Abstract
The harm of air pollution to public health has become a research hotspot, especially atmospheric fine-particulate matter (PM2.5). In recent years, epidemiological investigations have confirmed that PM2.5 is closely related to chronic kidney disease and membranous nephropathy Basic research has demonstrated that PM2.5 has an impact on the normal function of the kidneys through accumulation in the kidney, endothelial dysfunction, abnormal renin-angiotensin system, and immune complex deposition. Moreover, the mechanism of PM2.5 damage to the kidney involves inflammation, oxidative stress, apoptosis, DNA damage, and autophagy. In this review, we summarized the latest developments in the effects of PM2.5 on kidney disease in human and animal studies, so as to provide new ideas for the prevention and treatment of kidney disease.
Introduction
The latest released data from the Global Burden of Disease showed that air pollution became the fourth largest risk factor worldwide in 2019. The risk of exposure to air pollution increased significantly between 2010 and 2019, and there was an increasing trend. The main compound of air pollution is PM2.5, which consists of particulate matter with an aerodynamic diameter of less than 2.5 μm. 1 In recent years, a large number of evidences have shown that PM2.5 plays an important role in the occurrence and development of renal injury and increases the risk of nephropathy.
Data published by the World Health Organization (WHO) indicated that kidney disease was one of the top 10 causes of death worldwide in 2019. 2 Kidney disease not only directly causes death but CKD is also an important risk factor for cardiovascular disease. 3 It is necessary to study the effects of PM2.5 on kidney disease and conduct effective prevention and treatment to improve public health.
In this paper, we first introduce the sources, composition and toxic effects of PM2.5, then sort out the relationship between PM2.5 and kidney disease, and finally deeply explore the mechanism of PM2.5-induced kidney injury. In order to provide new ideas for the prevention and treatment of nephropathy.
Sources, composition, and hazards of PM2.5
With the development of modern industry, the main sources and components of PM2.5 have changed. Reports on PM2.5 and public health have also gradually attracted attention. 4
The sources of PM2.5 are classified into three categories: natural sources, secondary particulate matter, and anthropogenic sources. At this stage, PM2.5 mainly comes from anthropogenic sources. Anthropogenic sources are further divided into fixed and mobile sources. Epidemiological surveys focus on cooking smoke, welding smoke, cigarette smoke, smoke from heating emissions, and traffic exhaust from mobile sources in fixed sources.5–10
The composition of PM2.5 is very complex and uncertain. The main components include elemental carbon, biological substances, inorganic components, organic components, and trace elements. 11 The key components of PM2.5 that are harmful to health vary in different spaces. For example, PM2.5 in welding fumes is rich in metals. PM2.5 in cooking smoke is rich in organic matter. 12 It is reported that PM2.5 exposure could cause accumulation of chromium and cadmium in the kidney. The deposition of particulate matter and its components in the body cause a systemic inflammatory response and vasodilation disorder. 13 Organophosphate esters (OPEs) are found in PM2.5. However, little is known about their toxic potentials on kidney, except adverse effects on renal proximal tubule cells. 14 Due to the different main components in PM2.5, they have differences in toxic effects on the body. In addition to considering the health effects of the main components, attention should also be paid to the combined toxicity of each component.
As a pollutant, PM2.5 is very harmful to public health. It can easily enter the body and exert toxic effects. Specifically, it includes (1) PM2.5 is present in aerosols (characterized by long transmission distances and difficulty in sedimentation) and more easily inhaled by the human body.15,16 (2) PM2.5 has a high specific surface area and strong adsorption. Harmful substances such as bacteria and viruses are easily adsorbed by PM2.5.17–19 (3) the size of PM2.5 is small and the number is large. A large amount of granular material is retained in the terminal bronchioles and alveoli, even crosses the blood gas barrier, and reaches sites such as the liver and kidneys with blood circulation, causing varying degrees of damage. 17 (4) harmful substances in PM2.5 produce combined toxic effects in the body.20–22
PM2.5 and renal injury
Renal disease affected by PM2.5
Epidemiological studies on the association between PM2.5 exposure and kidney injury.

MN is a primary glomerular disease associated with autoimmunity. A study analyzed the renal pathological results of 71,151 patients from 938 hospitals in China. It found that the incidence of MN increased year by year during 11 years. The incidence increased more significantly in cities with more severe PM2.5 pollution. Studies confirmed that long-term exposure to high concentrations of PM2.5 increased MN incidence risk. 28 MN is mainly caused by circulating autoantibodies against podocyte antigens, including the M-type phospholipase A2 receptor (PLA2R) and thrombospondin domain-containing 7A (THSD7A). It is demonstrated that PM2.5-induced exposure of PLA2R outside the kidneys could increase the incidence of MN. 29
Notably, while studies revealed a relationship between PM2.5 exposure levels and incidence of kidney disease, its effects on kidney disease vary among regions due to the complex sources and composition of PM2.5. 26 It is certain that reducing exposure time and concentration in the PM2.5 environment can reduce the risk of kidney disease.
The pathways of PM2.5 affecting kidney
Studies showed that PM2.5 directly and indirectly affected the kidneys through multiple pathways. Specifically, it may include PM2.5 accumulation in the kidneys, endothelial dysfunction, renin-angiotensin system (RAS) abnormalities, and immune complex deposition.13,30
PM2.5 is mainly inhaled by the respiratory tract. Smaller particulate matter in PM2.5 crosses the blood gas barrier, then transfers to the kidneys through the blood circulation, and accumulates in the kidneys. When the amounts of harmful substances in PM2.5 exceeds the level of renal compensation, it will damage the kidneys. Using high resolution of the fluorescent imaging method, PM0.2 and PM2.0 particles were found in mice kidneys after an inhalation experiment lasted for 15 min. 31 Accumulation of cadmium and chromium also occurred in the kidneys of mice exposed to PM2.5 plus PM10.13 Although there is ample evidence for accumulation of PM2.5 components in the kidney, how they enter the kidney remains to be further explored. It may be that macrophages endocytose PM2.5, which allows PM2.5 to cross the blood gas barrier and eventually reach the kidneys. 32 It is also possible that PM2.5 adsorbs proteins such as albumin and hemoglobin and follows the blood circulation to the kidneys. 33
Endothelial cells form the inner wall of blood vessels and have the functions of exchanging substances, adjusting vascular permeability, synthesizing and secreting a variety of bioactive substances. 34 Since endothelial cells are in direct contact with blood, they are more vulnerable to be damaged by PM2.5. In addition, peripheral blood or urine tests of truck drivers, welding workers, and traffic commanders revealed an elevated risk of systemic inflammation in humans due to long-term exposure to PM2.5.35–38 During systemic inflammation, humoral and cellular inflammatory mediators are activated. PM2.5 and activated inflammatory mediators damage endothelial cells, which cause endothelial dysfunction, ultimately affecting vascular function. The kidney is a highly vascularized organ that receives 20% of cardiac output. Disturbances in endothelial function may affect renal microcirculation, which in turn leads to renal dysfunction. 39
PM2.5 causes renal injury through RAS imbalance. Male SD rats and BALB/c mice exposed to PM2.5 had an imbalance in RAS and developed renal injury.40,41 The expression of angiotensin receptor-1 (AT1R) and angiotensin-converting enzyme (ACE) was increased in mouse renal tissue, which preceded oxidative stress and inflammatory responses in the kidney. 42 Intervention experiments with compound essential oils (CEOs) in mice showed that CEOs first regulated RAS and then reduced renal injury. However, the mechanism by which PM2.5 affects RAS still needs to be further explored. 43
Currently, the pathway of renal injury caused by antibodies to M-type phospholipase A2 receptor (PLA2R) has not been fully clarified. It is generally accepted that PLA2R is expressed in both lung and kidney. PLA2R in the lungs is phagocytosed and presented to other immune cells by phagocytes activated by PM2.5. Eventually, antibodies directed against PLA2R that are secreted and released by plasma cells in the blood circulation bind to PLA2R in the kidney to form immune complexes. The deposition of immune complexes in the glomeruli may cause renal injury. 44
The mechanism of PM2.5 inducing renal injury
The toxicological mechanism by which PM2.5 causes damage to the kidney is very complex. Studies showed that most of the potential molecular mechanisms are related to inflammation, oxidative stress, apoptosis, DNA damage, and autophagy. These mechanisms are interactive and interdependent (Figure 1). Graphical abstract summarizing the mechanisms of PM2.5-induced kidney damage.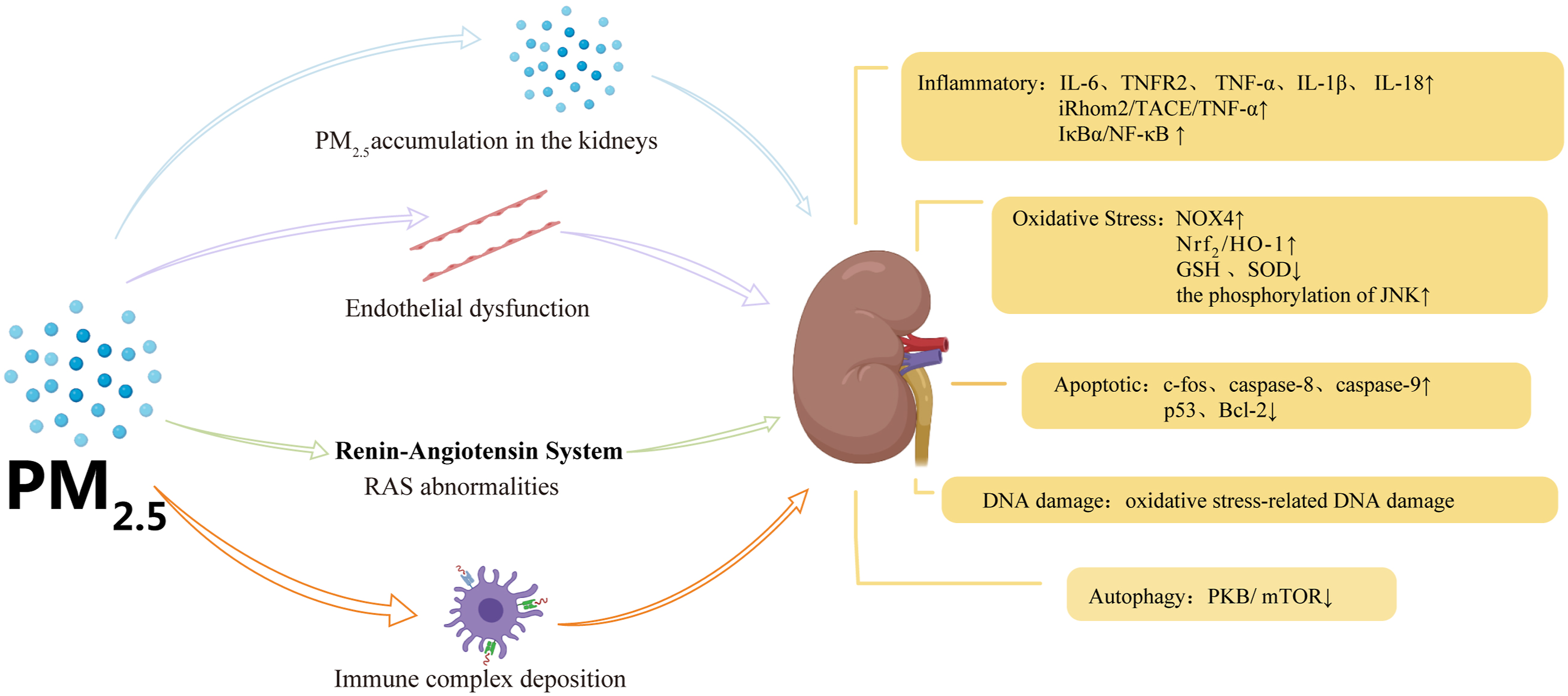
Inflammatory
Inflammatory response is a protective measure for the immune system to remove harmful irritants and promote body repair. But prolonged inflammatory response hinders the normal function of the body. PM2.5-exposed mice developed severe renal injury, accompanied by enhanced expression of interleukin-6 (IL-6), type-2 receptor (TNFR2), tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-18 (IL-18) in tissue samples. 45 Moreover, long-term exposure of workers to metal aerosols and cooking fumes resulted in systemic inflammation and renal dysfunction.46–48 Since PM2.5 directly or indirectly activated inflammatory cells in the kidney, a large number of those cells overexpress and release various inflammatory and chemical factors. They cause a cascade of reactions and inflammatory damage.
Macrophages are important inflammatory cells. Activated macrophages can infiltrate the kidney and promote the development of inflammation. 49 In the experiment, PM2.5 upregulated the mRNA of inactive rhomboid-like protein 2 (iRhom2) and TNF-α converting enzyme (TACE) levels in RAW264.7 cells. The expression of NF-κB (IκBα)/nuclear factor kappa-B (NF-κB) and TACE/TNFRs were promoted. Moreover, the macrophages secreted large amounts of cytokines after exposure of PM2.5, which in turn caused renal injury. 45
Podocytes, covering the outer layer of the glomerular basement membrane, are the last filtration barrier for the loss of urinary protein and urinary red blood cells in patients with nephritis. It was found that PM2.5 activated pathways such as iRhom2/TACE/TNF-α in renal macrophages. Abnormalities in these pathways led to the accumulation of IL-1β, IL-6, TNF-a, and caused damage to podocytes. 50 In addition, PM2.5 not only stimulated macrophages to release large amounts of inflammatory cytokines to cause podocyte injury but also activated proinflammatory pathways in podocytes. Those exacerbated the inflammatory response.51–53 Some vitro experiments confirmed that PM2.5 significantly increased the disintegrate of F-actin stress fibers, promoted cytoskeletal disorganization and dissociation, disrupted the integrity of nuclear structure. 44 And the results showed PM2.5 increased the apoptotic rate of podocytes by activating the IκBα/NF-κB inflammatory signaling pathway in MPC5 cells. 54
Oxidative stress
Oxidative stress plays an important role in the process of renal injury induced by PM2.5. Oxidative stress occurs when free radical production is excessive and insufficient to be eliminated by the antioxidant defense system, accompanied by accumulation of reactive nitrogen species (RNS) and reactive oxygen species (ROS). Metal and organic components in PM2.5 promoted free radical production and reduced antioxidant capacity within the cell. This led to peroxidation of lipids on the cell membrane as well as an increase in intracellular Ca2+ concentration, ultimately triggering cell damage.55–58 The kidney is more likely to suffer damage from oxidative stress. When the kidney is stimulated in a short time, it will excrete metabolites and poisons. These stimuli lead to overwhelming ROS production in the kidney as a result. 59
The NOX family have the capacity to transport electrons across the plasma membrane and to generate superoxide and other downstream ROS. They are the main makers of ROS in the kidney under the influence of PM2.5. Studies confirmed that short-term PM2.5 exposure elevated the level of ROS through sustained activation of NOX4. When the antioxidant system is not sufficient to remove excessive ROS, the kidney is damaged that is not visible to the naked eye while there are significant differences in biochemical parameters. 43
Nuclear factor E2-related factor-2 (Nrf2) signaling is one of the antioxidant defense systems. Nrf2 is activated by the influence of some factors (e.g., Kelch-like epichlorohydrin-associated protein 1 (KEAP1)), which regulates a series of downstream antioxidants and cytoprotective enzymes.60,61 Heme oxygenase-1 (HO-1) is one of the downstream antioxidant enzymes. It effectively scavenges ROS by degrading heme groups and promoting biliverdin. The reduced bilirubin in turn reverses oxidative stress. Short-term PM2.5 exposure activated the Nrf2/HO-1 pathway in renal tissue, which rescued kidney injury. 43 However, long-term exposure to PM2.5 inhibited the Nrf2/HO-1 pathway. Since after long-term exposure to PM2.5, the inflammatory factor TNF-α was maintained at higher levels for a long time, which inhibited the expression of Nrf2. 50
The antioxidant enzyme systems downstream of Nrf2 also include glutathione peroxidase (GSH-Px) and superoxide dismutase (SOD).62,63 They have the efficacy of converting free radicals to less toxic or harmless substances. The decrease of L-Glutathione (GSH) and SOD levels is a key factor leading to oxidative stress and triggers a series of cascades. In the experiment, PM2.5 increased ROS but decreased GSH and SOD storage in the kidney, ultimately leading to renal injury. 43
C-Jun N-terminal kinase (JNK) is a subfamily of mitogen-activated protein kinase (MAPK) pathways, which is an important antioxidant defense system. JNK can be activated by ROS and inflammatory factors to regulate cellular metabolism and apoptosis by inducing the transcription of downstream genes.64–66 PM2.5 could significantly increase the phosphorylation of JNK, decrease the antioxidant enzyme levels, and induce cell death, thereby enhance oxidative stress and damage the kidney. 45
Apoptotic
PM2.5 exposure not only causes renal inflammation and oxidative stress but even triggers apoptosis. It is certain that proper apoptosis helps to maintain the normal function of the body, while dysregulation of apoptosis causes serious consequences. 67
Tubular epithelial cells and podocytes are important cells in the kidney that perform reabsorption and filtration functions. Their excessive apoptosis is closely related to the occurrence and development of renal diseases. Studies showed that PM2.5 affected the expression of apoptosis-related genes and proteins in HK-2 cells. It could increase the levels of c-fos, caspase-8, and caspase-9, and decrease the levels of P53 and B-cell lymphoma-2 (Bcl-2). Also, in HK-2 cells, traffic-related diesel particulate matter (DPM) not only induced ER stress but also led to mitochondrial damage by increasing mitochondrial ROS. Eventually, the caspase pathway was activated and triggered apoptosis. It also proposed that PM2.5-induced apoptosis in MPC-5 cells may be related to NF-κB signaling pathways. 54 In the experiment, PM2.5 significantly increased the protein expressions of Bax, NF-κB/p65 and p-IκBα, but markedly decreased the protein expressions of Bcl-2, nephrin, and podocin in podocytes.
In addition, heavy metals have a long half-life period. They could accumulate in the body, especially in the kidney after long-term exposure.69 This may cause apoptosis of tissues and cells.69,70 The SD rats were exposed to PM2.5 plus PM10 for 6 months, then significantly higher levels of cadmium and chromium were found in their kidneys. When the content of cadmium and chromium exceeded the threshold that the kidney beard, it may cause excessive apoptosis of cells in the tubules.68,71,72
DNA damage
In recent years, studies have confirmed the effects of PM2.5 on renal DNA. Ordered DNA expression is essential for the normal development of the body, and its abnormal changes may lead to the development of diseases.73,74 The AJ mice were exposed to traffic-related PM2.5 for 3 months (from infancy to adulthood). 75 The levels of oxidative stress-related DNA damage increased in their renal tissue, which may be the result of the massive ROS attack on DNA caused by PM2.5.
Autophagy
Recently, studies have found the role of autophagy in renal injury caused by benzopyrene (BaP) (a common organic chemical in PM2.5) and diesel particulate matter. 76 Autophagy is an important mechanism by which cells cope with intracellular or extracellular stress. During autophagy, some toxic components were degraded by autolysosomes to prevent cell damage.77,78 However, the efficiency of autophagy could be reduced by PM2.5. In the experiment, BaP reduced the viability of podocytes in a dose-and time-dependent manner by inhibiting autophagy. And the expression of synaptopodin and desmin (podocyte maturation markers), nephrin and podocin (filtration barrier) were decreased. The autophagy inducer, rapamycin, rescued podocyte injury and restored its filtration function. Diesel particulate matter inhibited the autophagy induced by protein kinase B (PKB)/mammalian target of rapamycin (mTOR) pathway, which attenuated the protective role of autophagy in renal injury.79,80
Conclusion
This paper provides a solid rationale for PM2.5 being a key risk factor for kidney disease. Epidemiological studies showed that PM2.5 increases the risk of kidney disease. In vivo and in vitro studies explained that PM2.5 may affect renal function through many kinds of pathways. Further studies showed that the toxicological mechanism of PM2.5 on the kidney was related to inflammation, oxidative stress, apoptosis, DNA damage, and autophagy. However, how these mechanisms affect each other needs to be explored comprehensively and systematically. It is certain that effectively controlling air pollution can reduce the risk of kidney disease. In addition, the susceptible population can be recommended to use rich antioxidant animal and plant drug resources. Medical staff should be aware of the harm of PM2.5 and recommend patients to reduce relevant environmental exposure, so as to better control the development of kidney disease.
Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NSFC, 81872623), and the Liaoning Provincial Natural Science Foundation of China (20180550737).
