Abstract
Introduction
Diabetic nephropathy (DN), a global problem that threatens human health, is an important reason for chronic kidney disease and kidney failure. In our study, it was aimed to investigate the individual and combined effects of SA and EA in streptozotocin (STZ)-induced rats.
Methods
The groups are as follows: Control, untreated diabetic, diabetic treated with Sinapic acid (SA), diabetic treated with Ellagic acid (EA), diabetic treated with SA and EA, treated with SA, treated with EA, and treated with SA and EA. Total kidney volume, total glomerulus volume, total filtration space volume, caspase-3, and 8-OHdG immunoreactivity, Malondialdehyde (MDA), Glutathione (GSH), Catalase (CAT), serum urea, and creatinine levels were evaluated by stereological, immunohistochemical, and biochemical methods.
Results
The findings of the study showed that total kidney volume, total glomerulus volume, total filtration gap volume, caspase-3, and 8-OHdG immunoreactivity, MDA, serum urea, and creatinine levels significantly increased in the untreated diabetic group compared to the control group. Also, severe mesangial and glomerular enlargement, extracellular matrix accumulation, and glomerular and tubular basal membrane thickness were observed in the tubulointerstitial and glomerular of the diabetic rats. However, individual and combined treatments of SA and EA ameliorated these histological changes. Additionally, decreased GSH and CAT in the untreated diabetic group increased by SA and EA treatment.
Conclusions
The findings suggest that treatment of SA and EA prevent apoptosis and DNA damage and structural changes in STZ-induced DN. However, the combined treatment of SA and EA were more effective than their individual treatments in all parameters except serum urea and creatinine.
Introduction
Diabetes mellitus (DM) is characterized by hyperglycemia which is resulted from defects in insulin synthesis, insulin secretion and insulin action, or all of them. Persistent hyperglycemia is related to long-term damage, dysfunction, and failure of various organs such as the kidneys, nerves, and heart in the future and is the main factor in the development of many complications in diabetes patients. 1 Thus, secondary complications of DM will be one of the most prominent medical concerns of the future. 2 Diabetic nephropathy (DN), the most common cause of kidney failure, is one of the major complications of diabetes which threatens human health. 3 Approximately 15–25% of Type-1 diabetes patients and approximately 30–40% of Type-2 diabetes patients suffer from DN. 4
Although the mechanism underlying DN is not fully understood, factors such as oxidative stress, inflammation, and apoptosis play an important role in the development of DN. DN is also characterized by structural changes such as mesangial expansion, thickening of the basement membrane, accumulation of extracellular matrix, hypertrophy and glomerulosclerosis, and functional impairments such as increased serum urea and creatinine. 5
Hyperglycemia, an important factor in the development of DN, is associated with the generation of reactive oxygen species (ROS) and, consequently, causes oxidative damage in kidneys.2,6 Therefore, controlling hyperglycemia and the generation of ROS is essential to preventer to treat DN and other diabetic complications.4,7 Synthetic hypoglycemic agents which are used to reduce or to prevent the DN have some toxic effects that can cause serious health problems. 8 More effective alternative treatments are needed as there is no satisfactory treatment for DN in modern medicine. 9 World Health Organization (WHO) has suggested the assessment and implementation of traditional herbal treatments for diabetes and its complications because they are effective, non-toxic, have fewer side effects or have no side effects, and are considered to be excellent candidates for oral treatment. 16 In recent years, many researchers have investigated the application of these plants both individually and in combination to treat diabetes and its complications.3,10,11
Herbal compounds are widely used by humans as they have enormous potential effects in the treatment of many diseases such as nephrotoxicity. 33 Polyphenol compounds have attracted major attention in recent years due to their beneficial and therapeutic effects. 12 Among them, SA and EA are some of the beneficial polyphenolic compounds which are found naturally in vegetables and fruits. It has been reported that these compounds have many beneficial effects, such as antimicrobial, anti-inflammatory, antioxidant, anticancer, and antidiabetic effects.3,13–15 Previous studies have been demonstrated that SA has an antihyperglycemic effect by increasing GLUT4 expression and glucose uptake in muscle tissue of diabetic rats. 35 Alaofi also reported that SA ameliorated the DN via NRF2/HO-1 mediated pathways in rats. 4 Similarly, it was declared that EA showed an anti-diabetic effect by decreasing serum glucose levels and increasing insulin and C-peptide levels in diabetic rats. 3 Previous studies also reported that EA attenuated the DN via the regulation of oxidative stress and inflammatory signaling in diabetic rats. 16
But so far, in the literature, little has been reported regarding the possible protective effects of SA and EA in STZ-induced DN, especially with their combined treatment. Further studies are needed to fully elucidate the mechanism of action of individual and combined treatments of SA and EA in DN. It has been understood that further studies are needed to fully elucidate the mechanism of action of both individual and combined treatments of SA and EA in DN. Therefore, this study aims to investigate the possible renopreventive effects of SA and EA in STZ-induced DN by trying to explain the roles of apoptosis, DNA fragmentation, oxidative stress, morphometrical, and functional changes by using many methods such as stereological, histopathological, immunohistochemical, and biochemical methods in the kidney of diabetic rats.
Material and Method
Chemicals
Caspase-3 (sc-7272), 8-OHdG (sc-66036), EA (sc-202598A), and STZ (sc-200719) were obtained from Santa Cruz Biotechnology. SA (CAS number: 530–59-6) was obtained from Sigma Aldrich. The kit and other chemicals required for immunohistochemical examination were obtained from Thermo Fisher Scientific.
Animals and Experimental protocol
In the present study, fifty-six adult Albino Wistar female rats weighing 200–250 g were randomly divided into eight groups (n = 7): Control (C) group was received water, untreated diabetic (UTD) group was injected a single dose of 50 mg/kg of STZ by intraperitoneal (IP), the SA-treated diabetic (DSA) group was injected a single dose 50 mg/kg of STZ by IP and given 20 mg/kg/d SA by gavage, 10 the EA-treated diabetic (DEA) group was injected a single dose 50 mg/kg of STZ by IP and given 50 mg/kg/d EA by gavage 3 , SA- and EA-treated diabetic (DSAEA) group was injected a single dose 50 mg/kg of STZ by IP and given 20 mg/kg/d SA and 50 mg/kg/d EA by gavage, SA group was given 20 mg/kg/d SA by gavage, EA group was given 50 mg/kg/d EA by gavage, and SAEA group was given 20 mg/kg/d SA and 50 mg/kg/d EA by gavage. The experimental treatment period lasted 28 days. To confirm experimental diabetes, 72 h after STZ injection in rats, blood glucose was measured from the blood obtained from the tail veins. Rats with blood glucose levels of 250 mg/dL and above were considered to be diabetic rats and were selected for the experiment. Animals were obtained from Van Yüzüncü Yıl University Experimental Medicine Application and Research Center. Animals were maintained at normal light and dark cycles (12 h:12 h light/dark), at temperature (21 ± 2°C) and humidity (50% ± 10); their feed and water were given in accordance with the standards (ad libitum). All protocols of the experiment have been approved by Van Yüzüncü Yıl University Animal Experiments Local Ethics Committee (institutional approval number: 06.02.2020/03). All study protocol was also performed according to the guidelines of the Helsinki Declaration in 2000.
At the end of the experiment period, the rats were anesthetized with intraperitoneal ketamine (50 mg/kg) and xylazine (10 mg/kg). 17 The thoracic region was dissected and blood taken from the heart was transferred to heparinized tubes. The kidney was removed and fixed in 10% neutral buffered formalin for histopathological, stereological, and immunohistochemical evaluation. A part of the kidney was homogenized for the analysis of tissue oxidative stress parameters.
Stereology
For stereological analysis, kidney tissue was evaluated by random sampling and separated into fractions (f). For this, the kidney tissue was divided into two equal parts (f1 = 1/2). One of the two pieces was randomly selected and embedded in paraffin after histological tissue processing stages. 4 μm thickness were cut from paraffin blocks by a rotary microtome (Leica RM 2125, China). The first section was taken randomly, and the next sections were taken every 100 steps (f2 = 1/100). An average of 8–10 sections was taken. The sections were stained with Hematoxylin-Eosin (H-E) and examined under a light microscope (Olympus BX53, Japan) using a camera (Olympus DP27, Japan) and microscope imaging systems (Olympus cellSens Entry, Japan).
Cavalieri principle was used to estimate the total volume of the kidney, the total volume of the glomerulus, and the total volume of filtration space. For this, the total numbers of points hitting the kidney, a glomerulus, and filtration space were counted using the point-counting probe (Figure 1). The following formula was used to calculate the volume Placing the point-counting probe on the kidney.
where ‘‘V’’ is the volume of the structure, ‘‘∑p’’ is the total number of points hitting the structure, ‘‘a/p’’ is the area covered by one point, and ‘‘t’’ is the section thickness. Obtained total volume values are multiplied by the inverse of the fraction ratios (f1 and f2).
After the histological tissue processing stages (fixation, processing, sectioning, and staining), the volume of tissue is usually shrunk. To determine the final kidney volume, the tissue shrinkage value must be determined. Thus, the degree of tissue shrinkage (d(shr)) is calculated by the following formula
Coefficient error (CE) was calculated to determine whether sampling is sufficient for each animal. The coefficient of variation (CV) was calculated to determine whether the animals in the group were sufficient by taking the changes depending on the biological variation into account.
Histopathological and immunohistochemical evaluation
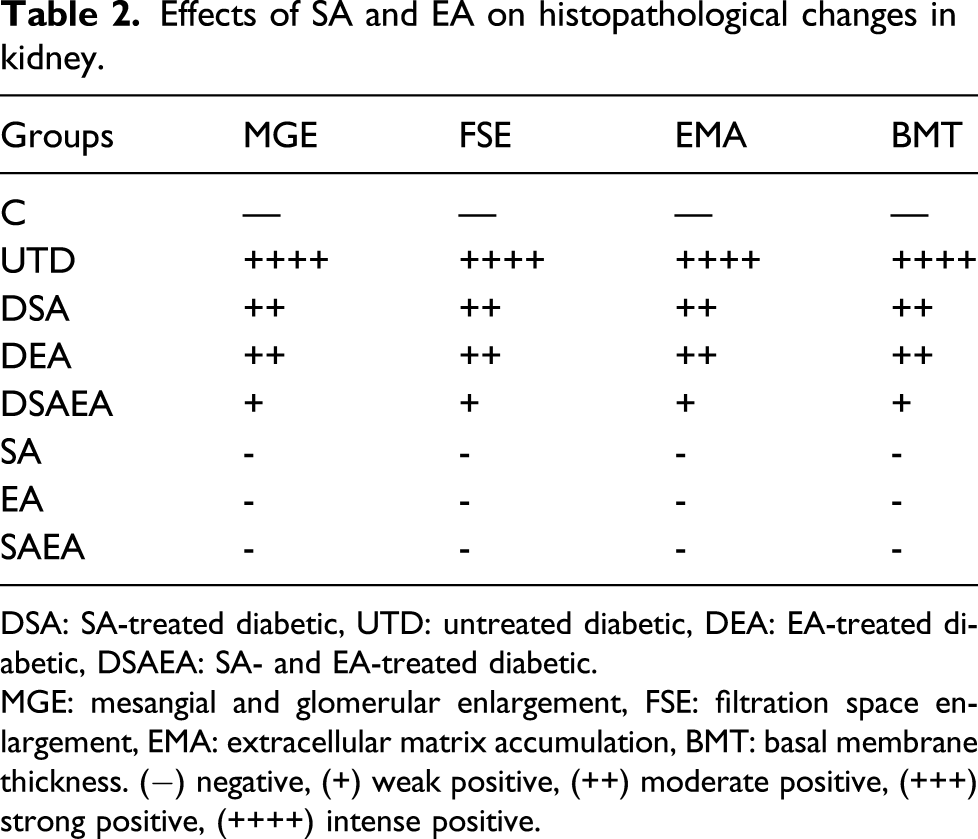
The kidney was fixed in buffered formalin and then embedded in paraffin. 4 μm thickness sections taken from paraffin blocks of the kidney were stained with periodic acid Schiff (PAS) and Masson’s trichrome stains for histopathological evaluation and examined by light microscopy (Olympus BX53, Japan). For histopathological evaluation, an average of 15–20 areas was evaluated by random sampling for each animal in the groups. The findings were semiquantitatively evaluated according to the number of lesions observed in examined areas; thus, it was performed as normal: (−) negative reaction, (+) weak positive reaction, (++) moderate positive reaction, (+++) strong positive reaction, and (++++) intense positive reaction.
In STZ-induced DN, the immunohistochemical method was used to elucidate the protective effects of SA and EA on apoptosis and oxidative damage in DNA. Caspase-3 (Santa Cruz Biotechnology, dilution ratio: 1/50) and 8-OHdG (Santa Cruz Biotechnology, dilution ratio: 1/50) primary antibodies were used, respectively, to determine apoptosis and DNA damage. After fixation, the tissue was washed with water, dehydrated with alcohol, and cleared with xylene, and embedded in paraffin. The sections of 4 μm thickness were cut from paraffin blocks by a rotary microtome. Sections taken for immunohistochemical examination were deparaffinized and dehydrated. To prevent nonspecific background staining due to endogenous peroxidase, incubate slide in 3% hydrogen peroxide (H2O2) for 10 min. To prevent antigen masking in the nucleus, the sections were heated twice in the microwave oven in antigen retrieval (citrate buffer, pH 6.1) solution for 5 min. To block nonspecific background staining, incubate slide in Ultra V Block for 10 min at room temperature. Slides were incubated in primary antibodies at +4 C in a humidity chamber overnight. After washing the sections with PBS, respectively, Biotinylated Goat Anti-Polyvalent and Streptavidin–Peroxidase conjugate were dropped, and both of them were incubated for 10 min. Diaminobenzidine (DAB) was used as a chromogen and then counterstained with Mayer’s hematoxylin. It was then examined under a light microscope (Nikon Y-IM 7551012, Japan). For immunohistochemical evaluation, in areas selected by random sampling, apoptotic cells which were calculated to their immunoreactivity grades were applied H-score and were statistically evaluated.
Biochemical analysis
At the end of the study, blood samples were taken by cannulation of the heart with an injector. Blood samples were centrifuged at + 4°C at 3000 r/min for 10 min. Serum urea and creatinine were evaluated in an autoanalyzer (Architect ci16200) using commercial kits (Archem Diagnostics).
Measurement of oxidative stress–associated enzymes in kidney
In order to evaluate the antioxidant, GSH and CAT activity was measured and MDA content was measured to determine lipid peroxidation. To measure the oxidative stress–associated enzymes, kidney tissues were homogenized by a homogenizer. Homogenates were centrifuged at 10000 × g for 15 min at + 4 C. The obtained supernatants were used for biochemical analysis. Analysis of GSH, CAT, and MDA was evaluated by a spectrophotometric method according to the previous studies. 21
Statistical analysis
Statistical analyses were accomplished by using SPSS 21.0 software. Differences between groups were evaluated by the Kruskal–Wallis test, and the Mann–Whitney U test was used to evaluate the difference between the two groups. p value ≤ 0.05 was accepted as statistically significant. All data were expressed as means ± standard deviations.
Results
Stereological findings
Effects of SA and EA on kidney volume, glomerulus volume, and filtration space volume in STZ-induced rats.

DSA: SA-treated diabetic, UTD: untreated diabetic, DEA: EA-treated diabetic, DSAEA: SA- and EA-treated diabetic.
Values are expressed as means ± SD. Significant differences as compared with the C group at *p <0.05, **p<0.01, and ***p<0.005.
Significant differences as compared with the DC group at #p < 0.05, ##p < 0.01, and ###p < 0.005.
Histopathological findings
The kidney tissues of the rats in the control group were observed to have normal histological architecture. Compared to the control group, severe mesangial and glomerular enlargement, filtration space enlargement, extracellular matrix accumulation causing fibrosis, and sclerosis in the tubulointerstitial and glomerular were observed in the diabetic group. In addition, glomerular and tubular basal membrane thickness increased. Moderate mesangial and glomerular enlargement, glomerular and tubular basement membrane thickness, and extracellular matrix accumulation in tubulointerstitial and glomerular rats were observed in diabetic rats with the individual treatment of SA and EA. However, mild mesangial and glomerular enlargement, glomerular and tubular basement membrane thickness, and extracellular matrix accumulation in tubulointerstitial and glomerular rats were observed in diabetic rats with combined treatment of SA and EA (Figure 2(a) and (b) and Table 2). Effect of SA and EA on histopathological changes in the kidney. Masson’s trichrome (a) and PAS (b) stains were used to evaluate renal histopathological changes. Blue areas (a) indicate Masson’s trichrome staining positivity, while pink areas (b) indicate PAS staining positivity in the glomeruli and interstitial areas. 400X. Note: PAS: periodic acid Schiff. Effects of SA and EA on histopathological changes in kidney. DSA: SA-treated diabetic, UTD: untreated diabetic, DEA: EA-treated diabetic, DSAEA: SA- and EA-treated diabetic. MGE: mesangial and glomerular enlargement, FSE: filtration space enlargement, EMA: extracellular matrix accumulation, BMT: basal membrane thickness. (−) negative, (+) weak positive, (++) moderate positive, (+++) strong positive, (++++) intense positive.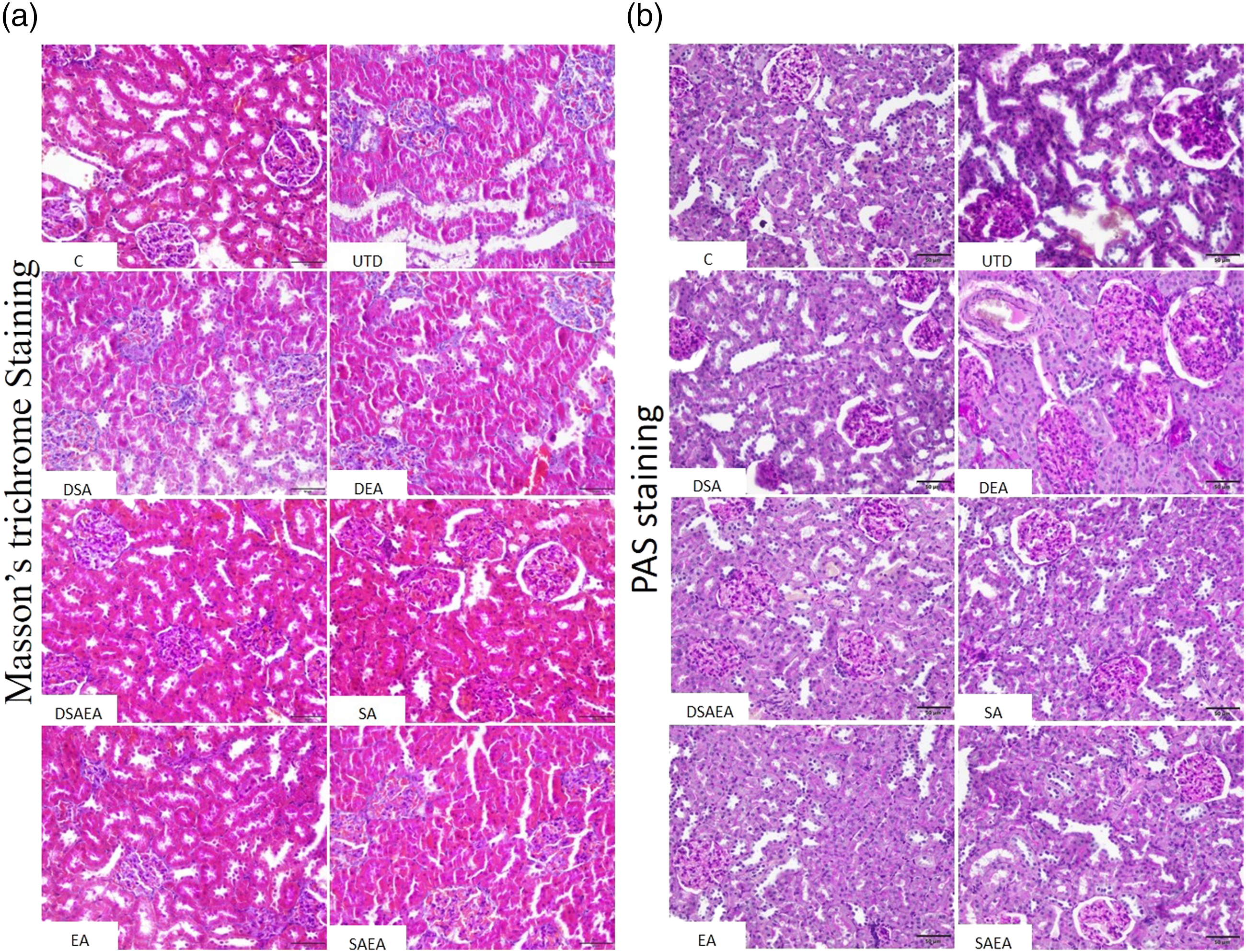
Expression of caspase-3 and 8-OHdG in the kidney of diabetic rats
In the present study, it was observed that caspase-3 and 8-OHdG immunoreactivity was significantly increased in the kidney of the UTD group compared to the control group. However, caspase-3 and 8-OHdG immunoreactivity significantly reduced in diabetic rats treated with SA and EA compared to the UTD group. In addition, it was observed that caspase-3 and 8-OHdG immunoreactivity in the combined treatment group of SA and EA were less than in the individual treatment group of SA and EA (Figures 3 and 4). Comparison of caspase-3 and 8-OHdG immunoreactivities by H-score in the kidney of diabetic rats. Values are expressed as means ± SD. Significant differences as compared with the C group at *p < 0.05, **p < 0.01, and ***p < 0.005. Significant differences as compared with the DC group at #p < 0.05 and ##p < 0.01. Immunohistochemical photomicrographs of caspase-3 and 8-OHdG expressions in the kidney. Arrows indicate caspase-3 and 8-OHdG immunoreactivity. Caspase-3 and 8-OHdG immunoreactivity increased in the untreated diabetic group. However, caspase-3 and 8-OHdG immunoreactivity reduced in diabetic rats treated with SA and EA. 400X.

Biochemical findings
The effects of SA and EA on serum kidney-function markers in diabetic rats.

UTD: untreated diabetic, DEA: EA-treated diabetic, DSAEA: SA- and EA-treated diabetic, DSA: SA-treated diabetic.
Values are expressed as means ± SD. Significant differences as compared with the C group at *p < 0.05, **p < 0.01, and ***p < 0.005.
Significant differences as compared with the DC group at #p < 0.05, ##p < 0.01, and ###p < 0.005.
The effects of SA and EA on oxidative stress–associated enzymes in kidney
Effects of SA and EA on MDA, GSH, and CAT in the kidney of diabetic rats.
DSA: SA-treated diabetic, UTD: untreated diabetic, DEA: EA-treated diabetic, DSAEA: SA- and EA-treated diabetic.
Values are expressed as means ± SD. Significant differences as compared with the C group at *p < 0.05, **p < 0.01, and ***p < 0.005.
Significant differences as compared with the DC group at #p < 0.05, ##p<0.01, and ###p < 0.005.
Discussion
DN, a serious complication of DM, is characterized by morphological, molecular, and functional changes in the kidney such as mesangial and glomerular enlargement, extracellular matrix accumulation, basal membrane thickness, hypertrophy, oxidative stress, apoptosis, DNA damage, and increased serum urea and creatinine. 22 In this study, we investigated the renoprotective effects of individual and combined treatments of SA and EA and their potential mechanisms on DN in STZ-induced diabetic rats. The stereological, histopathological, immunohistochemical, and biochemical data of this study revealed that individual and combined treatments of EA and SA prevented the progression of DN, especially their combined treatments been more effective. Therefore, the pathological changes observed in diabetic rats in the present study, such as mesangial and glomerular enlargement, extracellular matrix accumulation, basal membrane thickness, hypertrophy, oxidative stress, apoptosis, DNA damage, and increased serum urea and creatinine, which are typical features of DN in diabetic rats, were considerably ameliorated with SA and EA treatment. The findings of the current study are compatible with reports of many previous studies which have shown similar findings.10,23–25
It has been reported that long-term hyperglycemia led to the overproduction of reactive oxygen species (ROS) and oxidative stress in tissues. 26 Overproduction of ROS and oxidative stress can cause cell damage and trigger the production of MDA, a marker of lipid peroxidation. 27 Nevertheless, ROS overproduction and oxidative stress are widely accepted theory for explaining DN. 23 In the present study, we demonstrated that MDA level significantly increased, while the activity of GSH and CAT which are antioxidant defense enzymes significantly decreased in the kidney of STZ-induced diabetic rats. These findings of the present study suggest that SA and EA have a renoprotective effect against oxidative stress by down-regulating lipid peroxidation enzymes and up-regulating antioxidant defense enzymes. So, ROS produced in kidney cells is detoxified by increased GSH and CAT enzymes with SA and EA treatments in the kidney. Besides, the combined treatment of SA and EA has a stronger antioxidant and ROS scavenging effect than the individual treatment of SA and EA. Consistent with these findings of the current study, Alaofi revealed that MDA and NO2 increased, while GSH and CAT decreased in the kidney of STZ-induced diabetic rats. Moreover, SA treatment decreased MDA and NO2 activity and increased GSH and CAT enzymes in DN. 10 Zhou et al. reported that EA attenuated increased MDA levels and decreased SOD levels in STZ-induced diabetic mice. 16 Pradeep and Srinivasan reported that 8-OHdG immunoreactivity, which is an important marker of oxidative DNA damage, increased in kidney tissue in STZ-induced DN. 28 In this study, the significant increase in 8-OHdG expression in the kidney of STZ-induced diabetic rats is consistent with previous studies. 2 This increase in 8-OHdG expression in the kidney may be associated with increased lipid peroxidase (LPO) in DN. 4 In the present study, the treatments of SA and EA reduced the 8-OHdG expression in diabetic rats compared to the untreated diabetic group. This is estimated to be due to the increased ROS scavenging effects of SA and EA or may be due to their antioxidant effects.10,16 Thus, the lipid peroxidation, antioxidant, and 8-OHdG data of this study support each other. 8
Besides oxidative stress in inducing DN, apoptosis has also an important role in detecting kidney dysfunction and histological deterioration in the kidney 4 because oxidative stress and apoptosis are related situations. Caspases, which play a key role in the progression of apoptosis, have been reported to be responsible for various biochemical and morphological changes. 29 Ahmed et al. reported that caspase-3 expression increased in STZ-induced DN. 30 Similarly, Jing et al. demonstrated that there was a significant increase in caspase-3 expression in STZ-induced untreated diabetic rats. 13 In previous studies, it was reported that SA and EA exhibited antiapoptotic effects by decreasing caspase-3 and Bax expressions, which increased in nephrotoxicity.6,18 In this study, caspase-3 expression, a marker of apoptosis, which plays a crucial role in the pathogenesis of DN increased in the untreated diabetic kidney. Moreover, apoptosis can decrease the functional capacity of the kidney, which plays a significant role in the development of DN. 16 In the present study, SA and EA treatment significantly decreased caspase-3 expression in STZ-induced diabetic rats and it is compatible with previous studies.18,31 However, we found that the combined treatment of SA and EA is more effective in reducing caspase-3 expression in DN than their individual treatments. These findings reveal that SA and EA can have antiapoptotic effects in DN.
Transforming growth factor-β1 (TGF-β1) is an important factor that has been determined to play a role in the development of DN. In previous studies, it has been reported that increased expression of TGF-β1 in DN causes renal hypertrophy and extracellular matrix accumulation in the mesangium. Besides, TGF-β1 has been reported to cause albuminuria by increasing glomerular permeability and by decreasing reabsorption in proximal tubes. 24 Albuminuria is considered an early marker in progressive of DN. 12 Diabetic albuminuria is associated with histopathological changes such as glomerular basement membrane (GBM) thickness and mesangial enlargement. 24 DN is characterized by albuminuria, glomerular hypertrophy, tubular and glomerular basement membrane thickness, and extracellular matrix deposition causing tubulointerstitial and glomerular fibrosis and sclerosis.26,32 Therefore, structural changes in the kidney are one of the stages of DN. 9 Zangeneh et al. and Gorani et al. demonstrated that renal hypertrophy, proximal tubule, distal tubule, collecting duct, and vessels’ volumes increased in STZ-induced diabetic animals.19,33 Consistent with previous studies, quantitative data obtained with stereological methods which are used in the current study revealed to be a significant increase in kidney volume in diabetic rats. In addition, glomerulus volume and filtration space volume significantly increased in diabetic rats. 33 However, it was determined that there was a significant improvement in these parameters in SA- and EA-treated diabetic rats. In the present study, the increase in kidney volume of STZ-induced diabetic rats is estimated to be due to the increase in the volume of kidney subcomponents such as filtration space enlargement, extracellular matrix accumulation, basal membrane thickness, and mesangial and glomerular enlargement. Soetikno et al. 34 reported the increase in the expression of TGF-1, which causes hypertrophy of kidney subcomponents, but curcumin, a phenolic compound, decreased the expression of increased TGF-1 in STZ-induced DN. In the current study, an increase in kidney and glomerulus volume of untreated diabetic rats may be a result of the induction of TGF-1. 34 However, we estimate that the underlying mechanism of the volume increase in the filtration space may be due to a result of the increase in glomerular permeability (increase infiltration rate), which may be caused by increased TGF-1 in DN or the decrease in proximal tubular reabsorption. This may result in decreased reabsorption in the proximal tubules and albuminuria, an early marker in the progressive of DN.12,24, SA and EA treatments decreased the increase in renal and glomerular hypertrophy and expansion of filtration space in diabetic rats. This may be a result of suppressing the expression of TGF-1 by SA and EA. Alaofi reported that SA significantly reduces the increasing renal hypertrophy index in DN. 10 In this study, SA significantly reduced the increased kidney volume in diabetic rats; thus, it is compatible with the previous study. Lv et al. reported that the expressions of fibronectin and Collagen I, which are components of the extracellular matrix, were significantly increased in the STZ-treated diabetic rats’ kidney by immunohistochemical methods. 12 In this study, intense positivity of PAS and Masson’s trichrome stains were observed in the extracellular area in untreated diabetic rats. On the other hand, treatment of SA and EA significantly decreased the positivity of PAS and Masson’s trichrome stain. These findings of the current study support the previous study. 29 Tavafi et al. 27 reported that rosmarinic acid, a phenolic compound, inhibited the increased glomerulosclerosis in DN. According to the findings of this study, the treatments of SA and EA reduced the increasing glomerulosclerosis in STZ-induced DN and show compliance with the literature. 27
Serum urea and creatinine levels are important markers of kidney function, indirectly reflecting the glomerular filtration rate. 16 Zhou et al. 16 found that serum urea and creatinine levels significantly increased in STZ-induced diabetic animals. Similarly, Alaofi reported that serum urea and creatinine significantly increased in STZ-induced rats, but that SA treatment significantly improved serum urea and creatinine. In the present study, serum urea and creatinine levels considerably increased in diabetic rats; however, SA and EA treatments restored serum urea and creatinine levels; thus, it is compatible with the previous study.10,16 These findings of this study indicate that kidney function was clearly affected because of DN, which appeared in alterations of serum urea and creatinine levels. Also, SA and EA treatments improved the decreased kidney functions in DN.
Conclusions
The findings of this study confirmed the renoprotective effect of SA and EA and demonstrated the combined treatments of SA and EA have a synergistic effect against STZ-induced DN in rats. It is also demonstrated that their mechanisms of their renoprotective effect rely on suppression of apoptosis, DNA damage and oxidative stress, upregulation of antioxidant defense enzymes, and amelioration of structural and functional deterioration in diabetic rats’ kidneys. Therefore, SA and EA, especially their combined treatments, may be used as a potential therapeutic agent against DN. In addition, further studies should be conducted to elucidate the synergistic effect of combined treatment of SA and EA in DN.
Footnotes
Acknowledgments
This study was carried out with the resources provided by the Department of Histology and Embryology, Faculty of Medicine, and Vocational Higher School of Healthcare Studies, Van Yüzüncü Yıl University.
Author contribution
Fikret Altındağ: Conceptualization, Methodology, Software, Validation, Formal analysis, Investigation, Resources, Data Curation, Writing—Original Draft, Writing—Review and Editing, Visualization, Supervision, Project administration, Funding acquisition. Uğur Özdek: Validation, Formal analysis (biochemical parameters), Resources, Data Curation, Funding acquisition, Review and Editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



