Abstract
Cardiovascular disorders are the leading cause of death globally. Rosuvastatin is a member of statins (inhibitors of 3-hydroxy-3-methylglutaryl coenzyme A reductase) with many pleiotropic properties. This study investigated cardioprotective effects of rosuvastatin in isoprenaline-induced myocardial injury. Male rats were given rosuvastatin (1, 5, or 10 mg/kg, oral) daily for 1 week and on seventh and eighth day isoprenaline (150 mg/kg, subcutaneous) was given to induce cardiac injury. On ninth day, rats were euthanized and different samples were harvested for analysis. Isoprenaline administration resulted in increased cardiac mass, increased cardiac injury marker levels (cTnI, CK-MB, ALT, and AST), increased lipid/protein oxidation, and increased cardiac nitrite levels. It also decreased superoxide dismutase, CAT, GST, and glutathione reductase activities, and total antioxidant activity. Isoprenaline also increased TNF-α and IL-6 levels. Decreased mRNA expression of Nrf2 and Bcl-2 along with increased mRNA expression of Bax, eNOS and iNOS genes was observed in isoprenaline treated animals. Histopathological evaluations of rosuvastatin pre-treated groups showed reduction of myocardial necrosis. Pretreatment with rosuvastatin (5 and 10 mg/kg) reduced many of these pathological changes. The current study showed that rosuvastatin significantly reduces myocardial injury induced by isoprenaline.
Introduction
Isoprenaline (ISO) is a non-selective β-adrenergic receptor agonist. A very high dose of ISO causes myocardial stress resulting in infarct-like lesions. 1 Cellular infiltration, hypertrophy, apoptosis, necrosis, and fibrosis are seen after ISO administration. 2 ISO-induced cardiac injury has been widely used to study the effect of drugs on myocardial infraction. 3 Cardiovascular diseases (CVDs) have been a major health problem and the leading cause of death worldwide. According to the World Health Organization (WHO), among various cardiovascular problems, ischemic heart disease (IHD) will remain as a major disease-burden by the year 2060. 4
Statins are reversible inhibitors of 3-hydroxy-3-methylglutaryl coenzyme A reductase. Many clinical trials report relationship between statins and reduction of cardiovascular events.5,6 Statins have been used before cardiac surgery due to their pleiotropic actions, independent of cholesterol reduction. 7 Rosuvastatin has been reported to exert various pharmacological actions such as anti-inflammatory and anti-oxidative activities. It is reported, neuronal cell apoptosis was reduced and neurological deficit was improved by rosuvastatin in transient middle cerebral artery occlusion/reperfusion injury. 8 It also promoted angiogenesis in myocardial infarction in rats. 9 Also, statins may exert beneficial effects in Alzheimer’s dementia and pulmonary arterial hypertension.10,11
Most of the agents investigated for cardioprotection fail to show clinical relevance and many of them show cardiotoxicity. 12 Oxidative stress and inflammatory processes play an important role in pathophysiology of myocardial injury.13,14 Thus, rosuvastatin may act as an antioxidant and anti-inflammatory agent to prevent myocardial injury. Also, the mechanism by which it protects against myocardial injury is not elucidated. The present study was proposed to investigate the cardioprotective potential of rosuvastatin on ISO-induced myocardial injury in rats.
Methods
Chemicals
Rosuvastatin calcium was bought from AstraZeneca Pharma, India Limited and isoprenaline hydrochloride (powder, >99% purity) was purchased from TCI Chemicals, India. Rosuvastatin was dissolved in normal saline. All other chemicals and reagents used in this study were of analytical grade.
Animals
Wistar rats weighing 180–220 g and 8–10 weeks of age, purchased from the National Institute of Pharmaceutical Education and Research (NIPER), Sahibzada Ajit Singh Nagar, Punjab, India, were used. The animals were maintained under standard laboratory conditions at 25±2°C and 50 ± 15% humidity with a 12/12 h light/dark cycle. They were acclimatized to the conditions of animal house for 10 days before the experiment and allowed free access to standard laboratory diet (Ashirwad Industries, Chandigarh, India) and water. All procedures in this study were in accordance with the Institutional Animal Ethics Committee of Guru Angad Dev Veterinary and Animal Sciences University, Ludhiana, India.
Experimental protocol
The rats were randomly divided into five groups (n = 35). Control group: control rats received normal saline solution (P/O) for a period of 7 days. ISO group: received normal saline for 7 days followed by isoprenaline (150 mg/kg, S/C) on seventh and eighth day. ROSU1 group: rats received rosuvastatin (1 mg/kg, P/O) for a period of 7 days and isoprenaline on seventh and eighth day. ROSU5 group: rats received rosuvastatin (5 mg/kg, P/O) for a period of 7 days and isoprenaline on seventh and eighth day. ROSU10 group: rats received rosuvastatin (10 mg/kg, P/O) for 7 days followed by isoprenaline on seventh and eighth day. At the end of the experimental period, after 24 h of last ISO-injection, rats were anaesthetized and sacrificed humanely. Blood and tissue samples were collected and processed for downstream analysis.
Cardiac injury markers
Serum level of cardiac troponin I (cTnI) was estimated using a standard ELISA kit (DiaMetra SRL, Italy) following the manufacturer’s instructions. Also, serum activities of AST, ALT, and CK-MB were analyzed using commercial kits, purchased from Coral Clinical systems, India.
Lipid peroxidation and protein carbonylation
Cayman’s TBARS (TCA Method) assay kit (USA) was used for estimation of lipid peroxidation (LPO) while carbonyl content which indicates protein oxidation was estimated by using Cayman’s protein carbonyl colorimetric assay kit (USA).
Nitric Oxide
An improved Griess method was used to measure nitric oxide in heart homogenate by estimating the end products of nitric oxide production. 15
Total antioxidant activity and antioxidant enzymes
Total antioxidant activity (TAA) in tissue homogenate was measured according to Re et al. 16 (1999). Activities of superoxide dismutase (SOD), catalase (CAT), glutathione S-transferase (GST), and glutathione reductase (GR) were measured according to the methods of Madesh and Balsubramanian (1998), 17 Aebi (1984), 18 Habig et al. (1974), 19 and Carlberg and Mannervik 1985, 20 respectively.
Cytokines in heart
TNF-α and IL-6 levels in tissue homogenates were measured using enzyme-linked immunosorbent assay kits (Krishgen BioSystems, USA) according to the manufacturer’s protocol.
Histopathological examination
The heart tissues were removed, then the cardiac apex was immediately fixed in 10% formalin for at least 24 h, processed in ethanol, embedded in paraffin wax, sliced (5 μm), stained with hematoxylin and eosin (H&E), and examined under a light microscope (Olympus, Tokyo, Japan).
qRT-PCR
Primer sequences for qRT-PCR.

Statistical analysis
Data are expressed as mean ± SEM and values were evaluated using GraphPad Prism 5.01 software (GraphPad, San Diego, CA, USA). Differences between groups were assessed by one-way analysis of variance (ANOVA) followed by Tukey’s post hoc comparisons to determine differences between the groups. A value of p<0.05 was considered to be statistically significant.
Results
Effect of rosuvastatin on heart weight to body weight
The ratio of heart weight to body weight was significantly increased in the ISO group when compared with the normal control group. Rats pretreated with rosuvastatin @ 5 mg/kg and 10 mg/kg showed a significant reduction in the heart weight to body weight ratio as compared to the ISO group (Figure 1). Effect of rosuvastatin on heart weight to body weight ratio. Results are expressed as Mean ± SEM, n = 7. Compared with control: **p < 0.01; compared with ISO: #p < 0.05. Control: normal control group, ISO: isoprenaline group, ROSU (1,5,10 mg/kg): rosuvastatin groups.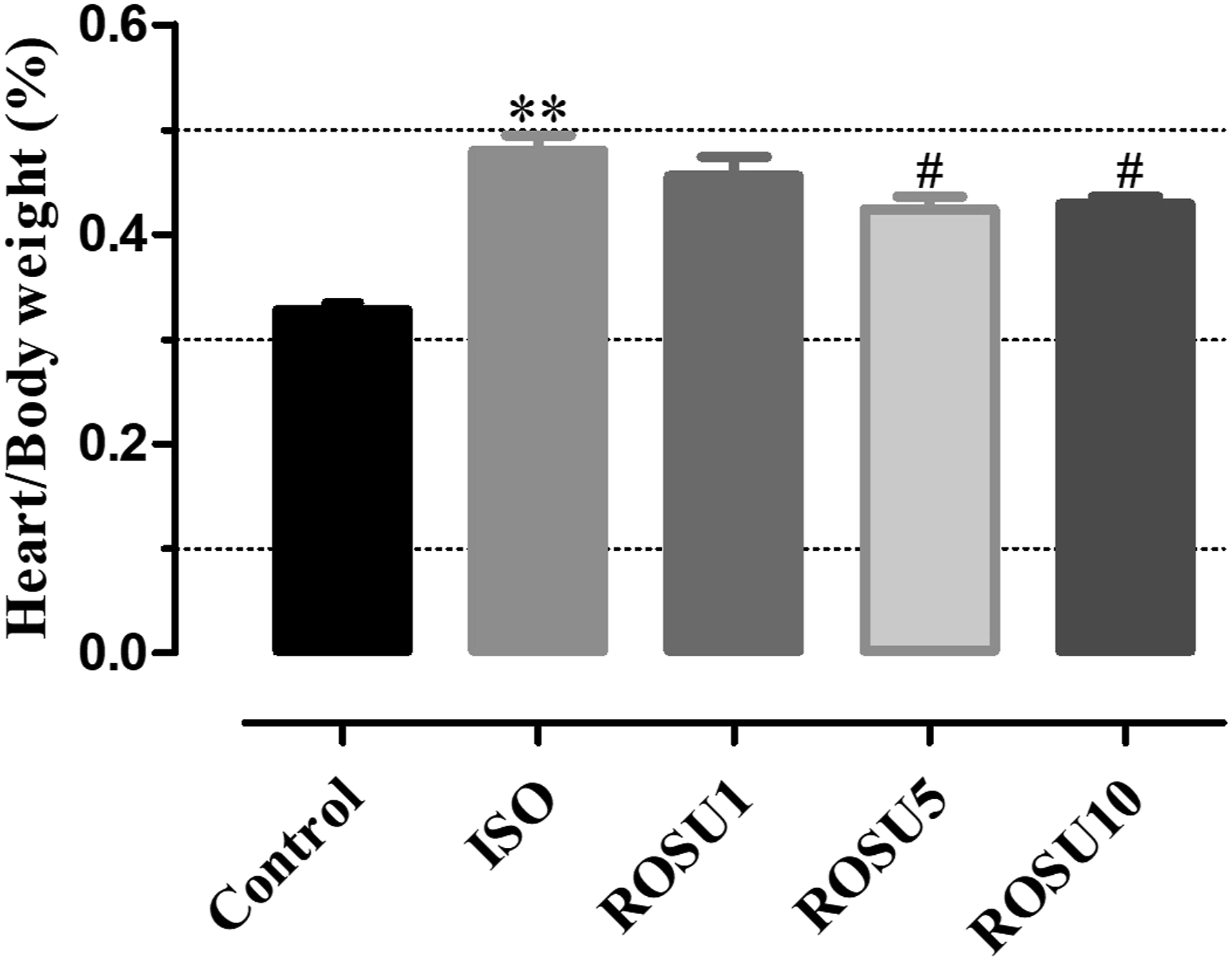
Effect of rosuvastatin on cardiac markers
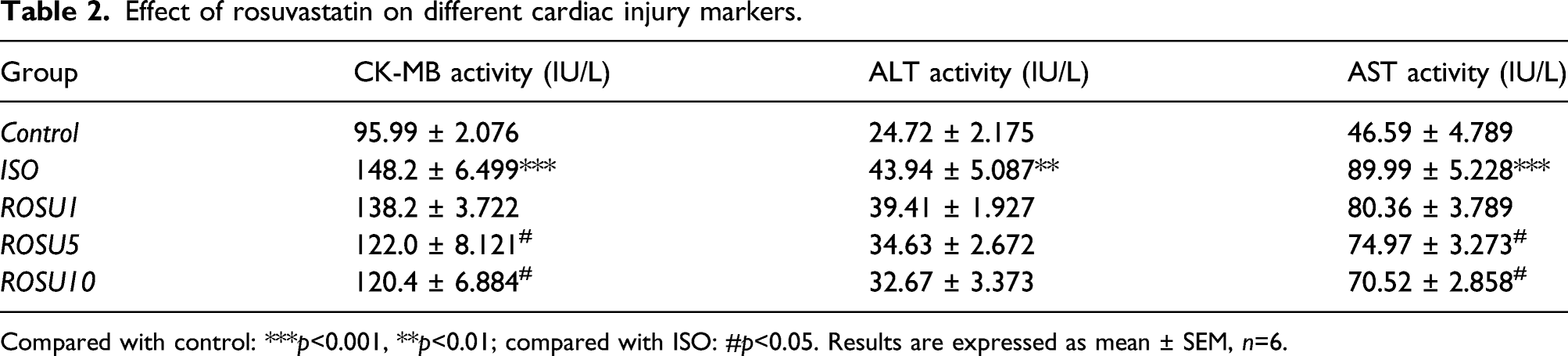
ISO group exhibited higher values of CTnI but pretreatment with rosuvastatin (ROSU10) decreased its value in contrast to ISO group (Figure 2). While ISO group animals showed a significant increase in the activities of serum myocardial injury marker enzymes (CK-MB, ALT, and AST) when compared with that of the control group, pretreatment with rosuvastatin (ROSU5 and ROSU10) maintained significantly lower values of CK-MB and AST (Table 2). Even though ROSU1 group showed lesser values of these markers but these were statistically insignificant. Effect of rosuvastatin on CTnI. Results are expressed as Mean ± SEM, n = 7. Compared with control: **p < 0.01; compared with ISO: #p < 0.05. Control: normal control group, ISO: isoprenaline group, and ROSU (1, 5, and 10 mg/kg): rosuvastatin groups. Effect of rosuvastatin on different cardiac injury markers. Compared with control: ***p<0.001, **p<0.01; compared with ISO: #p<0.05. Results are expressed as mean ± SEM, n=6.
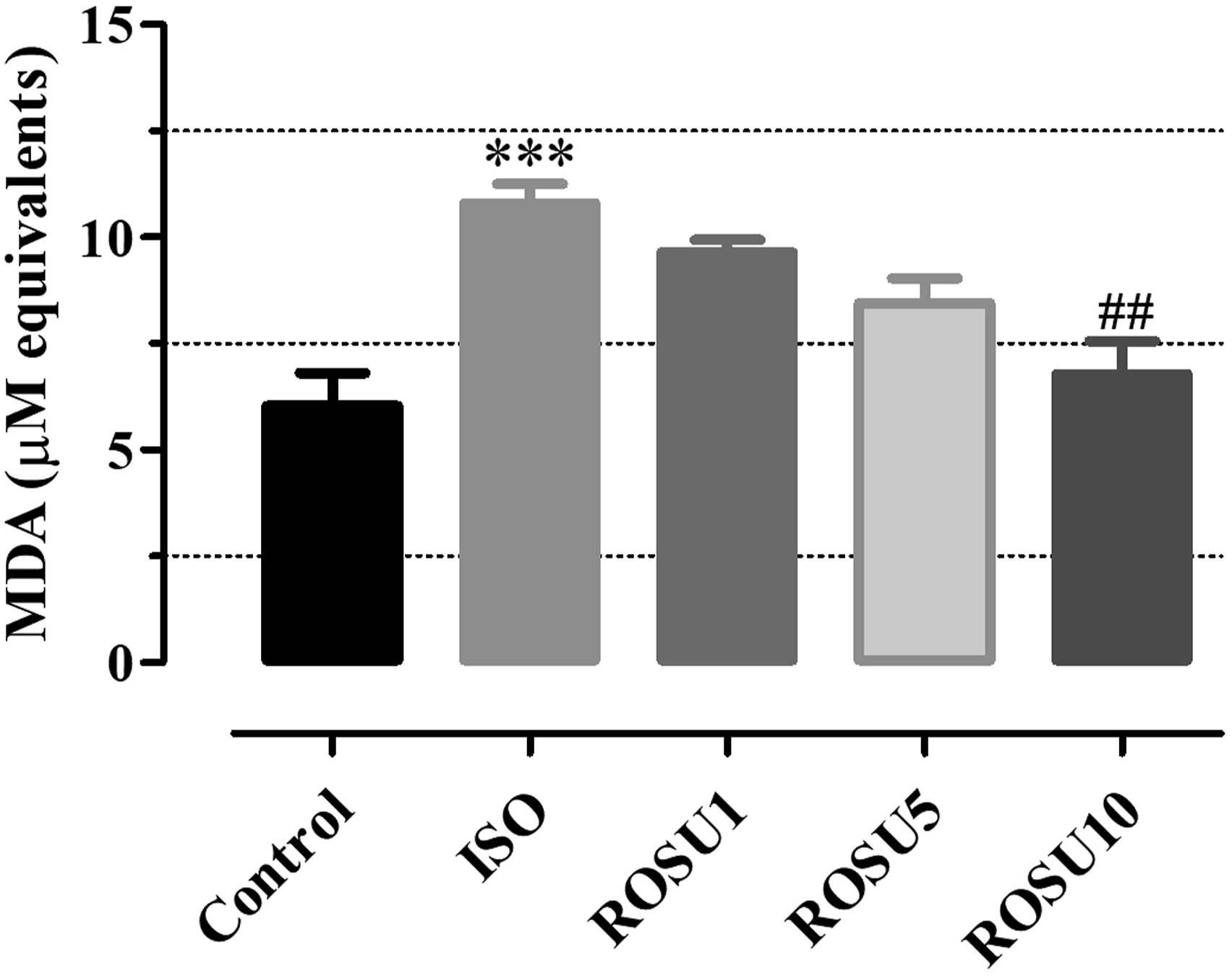
Effect of rosuvastatin on protein carbonylation and lipid peroxidation
Protein carbonyl levels showed a significant increase in the ISO group, which was reduced to a significant extent by pretreatment with rosuvastatin (ROSU10) (Figure 3). Marked increase of malondialdehyde, a lipid peroxidation byproduct, was observed in the heart of ISO group. The increase in lipid peroxidation was minimized significantly by pretreatment of rosuvastatin (ROSU10) (Figure 4). Effect of rosuvastatin on protein oxidation. Results are expressed as Mean ± SEM, n = 6. Compared with control: **p < 0.01; compared with ISO: #p < 0.05. Control: normal control group, ISO: isoprenaline group, and ROSU (1, 5, and 10 mg/kg): rosuvastatin groups. Effect of rosuvastatin on lipid oxidation. Results are expressed as Mean ± SEM, n = 6. Compared with control: ***p < 0.001; compared with ISO: ##p < 0.01. Control: normal control group, ISO: isoprenaline group, and ROSU (1, 5, and 10 mg/kg): rosuvastatin groups.
Effect of rosuvastatin on myocardial nitric oxide
Nitric oxide in heart homogenates was measured by estimating concentration of nitrites. Isoprenaline significantly increased nitrite levels and this increase was minimized significantly by pretreatment of rosuvastatin at 10 mg/kg (Figure 5). Effect of rosuvastatin on myocardial nitric oxide. Results are expressed as Mean ± SEM, n = 6. Compared with control: ***p < 0.001; compared with ISO: #p < 0.05. Control: normal control group, ISO: isoprenaline group, ROSU (1, 5, and 10 mg/kg): rosuvastatin groups.
Effect of rosuvastatin on antioxidant enzymes
Effect of rosuvastatin on antioxidant enzymes.
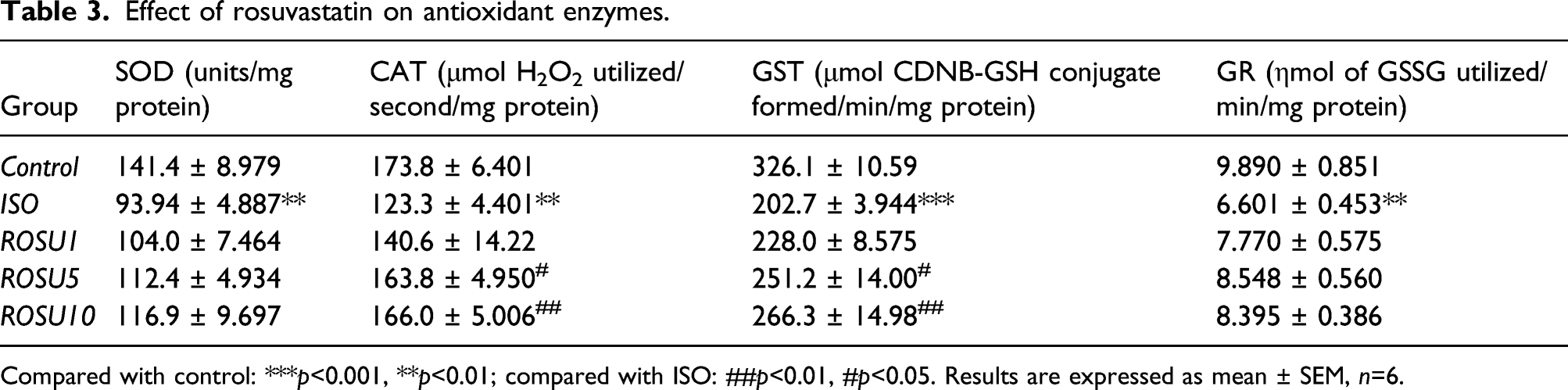
Compared with control: ***p<0.001, **p<0.01; compared with ISO: ##p<0.01, #p<0.05. Results are expressed as mean ± SEM, n=6.
Effect of rosuvastatin on total antioxidant activity
TAA was estimated in terms of Trolox equivalents Figure 6. Isoprenaline caused significant decrease in TAA. However, this decrease was significantly reduced by the pretreatment with rosuvastatin at 10 mg/kg only. Effect of rosuvastatin on total antioxidant activity. Results are expressed as Mean ± SEM, n = 6. Compared with control: **p < 0.01; compared with ISO: #p < 0.05. Control: normal control group, ISO: isoprenaline group, ROSU (1, 5, and 10 mg/kg): rosuvastatin groups.
Effect of rosuvastatin on cytokines in heart
Compared to the normal control group, administration of ISO significantly increased the secretion levels of TNF-α and IL-6 in the heart. Pretreatment with rosuvastatin decreased ISO-induced elevation of cardiac TNF-α at 5 mg/kg and 10 mg/kg dose level (Figure 7). There was no effect of rosuvastatin on the increased levels of IL-6 at any dose level of this study (Table 4). Effect of rosuvastatin on myocardial TNF-α levels. Results are expressed as Mean ± SEM, n = 6. Compared with control: ***p < 0.001; compared with ISO: #p < 0.05, ##p < 0.01. Control: normal control group, ISO: isoprenaline group, ROSU (1,5,10 mg/kg): rosuvastatin groups. Effect of rosuvastatin on tissue IL-6 levels. Compared with control: ***p<0.001. Results are expressed as mean ± SEM (n = 6).

Effect of Rosuvastatin on Histopathology
Isoprenaline caused significant loss of tissue architecture in comparison to the normal control group. It resulted in cellular infiltration, interstitial edema, and myofibrillar degeneration. Furthermore, results of histopathological examination confirmed protective effect of rosuvastatin in ISO-induced myocardial injury. The sections from ROSU5 and ROSU10 groups did show cellular infiltration, interstitial edema and but these changes were milder in magnitude and the tissue architecture was relatively well preserved in comparison to ISO group (Figure 8). Effect of rosuvastatin on histopathological changes in different groups. Cardiac tissues were stained with hematoxylin and eosin. Visualized under a light microscope at × 200 magnification. The dark bar is equal to 100 μm. Asterisks indicate myocardial damage and arrows depict inflammatory process. Control: normal control group, ISO: isoprenaline group, ROSU (1,5,10 mg/kg): rosuvastatin groups.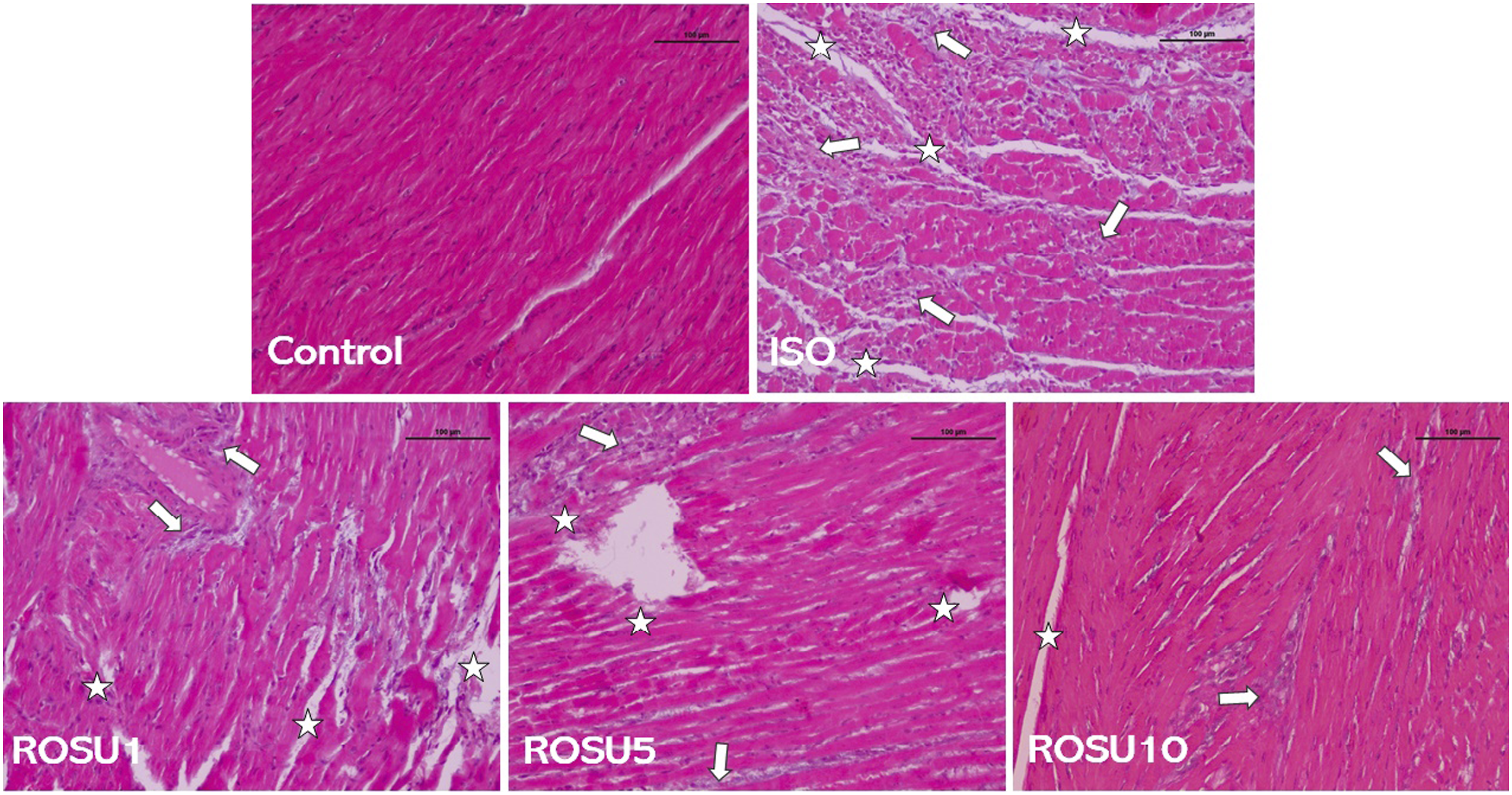
Effect of Rosuvastatin on mRNA Expression of Nrf2,, Bcl-2/Bax, eNOS, and iNOS
Tissue mRNA expression of Nrf2 was significantly lower in ISO group than the control group. While significantly lesser depression of Nrf2 mRNA was seen in ROSU10 group (Figure 9 Effect of rosuvastatin on mRNA expression levels of Nrf2 gene in rats. Compared with control: ***p < 0.001; compared with ISO: #p < 0.05. Results are expressed as mean ± SEM, n = 6. Control: normal control group, ISO: isoprenaline group, ROSU (1,5,10 mg/kg): rosuvastatin groups. Effect of rosuvastatin on Bcl-2/Bax expression ratio. Compared with control: ***p < 0.001; compared with ISO: #p < 0.05. Results are expressed as mean ± SEM, n = 6. Control: normal control group, ISO: isoprenaline group, ROSU (1, 5, and 10 mg/kg): rosuvastatin groups. Effect of rosuvastatin on mRNA expression levels of iNOS gene in rats. Compared with control: ***p < 0.001; compared with ISO: #p < 0.05. Results are expressed as mean ± SEM, n = 6. Control: normal control group, ISO: isoprenaline group, ROSU (1, 5, and 10 mg/kg): rosuvastatin groups.


Discussion
This study indicates the cardioprotective potential of rosuvastatin against ISO-induced myocardial injury. Pretreatment with rosuvastatin significantly decreased serum levels of cardiac marker enzymes in ISO-induced cardiac injury in rats. Also, rosuvastatin reduced ISO-induced decline in antioxidants and minimized lipid peroxidation. In our study, a rat model of myocardial injury was successfully established, as demonstrated by significantly increased serum levels of cardiac troponin, marker enzymes, and abnormal cardiac architecture observed in histopathology. These results are in accordance with previous in vivo studies. 22 ISO-induced injury caused increased cellular infiltration, increased water content, interstitial edema, and necrosis which may be responsible for the increased heart to body weight ratio. Higher levels of serum cTnI measured in ISO group indicate leakage of this protein because of necrosis and injured cellular membranes caused by high levels of isoprenaline. Rosuvastatin prevented the increase in cTnI levels indicating that rosuvastatin may have protective potential against acute cardiac injury.
Myocardial injury and oxidative stress are related. Metabolic disturbance in oxidants and antioxidants has been noted in patients with acute myocardial infarction. 23 We observed increased malondialdehyde and decreased antioxidants in ISO-treated rat heart tissues. Protein carbonylation, an irreversible oxidative modification of proteins, is considered an indicator of oxidative damage. Our data indicate that the oxidative stress produced by isoprenaline caused carbonylation of proteins and these findings are in agreement with those of Dhivya et al. 24 (2017). Thus, it suggests that oxidative stress may be involved in ISO-induced myocardial injury of rats. However, the treatment of rosuvastatin reduced ISO-induced oxidative stress by increasing antioxidant activities, decreasing lipid peroxidation and having a sparing effect on proteins, thus preventing cellular damage. SOD, CAT, GST, GPx, and GR are considered as first-line cellular defense against oxidative stress. 25 In our study, significantly lower activities of SOD, CAT, GST, GPx, and GR were detected in heart of ISO-treated rats when compared to control rats. Such a decrease in the activities might be due to their increased utilization for scavenging ROS and their inactivation. Their decreased activities make myocardial cell membranes more susceptible to oxidative damage. Redox balance in the cell is maintained by its endogenous antioxidant defense system, which includes antioxidant enzymes and non-enzymatic compounds. 26 This represents the total antioxidant activity of a system as it considers a cumulative antioxidant effect within a sample. Tissue homogenates from ISO group showed significantly lower Trolox equivalents. These results are in accordance to those of Kocak et al. 27 (2016). Pretreatment with rosuvastatin maintained TAA at a higher level compared to the ISO group. These results back the findings with oxidant and antioxidant parameters in this study.
The endogenous nitric oxide is produced from L-arginine, oxygen, NADPH, and NO synthase. Low levels of NO commonly target soluble guanylate cyclase and activate cGMP-mediated signaling, which mediates diverse physiological effects. However, high levels of NO interact with molecular oxygen and anion superoxide resulting in the production of reactive nitrogen species like dinitrogen trioxide and peroxynitrite. 28 These agents having oxidizing potential, can deplete sulfhydryl groups, and oxidize most biomolecules, acting similar to hydroxyl radical. The peroxynitrite species can rapidly react with manganese-superoxide dismutase and inactivate it. 29 Our study indicates that ISO-induced injury results in a buildup of myocardial NO and generates radical cascade that may disturb various cellular targets and mediates cardiomyopathy. Rosuvastatin reduced this NO buildup in cardiac tissue signifying that it may act as a protective agent.
TNF-α is an important mediator in cardiovascular disorders as it causes contractile dysfunction, apoptosis, and activates other inflammatory cytokines. 30 ROS induce apoptosis by activating p38 MAPK which results in the activation of NF-κB and TNF-α production. 31 The ISO-induced myocardial injury resulted in very high levels of TNF-α and IL-6 as supported by previous studies. 32 Rosuvastatin mitigated this rise in the levels of TNF-α. However, rosuvastatin pretreatment had no effect on the elevated levels of IL-6. Keep in mind that IL-6 signaling is cardioprotective during acute responses and induces cardiac dysfunction under chronic conditions. 33 Such a temporal regulation of IL-6 is apparent in cardiac tissue. 34 In accordance with cardiac injury markers, cytokines, and redox parameters, histological studies of ISO group showed marked degenerative changes with cellular infiltration, necrosis, interstitial edema, and debris. Such pathological changes are in line with the earlier studies. 35 The present study established that rosuvastatin pretreatment preserved the tissue architecture which might be ascribed to its antioxidant and anti-inflammatory potential.
Nrf2 maintains redox balance in response to toxic or oxidative insults. We expected higher mRNA expression of Nrf2 after isoprenaline but this was not the case. We observed a reduction in isoprenaline-dosed rats. This might relate to the TNF-α rise. TNF-α affects Nrf2 signaling in a concentration-dependent fashion as high levels of TNF-α markedly suppress Nrf2 system and mediate cellular toxicity. 36 Such a relationship is indicated in our study where the elevated levels of TNF-α by isoprenaline may cause depression of Nrf2 signaling at different levels, that may be involving the mRNA depression also. Other factors which may play a role include dose and time. Since we used a very high dose level of isoprenaline over a short period of time, this may result in reduction of Nrf2 levels. Rosuvastatin pretreatment minimized this mRNA depression of Nrf2 indicating protective potential. It has been reported that upregulation of proapoptotic factors (Bax) with or without the down-regulation of antiapoptotic factor (Bcl-2) is associated with apoptosis and cell survival. Both necrosis and apoptosis mediate acute myocardial injury. 37 Imbalance in Bcl-2 and Bax expression (here decreased Bcl-2/Bax ratio) may result in mitochondrial dysfunction. This balance of Bcl-2/Bax expression is a chief factor in determining the degree of apoptosis. In this study, rosuvastatin pretreatment increased Bcl-2/Bax ratio, indicating that it restores the balance between Bcl-2 and Bax.
During ischemia, there is a buildup of tissue NO from different sources and upon reperfusion, due to burst of ROS release, NO gets converted to peroxynitrite, thus causing reperfusion injury. Use of NOS inhibitors has been shown to decrease reperfusion injury by reducing peroxynitrite formation. 28 Excess production of NO from upregulation of iNOS results in myocyte apoptosis, cardiac remodeling, and dysfunction.38,39 In our study increased iNOS expression was observed on isoprenaline administration as seen in other studies. 40 However, isoprenaline treatment also increased the expression of eNOS which was contrary to our expectations. It has been reported that administration of isoprenaline in rats caused upregulation of eNOS and β2 stimulation resulted in elevated eNOS mRNA levels. 41 These findings are supporting our results in the ISO group. However, pretreatment with rosuvastatin did not alter this increase in eNOS mRNA level. iNOS gene expression in collaboration with the nitrite levels support each other, indicating the protective potential of rosuvastatin against acute cardiac injury.
Conclusion
The present study indicates that rosuvastatin reduced ISO-induced myocardial injury in rats. The effect is associated with its influence on the ROS generation, immunomodulation, membrane stability, Nrf2 mRNA preservation, and iNOS and Bcl-2/Bax expression levels. However, further studies are needed to explore the exact mechanism, tackle the limitations, and determine the clinical utility of the drugs in veterinary and human medicine. Our study suggests that rosuvastatin may have the potential for the prevention of myocardial injury.
Footnotes
Acknowledgments
The authors are thankful to Dr(s) Javeed Ahmad Dar, Ishfaq Maqbool and Arshad Hussain for their help in histopathological and PCR studies. Dr Faheem Sultan received research fellowship from Indian Council of Medical Research during this study and we acknowledge that.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
