Abstract
N-nitrosodimethylamine (NDMA) is a xenobiotic widespread in human environment capable of regulating the lifespan of immune cells. In this study, we examined the roles of the tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)/death receptor 5 (DR5) complex and the Fas molecule in the induction of the extrinsic apoptosis pathway in human neutrophils (polymorphonuclear neutrophils (PMNs)) and peripheral blood mononuclear cells (PBMCs) exposed to NDMA. Also we assessed these proteins ability to trigger the intrinsic apoptosis pathway in those cells. For this purpose, we examined the expression of Fas-associated protein with death domain, truncated Bid (tBid) proteins, and apoptogenic factors such as apoptosis-inducing factor, Smac/Diablo, Omi/HtrA2, and caspase-3 as an indication of accomplished apoptosis phenomenon. PMNs and PBMCs were isolated from whole blood by density gradient centrifugation using Polymorphrep. Apoptotic cells were assessed with flow cytometry using a ready-made kit. The expression of proapoptotic molecules was investigated by Western blot analysis of PMNs and PBMCs treated with NDMA and/or rhTRAIL. The obtained results confirm the proapoptotic effects of NDMA on the examined human leukocytes and indicate an active participation of the TRAIL/DR5 complex and Fas protein in the process of apoptosis. Moreover, the research revealed distinct mechanisms of intrinsic apoptosis pathway activation between PMNs and PBMCs exposed to NDMA, as confirmed by the different levels of tBid, Smac/Diablo, Omi/HtrA2, and caspase-3 expression in those cells.
Introduction
Apoptosis––programmed cell death––is an essential element of homeostasis maintenance in the immune system. The intensity of this process is regulated by various endo- and exogenous factors. 1
Among the exogenous factors affecting apoptosis, there are xenobiotics widespread in human environment such as N-nitrosodimethylamine (NDMA). The occurrence of this xenobiotic in drinking water, certain food products, or rubber items creates the risk of constant exposure. 2 Earlier studies showed that NDMA, having strong carcinogenic properties, is able to induce apoptosis in human leukocytes. 3 Increased induction of apoptosis and changes in the biological availability of those cells which were exposed to NDMA can consequently impair the early and late phase response to various pathogenic factors. 4
The induction of programmed death of peripheral blood leukocytes, including neutrophils and mononuclear cells, can involve both the extrinsic and intrinsic pathway. The former depends on receptors from the tumor necrosis factor (TNF) superfamily, while the latter is connected with the expression of mitochondrial pro- and antiapoptotic proteins of the Bcl-2 family. 5,6
The extrinsic apoptosis pathway involves many proteins, including a TNF-related apoptosis-inducing ligand (TRAIL) molecule. Being a member of the TNF family, the molecule is also a ligand of proapoptotic death receptors (DRs): TRAIL-R1 (DR4) and TRAIL-R2 (DR5). 7
The binding of the TRAIL ligand with the receptor leads to the creation of a death-inducing signalling complex (DISC), which contains a Fas-associated death domain (FADD) and a caspase-8 initiator. The interaction between the DR5 receptor and the FADD molecule has the characteristic of a homotypic interaction between identical death effector domains (DEDs) in the two proteins. 8 This phenomenon initiates a two-stage proteolytic splitting of procaspase 8 in the DISC complex, leading to its activation and triggering a cascade of effector caspases. 9 However, the connection of the TRAIL molecule with the receptor does not always lead the cell to apoptotic death. This is because the ligand can connect with decoy receptors that lack functional death domains, such as TRAIL-R3 (DcR1/TRID), TRAIL-R4 (DcR2/TRUNDD), and osteoprotegerin (OPG), which occurs only in dissolved form. Therefore, the relationship between the death and decoy receptors is a significant factor determining the process of programmed neutrophil death induced by TRAIL. 7
A significant part of the intrinsic pathway includes the interactions between the pro- and antiapoptotic proteins of the Bcl-2 family, and the proteins and mitochondrial membrane that allow the formation of its mega channels. 10 The mitochondrial pores that develop serve as channels through which the apoptogenic factors, such as Smac/Diablo and Omi/HtrA2, are released to the intracellular environment. These factors release the activity of caspases 3, -7, and -9 from the suppression produced by the inhibitor of apoptosis proteins. 11
Moreover, in response to the apoptotic stimulus from the mitochondrial intermembrane space, the apoptosis-inducing factor (AIF) is also released. The factor is transported to the cytosol, and then, along with the cofactor cyclophilin A, to the nucleus. The AIF binds with the DNA, inducing a peripheral condensation of chromatin and fragmentation of the DNA, which, consequently, leads to apoptotic changes in the nucleus. 12,13
Our earlier research indicated the influence of NDMA on the induction of apoptosis with the participation of elements of the intrinsic pathway. We found that it is related to an increased expression of proapoptotic Bax protein and a decreased expression of the antiapoptotic protein Mcl-1. In the process we also noticed the participation of the extrinsic pathway, connected with the expression of the DR5 receptor. 14 –16
The performed research specified the role of the TRAIL/DR5 complex in the induction of apoptosis in human leukocytes exposed to NDMA and its relation to the activation of the intrinsic pathway in these cells. Also it assessed the influence of NDMA on the expression of the Fas receptor, essential in apoptosis induction in neutrophils.
Methods
Isolation and incubation of leukocytes
The research material was composed of venous blood from 15 healthy volunteers (seven males and eight females) aged 23–42. After the volunteer consent, the blood samples were collected from the basilic vein, 8 ml of which were stored with ethylenediaminetetraacetic acid and 2 ml without any anticoagulant to extract serum. The study was approved by the Ethics Committee of the Medical University of Białystok (R-I-002/84/2012).
The cell fractions containing neutrophils and mononuclear cells were isolated in a concentration gradient using Polymorphrep (Axis-Shield, Oslo, Norway). The obtained mono- and polynuclear cells populations were suspended in Hank’s fluid and Rosewell Park Memorial Institute-160 medium, respectively, with a culture medium (BIOMED-Lublin, Poland) containing 7.4% (20/270 µl) of autologous serum, 100 U/ml of penicillin, and 50 ng of streptomycin (Polfa Tarchomin S.A., Poland), leading to a density of 5 × 106 cells/ml. The cells were exposed to NDMA in a concentration of 10 µg/ml (Sigma Aldrich, Steinheim, Germany, CAS no. 62-75-9) and/or rhTRAIL in a concentration of 80 ng/ml were incubated for 20 h on microplates (Microtest III-Falcon, BD Biosciences, Bedford, Massachusetts, USA) at a temperature of 37°C in an incubator with a maintained 5% carbon dioxide flow (NUAIRE™). Cytotoxic effect of NDMA was analyzed using the tetrazolium-based colorimetric assay (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide reduction assay).
Western blot analysis of protein expression
The mono- and polynuclear cell fractions were lysed via sonification (Sonic Vibra Cell) in the presence of a Protease Inhibitor Cocktail (Sigma Aldrich, Steinheim, Germany). The obtained lysate was suspended in a Laemmli buffer (Bio-Rad Laboratories, Hercules, California, USA). Fifty micrograms of the protein were used for electrophoresis with sodium dodecyl sulfate–polyacrylamide gel electrophoresis gel (Bio-Rad Laboratories Mini PROTEAN®Tetra Cell). The separated protein fractions were transferred onto 0.2 μm pore-sized nitrocellulose membranes (Bio-Rad Laboratories). Next, the membranes were incubated at 37°C for 4 h with specific polyclonal antibodies: anti-DR5 (cas no. AF631), anti-FADD (cas no. AF2938), anti-Smac/Diablo (cas no. AF7891) (R&D Systems, Minesota, Minneapolis, USA), anti-cleaved Bid (ca no. sc-34325), and anti-cleaved caspase-3 (ca no. sc-22171-R) (Santa Cruz Biotechnology, Heidelberg, Germany) and monoclonal antibodies: anti-Fas (ca no. sc-52394), anti-Omi/HtrA2 (ca no. sc-58371), and anti-AIF (ca no. Sc-13116), anti-β-actin (ca no. sc-130301) (Santa Cruz Biotechnology, Heidelberg, Germany). After washing with 0.1% Tris-buffered saline–Tween, the membrane was incubated at room temperature for 1 h with antibodies marked with alkaline phosphatase directed against immunoglobulin G (Vector Laboratories, Burlingame, California, USA). Immunoreactive protein bands were obtained by adding 5-bromo-4-chloro-3-indolyl-phosphate/nitro blue tetrazolium liquid substrate system (Sigma Aldrich). The intensity of the bands was assessed using ImageJ software (public domain, Java-based image processing program developed at the National Institutes of Health, Maryland, Bethesda USA) and estimated by arbitrary units (AU). The antibody against β-actin (Santa Cruz Biotechnology), which detects the expression of β-actin in cells lysates, was used for standardization.
Flow cytometry apoptosis assessment
Apoptotic cells were assessed by flow cytometry using a ready-made kit (Apotest™-Fluorescein isothiocyanate (FITC), Nexins Research, Holland). The polymorphonuclear neutrophils (PMNs) and peripheral blood mononuclear cells (PBMCs) fractions immediately after isolation and after 20-h incubation with or without the presence of NDMA were stained with FITC conjugated with annexin V and propidium iodide. The samples were analyzed with CellQest Analysis software (Becton Dickinson, Epics Coulter, San Jose, California, USA). The results were presented in the form of the percentage of annexin V-positive and propidium iodide-positive cells in the case of late apoptosis, and the percentage of annexin V-positive and propidium iodide-negative cells for early apoptosis.
Statistical analysis
The results were analyzed using Statistica version 9.1 (StatSoft, Inc., Tulsa, Oklahoma, USA). The normality of data distribution was examined using the Kolmogorov–Smirnov test. The data were not normally distributed, therefore Mann–Whitney U nonparametric tests were applied. Flow cytometry data were presented as median and ranges (minimum and maximum). The data in Figures 3 and 4 was presented as mean ± SD. Statistical significance level was set at p < 0.05.
Results
Apoptosis of neutrophils and mononuclear cells assessed via flow cytometry
Pursuant to earlier observations, after a 20-h incubation of neutrophils, both with and without the presence of NDMA, there was an observed increase in the percentage of apoptotic cells in comparison to cells that were not exposed to the xenobiotic. Similar changes were observed after exposing the cells to rhTRAIL. Simultaneous exposition of PMNs to NDMA and rhTRAIL resulted in the highest number of apoptotic cells (Figure 1).

Median ± maximum and minimum values of apoptotic PMNs cells after isolation, after 20 h incubation exposed to NDMA (10 µg/ml), rhTRAIL (80 ng/ml), NDMA + rhTRAIL and not exposed. aStatistical difference between the percentage of apoptotic cells immediately after isolation and after 20-h incubation with NDMA or rhTRAIL (p < 0.05). bStatistical difference between percentage of unexposed apoptotic cells and exposed to NDMA or rhTRAIL cells (p < 0.05). cStatistical difference between the percentage of apoptotic cells exposed to NDMA and rhTRAIL simultaneously and the percentage of unexposed apoptotic cells (p < 0.05). dStatistical difference between the percentage of apoptotic cells exposed to NDMA or rhTRAIL and cells exposed to NDMA and rhTRAIL simultaneously. PMNs: polymorphonuclear neutrophils; NDMA: N-nitrosodimethylamine; TRAIL: tumor necrosis factor-related apoptosis-inducing ligand.
However, incubation of mononuclear cells in the presence of NDMA or rhTRAIL did not cause any significant changes when compared to cells that were not exposed to these factors. The highest number of apoptotic PBMCs was noticed after simultaneous incubation with NDMA and rhTRAIL (Figure 2).
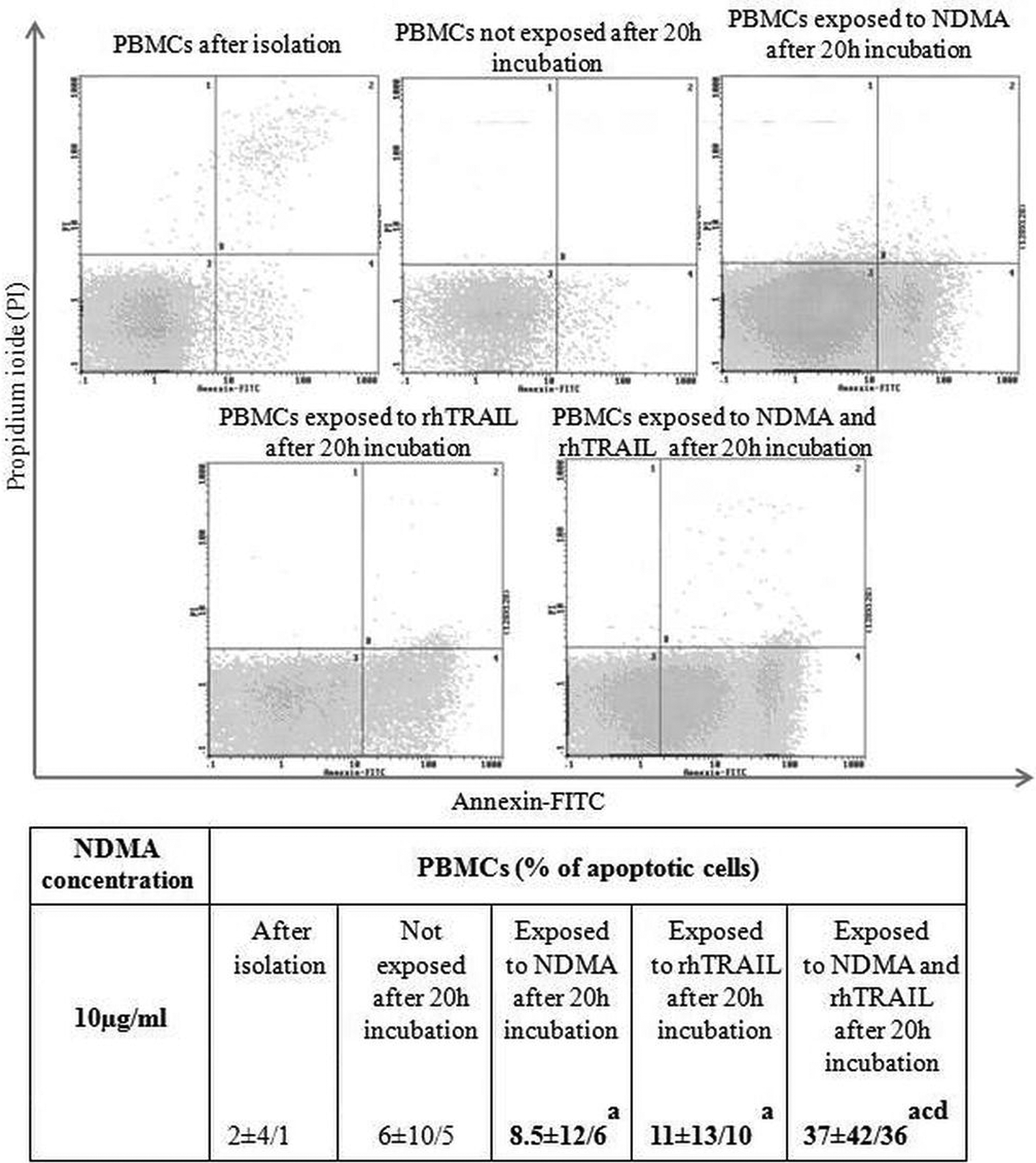
Median ± maximum and minimum values of apoptotic PBMCs cells after isolation, after 20 h incubation exposed to NDMA (10 µg/ml), rhTRAIL (80 ng/ml), NDMA + rhTRAIL and not exposed. aStatistical difference between the percentage of apoptotic cells immediately after isolation and after 20-h incubation with NDMA or rhTRAIL (p < 0.05). bStatistical difference between the percentage of unexposed apoptotic cells and exposed to NDMA or rhTRAIL cells (p < 0.05). cStatistical difference between the percentage of apoptotic cells exposed to NDMA and rhTRAIL simultaneously and the percentage of unexposed apoptotic cells (p < 0.05). dStatistical difference between the percentage of apoptotic cells exposed to NDMA or rhTRAIL and cells exposed to NDMA and rhTRAIL simultaneously. PBMCs: peripheral blood mononuclear cells; NDMA: N-nitrosodimethylamine; TRAIL: tumor necrosis factor-related apoptosis-inducing ligand.
Expression of extrinsic and intrinsic pathway apoptosis proteins assessed via Western blot
Death receptor 5
Western blot analysis showed an increased expression of the DR5 receptor in neutrophils incubated with NDMA, as confirmed by previous own observations. 16 The expression of DR5 after incubation of the PMNs with a specific ligand––an rhTRAIL molecule and NDMA––was increased in comparison to unstimulated cells and cells that were exposed to NDMA and rhTRAIL separately. An increased expression of DR5 protein was indicated in mononuclear cells exposed to NDMA. The expression of the receptor due to simultaneous and separate exposition to rhTRAIL and NDMA was higher than in unstimulated cells (Figure 3).

Western blot analysis of DR5, Fas, FADD, and tBid proteins in PMNs and PBMCs after 20 h incubation exposed to NDMA (10 µg/ml), rhTRAIL (80 ng/ml), NDMA + rhTRAIL and not exposed. aStatistical difference between not exposed and exposed cells (p < 0.05). bStatistical difference between cells exposed to NDMA and cells exposed to rhTRAIL (p < 0.05). cStatistical difference between cells exposed to NDMA and rhTRAIL simultaneously and cells exposed to NDMA (p < 0.05). dStatistical difference between cells exposed to NDMA and rhTRAIL simultaneously and cells exposed to rhTRAIL (p < 0.05). DR5: death receptor 5; PMNs: polymorphonuclear neutrophils; NDMA: N-nitrosodimethylamine; PBMCs: peripheral blood mononuclear cells; TRAIL: tumor necrosis factor-related apoptosis-inducing ligand; FADD: Fas-associated protein with death domain; tBid: truncated Bid.
Fas
The exposition of PMNs to rhTRAIL led to an increased expression of the Fas receptor. Higher expression of Fas was observed in cells subjected to rhTRAIL than in PMNs subjected to NDMA. The highest expression of this protein was observed in populations exposed to the two factors simultaneously. The changes in Fas expression in the PBMC population were statistically insignificant (Figure 3).
Fas-associated protein with death domain
The study indicated an increased expression of the FADD adaptor protein in neutrophils exposed to NDMA. Increased expression of FADD was also observed in PMNs incubated with rhTRAIL molecules, compared with unstimulated cells and those exposed to NDMA. The highest expression of this protein was noticed in PMNs incubated with rhTRAIL and NDMA simultaneously. The same changes in the expression of FADD protein were observed in mononuclear cells (Figure 3).
truncated Bid
Expression of truncated Bid (tBid) was lower in PMNs exposed to NDMA than in unexposed cells. However, the incubation of PMNs with rhTRAIL led to increased expression of the tBid protein, compared with the cells incubated with NDMA. Simultaneous exposition to rhTRAIL and NDMA resulted in the highest expression of tBid in neutrophils. In the case of mononuclear cells, the expression of tBid under the influence of NDMA was lower than in unexposed cells. The expression of tBid in PBMCs incubated with NDMA and rhTRAIL simultaneously was also lower in comparison to unstimulated cells (Figure 3).
AIF
The research pointed to a decreased expression of the apoptogenic factor AIF in PMNs incubated with NDMA and rhTRAIL in comparison to unstimulated cells. Incubation of cells in the presence of NDMA and rhTRAIL simultaneously led to a statistically significant increase in AIF expression, compared to cells which were exposed to NDMA and rhTRAIL separately. In the population of mononuclear cells the expression of AIF increased when exposed to NDMA and rhTRAIL, in comparison to cells unexposed to the xenobiotic and cells subjected to rhTRAIL only (Figure 4).
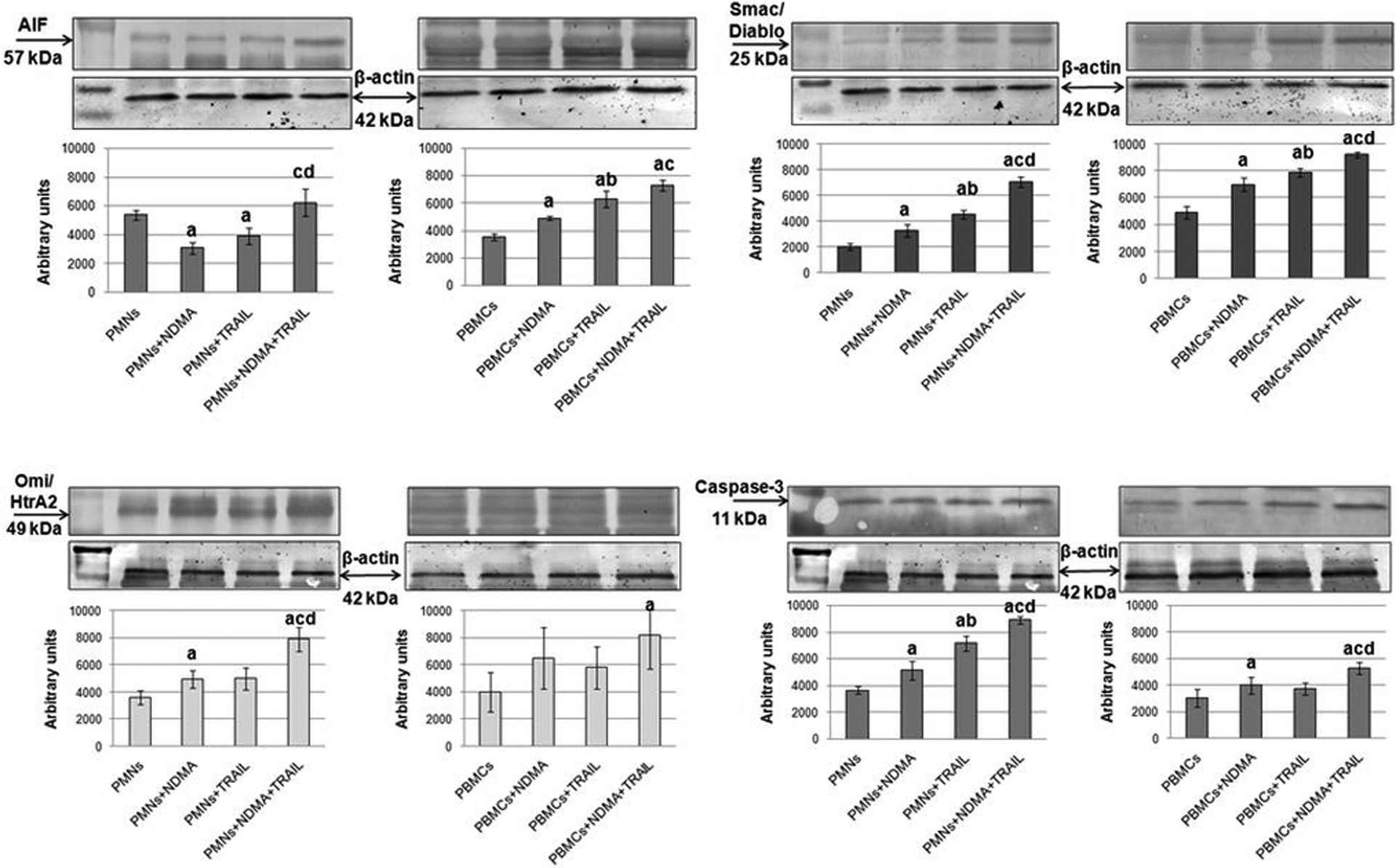
Western blot analysis of AIF, Smac/Diablo, Omi/HtrA2, and caspase-3 proteins in PMN and PBMCs after 20 h incubation exposed to NDMA (10 µg/ml), rhTRAIL (80 ng/ml), NDMA + rhTRAIL and not exposed. aStatistical difference between not exposed and exposed cells (p < 0.05). bStatistical difference between cells exposed to NDMA and cells exposed to rhTRAIL (p < 0.05). cStatistical difference between cells exposed to NDMA and rhTRAIL simultaneously and cells exposed to NDMA (p < 0.05). dStatistical difference between cells exposed to NDMA and rhTRAIL simultaneously and cells exposed to rhTRAIL (p < 0.05). AIF: apoptosis-inducing factor; PMNs: polymorphonuclear neutrophils; NDMA: N-nitrosodimethylamine; PBMCs: peripheral blood mononuclear cells; TRAIL: tumor necrosis factor-related apoptosis-inducing ligand.
Smac/Diablo
In neutrophils, the expression of Smac/Diablo after incubation with NDMA or rhTRAIL was increased in comparison with unstimulated cells. However, incubation of PMNs with rhTRAIL was higher than those exposed to NDMA. The highest expression of Smac/Diablo was noticed in cells incubated with NDMA and rhTRAIL simultaneously. Similar changes of Smac/Diablo expression as in neutrophils were observed in mononuclear cells (Figure 4).
Omi/HtrA2
In neutrophils, the expression of the Omi/HtrA2 protein was significantly increased under the influence of exposition to NDMA in comparison to cells unexposed to the xenobiotic. Simultaneous use of NDMA and rhTRAIL on PMNs caused increased expression of Omi/HtrA2, compared to the effects which NDMA and rhTRAIL had separately. The highest expression of the protein was observed in PMNs that were subjected to NDMA and rhTRAIL simultaneously. The PBMC population showed a statistically significant increase in the expression of Omi/HtraA2 in cells simultaneously exposed to NDMA and rhTRAIL than in those which were not stimulated (Figure 4).
Caspase-3
The expression of caspase-3 was higher in PMNs exposed to NDMA than in unstimulated cells. Neutrophils indicated similar changes also after incubation in the presence of rhTRAIL. Expression of caspase-3 in PMNs under the influence of rhTRAIL was higher than after exposing the cells to NDMA. Moreover, there was an observed increase in the expression of caspase-3 in PMNs incubated in the simultaneous presence of rhTRAIL and NDMA, in comparison with unstimulated cells or those which were exposed to NDMA and rhTRAIL separately. Mononuclear cells showed a larger increase in the expression of caspase-3 in cells exposed to NDMA than in those which were not exposed. The highest expression of caspase-3 was observed in PBMCs exposed to NDMA and rhTRAIL simultaneously (Figure 4).
Discussion
The biological availability of neutrophils is determined by numerous factors and is regulated mainly through the process of apoptosis. The lifespan of these cells depends on the balance between the pro- and antiapoptotic proteins in the intrinsic pathway and the ligands and receptors of the extrinsic pathway. 17
Intensified induction of apoptosis leading to a shorter lifespan of leukocytes exposed to NDMA, neutrophils in particular, can therefore contribute to the impairment of early phase response, controlled by these cells. 18 Akgul et al. proved that apoptotic neutrophils lose certain abilities such as chemotaxis, degranulation, and generation of reactive oxygen species. 19 Clinical manifestations of this situation may include recurring fungal and bacterial infections. 20
The proapoptotic properties of nitroso compounds were revealed in the research performed by Garcia et al., who studied the HL60 human leukaemia line of cells. They demonstrated the morphological changes that are characteristic of the process of apoptosis in cells subjected to N-nitrosopiperidine and N-nitrosodibutylamine, both belonging to a group of N-nitrosamines. 21 Hatanaka et al. demonstrated the impact of xenobiotic on the expression of antiapoptotic protein of the Bcl-2 family. They suggested that the decrease of Bcl-x expression in rat liver after exposure to diethylonitrosamine might be an indicator of preneoplastic lesions. 22
The results obtained in our own research confirm the proapoptotic effects of NDMA on human leukocytes and indicate an active participation of the TRAIL/DR5 complex in the process of apoptosis. The increase of DR5 expression in this study was accompanied by elevated expression of FADD adaptor protein in neutrophils and mononuclear cells. The involvement of rhTRAIL in the process seems to be confirmed by higher expression of FADD protein in cells exposed to NDMA and rhTRAIL simultaneously than in those which were exposed only to NDMA (Figure 5). One of the results of changes in DR5 and FADD expression is the activation of initiator caspases such as caspase-8, which undergoes proteolytic degradation and interacts with FADD to create the DISC complex and activates executioner caspases, mainly caspase-3. 23 FADD, an adaptor protein, may also participate in apoptosis induction on a pathway depending on the Fas/FasL complex. The connection of the Fas ligand with the receptor triggers an apoptotic cascade in infected and auto-reactive cells and cells with a constitutively short lifespan. 24 Research from various authors has indicated that this complex plays a special role in inducing apoptosis in neutrophils. Paunel-Görgülü et al. showed that apoptosis initiated in the extrinsic pathway involving the Fas/FasL complex relies also on the activity of caspases. The researchers observed a decrease in the percentage of apoptotic neutrophils incubated in the presence of a soluble Fas agonist (CH-11) and caspase inhibitor, in comparison to cells incubated only in the presence of CH-11 in patients with Systemic Inflammatory Response Syndrome. 25 In our research, we did not find any increased expression of Fas in PMNs and PBMCs subjected to NDMA, that is, the Fas receptor is uninvolved in apoptosis in cells exposed to NDMA (Figure 5).
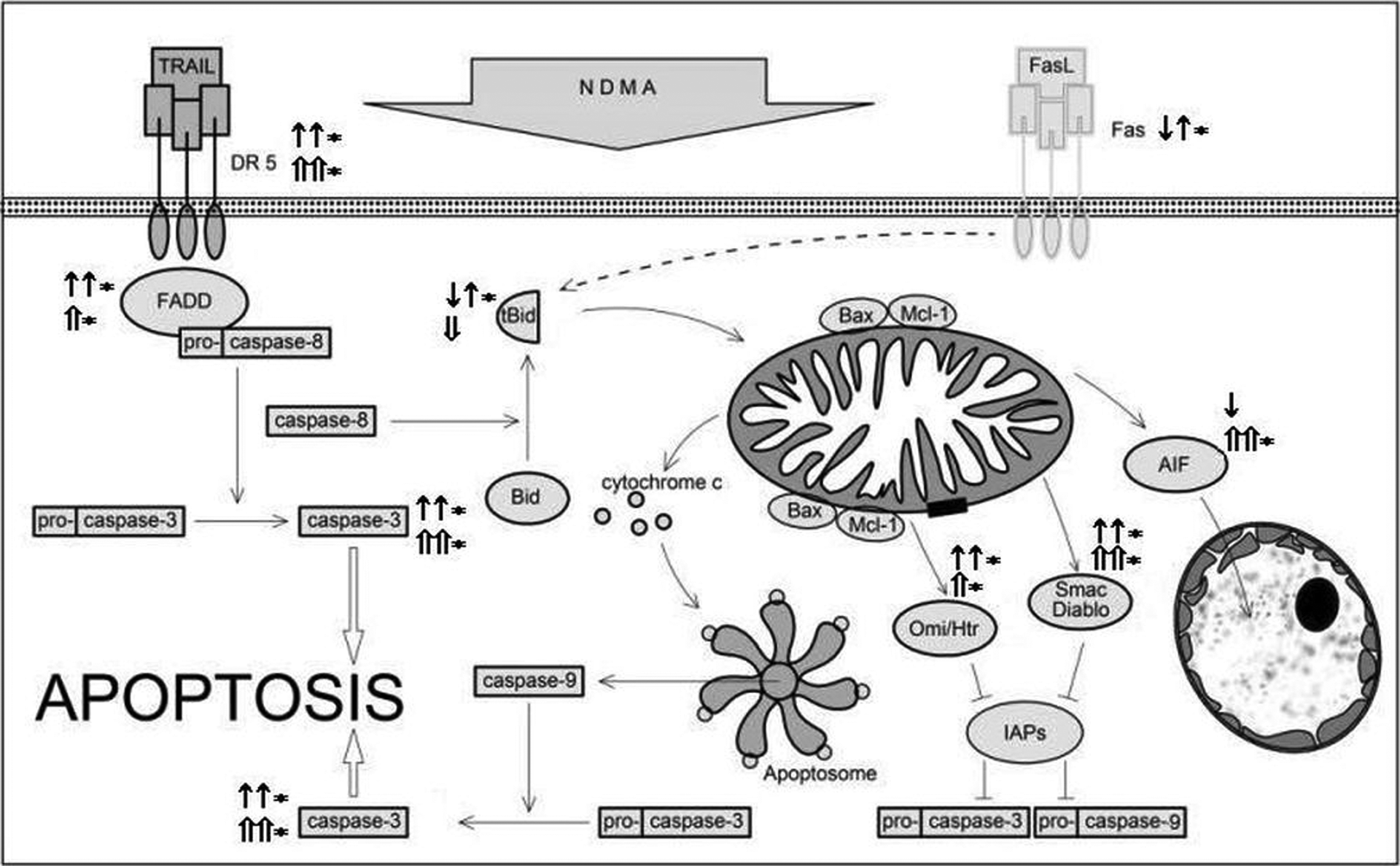
The effect of NDMA on the expression of selected proteins of the extrinsic and intrinsic pathways of apoptosis in human neutrophils (according to Simon, 1 Maianski et al., 6 Leber et al., 10 Van Loo et al., 13 Luo and Loison, 17 and Geering and Simon 20 ). ↑: An increased expression of the apoptosis proteins in PMNs exposed to NDMA. ↑*: An increased expression of the apoptosis proteins in PMNs exposed to the NDMA + rhTRAIL. ↓: A decreased expression of the apoptosis proteins in PMNs exposed to NDMA. ⇑: An increased expression of the apoptosis proteins in PBMCs exposed to NDMA. ⇑*: An increased expression of the apoptosis proteins in PBMCs exposed to NDMA + rhTRAIL. ⇓: A decreased expression of the apoptosis proteins in PBMCs exposed to NDMA. PMNs: polymorphonuclear neutrophils; NDMA: N-nitrosodimethylamine; PBMCs: peripheral blood mononuclear cells; TRAIL: tumor necrosis factor-related apoptosis-inducing ligand.
The death signal induced in the extrinsic pathway, for example, via TRAIL/DR5, can be enhanced through the mitochondria. 26,27 The strengthening of the signal is related to the ability of caspase-8 to proteolytically cleave Bid, a cytosolic protein, into tBid. The proapoptotic tBid, a member of the Bcl-2 family, transfers to the mitochondria where it enables other proteins of the same family to integrate with the outer mitochondrial membrane, create pores, and release, among other things, such as cytochrome c. 28,29
Earlier own research has shown significant effects of NDMA on decreasing the lifespan of neutrophils depending on changes in protein expression in the intrinsic pathway, that is, high expression of the proapoptotic Bax protein and low expression of antiapoptotic Mcl-1 protein. 15 One of the effects of Bax may be the increase of expression of apoptogenic molecules such as Omi/HtrA2, Smac/Diablo, or AIF, observed in the presented research. The integration of Bax into the mitochondrial membrane is one of the main factors enabling the creation of its mega channels, through which the mitochondria release apoptogenic agents. 30,31 Our demonstration of the increased expression of caspase-3 in neutrophils confirms the elevated expression of apoptogenic factors (Figure 5).
Hebels et al. in their research have also observed that exposing human blood cells to nitrosamines, including NDMA, causes the activation of a caspases cascade, showing an increased expression of genes encoding caspase-8 to the initiator. 32
The presented results confirm the significance of NDMA in the induction of apoptosis involving both the extrinsic and intrinsic pathway. Moreover, the results suggest a different course of activation of apoptosis in neutrophils and in mononuclear cells, signified by the differences between the numbers of apoptotic cells exposed to NDMA, and the expression of apoptogenic factors and caspase-3 in PMNs and PMBCs. Based on the obtained results we can assume that neutrophils, compared to mononuclear cells, are more susceptible to apoptosis activation via the intrinsic pathway. These observations suggest a different character of NDMA-induced apoptosis activation in PMNs and PMBCs, and confirm the complexity of their programmed death.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
