Abstract
Carbon disulfide (CS2) has reproductive toxicity but the mechanism remains unclear. The aim of the present study was to investigate the effect of oxidative stress and DNA damage on embryo implantation of mice exposed to CS2 at peri-implantation. CS2 exposure was on gestational day 3 (GD3), GD4, GD5 and GD6, separately, and the number of embryonic day 9 (E9) mouse embryos was obtained. DNA damage of endometrial cells, oxidative stress and 8-hydroxy-2′-deoxyguanosine (8-OH-dG) level in uterus tissues were tested with time series at different end points after exposure. The number of E9 mouse embryos significantly decreased in all CS2 exposure groups, especially on GD4 exposure. The rates of embryo implantation decreased by 43.05%, 63.74%, 60.45% and 47.26% for CS2 exposure on GD3, GD4, GD5 and GD6, respectively. Oxidative stress significantly increased within 24 h and reached the top level at 18 h after exposure. The same time-dependent trend was observed no matter when the exposure happened at peri-implantation. 8-OH-dG significantly increased at 18 h and 24 h after exposure by 893.8% and 647.4%, respectively, when compared with the control. The indexes of DNA damage significantly increased at 6 h after exposure, which appeared earlier than the changes of oxidative stress and 8-OH-dG. Besides, both oxidative stress and DNA damage showed a strong negative correlation with the number of E9 mouse embryos. The present study illustrated that CS2 directly induced DNA damage in endometrial cells and enhanced the action through oxidative stress, both of which were responsible for CS2-induced embryo loss.
Introduction
Carbon disulfide (CS2), a volatile organic solvent, is often used in various industrial processes, such as vulcanizing rubber, fumigating grain, oil extraction and manufacturing viscose rayon fibers. 1 CS2 has multisystem toxicity and long-term exposure to low concentrations of CS2 can cause neurological, cardiovascular, psychological and hormonal effects. 2 Furthermore, it is noteworthy that wide use of CS2 is associated with various types of reproductive disturbance. CS2 could induce atrophy of testis, dyszoospermia, sexual disorder, decline of sex hormone and decrease in semen quality. 3 Many studies have shown that CS2 could induce menstrual disorders and abnormal labor, such as spontaneous abortion and premature birth among female employees. 4 In a prospective cohort study, we found that the incidence of clinically unrecognized pregnancy loss increased markedly, and the time-to-pregnancy was extended in the women employees exposed to CS2. 5 We also found that the number of implanted embryos significantly decreased when mice were exposed to CS2 at the phase of embryo implantation. 6 These results suggested that CS2 could disturb embryo implantation and induce embryo loss, but the mechanism was still unclear.
Oxidative stress induces lipid peroxidation, structurally and functionally alters protein and DNA and promotes apoptosis. 7 It was reported that reduced plasma levels of antioxidant increased risk of spontaneous abortion. 8,9 Chronic exposure to CS2 could cause coronary heart disease by oxidative stress. 10 Our previous study revealed that CS2 exposure could induce DNA damage in the endometrial cells of mice at the phase of implantation. 11 Therefore, we hypothesized that oxidative stress and DNA damage were a single or joint participation in disturbing embryo implantation after CS2 exposure. In the present study, the mice were exposed to CS2 at peri-implantation. Oxidative stress, DNA damage and the oxidative DNA damage product 8-hydroxy-2′-deoxyguanosine (8-OH-dG) 12 were observed and evaluated with time series to explore the mechanism of CS2-induced embryo loss.
Materials and methods
Animals
Adult Kunming mice, female (7–9 weeks old, 23–25 g) and male mice (8–10 weeks, 30–35 g), were provided by Vital River Laboratories (Beijing, China). All the animals were kept under controlled environmental conditions at room temperature (22 ± 2°C) with 50% ± 10% humidity and an automatically controlled cycle of 12 h light and 12 h dark. Standard laboratory animal feed (purchased from commercial supplier) and water were provided ad libitum. Animals were acclimatized to the experimental conditions for a period of one week prior to the commencement of the experiment. Then the female mice were mated with the male mice. The day on which sperm plug was found after the mating procedure was designated as gestational day 1 (labeled GD1) and the mice were randomly grouped. All the animal experiment protocols were approved by the Medical Ethics Committee of Shandong University.
Experimental design
The experiment was divided into two parts. Part 1 was designed to establish the animal model of CS2 exposure at different time of peri-implantation in order to find the sensitive exposure time, and to determine the association between embryo implantation and oxidative stress. Part 2 was to evaluate the levels of DNA damage and 8-OH-dG with time series after exposure at the sensitive exposure time obtained from part 1.
Animal treatment
In part 1, mice on GD1 were randomly divided into four groups and each group contained two types of exposure (CS2 exposure and solvent exposure). Mice in group 1 were exposed on GD3, group 2 on GD4, group 3 on GD5 and group 4 on GD6, respectively. There were a total of 26 end points in four groups. The end points were introduced in detail as follows. Eight end points in group 1 were designed at 6 h, 12 h, 18 h, 24 h, 48 h, 72 h, 96 h and 144 h (GD9) after exposure. Seven end points in group 2 were at 6 h, 12 h, 18 h, 24 h, 48 h, 72 h and 120 h (GD9). Six end points in group 3 were at 6 h, 12 h, 18 h, 24 h, 48 h and 96 h (GD9). Five end points in group 4 were at 6 h, 12 h, 18 h, 24 h and 72 h (GD9) after exposure. They are illustrated in Figure 1. The aim of the last end point (GD9) was to observe the maternal toxicity and embryo implantation, and the aim of other end points was to measure the level of oxidative stress in the uterine tissue.

A schematic diagram of the arrangements of exposure points and end points in part 1. The exposure points of mice in group 1 were on GD3, group 2 on GD4, group 3 on GD5 and group 4 on GD6, respectively. Eight end points in group 1 were at 6 h, 12 h, 18 h, 24 h, 48 h, 72 h, 96 h and 144 h after exposure. Seven end points in group 2 were at 6 h, 12 h, 18 h, 24 h, 48 h, 72 h and 120 h. Six end points in group 3 were at 6 h, 12 h, 18 h, 24 h, 48 h and 96 h. Five end points in group 4 were at 6 h, 12 h, 18 h, 24 h and 72 h. GD: gestational day.
In part 2, CS2 exposure was arranged on GD4 according to the results of part 1. Four end points were designed at 6 h, 12 h, 18 h and 24 h after CS2 or solvent exposure, respectively. There were six gestational mice in each end point in part 1 and part 2.
Each animal received a single dose intraperitoneal injection of CS2 (Guangcheng chemical reagent corporation, Tianjin, China) or olive oil (Shanghai Kerry Food Industries Co., Ltd, Shanghai, China) at the designed exposure time. CS2 dissolved in olive oil was 631.4 mg/kg body weight, 0.4 median lethal dose (LD50), based on the data from the acute toxicity test for female Kunming mice in our previous research. 11 The injection volume was 0.1 mL/10 g body weight.
Sample collection
The mice for the last end point (GD9) in all groups in part 1 were weighed and killed on GD9. The utero weight on GD9 was harvested and weighed, and the number of embryonic day 9 (E9) mouse embryos for each litter was counted. Uterus, ovary, liver, spleen and kidney were isolated and weighed and then frozen at −80°C in a refrigerator for further analysis.
The mice in other end points, except for the last one in all groups in part 1, were killed at the designed end points. Half of each uterine tissue was separately frozen at −80°C for quantitating the protein content of the homogenate and testing oxidative stress and 8-OH-dG levels. The endometrium of the other half uterine was manually scraped with a flat bamboo stick gently and isolated into a single cell for assessing DNA damage.
Measurement of malondialdehyde, superoxide dismutase, glutathione peroxidase and catalase in uterine tissue
Malondialdehyde (MDA), superoxide dismutase (SOD), glutathione peroxidase (GSH-PX) and catalase (CAT) in the uterine tissue homogenate were measured with commercial chemical assay kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) by an UV-2450 spectrophotometer (Shimadzu Co., Kyoto, Japan). The following wavelengths were used: 532 nm for MDA, 550 nm for SOD, 412 nm for GSH-PX and 405 nm for CAT. The results were expressed as nanomoles of MDA per milligram of protein (nmol/mgprot) for MDA, units per milligram protein (U/mgprot) for SOD, GSH-PX and CAT. The experimental procedures were in strict accordance with the kit instructions. Results were determined according to the manufacturer’s guide.
Determination of the total protein content
The total protein content was determined with Coomassie blue method assay kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) by an UV-2450 spectrophotometer. Wavelength used was 595 nm, and the result was expressed as gram per liter (g/L). Results were determined according to the manufacturer’s guide. Determination of the total protein was to standardize the indexes of oxidative stress.
Endometrial cell comet assay
The comet assay was carried out with slight modifications of the standard protocol as described by Singh et al. 13 Two solutions, one containing 0.8% normal melting point agarose (NMA; Amresco, Solon, Ohio, USA) and the other containing 0.8% low melting point agarose (LMA; Amresco) dissolved in Ca2+, Mg2+-free Phosphate Buffered Saline (PBS), were prepared. Endometrial cell suspension of 25 µL and 75 µL LMA were distributed onto microscope slides precoated with 100 µL of 0.8% NMA. After solidifying the agarose, slides were immersed in ice-cold freshly prepared lyses solution (2.5 M sodium chloride, 100 mM diaminetetraacetic acid (Na2EDTA) (Amresco), 10 mM Tris (Sigma, St. Louis, Missouri, USA), pH 10.0 containing 1% Triton X-100 (Amresco) and 10% dimethyl sulfoxide (Amresco)) for 1 h to lyse the cells and to allow DNA unfolding. Then the slides were placed on a horizontal electrophoresis chamber and DNA was allowed to unwind for 20 min in an alkaline solution containing (300 mM sodium hydroxide and 1 mM Na2EDTA, pH 13). Electrophoresis was conducted at 20 V, 150 mA for 20 min. After electrophoresis, the slides were gently washed three times for 5 min with neutralization buffer (0.4 M Tris-base, pH 7.4). The slides were drained and SYBR-Green I (30 μL/slide; Sigma) was added. To prevent additional DNA damage, all the steps described above were run under red light. Analyses were performed immediately under a fluorescence microscope (Nikon, Tokyo, Japan) and the photographs were individually measured at 200× magnification using a calibration scale. A total of 50 individual cells were screened per slide to determine the following parameters for the DNA damage analysis: tail length (TL, in μm), tail moment (TM), tail moment olive (OTM) and tail DNA% (TDNA%) with image analysis software (CASP, Poland 14 ). TL is the maximum distance that the damaged DNA migrates from the center of the cell nucleus. TDNA% is the total DNA that migrates from the nucleus into the comet tail. TM is the product of the TL and TDNA%, which gives a more integrated measurement of overall DNA damage in the cell. OTM is the product of TDNA% and median migration distance. All slides were coded and scored blindly, and each experiment was reproduced independently at least three times.
Measurement of 8-OH-dG in uterine tissue
The mouse 8-OH-dG ELISA kit (Shanghai BlueGene Biotech Co., Ltd, Shanghai, China) was used to measure the 8-OH-dG in uterine tissue homogenate. The whole experiment process was according to the manufacturer’s guide. Reading the optical density (OD) at 450 nm within 5 min after the termination of the reaction by Thermo Scientific Multiskan MK3 (Thermo Fisher, Boston, Massachusetts, USA). The result was expressed as nanograms per milliliter 8-OH-dG.
Statistical analysis
Data were shown as mean ± SD for each group. Statistical analysis was performed using the SPSS 20.0 statistical software. The data were first analyzed by the homogeneity test for variance. If variances were equal, one-way analysis of variance was performed followed by Dunnett’s t test if the results were significant. If variances were unequal, statistical differences were evaluated by the Brown–Forsythe test. p < 0.05 was considered to be statistically significant. Linear correlation analysis was performed in order to observe the correlation between the number of E9 mouse embryos and different other parameters.
Results
Maternal toxicity and embryo implantation
CS2 exposure on GD3, GD4, GD5 and GD6 all failed to induce a statistically significant difference in maternal net body weight on GD9 (Figure 2(a)). Furthermore, no statistically significant changes were observed in the weight of the uterus, ovary, liver, spleen and kidney between CS2 exposure and solvent control in four groups (Figure 2(b)). Yet, a statistically significant reduction was observed in the number of E9 mouse embryos when compared with the control (Figure 2(c)). The rates of embryo implantation were 56.95%, 36.26%, 39.55% and 52.74% for CS2 exposure on GD3, GD4, GD5 and GD6, respectively, when compared with each corresponding control. No statistically significant change was found in the mean weight of conceptus in each CS2 exposure when compared with the control (Figure 2(d)).

Maternal toxicity and blastocyst implantation after exposing mice to CS2 at different time in peri-implantation. (a) Maternal net body weight gain in each group; (b) the weight of maternal uterus, ovary, liver, spleen and kidney in each group; (c) the number of E9 mouse embryos in each group; (d) mean weight of conceptus in each group. All the values are expressed as mean ± SD. **p < 0.01 compared with control. The control is the merged control of four groups. CS2: carbon disulfide; E9: embryonic day 9.
Oxidative stress after mice exposed to CS2 at peri-implantation
The level of MDA, SOD, GSH-PX and CAT in uterine tissue
MDA content as an indicator of lipid peroxidation had a significant increase at 18 h after CS2 exposure on GD3, GD4, GD5 and GD6 when compared with the control (Figure 3(a)). At 18 h after CS2 exposure, the MDA level increased by 131.4% in GD4, which is higher than 120.8%, 121.6% and 104.9% in GD3, GD5 and GD6 CS2 exposure groups when compared with the control. In addition, SOD, GSH-PX and CAT initially resulted in no significant increase, whereas subsequent statistically significant decreases at different time points were observed: SOD at 18 h, GSH-PX at 12 h and 18 h and CAT at 12 h when compared with each control (Figure 3(b) to (d)).
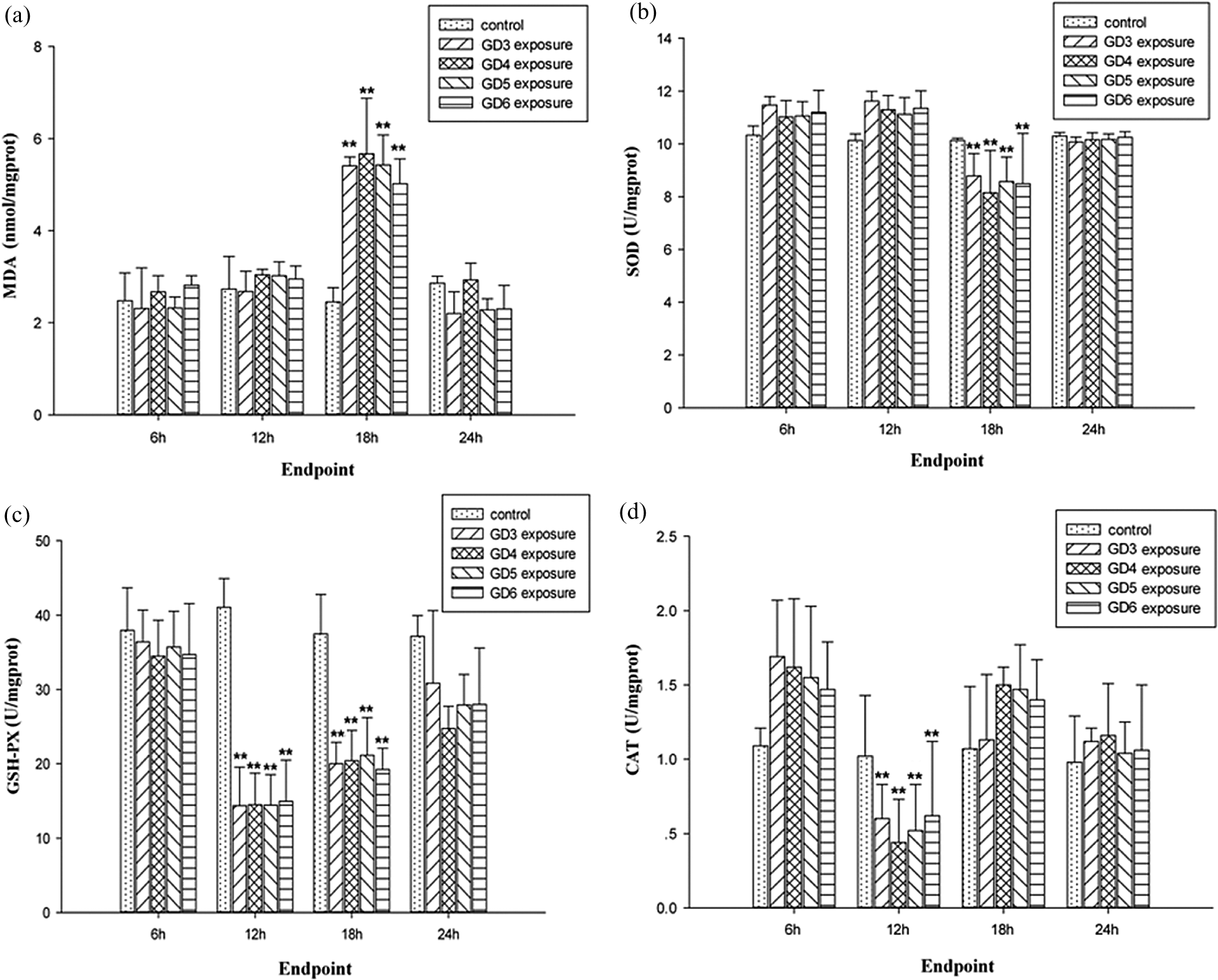
Oxidative stress levels in uterine tissue after exposing gestational mice to CS2 at different time in peri-implantation. (a) MDA level in each group; (b) SOD level in each group; (c) GSH-PX level in each group; (d) CAT level in each group. All the values are expressed as mean ± SD. **p < 0.01 compared with control. CS2: carbon disulfide; MDA: malondialdehyde; SOD: superoxide dismutase; GSH-PX: glutathione peroxidase; CAT: catalase.
The trends with time for oxidative stress
MDA levels, in all CS2 exposure groups, significantly increased within 24 h after CS2 exposure and then returned to normal after 24 h. No statistically significant changes were observed at 24 h and after 24 h between each CS2 exposure and their control. The variation trend of MDA was the same no matter when CS2 exposure happened on GD3, GD4, GD5 or GD6. SOD, GSH-PX and CAT levels all decreased in 24 h and returned to normal after 24 h (Figure 4(a) to (d)).

Trends with time for oxidative stress levels in uterine tissue after exposing mice to CS2 at different time in peri-implantation. (a) The trends with time for MDA in each group; (b) the trends with time for SOD in each group; (c) the trends with time for GSH-PX in each group; (d) the trends with time for CAT in each group. The values are expressed as mean. CS2: carbon disulfide; MDA: malondialdehyde; SOD: superoxide dismutase; GSH-PX: glutathione peroxidase; CAT: catalase.
The relationship between oxidative stress and embryo implantation
The correlation analysis showed a strong negative correlation between MDA levels at 18 h after exposure and the number of E9 mouse embryos in each group (r = −0.783, p < 0.01; Figure 5).

The correlation analysis between MDA levels at 18 h after exposure and the number of E9 mouse embryos in each group. CS2: carbon disulfide; MDA: malondialdehyde; E9: embryonic day 9.
DNA damage of endometrial cells in mice after CS2 exposure at peri-implantation
After CS2 exposure on GD4, the parameters of comet assay such as TL, TM, OTM and TDNA% all showed statistically significant increases when compared with the control in part 2 (Table 1). Comet parameters appeared to significantly increase at 6 h after CS2 exposure and reached the top level at 18 h, and then these comet parameters decreased gradually (Figure 6(a) to (e)). The correlation analysis showed a strong negative correlation between the number of E9 mouse embryos and TM, OTM, TL and TDNA% after CS2 exposure at 18 h (r = −0.804, −0.847, −0.934 and −0.863, respectively, p < 0.01).

DNA damage of endometrial cell after exposing mice to CS2 on GD4. Control (a), 6 h (b), 12 h (c), 18 h (d) and 24 h (e) of the comet assay in the mice endometrial cell at ×200, respectively.
DNA damage of endometrial cells for mice exposed to CS2 on GD4.

CS2: carbon disulfide; GD4: gestational day 4; 8-OH-dG: 8-hydroxy-2′-deoxyguanosine; TL: tail length; TM: tail moment; OTM: tail moment olive; TDNA%: tail DNA%.
a The control is the merged control of four groups.
b p < 0.01 compared with control.
The level of 8-OH-dG in uterine tissue after CS2 exposure at peri-implantation
Statistically significant increases in the 8-OH-dG were observed at 18 h and 24 h after exposure when compared with the control in uterine tissue (Figure 7). The increased rate of the 8-OH-dG level was 893.8% at 18 h and 647.4% at 24 h after exposure when compared with the control.

Oxidative DNA damage induced by CS2 in uterine tissue after exposing mice to CS2 on GD4. The 8-OH-dG in uterine tissue of pregnant mice killed at the end point of 6 h, 12 h, 18 h and 24 h, respectively, and their controls. **p < 0.01 compared with control. CS2: carbon disulfide; GD4: gestational day 4; 8-OH-dG: 8-hydroxy-2′-deoxyguanosine.
The relationship between oxidative stress, DNA damage and 8-OH-dG level
TDNA%, TL, TM and OTM showed statistically significant difference at 6 h after exposure, whereas MDA showed a statistical increase at 18 h after exposure. MDA change that indicated oxidative stress occurred after DNA damage. Then TDNA%, TL, TM, OTM and MDA all showed a decrease at 24 h after exposure. The correlation analysis showed a strong positive correlation between the 8-OH-dG level and TDNA%, TL, TM and OTM (r = 0.766, 0.688, 0.738 and 0.771, respectively, p < 0.01). Figure 8 shows the relationship between oxidative stress, DNA damage and 8-OH-dG after exposing mice to CS2 on GD4.

The relationship between oxidative stress, DNA damage and 8-OH-dG after exposing mice to CS2 on GD4. Point 0 on the X-axis is the merged control for each index. CS2: carbon disulfide; GD4: gestational day 4; 8-OH-dG: 8-hydroxy-2′-deoxyguanosine.
Discussion
Embryo implantation in rats and mice occurs between days 4 and 5 after mating, 15 which is called the implantation window. 16 In the present study, CS2 exposure was arranged on GD3, GD4, GD5 and GD6, respectively, to find out the effect of CS2 exposure on embryo implantation at different exposure time. Therefore, the mice only received a single exposure, which could not induce maternal toxicity, but had a significant effect on embryo implantation. Based on our previous study, 11 0.4 LD50 (631.4 mg/kg) was the appropriate dose. The results showed a statistically significant reduction in the number of E9 mouse embryos at single dose exposure, especially on GD4. Meanwhile, no significant maternal toxicity and the changes in mean weight of conceptus were observed. These results illustrated that CS2 exposure at different time of peri-implantation disturbed embryo implantation, and the sensitive exposure time was GD4. According to the results of embryology studies, successful embryo implantation depends on endometrial receptivity in the implantation window period, 17 and many signals and molecular pathways that induce or regulate the complex series of interactions required for implantation. 18 If the condition of maternal uterus was disturbed, the loss of embryos would happen. 19 In our previous studies, CS2 exposure at peri-implantation disturbed expression of MMP(metalloproteinase)-2, MMP-9, 20 leukemia inhibitory factor, 21 adhesion molecule fibronectin and its ligand, and integrin β1 in the uterus of gestational mice, 22 all these are related to implantation failure. Therefore, we speculated that CS2 exposure might affect the environment of the uterus and disturb the development of the implantation window to disturb the process of embryo implantation.
Luo et al. 23 reported that CS2 exposure could induce oxidative stress and reduce the levels of antioxidative enzymes in the blood. Song et al. 24 showed that CS2 exposure could induce oxidative–antioxidative system disorders and significant changes in oxidative–antioxidative function in rat nerve tissues. Determination of free radicals that could indicate oxidative stress is difficult because of their relatively short half-lives. Instead, they can be evaluated indirectly by measurement of the levels of some antioxidant enzymes such as SOD, CAT and GSH-PX, and by measurement of products of lipid peroxidation such as MDA. 25,26 It has been reported that the increase in SOD in human endometrial stromal cells with decidualization is an important component of implantation, 27,28 and both ROS and SOD may act as second messengers to regulate endometrial function. 29 In the present study, oxidative stress was assessed using MDA and antioxidant enzymes. Within 24 h after CS2 exposure, MDA increased significantly in uterine tissue; the antioxidant enzymes were initially induced and then significantly inhibited and GSH-PX decreased before SOD and CAT. The reasons for the above evidence may be that ROS increased rapidly, which led to tissue cell damage and produced MDA by lipid peroxidation after CS2 exposure. 24 The increased ROS induced compensation function and resulted in the increase of enzyme activity. However, the increased ROS not only consumed a lot of enzymes but also changed the structure and function of protein and DNA, 30 which significantly reduced the activities of the antioxidant enzyme. Furthermore, the GSH-PX may be more sensitive to CS2 exposure. The result showed a strong negative correlation between the number of E9 mouse embryos and the level of MDA. Moreover, positive correlations between the number of E9 mouse embryos and the level of SOD, CAT and GSH-PX were observed. The above results showed that oxidative stress was one of the mechanisms responsible for CS2-induced embryo loss. In addition, no matter when the exposure happened on GD3, GD4, GD5 or GD6, the trend of change for oxidative stress parameters above in uterine tissue of gestational mice was the same along with the reduced number in each exposed group. Coincidentally, oxidative stress was more severe in uterine tissue of mice on GD4 exposure that was corresponding to the least number of E9 mouse embryos on GD4 exposure. It can be speculated that oxidative stress destroyed the endometrial receptivity, and GD4 was the most sensitive period for CS2 exposure.
Reactive oxygen species is known to produce membrane peroxidation and MDA formation and attack on DNA. 31 Studies reported that exposure to lower concentration of CS2 could cause certain damage to human buccal cell DNA 32 and mice sperm cell DNA. 33 Based on the evidence and the results in part 1, GD4 exposure was chosen to further assess the relationship between oxidative stress, DNA damage and embryo implantation. Endometrial cell comet assay revealed that CS2-induced DNA damage began at 6 h after exposure and reached the top level at 18 h. To ensure the role of the increased oxidative stress in the induction of oxidative DNA damage, quantification of 8-OH-dG was performed. It has been reported that the levels of the oxidative DNA damage marker 8-OH-dG were significantly elevated in infertile male patients. 34 In the present study, a significant increase in 8-OH-dG was observed at 18 h and 24 h after exposure and has been well correlated with the parameters of endometrial cell DNA damage. Furthermore, the linear correlation analysis showed a strong negative correlation between the number of E9 mouse embryos and DNA damage. Thus, it can be inferred that CS2 destroys the function of endometrium to accept embryo implantation by DNA damage and oxidative stress. Interestingly, DNA damage was found earlier than oxidative stress, and the 8-OH-dG and DNA damage increased significantly after oxidative stress returned to basal levels. This result was inconsistent with the traditional theory that DNA damage was caused by oxidative stress. Hence, we conjecture that CS2 exposure can directly cause DNA damage, which was strengthened by the oxidative stress action. Additionally, further research are required to explain why oxidative DNA still had statistically significant changes after oxidative stress return to basal levels.
The part 1 of this study can provide the optimum exposure point for further confirmation and mechanistic evaluation, and the part 2 can provide better and more informative results. Based on the results of the present study, if the women at peri-implantation stage cannot avoid contacting with CS2, we recommend taking interventions on oxidative stress to reduce the damage caused by CS2. Embryo implantation is a complex process, it is essential to characterize the embryotoxicity of CS2 using other surrogate end points in order to reduce the potential risk in women who are exposed to CS2 in workshop, especially those of reproductive age.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by the projects of National Natural Science Foundation of China [grant numbers 30671745 and 81072275].
