 was found to be increased only in pruritic atopic skin, while expression of genes for TRPM8, TRPV3, phospholipase C, and IL-36α/γ was elevated only in pruritic psoriatic skin. 36 Moreover, TRP channels are associated with pain in arthritis and mediate neurogenic inflammation in the skin. 37
We have previously studied the mRNA expression levels of TRP channels in PBMCs of multiple sclerosis patients. In this study, we found that while TRPV1, TRPV3, TRPV4, TRPM7, and TRPC6 mRNA expression levels decreased in patients with multiple sclerosis, mRNA expression levels of other TRP channels did not change. 38 Although multiple sclerosis is an autoimmune disease such as psoriasis, it is a neurological disorder with a different pathology. It is therefore not surprising that there are both similarities and differences in gene expression levels.
In conclusion, these are the first data to demonstrate that, decreased TRPM4, TRPM7, TRPV3, TRPV4, and TRPC6 genes expression levels were found in the patient group compared to controls whereas, significantly increased expression level was found in TRPV1 and TRPM2 genes in the patient group. Additionally, this is the first study showing the TRP channel mRNA expressions in PBMCs of psoriasis patients. The present study had several limitations. First, it is necessary to validate these findings with a larger cohort to reach a more certain conclusion, additional research on protein expressions. Second, mRNA expressions in PBMC samples should be compared with tissue samples obtained from psoriasis patients. Different expression patterns of TRP channels may cause the development of several diseases. Thus, these channels are important pharmacological targets for novel therapeutic treatments. Furthermore, in future studies with larger patient numbers are needed to confirm the present findings as well as to clarify the contribution of TRP channels expression in psoriasis pathogenesis.
Footnotes
Author's note
G Gürel is now with the Department of Dermatology, Faculty of Medicine, Afyonkarahisar Health Sciences University, Afyonkarahisar, Turkey.
Acknowledgement
We thank Yozgat Bozok University Microbiology Laboratory and Professor Neziha Yılmaz for contributions to this study.
Author contributions
SSO and MÇ conceived of the presented idea. GG was involved in collecting blood samples and clinical data. SSO carried out the experiments. GG, SSO, and MÇ performed the analysis. SSO took the lead in writing the manuscript. All authors discussed the results and contributed to the final manuscript.
Ethical approval
The study was approved by the ethical committee (Decision number: 2017-KAEK-189_2020.02.12_15), and written informed consents from the subjects were also obtained for all cases.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
