Abstract
Transient receptor potential channels have responsibilities in many cellular processes such as cytokine production, cell differentiation, and cytotoxicity by affecting intracellular cation levels or intracellular signal pathways. Multiple sclerosis is a chronic autoimmune central nervous system (CNS) disease caused by environmental and genetic factors. In this study, we aim to investigate TRPV1-TRPV4, TRPM2, TRPM4, TRPM7, TRPC6, and TRPA1 mRNA expression levels, which are associated with the inflammatory process, in the peripheral blood mononuclear cells (PBMCs) of relapsing-remitting multiple sclerosis (RRMS) patients. Thirty-five healthy controls and age–gender matched thirty patients with RRMS were involved in the study. TRPC6, TRPA1, TRPM2, TRPM4, TRPM7, TRPV1, TRPV2, TRPV3, and TRPV4 PBMCs mRNA expression levels were determined by qPCR. In the present study, the TRPC6, TRPM7, TRPV1, TRPV3, and TRPV4 mRNA expressions of RRMS patients in PBMCs decreased at a significant level compared to the healthy control group (p = .000, p = .000, p = .044, p = .000, p = .004, respectively). The decreased expression of TRPC6, TRPM7, TRPV1, TRPV3, and TRPV4 in PBMCs may be associated with the pathogenesis of MS. Further studies are required to understand the mechanism of the relation between these TRP channels and MS and other autoimmune diseases.
Keywords
Introduction
Multiple sclerosis is a chronic autoimmune central nervous system (CNS) disease triggered by environmental and genetic factors. There are more than two million MS patients in the world. 1 The pathological process in MS involves the disruption of the blood–brain barrier, multifocal inflammation, demyelination, oligodendrocyte loss, reactive gliosis, and neuroaxonal degeneration. 2 It includes clinical symptoms such as muscle weakness, blurred vision, vertigo, fatigue, and balance problems that may change depending on the load and location of the lesion. 3 In MS, microglial activation is accompanied by blood–brain barrier disturbance and lymphocyte and macrophage infiltration. 4 The success of clinical trials that target the immune molecules or specific cell types in MS strengthens the idea arguing that the immune system plays significant roles in MS. There is strong proof showing that certain immune pathways are associated with the pathogenesis of the disease. 5
The change in intracellular Ca2+ concentration plays an important role in controlling the inflammation and regulating the immune cell function. Intracellular Ca2+ flow plays key roles in the activation and proliferation of lymphocyte and in cytokine production. 6 Transient receptor potential (TRP) channels are Ca2+-permeable non-selective cation channels. 7 As a result of previous studies, the presence of six subfamilies and 28 superfamilies of TRP channels was shown in mammals. These subfamilies are (1) TRP canoncial (TRPC) with seven different subchannel groups, (2) TRP vanilloid (TRPV) with six different subchannels, (3) TRP polycystin (TRPP) with three different subchannels, (4) TRP mucolipin (ML) with three different subchannels, (5) TRP ankyrin (TRPA) with one different sub-channel, and (6) TRP melastatin (TRPM) with eight different sub-channel groups. 8
TRP channels have been expressed in immune cells. These channels play active roles in many cellular processes like cytokine production, cell differentiation, and cytotoxicity by influencing intracellular cation levels or intracellular signal pathways. TRP channels have roles in the formation of several pathological conditions with the effect of the immune response. 9 Previous studies reported that some TRP channels accelerate the inflammatory process, while several others suppress it. 10
In this study, we investigated TRPV1-TRPV4, TRPM2, TRPM4, TRPM7, TRPC6, and TRPA1 mRNA expression levels, which are associated with the inflammatory process in the peripheral blood mononuclear cells (PBMCs) of relapsing-remitting multiple sclerosis (RRMS) patients.
Materials and methods
Subjects
The study protocol was approved by the local ethics committee (2017-KAEK-189-2019.01.02-04). This prospective and cross-sectional study included 30 RRMS patients (17 females and 13 males) and 35 healthy controls (20 females and 15 males) aged 24 to 58 who applied to the neurology clinic. The sample size of the study was determined using statistical power analysis. The disease duration, frequency of attacks, and drug use of the patients were recorded. Those with other diseases in RRMS were excluded from the study. In the study, seven patients did not receive prophylactic treatment, while the other seven patients received second-line treatment (natalizumab and fingolimod); and 16 patients received primary-line treatment (dimethyl fumarate, teriflunomide, and interferon). The control group consisted of healthy, body mass index (BMI)– (kg/m2), age-, and gender-matched volunteers.
Neurological examination of all patients was performed, and disabilities were defined in line with the Expanded Disability Status Scale (EDSS) score and by employing scores for functional systems. Disabilities were measured with the EDSS, which is an ordinal scale of 0.0–10 (from “no disability” to “death due to MS”), with higher scores, which reflected the greater level of disability. 11 The RRMS was defined in line with the revised McDonald criteria (2010) and with the attacks or steroid use in the month prior to the interviews. Patients with a known history of metabolic diseases (diabetes mellitus, hypertension, etc.), peripheral neuropathy, acute-chronic infection, collagen tissue disease, cardiac-thyroid-lung diseases, renal failure, and liver diseases were excluded from the study.
PBMCs isolation
Peripheral blood samples were collected in EDTA vacutainer tubes following the informed consents of the patients and healthy subjects, and PBMCs were isolated using lymphocytes’ separation media (Capricorn Scientific, Ebsdorfergrund, Germany) by gradient centrifugation (400g, 30 min) and the PBMCs’ layer was harvested with a pipette. PBMCs were repeatedly washed with phosphate-buffered saline. Later, ambion RNA was added to the pellet, and samples were then frozen at −80°C.
Real-time RT-PCR
Total cellular RNA was purified using a TRIzol reagent by following the manufacturer’s instructions (Tri Pure Isolation Reagent, Roche Diagnostics, Mannheim, Germany). The samples were then reverse transcribed into cDNA templates as recommended by the manufacturer (Transcriptor First Strand cDNA Synthesis Kit, Roche Diagnostics, Mannheim, Germany). For real-time RT-PCR, cDNA (500 ng) was amplified (45 cycles) using LightCycler 480 SYBR Green I Master (Roche Diagnostics, Mannheim, Germany) on the LightCycler 480 platform (Roche Diagnostics). Primers were designed using NCBI primer-BLAST. A list of PCR primers for TRP channels and internal reference gene (β-actin) is given in Table S1. The cycle conditions were 95°C for 5 min, followed by 45 cycles at 95°C for 10 s, 60°C for 30 s, and 72°C for 5 s. At the end of the cycles, a cooling step at 40°C was performed for 10 s for each reaction. All runs included one negative cDNA control consisting of DNase- and RNase-free water. Target genes’ expression relative to control expression was calculated by LightCycler 480 software. TRP channels’ mRNA expressions were analyzed relative to the expression of the housekeeping genes β-actin in each run, and final differences in expression levels were calculated using the ΔΔCt method. The relative mRNA expression was expressed as fold change with regard to the average control gene expression. A difference was considered significant at p < .05.
Statistical analysis
The study data were analyzed statistically using the SPSS 20 software package (IBM Corporation, Armonk, NY, USA). The nonparametric Mann–Whitney U-test was used for the comparison between the groups that had non-normally distributed data. The independent samples t-test was used to compare the normally distributed data among the groups. The Kruskal–Wallis test and Mann–Whitney U-test were used to compare subgroups of MS patients (no prophylactic treatment, primary-line treatment, and second-line treatment) with the control group. The arithmetic mean ± standard deviation (SD) and standard error of mean (SEM) was employed to define the quantitative data. We used SD for biochemical and demographic data such as age and sex. Since the PCR analysis was performed in three replicates, we used the SEM value, which is the average of these replicates. A difference was considered significant at p < .05. Spearman’s correlation analysis was used for correlation analysis of non-normally distributed data.
Results
Baseline clinical and laboratory characteristics.
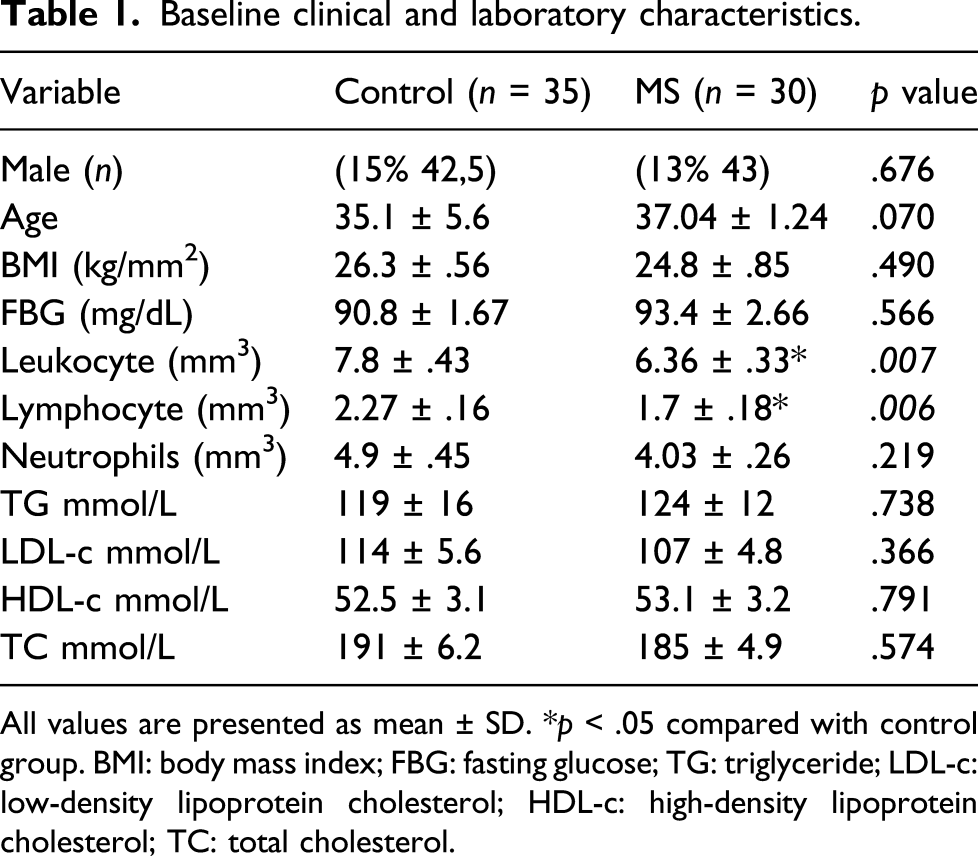
All values are presented as mean ± SD. *p < .05 compared with control group. BMI: body mass index; FBG: fasting glucose; TG: triglyceride; LDL-c: low-density lipoprotein cholesterol; HDL-c: high-density lipoprotein cholesterol; TC: total cholesterol.
In this study, it was observed that there were some significant changes in the TRPC6, TRPA1, TRPM4, TRPM2, TRPM7, TRPV1, TRPV2, TRPV3, and TRPV4 gene expression levels in the control and RRMS groups (Figure 1). The TRPC6, TRPM7, TRPV1, TRPV3, and TRPV4 gene expression levels decreased significantly in the RRMS group when compared to the control group (p = .000, p = .000, p = .044, p = .000, p = .004), respectively. There was no statistically significant difference between the RRMS and control groups in terms of TRPV2, TRPM2, TRPM4, and TRPA1 gene expression levels (p > .05). The levels of mRNA expression were compared in the control group separately with the patients receiving first-line treatments, second-line treatments, and without prophylactic treatment. TRPV1, TRPV3, TRPV4, TRPM7, and TRPC6 mRNA expression levels of the patients receiving first-line treatments, second-line treatments, and without prophylactic treatment were significantly lower compared to the control group. Therefore, it was confirmed that changes in mRNA expression levels in the MS group were not due to the treatments. TRP channel mRNA levels (fold change) in PBMCs of multiple sclerosis and controls. The bars represent means ± SEM (n = 35 for control and n = 30 for the patient group). The data were analyzed by the Mann–Whitney U-test and significant main effects were indicated as follows: (*) p ≤ .05, (**) p ≤ .01, (***) p ≤ .001, and (****) p ≤ .0001, ns: non-significant.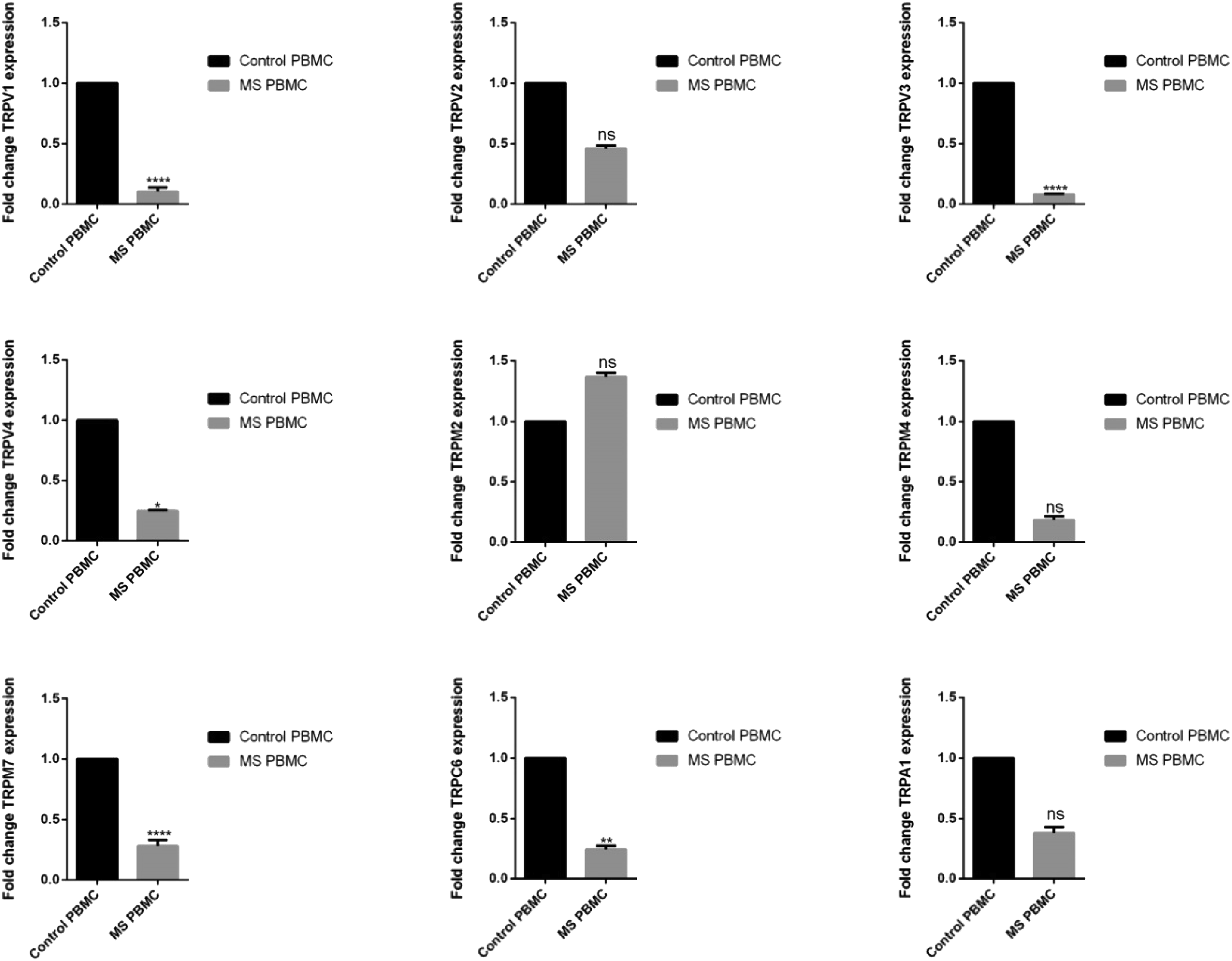
In the study, it was found that there were negative correlations between the TRPM2 (r = −568, p = .001) and TRPV4 (r = −384, p = .036) mRNA levels and the EDSS score (Figure 2). A positive correlation was found between the TRPV1 (r = 363, p = .006), TRPV3 (r = 498, p = .000), TRPM7 (r = 307, p = .023) mRNA levels, and lymphocyte count (Table 2). There was also a positive correlation between the TRPV3 level and leukocyte count (r = 446, p = .001) and the TRPM7 and leukocytes count (r = 338, p = .012). Correlation between TRPM2 and TRPV4 gene expression and EDSS score in the patient group. Spearman’s correlation analysis was used to evaluate the correlation of the data. Correlations of TRP genes with lymphocytes and leukocytes in the patient and control groups.

Discussion
MS is an immune-mediated, inflammatory, demyelinating, and neurodegenerative disease of the CNS. 12 The infiltration of immune cells into the CNS causes further inflammation and damage. The most important cells that infiltrate the tissues in MS are the T-lymphocytes. The tissue infiltration of the B-cells and plasma cells is also present in smaller amounts. 13 Meanwhile, the antibodies that are produced by the B-cells against the myelin sheath are also involved in the inflammatory process. 14 The release of the proinflammatory cytokines from the activated lymphocytes increases the activation of the microglia and macrophages even more, which leads to increased phagocytic properties of the microglia and macrophages, and to the production of various cytotoxic agents like reactive oxygen species (ROS) and reactive nitrogen species (RNS). 15 For the success of the treatment in MS, immunomodulatory applications in RRMS, which is the initial stage of the disease, are very important because neurological deficits increase more in the further stages of the disease. 16
Ion channels, which play roles in nerve conduction in myelinated axons, intensify in the nodes of Ranvier. With the help of the ion channels here, the action potential is transferred from one node to the other with less energy consumption. In MS, the same ion channels in the demyelinated axons cause that dangerous inflammation accelerating neurodegeneration occurs. 17 Intra-axonal sodium and calcium deposition end up with the activation of the proteolytic enzymes, ROS and RNS production, and finally, with mitochondrial damage and bioenergetic failure. 18
The TRP channels are non-selective cation channels. The evidence obtained in the recent years on the dysfunction of the TRP channels demonstrates that they are associated with cardiovascular, neurological, metabolic, or neoplastic disorders. 19 The change in the intracellular [Ca2+] level is the basic mechanism in the regulation of the inflammation and immune cell function. Since the TRP channels are the cation channels supporting the Ca2+ permeability, it may be considered that the TRP channels—together with the other important molecular pathways—might contribute to immune and inflammatory responses. 9
The immunoregulatory properties of the TRPV1-TRPV4 channels activated by heat, PH, and lipoxygenase metabolites of arachidonic acid, were identified.20,21 Different data were also presented on the effect of the TRPV1 activation. In some studies, it was reported that TRPV1 activation had a pro-inflammatory effect, while in some other studies, it was reported that it had protective properties. 22 Fernandes et al. 23 showed that the systemic inflammatory response increased in the TRPV1-knockout mice with sepsis. Another study reported that the disruption in the blood–brain barrier and immune cell infiltration decreased in TRPV1 KO mice, and meanwhile, a correlation was found between the TRPV1 expression and demyelination in the EAE model. 24 Billeter et al. 25 showed that TRPV1 activation decreased the level of inflammatory cytokines. Furthermore, in another study, it was reported that TRPV1 inhibition had an anti-inflammatory effect.26,27 Du et al. was observed that TRPV1 expression decreased in damaged nerves. 28 Similarly, we found that TRPV1 gene expression was decreased in PBMCs of MS patients.
It was shown that TRPV3 transgenic mice developed spontaneous dermatitis. 29 It was reported that the activation of the TRPV3 results in the release of the inflammatory mediators.29,30 In studies that were conducted on TRPV3, it was observed that the decrease in the TRPV3 expression levels resulted in inflammatory reactions in the skin. 31 In our study, the TRPV3 mRNA expression was lower in RRMS patients when compared to that of healthy individuals. In our previous study, TRPV3 gene expression was decreased in PBMCs of psoriasis patients. 32 Similarly, in this study, the TRPV3 mRNA expression was lower in RRMS patients when compared to that of healthy individuals.
In research conducted with TRPV4, whose physiologic role has not been fully understood, different results that seem to contradict each other have been observed. 33 For instance, while the inhibition of TRPV4 has been reported to reduce neuron degeneration and cause an anti-inflammatory response, the activation of it causes an inflammatory response and neurodegeneration.21,33 On the other hand, in different studies, findings were the exact opposite. For example, in our previous study, we discovered that the TRPV4 expression level in the PBMCs cells decrease in patients with psoriasis, an autoimmune disease. 32 In another study, TRPV4 overexpression and pharmacological activation were observed to increase the neurotigenesis during neuronal development. It has also been reported that there is a relation between TRPV4 channels and axonal growth. 34 It is known for a fact that the mutations of the TRPV4 genes are related to various axonal neuropathies. 35 This could support our idea that in the patient group of our study, the decrease in the TRPV4 expression and the negative correlation between EDDS score and TRPV4 gene expression may possibly be related to the axonal degeneration in MS disease.
The densely expressed TRPM2 channels in the immune cells and in the brain tissue are sensitive to oxidative stress. 36 Furthermore, one study showed that TRPM2 deletion and TRPM2 inhibition improved the clinical symptoms in the EAE model. In the same study, it was reported that TRPM2 deficiency did not change the T-cell infiltration into CNS and decreased the CXCL2 level. 37 In the present study, although the TRPM2 mRNA expression increased in the PBMCs in the RRMS group, it was not significant. It has been shown in previous studies that TRPM2 channels play a role in synaptic plasticity. 38 In the study conducted by Li et al., it has been observed that the TRPM2 level in mice’s cerebral cortex increased when they were exposed to heat stress. In the same study with TRPM2-deficient mice, the neural stem cell (NSC) proliferation was shown to reduce. 39 Based on these findings, it can be said that TRPM2 level increases under stress, but in coping with stress, it contributes to the protection of homeostasis. This deduction supports the idea in our study that there is a negative correlation between TRPM2 gene expression level and EDDS score.
The TRPM7 is an ion channel, which is associated with the protein and neurotransmitter release in synaptic vesicles in the neurons. 40 Kamermans et al. 41 reported that the expression of TRPM7 increased in the reactive astrocytes in the MS lesions, and in the primary astrocytes in chronic inflammatory conditions. It has been shown that in cells with low levels of TRPM7 channels, growth and proliferation slows down. 42 In animals with TRPM7 knockdown, learning and memory disorders, and reduction in synapse density and plasticity have been observed. 43 In another study, it was shown that the neuron development is important in axonal and maturation. 44 It was stated that TRPM7 mutations cause motor deficits. 45 In our current study, TRPM7 gen expressions reduced in the PBMCs of MS patients in line with literature.
The TRPC channels are widely expressed in neuronal tissues and immune cells. It was found that TRPC6 channels play roles in immune cell activation, adhesion, and cytokine and chemokine release. 46 It was shown that the TRPC6 mRNA levels in peripheral leucocytes decreased in Alzheimer’s disease, which is in correlation with the impairment in cognitive functions. 47 In another study, it was found that both the TRPC6 mRNA and protein expression in the brain tissue and in the peripheral leucocytes decreased in patients who had Alzheimer’s disease. 48 It was shown that the TRPC6 downregulated in the hippocampus of chronic epileptics 49 and in the rats exposed to stress. 50 In the present study, it was found that the TRPC6 mRNA level decreased, which is similar to the results of other studies.
None of the patients that participated in the study was in attack period or had active inflammation tables. The level of leukocytes and lymphocytes in the patient group was low compared to the control group. The decrease of leukocyte and lymphocyte levels in the patients may be a result of receiving long-term protective treatment. In our study, we also do not believe that the correlation we found between TRPV1, TRPV3, TRPM7, and leukocyte and lymphocyte is related to the inflammation in patients. After all, this correlation included not only MS patients but also the control group. In our study, we divided the patients into subgroups to prove that the change in the expression of TRP channels in the MS group was not caused by the drug treatment administered to the patients (receiving first-line treatments, second-line treatments, and without prophylactic treatment). When we compared the expression of the TRP channels of these subgroups with the control group, the results in treated and untreated patients were similar.
In conclusion, we discovered in this study for the first time that TRPV1, TRPV3, TRPV4, TRPM7, and TRPC6 mRNA expression in the PBMCs of RRMS patients reduces. When we analyzed the gene expressions of TRP channels in PBMCs of the autoimmune disease psoriasis patients before, we got results similar to this study. 32 The extent of our study is limited because there is a need for more extensive research in order to understand the mechanisms behind these changing gene expressions. Besides, the fact that the patients in our study are in the RRMS stage, the early stage of MS disease, is also a limitation. More extensive studies with all different stages of MS and with more participants could be more enlightening. Research that analyzes the protein levels in MS patients’ PBMCs and TRP channels in the brain tissue can be very useful for us to confirm the data of our study.
Supplemental Material
sj-pdf-1-het-10.1177_09603271211043476 – Supplemental Material for Selected transient receptor potential channel genes’ expression in peripheral blood mononuclear cells of multiple sclerosis
Supplemental Material, sj-pdf-1-het-10.1177_09603271211043476 for Selected transient receptor potential channel genes’ expression in peripheral blood mononuclear cells of multiple sclerosis by Murat Çakır, Hikmet Saçmacı and Seda Sabah-Özcan in Human & Experimental Toxicology
Footnotes
Acknowledgments
We thank Yozgat Bozok University Microbiology Laboratory and Professor Neziha Yılmaz for contributions to this study.
Author contributions
MÇ and SSO conceived of the presented idea. HS was involved in collecting blood samples and clinical data. SSO carried out the experiments. MÇ, SSO, and HS performed the analyses. MÇ took the lead in writing the article. All authors discussed the results and contributed to the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
