Abstract
Current human immunodeficiency virus treatments need to be periodically administered lifelong. In this study we assess the effect of repeated doses of an anti-HIV peptide drug candidate in C57BL6 strain. Two schemes of up to 15 administrations and one of 30, daily dosing for 5 days per week, all by the subcutaneous route were evaluated. Different dose concentrations of the peptide were assayed. CIGB-210 treated animals showed no symptoms or abnormal behavior as compared with placebo. All the animals gained weight during the study. Macroscopic evaluation showed no alterations in any of the organs studied. Microscopic analysis of the tissues did not show morphological changes in thymus, stomach, small and large intestines, kidney, brain, or cerebellum. The proliferative response of splenocytes and their capacity to secrete gamma interferon were not compromised by the repeated administration of CIGB-210. There were not statistically significant differences for any of the parameters evaluated during the study among treated and non-treated groups. We can conclude that CIGB-210 is well tolerated in C57BL6 mice in the dose concentration range explored and merits subsequent toxicological studies.
Introduction
The human immunodeficiency virus (HIV), the causative agent of AIDS, is still one of the main scourges for mankind. According to the Joint United Nations Programme on HIV/AIDS (UNAIDS) 2021, 37.6 million people lived with the disease, 1.5 million of them became newly infected and 690,000 died from AIDS-related illnesses in 2020. 1 HIV/AIDS is still one of the major causes of death worldwide and specially in developing countries. 2
HIV/AIDS is characterized by a profound immunosuppression followed by the onset of opportunistic infections, associated neoplasias, and degeneration of the central nervous system. 3 Antiretroviral therapy (ART), through the combined use of two, or three HIV inhibitors, is the recommended treatment for all persons living with AIDS (PLWA).4,5 ART has greatly improved the survival and the quality of life of PLWA and reduced the number of deaths due to AIDS.6,7,8
However, the prolonged use of ART is still limited by the appearance of drug resistant mutants.9,10 Resistant HIV isolates have been found for every one of the ART drugs in the clinic and even in ART naive patients.11,12 Other limitations of ART are its incapacity to completely eliminate the virus from the organism (life time use) and the elevated cost of these medicines, which still limits the number of PLWA with full access to ART, especially in developing countries.13,14
The antiretroviral drugs currently approved for clinical use target different viral proteins and only two of them acts on a cellular protein.11,15,16 The need to keep improving the efficacy of ART has led to novel biological approaches in the quest for new drugs with antiviral activity. One of these approaches is the use of new host proteins involved in HIV replication as targets for antiviral drugs development. 11
Our group has identified vimentin as one of the cellular proteins involved in HIV replication. Vimentin is the main intermediate filaments (IF) forming protein in mesenquimal cells. Vimentin knockdown MT4 cells are refractory to HIV replication. 17 A 18 mer synthetic peptide derived from the human keratin 10 was first reported to disassemble vimentin intermediate filaments in human fibroblasts after intracellular microinjection. 18 Experiments carried out in our laboratory have shown that exogenous incubation of this peptide, entitled as CIGB-210, in MT4 cells triggers a redistribution of the vimentin IF. 17 CIGB-210 is capable to inhibit HIV replication in MT4 cells at the nanomolar range and exhibits a very low toxicity profile in this cell line.
The safety evaluation in animals is one of the most important stages during the development of a new drug candidate. 19 In particular, for an anti-HIV drug, it is extremely important to rule out any possible toxic effects on the immune system since it is the main target for the virus replication and pathogenicity. Additionally, CIGB-210 acts on the vimentin network being vimentin the major IF forming protein in lymphocytes and monocytes, the main targets of HIV replication.
The aim of this work was to preliminary assess the general safety of CIGB-210 in C57BL/6 mice. The specific goals were to look for the presence of systemic adverse reactions after multiple administrations of the product, to examine different organs for pathological findings that could be associated with the administration of this peptide, and to study the effect of CIGB-210 on the Concanavalin A (ConA) induced proliferation of splenocytes ex vivo, as well as the capacity of ConA activated splenocytes to secrete gamma IFN.
Materials and methods
Chemicals and solutions
RPMI 1640 medium (Hyclone, Switzerland); gentamicin (Sigma-Aldrich, USA); ammonium chloride (NH4Cl, 0.83%) (Sigma-Aldrich, USA); fetal bovine serum (FBS) (PAA, Canada); Concanavalin A (ConA) (Sigma-Aldrich, USA); carboxyfluorescein diacetate succinimidyl ester (CFSE) (Molecular Probes, United Kingdom); 3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) (Sigma-Aldrich, USA); anti-murine IFN gamma monoclonal antibody (MAb) AN18 and biotin-conjugated anti-murine IFNγ MAb R4-6A2 (Mabtech, Sweden); HRP-conjugated streptavidin (Amersham, United Kingdom); amino ethyl-carbazol (AEC) (Sigma-Aldrich, USA); hydrogen peroxide (H2O2) (Merck, Germany). Phosphate buffer saline (PBS), 137 mM NaCl; 3.8 mM KCl; 8.1 mM Na2HPO4; 1.5 mM KH2PO4, pH 7.2; formaldehyde 2% in PBS; formaldehyde 4% in PBS; PBS-Tween 0.05% and Trypan blue solution 0.4% (0.85 g NaCl; 0.4 g Trypan blue in 100 mL of H2O) (Gibco, USA).
CIGB-210
CIGB-210 is an 18 mer synthetic peptide derived from human keratin 10. The sequence is RVTQMNLNDRLASLYDKV; single letter amino acids code and its molecular weight is 2134.21 Da. It was synthesized at the Peptide Synthesis Department of CIGB, Havana, Cuba. Synthesis was performed manually on MBHA resin by stepwise solid-phase procedure using Fmoc/tBu strategy. 20 Peptides were analyzed by HPLC (GE Healthcare, USA).
Analytical separation was achieved in reversed-phase (RP) C18 column (Zorbax, 4.6 x 150 mm, 5 μm). A linear gradient from 5% to 60% of solvent B over 35 min at flow rate of 0.8 mL/min was used. The preparative purification was performed on the HPLC system LaChrom (Merck Hitachi, Germany) in a RP C18 column (Vydac, 25 × 250 mm, 25 μm). A linear gradient from 15% to 60% of solvent B over 50 min at a flow rate of 5 mL/min were used. Detection was accomplished at 226 nm. Solvent A: 0.1% (v/v) TFA inH2O. Solvent B: 0.05% (v/v) TFA in acetonitrile. The identity of the peptide was verified by Electrospray Ionization Mass Spectrometry (ESI-MS) in a hybrid quadrupole-time-of-flight instrument (QTOF1, Waters, USA) fitted with a nanospray ion source. The doses range studied is comprised between 0.64 mg/kg and 3.57 mg/kg. These doses are being considered for the clinical application of CIGB-210 based on their potentialities to achieve CIGB-210 blood concentrations in the nanomolar range, in correspondence to the inhibitory concentration 50 for the peptide. 17
Placebo
Sodium chloride 0.9% saline solution (DELMED, El Salvador) was used as placebo.
Experimental animals
Specific pathogen free C57BL/6 female mice between 8 and 10 weeks old, ranging from 18 to 23 g of weight were used. All animals were purchased from CENPALAB (National Center for Laboratory Animal Production, Havana, Cuba). Animals were allocated according to the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purpose (Strasbourg, Council of Europe). 21 The standard of care required by the US Department of Health and Human Service Guide for the Care and Use of Laboratory Animals, National Research Council; Animal Alternatives, Welfare and Ethics and EUROGUIDE on the Accommodation and Care of Animals Used for Experimental and Other Scientific Purpose was achieved.22–26 All animals were maintained in controlled animal facilities at temperature (21 ± 2°C) and humidity (55–65%), which were monitored daily. Light cycle control was automatic. Animals were adapted during 14 days, and then separated for dosing and food consumption measurement using a random list generated by the software Randomization Program Animals Over Cages, R.F. Marsman Bilthoven, RIVM, The Netherlands (version 94-1), World Health Organization. The Institutional Committee for the Care and Use of Laboratory Animals of the Center for Genetic Engineering and Biotechnology approved all the experimental protocols.
Experimental design
Experiment 1
Mice were randomly assigned to 4 experimental groups of 15 mice each to receive either CIGB-210 in 200 μL H2O or 200 μL placebo by the subcutaneous route. Five animals from each group received 5 inoculations in 1 week, the other 5 received 10 inoculations in 2 weeks and the other 5 received 15 inoculations in 3 weeks (daily administration from Monday to Friday). Animals in group 1 received placebo while groups 2, 3 and 4 were injected with 0.71 mg/kg, 1.29 mg/kg, and 2.57 mg/kg of CIGB-210, respectively. Five mice were euthanized 1 day after the fifth, tenth or fifteenth injections and their organs were processed as described later. Spleen cells were isolated after 15 inoculations for proliferation studies.
Experiment 2
The second experiment also included four experimental groups of 10 C57BL6 mice each one. Animals in group 1 received placebo while groups 2, 3 and 4 were injected with 0.64 mg/kg, 1.51 mg/kg and 3.57 mg/kg of CIGB-210, respectively. Five animals from each group received 15 inoculations of the peptide in 3 weeks and the other 5 received 30 inoculations in 6 weeks. All mice were euthanized 1 day after the fifteenth or thirtieth and their organs were processed as described later.
Experiment 3
Two experimental groups of 6 animals each were included. One group was treated with 1.51 mg/kg of CIGB-210 and the other one received placebo. Mice received 15 administrations of the peptide, from Monday to Friday during 3 weeks. All mice were euthanized 1 day after the last injection and the spleen was removed to proceed with splenocytes extraction.
Clinical evaluation
Animals in the 3 experiments were observed daily from Monday to Friday for the presence of physical, functional and general clinical signs or changes in behavior. Among the symptoms evaluated were emaciation, inflammation, hyperactivity, lethargy, aggressiveness, salivation, dyspnea, lesions, bleeding, and cyanosis. The inoculation site was monitored for edema, induration or any other sign indicative of a damage caused by the injected substance such as congestion or redness. The body weight was monitored once a week.
Morphological and histopathologic assessment
The study of the organs from mice in experiment 1 was conducted after 5, 10, or 15 administrations of CIGB-210 or placebo, while in experiment 2 it was conducted after 15 or 30 administrations. Mice were anesthetized by CO2 inhalation and subsequently euthanized by cervical dislocation according to the Good Practices for Laboratory Animals. All organs were observed macroscopically. The external characteristics of the organs such as size, surface, consistence, and color were described. Internal features, such as compactness, contents, and brilliance were also assessed after practicing different cuts in the organs. Samples from heart, lungs, mesenteric lymph nodes, thymus, spleen, stomach, small and large intestines, liver, kidney, brain, cerebellum, and thyroid were taken and fixed in 4% formaldehyde for the histological evaluation. A similar procedure was performed to non-treated C57BL6. Fragments from the organs were processed by paraffin inclusion method, stained with hematoxylin-eosin, and observed using optical microscopy at 10X and 40X for histopathological diagnosis (Carl Zeiss, Germany).
Splenocytes isolation
Mice spleen cells were isolated 1 day after the last inoculation through the perfusion method. The spleen was removed from each animal under sterile conditions and was perfused with 10 mL of RPMI 1640 medium supplemented with gentamicin and 5% FBS using a 5 mL syringe. Erythrocytes were lysed by incubation with 0.83% NH4Cl for 10 min at 4°C. The spleen cells were washed 3 times with PBS and centrifuged at 300 x g for 10 min. Cell viability was determined by Trypan blue staining.
MTT proliferation assay for splenocytes
The proliferative response was determined according to the method of MTT. 27 Spleen cells were seeded at 2.5 x 105 cells/mL in RPMI 1640 medium supplemented with antibiotic and 10% FBS in 96 wells flat bottom microplates (Costar, USA). Cells were stimulated with 1 μg/mL ConA and incubated for 72 h at 37°C in 5% CO2 atmosphere and 95% relative humidity. Then 5 mg/mL of MTT were added to each well and plates were further incubated for 4 h in the dark. A solution of isopropanol acid containing Triton X-100 and HCl was used to solubilize the crystals. Plates were stirred at room temperature (RT) for several minutes in the dark to accelerate the solubilization process. Absorbance at 578 nm was measured in an ELISA reader (Multiskan Spectrum Thermo Scientific, Finland). Each sample was evaluated in duplicate. Proliferation index (PI) was defined as the ratio between the optical density in the presence of ConA and the same parameter in the absence of the mitogen.
Carboxyfluorescein diacetate succinimidyl ester titration in splenocytes cultures
To determine the optimal concentration of CFSE28,29 to be used in the proliferation assays 1, 2.5, 5, 7.5, and 10 μM of the reagent were evaluated: A cell culture without CFSE was used as negative control of fluorescence. The cell concentration used was 10 x 106 cells/mL in PBS. Splenocytes were incubated with CFSE for 8 min at room temperature in the dark. The process of labeling was arrested with the addition of FBS. Cells were washed twice with PBS and once with RPMI 1640 supplemented with 5% FBS. Cells were incubated in RPMI 1640 supplemented with 10% FBS at 37°C in 5% CO2 and 95% relative humidity during 7 days.
Carboxyfluorescein diacetate succinimidyl ester proliferation assay for splenocytes
Spleen cells (10 x 106 cells/mL in PBS) were incubated with 5 μM CFSE for 8 min at RT in the dark. The reaction was arrested by adding one volume of FBS. The cells were next washed twice with PBS, one final time with RPMI 1640 medium and were incubated for 16 h at 37°C in 5% CO2 and 95% relative humidity. Afterward, 2 x 106 cells/mL were stimulated with ConA at 5 μg/mL. CFSE marked cells without ConA stimulation were included in the evaluation. Non-marked splenocytes cultured in RPMI were used as negative control. Cells were then cultured for 6 more days under the same conditions. PI was defined as the ratio between the percentage of proliferation in the presence of the mitogen and the percentage of proliferation in its absence.
Detection of IFNγ secreting cells by ELISPOT assay
The enzime-linked immunospot (ELISPOT) assay for the detection of interferon gamma (IFNγ) secreting cells was conducted as previously described. 30 Briefly, Immubilon-P 96 wells plates (Millipore, France) were coated with AN18 anti-murine IFNγ MAb at 5 μg/mL (Mabtech, Sweden) in PBS for 12°h at 4°C. The plates were washed 6 times with RPMI and incubated for 1°h at 37°C, in 5% CO2 atmosphere, and 95% relative humidity. A total of 2 x 105 splenocytes were seeded and stimulated with 10 μg/mL ConA. Plates were incubated for 48 h in 5% CO2 atmosphere and 95% relative humidity. After this incubation period the plates were washed twice with water, followed by 3 more washes with PBS and 5 washes with PBS-Tween. The biotin-conjugated anti-murine IFNγ MAb R4-6A2 was then added and incubated for 2 h at RT. After 3 washes with PBS-Tween, a 1:800 dilution of the HRP-conjugated streptavidin was added for 1 h at 25°C. After additional 3 washes with PBS-Tween the reaction was stained with 10 mg/mL of AEC and 0.05% H2O2 for 5 min. Finally the substrate was discarded and plates were washed with tap water. The plate was allowed to dry in the darkness at RT. The spots were counted in an Eli-Scan reader (AELVIS, Germany) and processed with the Eli-Expert analysis software V5.1. The results are expressed as the number of spot forming cells (SFC) per 106 splenocytes.
Statistical analysis
GraphPad Prism software, version 6.01 (GraphPad Software, USA) was used for all the statistical analysis. The normality of the data was checked using D′ Agostino-Pearson omnibus normality test, Shapiro–Wilk normality test or Kolmogorov–Smirnov test and the variance homogeneity with the Bartlett test or Levene’s test. Student’s t-test was applied to compare the results between two experimental groups. One-way ANOVA was used to look for statistically significant differences among four experimental groups. Mann–Whitney test was used when the data was found to be non-normally distributed. Fisher exact test was applied to determine if there are nonrandom associations between two categorical variables. Statistical differences were considered significant for p < 0.05.
Results
Clinical monitoring of the animals
Body weight of mice receiving 15 inoculations maximum. Experiment 1.
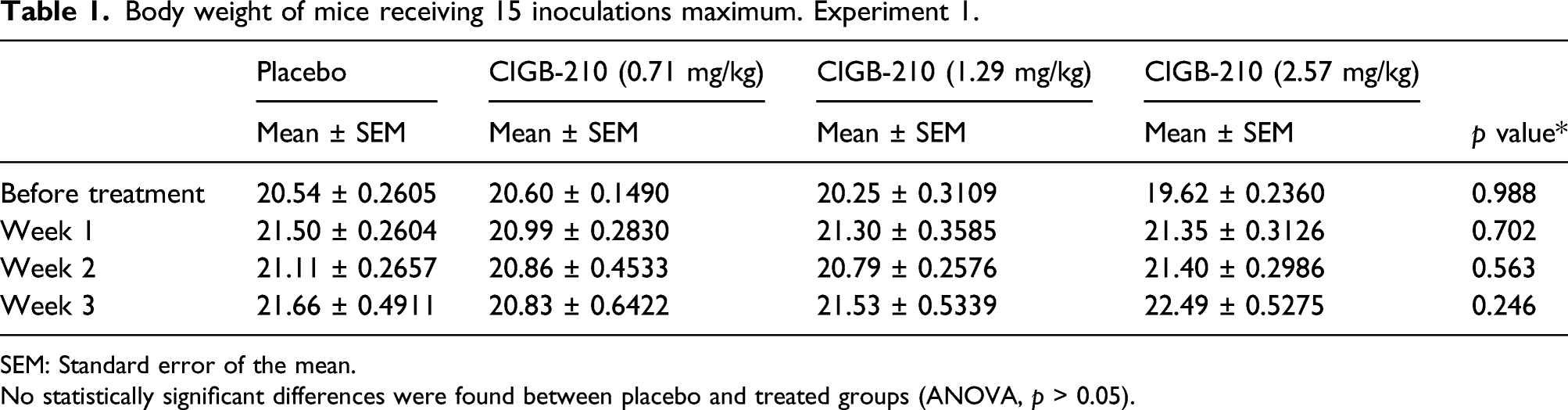
SEM: Standard error of the mean.
No statistically significant differences were found between placebo and treated groups (ANOVA, p > 0.05).
Body weight of mice receiving 30 inoculations maximum. Experiment 2.
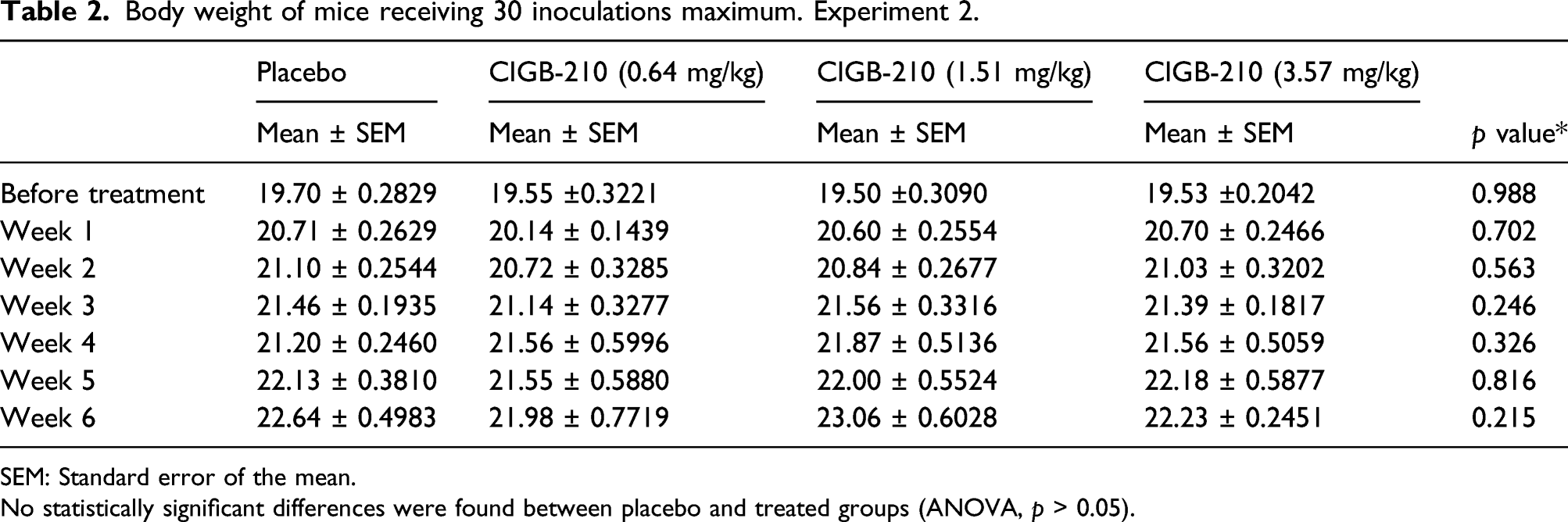
SEM: Standard error of the mean.
No statistically significant differences were found between placebo and treated groups (ANOVA, p > 0.05).
Body weight of mice receiving 15 inoculations. Experiment 3

SEM: Standard error of the mean.
No statistically significant differences were found between placebo and treated groups (Mann–Whitney test, p > 0.05).
Macroscopic evaluation of organs
The macroscopic evaluation of organs from placebo or CIGB-210 treated mice in experiments 1 and 2 showed no alterations of any the organs studied: heart, lungs, mesenteric lymph nodes, thymus, spleen, stomach, small and large intestines, liver, kidney, brain, and thyroid, independent of the number of inoculations or the dose of peptide administered. Even mice that received 30 administrations of the highest dose of the peptide (3.57 mg/kg of body weight) showed no macroscopic alterations in their organs.
Histological analysis
Experiment 1
The histopathological analysis of the tissues collected from different organs showed no alterations in thymus, stomach, small and large intestines, kidney, and brain. Morphological changes were observed in spleen, liver and mesenteric lymph nodes in some mice of the four experimental groups. Changes in heart and thyroid were also observed although in a very low frequency.
The Figure 1 shows representative microphotographs from spleen, and liver sections (Figure 2) from CIGB-210 treated animals showing megakaryocytes in the red pulp of the spleen and focal niches of immature myeloid cells in the liver parenchyma, indicating the presence of extramedullary hematopoiesis in these two organs. Similar findings were found in placebo group. All these events were observed in CIGB-210 treated groups and placebo as early as 5 inoculations. Spleen examination showed megakaryocytes in 15/20, 8/20 and 12/20 animals after 5, 10 and 15 administrations, respectively. Liver analysis evidenced this finding in 11/20 animals after 5 injections and 12/20 after 10 and 15 administrations. The Supplementary Figure 1 shows representative images of extramedullary hematopoiesis and germinal centers in the spleen of non-injected C57BL6. Germinal centers were observed in the cortex of mesenteric lymph nodes and in the periarteriolar lymphoid sheath of the spleen in all the animals from all the groups of the study (Figure 3). No statistically significant differences were found for the most frequently observed findings (Fisher’s exact test, p > 0.05, Supplementary Table). The presence of endocarditis was observed in heart sections of 1 animal from group 2 (0.71 mg/kg of CIGB-210) after 10 inoculations of the peptide. Additionally, 1 animal from group 4 (2.57 mg/kg of CIGB-210) showed signs of focal pericarditis of proliferative nature and prevalence of mononuclear cells after 15 inoculations. One animal from the same group showed focal necrosis in liver after 15 inoculations. Histopathologic analysis also revealed inflammation of the thyroid in one animal from group 2 (0.71 mg/kg of CIGB-210) and group 3 (1.29 mg/kg of CIGB-210) after 15 and 10 inoculations, respectively. Microphotographs of spleen sections of mice from experiment 1 showing megakaryocytes. A. Representative image of placebo treated animals. B: Representative image of CIGB-210 treated animals (2.57 mg/kg). Black arrows indicate the presence of megakaryocytes characteristic of extramedullary hematopoiesis. Hematoxylin-eosin staining; magnification 40X. Microphotograph of liver sections of mice from experiment 1. A. Representative image of placebo treated animals. B: Representative image of CIGB-210 treated animals (2.57 mg/kg). Black ellipses indicate focal niches of immature cells in the myeloid trunk characteristic of extramedullary hematopoiesis. Hematoxylin-eosin staining; magnification 40X. Microphotographs of spleen sections of mice from experiment 1 showing germinal centers. A: Representative image of placebo treated animals. B: Representative image of CIGB-210 treated animals (2.57 mg/kg). Black ellipses show the germinal centers. Hematoxylin-eosin staining; magnification 10X.


Experiment 2
The microscopic observation of sections from different organs showed no alterations in heart, thymus, stomach, small and large intestines, kidney, brain, cerebellum, thyroid, and liver. Morphological changes were observed in the mesenteric lymph nodes, spleen, and lungs in all experimental groups receiving either 15 or 30 inoculations of the peptide
Germinal centers were observed in the cortex of mesenteric lymph nodes in all animals of the study, independent of the treatment received and the number of inoculations. The same finding was observed in the periarteriolar lymphoid sheath of the spleen and megakaryocytes were found in the red pulp of this organ, which is indicative of extramedullary hematopoesis (16/20 animals after 15 inoculations and 20/20 after 30 administrations. These observations were noted in all groups of the study including non-treated and placebo.
There were findings in the lungs after 30 inoculations of either CIGB-210 or placebo that were not previously observed after 15 administrations; perivasculitis and focal pneumonia. The inflammatory process was characterized by a mixed infiltrate of mononuclear cells and neutrophils. The frequency of these alterations differed within groups, perivasculitis was evident in all the groups in study including placebo treated animals, and the lower frequency corresponded to group 4, where only one out of five mice exhibited this morphological alteration (Figure 4). On the other hand, focal pneumonia was found only in groups 1 and 2, and with low incidence: 2 animals in the placebo group and only one in group 2. Microphotograph of lung sections of mice from experiment 2. A. Representative image of placebo treated animals. B: Representative image of CIGB-210 treated animals (3.57 mg/kg). Black arrows indicate the presence of perivasculitis. Hematoxylin-eosin staining; magnification 40X.
No statistically significant differences were determined for the treated groups in comparison with placebo group for the most frequently observed findings (Fisher’s exact test, p > 0.05, Supplementary Table).
Effect of CIGB-210 treatment on the proliferative capacity of lymphocytes
Next, we evaluated if CIGB-210 treatment alters the proliferative capacity of mice splenocytes in the presence of ConA by the MTT colorimetric assay. The cells of the spleens of animals from experiment 1 were analyzed.
The proliferation index calculated for placebo group was 1.60. The proliferation index for the test groups receiving 0.71 mg/kg, 1.29 mg/kg and 2.57 mg/kg of CIGB-210 were 1.72, 1.64 and 1.48, respectively. The statistical analysis showed no significant differences in the mitogen induced proliferation among groups (p > 0.05) (Figure 5). ConA induced ex vivo proliferative response of splenocytes of mice from experiment 1. The graphic shows the proliferation indexes, calculated as the ratio between the proliferation in the presence of ConA and the same parameter in the absence of the mitogen. Bars indicate the arithmetic mean of the proliferation index for each group (n = 5). Error bars represent standard error of the mean. No statistically significant differences among groups were detected (one-way ANOVA; p > 0.05).
In the next experiment, the proliferative capacity of ConA stimulated splenocytes in animals from experiment 3 was evaluated using the CFSE method. The analysis was carried out in the region R1 selected from the forward scatter vs side scatter representation for the splenocytes (Supplementary Figure 2).
Several CFSE concentrations (1 μM, 2.5 μM, 5 μM, 7.5 μM and 10 μM) were explored to select the concentration with optimal balance between fluorescent staining and toxicity. This preliminary assay was carried out in splenocytes isolated from untreated mice. The microscopic visualization of CFSE labeled cells showed fluorescent cells at all the concentrations explored. No fluorescence was detected in splenocytes cultured in the absence of CFSE. The mean fluorescence intensity was measured by flow cytometry (Figure 6). The analysis demonstrated the direct relationship between CFSE concentration and the mean fluorescence intensity. The concentration of 5 μM was the lowest at which the mean fluorescence intensity resulted between 102 and 103 fluorescence units (highest fluorescence range). Therefore, this concentration was selected for further experiments. Flow cytometry histograms of CFSE stained murine lymphocytes. From left to right: controls without CFSE (pale blue lines), 1 μM CFSE (blue lines); 2.5 μM CFSE (violet lines); 5 μM CFSE (pink lines); 7.5 μM CFSE (green lines) and 10 μM CFSE (black lines).
Next we evaluated the effect of CIGB-210 on ConA induced lymphocyte proliferation in placebo controls and the group treated with 1.51 mg/kg of CIGB-210. An increment in the number of proliferating lymphocytes was observed in animals treated with CIGB-210 and placebo in the presence of ConA (Supplementary Figure 3). The mean proliferation indexes calculated for the placebo and CIGB-210 treated group was 10.25 and 9.32, respectively. The statistical analysis showed no significant differences among groups (unpaired Student T test; p > 0.05) (Figure 7). ConA induced ex vivo proliferative response of splenocytes of mice from experiment 3. Murine splenocytes were stained with CFSE and stimulated with ConA. The results show the proliferation index, calculated as the ratio between the frequency of proliferation (%) in the presence of ConA and the same parameter in the absence of the mitogen. Bars indicate the arithmetic mean of the proliferation index of 6 animals per group. Error bars represent the standard error of the mean. No statistically significant differences among group were detected (non-paired Student T Test; p > 0.05).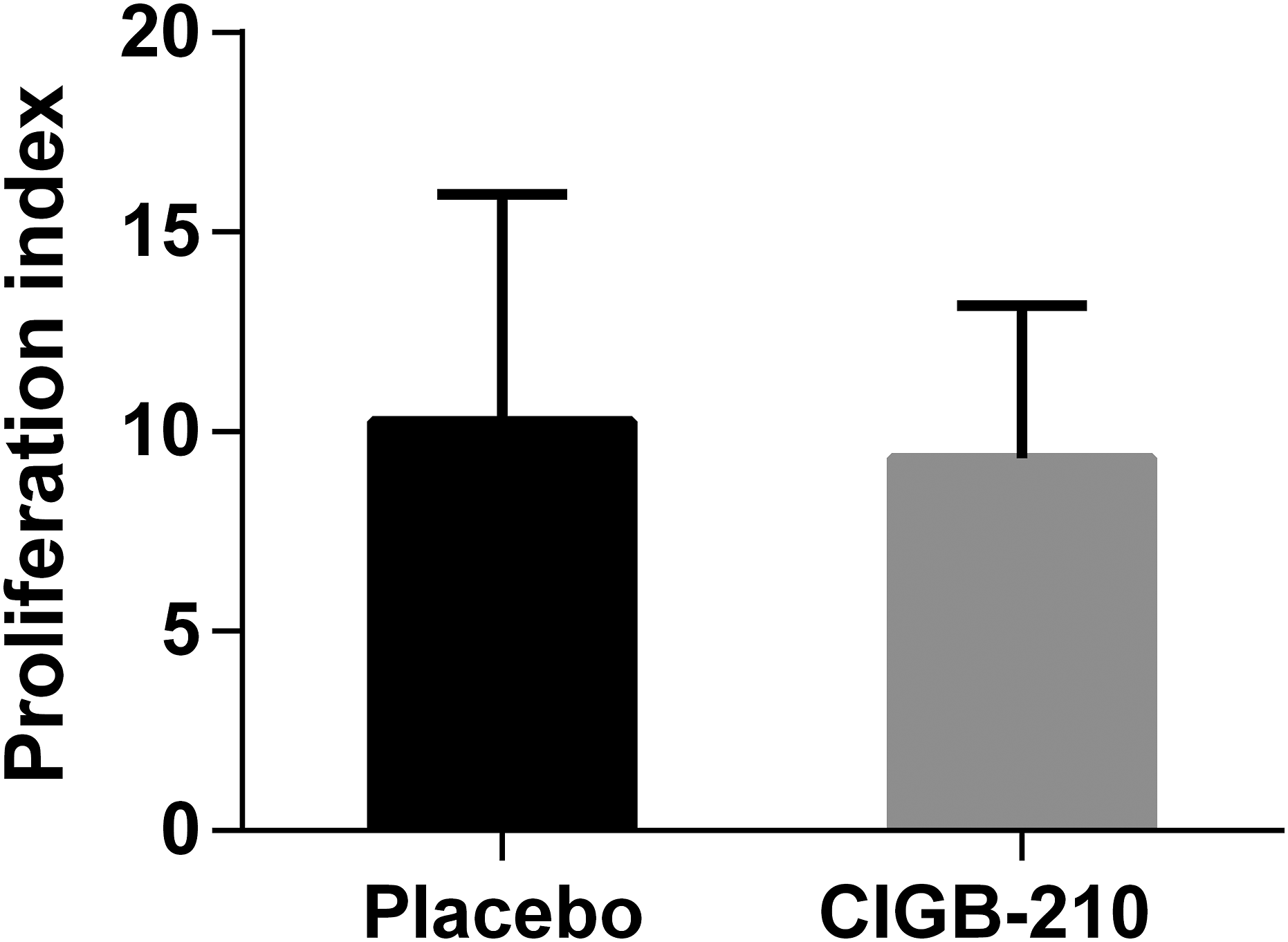
Determination of IFNγ secreting cells in mice treated with CIGB-210
The effect of CIGB-210 treatment on the capacity of murine lymphocytes to secrete IFNγ in response to a mitogen was evaluated by ELISPOT. SFC in response to ConA as indicator of the functionality of cells used in the study were detected in splenocytes culture from placebo and CIGB-210 treated group. Non ConA stimulated cultures showed no IFN-γ production which was indicated by a lack of SFC. The mean SFC per million for the placebo group was 1640 ± 68.66. The mean SFC per million for CIGB-210 treated group was 1676 ± 34.2 (Figure 8). No statistically significant differences were found between both groups (Mann Whitney test; p > 0.05). ConA induced IFNγ secreting response of splenocytes of mice from experiment 3. Murine splenocytes were stimulated with ConA and capacity of cells to secrete IFNγ was evaluated by ELISPOT. Bars represent the mean spot forming cells (SFC)/106 (n = 6). Error bars represent the standard error of the mean. No statistically significant differences between placebo and CIGB-210 treated groups (Mann Whitney test; p > 0.05).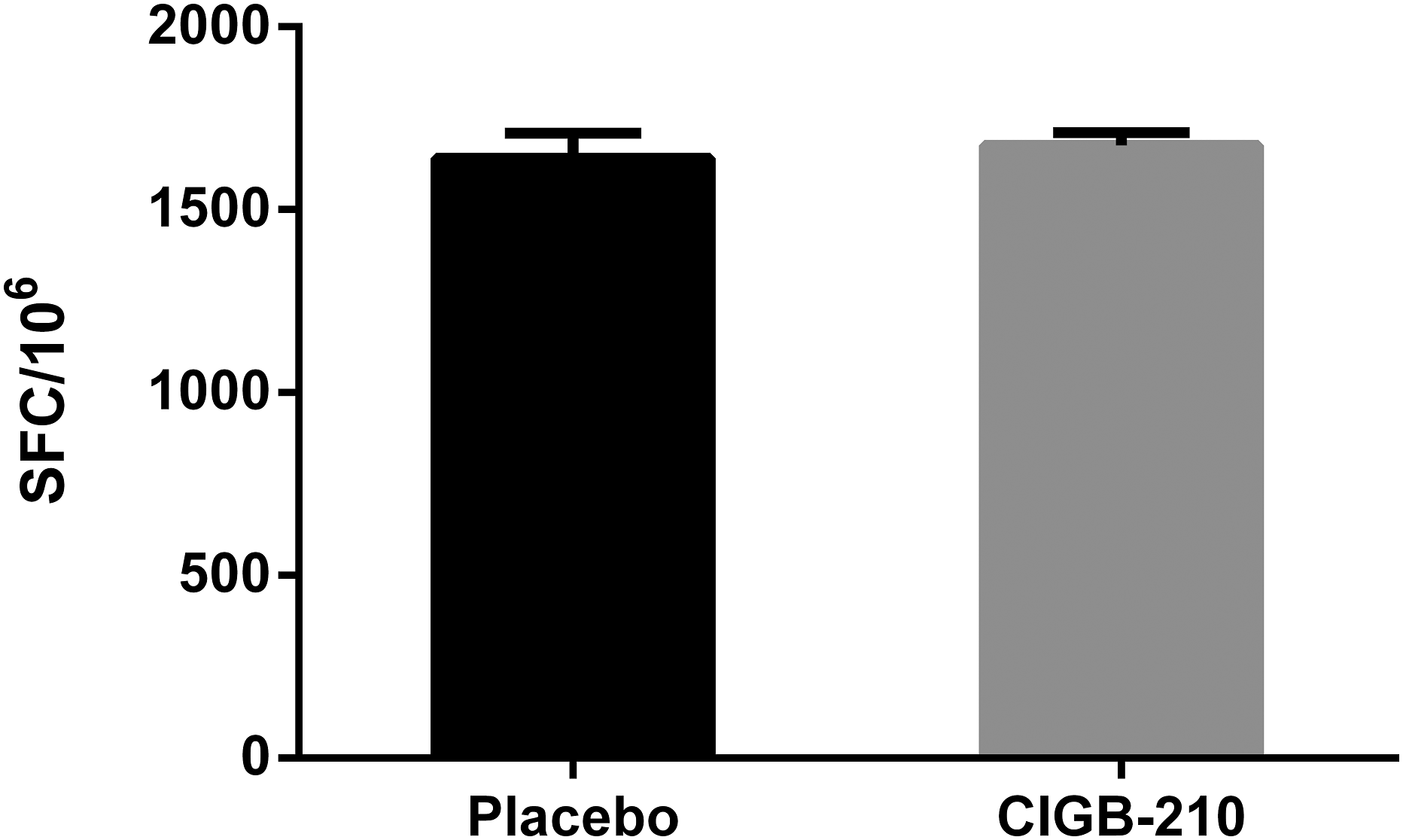
Discussion
CIGB-210 is a peptide drug candidate which inhibits HIV replication. The mechanism of action of this peptide has been related with a redistribution of the vimentin intermediate filaments network in the host cells. 17
The chances that the drug or some of its metabolites could display deleterious effects on cells and organs must be carefully examined in the pre-clinical phase. 31 In the present study, we conducted a preliminary safety assessment of CIGB-210 in mice to evaluate the impact of multi-dose peptide administration in the organism and to study the effect of this peptide in the functionality of lymphocytes.
CIGB-210 was assessed subcutaneously in three independent experiments in C57BL/6 mice. The subcutaneous route was selected as being currently the most common administration for peptides; it avoids the degradation by hepatic or gastrointestinal enzymes, 32 is the least painful of the parenteral administration routes and allows self-medication by the patients.33,34
A potential therapeutic doses range from 0.64 mg/kg to 3.57 mg/kg was evaluated. According to a pharmacokinetic study of CIGB-210 in mice (unpublished data) blood concentrations of CIGB-210 higher than its IC50 could be detected after 4 h of injection, even at the lowest dose.
The peptide was well tolerated and no systemic side effects were documented during 30 administrations. It was considered highly positive the fact that all the animals gained weight during the study and the absence of body weight loss as result of the repeated administration of CIGB-210. Macroscopic observations supported the results from clinical observations and evidenced that there was no damage to any of the examined organs. Microscopic analysis evidenced no alterations in thymus, stomach, small and large intestines, kidney, brain, and cerebellum.
The presence of extramedullary hematopoiesis and germinal centers was considered non-drug related since they were observed in all the experimental groups including placebo and non-treated. We consider these findings as characteristics of the mouse strain and the stress experienced by the animals due to the repeated inoculations programmed for the study.
Perivasculitis was considered independent of peptide administration as presented in all experimental groups with the lowest frequency at the highest concentration. The very low frequency of findings in heart and thyroid along with the fact that similar events were not found after 30 inoculations even at the highest dose, led us to consider these effects as not related to the administration of the product. Particularly regarding heart, it has been reported that some of normal non-treated laboratory mice are prone to develop abnormal cardiovascular alterations. 35
In the present study, the health and welfare of specific pathogen free mice kept under controlled conditions were verified. Our results evidenced that there were no macroscopic or microscopic lesions that could be attributed to the administration of CIGB-210 even at the highest dose administered. This dose would correspond to a human dose of 17.4 mg of CIGB-210 according to the allometric calculations based on the body weight/body surface relationship between human and mice 36 and is comprised in the potential therapeutic dose range of CIGB-210.
The in vitro evaluation of the capacity of lymphocytes to proliferate in response to a mitogen as an approximation to study the competence of cell mediated immune responses 37 evidenced no alterations after the repeated administration of CIGB-210. These results indicate that the complex array of signals in response to mitogens or antigens, including the recognition by the T cell receptor, the activation of genes and the secretion of lymphokines leading to the DNA replication and cellular division are not affected by the dose-repeated treatment with CIGB-210. On the other hand, the administration of CIGB-210 did not impair the capacity of mouse splenocytes to secrete IFNγ after ConA stimulation. This result suggests a favorable scenery for the occurrence of T cell mediated immune responses as mediated by the cytokine38,39 including the preservation for the functions of IFNγ secretor cells like CD8+ T cells, CD4+ Th1 lymphocytes, 40 macrophages, 41 and NK cells. 42
The demonstration that CIGB-210 treatment does not impair the mentioned above functionalities of the immune system is especially relevant for the safety of this potential drug, because it is intended to treat patients where immunosuppression is one of the hallmarks of the disease.
All these results together evidence that no functional damages are caused as consequence of the repeated administration of CIGB-210 and contribute important evidences to the preclinical safety of CIGB-210 as drug candidate.
Supplemental Material
sj-pdf-1-het-10.1177_09603271211073708 – Supplemental Material for Preliminary safety assessment of CIGB-210, an investigational peptide for HIV infection
Supplemental Material, sj-pdf-1-het-10.1177_09603271211073708 for Preliminary safety assessment of CIGB-210, an investigational peptide for HIV infection by Anna C Ramírez-Suárez, Taimi Paneque-Guerrero, Dionne Casillas-Casanova, Karelia Cosme, Dania Bacardí, Carlos A Duarte, Julio Ancízar, Emma Brown, Jorge Castro, José Suárez-Alba, Hilda Garay, Karla Pereira and Celia Fernández-Ortega in Human & Experimental Toxicology
Footnotes
Acknowledgements
The authors are grateful to Daylén Aguilar for her technical support and helpful recommendations for lymphoproliferation and ELISPOT experiments. We also thank Freya Freyre for her support in flow cytometry experiments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
