Abstract
This study explored the effects of indole-3-carbinol on the proliferation of human nasopharyngeal carcinoma, both in vitro and in vivo, and the underlying mechanisms in inducing apoptosis of CNE1 cells. Proliferation, apoptosis, malondialdehyde, superoxide dismutase, glutathione peroxidase, expressions of caspase-9, and caspase-3 in human nasopharyngeal carcinoma cells CNE1 were examined. Indole-3-carbinol suppressed proliferation, induced apoptosis, decreased malondialdehyde level, increased the activity of superoxide dismutase and glutathione peroxidase, and up-regulated the expression of active fragments of caspase-9 and caspase-3 both in vitro and in vivo. It was concluded that indole-3-carbinol could inhibit proliferation and induce apoptosis of CNE1 cells and inhibit tumor growth in mice. Increased activity of superoxide dismutase and glutathione peroxidase and activated expression of caspase-9 and caspase-3 were also observed in indole-3-carbinol–treated tumors or tumor cells, suggesting that stress- and apoptosis-related molecules are involved in the indole-3-carbinol–induced apoptosis and inhibition of tumor growth.
It is well known that dietary agents have a profound influence on the initiation and progression of cancers. 1 Although many foods and dietary ingredients have a proven relationship with the reduction of cancers, epidemiological data suggest that eating vegetables is perhaps the strongest way to protect against cancers. 2,3 Several types of plant ingredients or phytochemicals have been associated with the protective characteristics of vegetables against cancers, especially those found in many kinds of cruciferous vegetables known as glucosinolates. 3,4 Glucosinolates are composed of β-D-thioglucose and moieties containing a sulfate-attached glucose with a variable side chain. In the acidic environment of the stomach, myrosinase hydrolyzes glucosinolates of the plant tissue at the β-glucosyl moiety and thus generates many different kinds of breakdown products that have been found to be the underlying mechanisms of chemopreventive agents against several experimental cancer models, such as lung cancer, 5 mammary tumors, 6 liver cancer, 7 prostate cancer, 8 and colon cancer. 9 Although many glucosinolate breakdown products exist in the circulatory system, only a few have been well established for their chemoprotective activity. Indole-3-carbinol (I3C), found in broccoli and cauliflower, is the major breakdown products of the glucosinolate glucobrassicin. 10 In vitro experiments have shown that I3C can induce the expression of metabolic enzymes that facilitate the degradation and removal of cancer-causing agents from the body. 11 I3C can also inhibit proliferation of a variety of tumor cells and induce cell cycle arrest and apoptosis through regulation of DNA repair and nuclear factors. 12-15 Animal experiments also have shown that I3C can reduce the incidence of tumors. 16 However, the effect of I3C on nasopharyngeal carcinoma tumor, which has a high incidence in the southern regions of China, has not been reported. In this study, we examined the effects of I3C in a poorly differentiated human nasopharyngeal carcinoma cell line CNE1 and in nude mice transplanted with CNE1 cells. Oxidative stress–related enzymes and products as well as the expression of key apoptotic signaling molecules (caspase-9 and caspase-3) were examined to provide fundamental information for the mechanistic studies with regard to the role of I3C in nasopharyngeal carcinoma cells.
Methods
Cell Culture
CNE1 human nasopharyngeal carcinoma cell line was purchased from Shanghai Institute of Cell Biology of the Chinese Academy of Sciences (Shanghai, China) and was cultured in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (Invitrogen, Carlsbad, CA, USA), penicillin, and streptomycin (Gibco, Carlsbad, CA, USA). CNE1 cells were incubated in a CO2 (5%) incubator at 37°C with a relative humidity of 95%. For in vitro experiments, I3C was dissolved in DMSO (Sigma-Aldrich, St. Louis, MO, USA) to make a stock solution of 50 mmol/L. For in vivo experiments, I3C was dissolved in 0.5% (vol/vol) sodium carboxymethyl cellulose solution (Shanghai Sangon, Shanghai, China) to desired concentrations.
Experimental Animal
BALB/C nude mice 4 weeks of age (n = 48, 24 males and 24 females, weight 16.2 ± 0.9 g) were provided by the Shanghai Laboratory Animal Center (permission no. SCXK [Shanghai] 2003-0003 and qualification no. 0034193). The experimental protocol followed the Guiding Principles for Research Involving Animals and Human Beings described in the Declaration of Helsinki and was approved by the Ethical Committee for Experimental Animals of Guangzhou Center for Disease Control and Prevention.
In Vitro Proliferation Inhibition Test
Cells were seeded in 96-well plates with 4 × 103 cells per well and cultured for 48 hours before switching to culture medium containing 1% fetal bovine serum for experiment. 17 In addition to blank control and vehicle (DMSO) treatment groups, 8 experimental groups were set up with final I3C concentrations at 200, 100, 50, 25, 12.5, 6.3, 3.2, and 1.6 µmol/L, respectively. Each group consisted of 6 wells, and the experiment was repeated 3 times. After 24 hours of treatment, 20 µL of tetrazolium salt (WST-1 reagent, Roche, Mannheim, Germany) was added to each well and cultured for additional 2 hours to detect cell proliferation. The plate was then shaken for 10 minutes and optical density was measured at 570 nm.
Assays for Apoptosis Detection
CNE1 cells (5 × 105/mL) were seeded in a 6-well plate with cover slips with 1 × 105 cells in each well. The cells were allowed to attach to the cover slips for 2 hours before 3 mL of culture medium was added to each well. After 46 hours of incubation, the culture medium was replaced with fresh medium containing 1% fetal bovine serum before experiment. Experimental grouping, dosage, and treatment time were the same as described above. For Hochest 33258 staining, cells were washed with phosphate-buffered saline after treatment, fixed with methanol/glacial acetic acid (3:1) at 4°C for 10 minutes, and then stained with Hochest 33258 (5 mg/mL) (Sigma-Aldrich, St. Louis, MO, USA) for 15 minutes in the dark. After mounting, the cells were observed, counted, and photographed under a fluorescence microscope (Ex 340 nm, Em 420 nm). For TUNEL analysis, cells in each group were fixed in 4% paraformaldehyde for 30 minutes, permeabilized with hydrogen peroxide, blocked with bovine serum albumin, incubated with primary antibody (SABC-AP), and colored with BCIP-NBT staining. After mounting, the cells were observed, counted, and photographed under a light microscope.
Measurement of MDA Level and Activities of SOD and glutathione peroxidase
Malondialdehyde (MDA) level (thiobarbituric acid method), superoxide dismutase (SOD) activity (xanthine oxidase method), and glutathione peroxidase (GSHPx) activity (5, 5′-dithiobis) (2-nitrobenzoic acid method) in the cell lysates were measured using commercial kits (R&D, Minneapolis, Minn) according to the manufacturer’s instructions. Experimental grouping, dosage, and treatment time were described as above.
Detection of Cleaved Caspase-9 and Cleaved Caspase-3
Cells were seeded in a 3.5-cm diameter Petri dish (4 × 105 cells per dish) and incubated in the CO2 incubator for 48 hours before the medium was replaced with fresh medium containing 1% fetal bovine serum for the experiment. Four experimental groups in which the cells were treated with 200, 80, 16.0, and 6.4 µmol/L of I3C, respectively, in addition to a blank control group and vehicle (DMSO) control group were included in this experiment. After 24 hours of treatment, cleavage of caspase-9 and caspase-3 was determined by Western blot analysis. Briefly, protein samples from each group of cells were extracted and quantified before loading to an SDS-PAGE gel (12% separation gel and 5% stacking gel) for pre-electrophoresis (constant voltage 120 V, 80 mA, 10 minutes) and electrophoresis (constant voltage 120 V, 80 mA, 45 minutes). After electrophoresis, protein samples were transferred (constant voltage 100 V, 350 mA, 1 hour) into a nitrocellulose membrane. The membrane was blocked at room temperature for 2 hours and incubated with anti-β-actin (Boster, Wuhan, China), anti-cleaved caspase-3 antibody, or anti-cleaved caspase-9 antibody (Cell Signaling Technology, Danvers, Mass) at 4°C overnight. After incubation with goat anti-rabbit antibody (Beyotime, Jiangsu, China) at room temperature for 2 hours, the membrane was developed by chemiluminescence and x-ray film exposure. Bands were scanned, and the integral optical densities (IODs, the product of area and average optical density) were determined. Relative protein expression of cleaved caspase-3 or caspase-9 was expressed as ratio of the IODs of cleaved caspase-3 or caspase-9 and internal reference β-actin.
Animal Grouping
BALB/C nude mice (n = 48) were randomly divided into low (0.02 g/kg), medium (0.1 g/kg), and high (0.5 g/kg) I3C dose groups and a control group (0.5% vol/vol sodium carboxymethyl cellulose), with each group consisting of 12 mice (6 males and 6 females).
Tumor Transplantation and Measurements
Mice were administered by gavage (volume of 0.1 mL/10 g) the appropriate dose of I3C or sodium carboxymethyl cellulose solution daily for 10 days. Each nude mouse was then transplanted subcutaneously on the back near the armpit with 2 × 106 human nasopharyngeal carcinoma cells (CNE1). After transplantation, the mice were gavaged with I3C or sodium carboxymethyl cellulose solution for another 20 days. On the 31st day, tumor tissue was separated and weighed, and the long diameter (A) and short diameter (B) were measured using a Vernier caliper. The approximate size of the tumor was calculated according to the formula V = π / 6 · [(A + B) / 2]. 18 Tumor inhibition rate (IR) was calculated as IR = (1 – mean tumor weight of experimental group / mean tumor weight of the control group) × 100%. Tumor tissue of each mouse was divided into 2 parts: 1 part for measurement of MDA level and activities of SOD and GSHPx and the other part for determination the expression of cleaved caspase-3 and cleaved caspase-9.
Statistical Analysis
Data were expressed as mean ± standard deviation, and SPSS 11.0 software was used for statistical analysis. Single-factor analysis of variance and Dunnett t test were used for comparison between experimental groups and control group, with significance level set at α = 0.05. Pearson correlation analysis was performed for tumor size and the expression level of cleaved caspase-9 as well as cleaved caspase-3.
Results
I3C Inhibited Cell Proliferation In Vitro
The inhibitory rate of proliferation of human nasopharyngeal carcinoma CNE1 cells treated with more than 12.5 µmol/L of I3C for 24 hours was significantly higher than that of the untreated cells (F = 30.864, P < .01). Regression analysis showed that I3C inhibited the proliferation of CNE1 cells in a dose-dependent manner. The regression equation was Y = 16.529 + 0.226 X (P < .01, R 2 = 0.738, SE = 0.01) in which X (independent variable) indicated the dose and Y (dependent variable) indicated the inhibitory rate (Figure 1A).
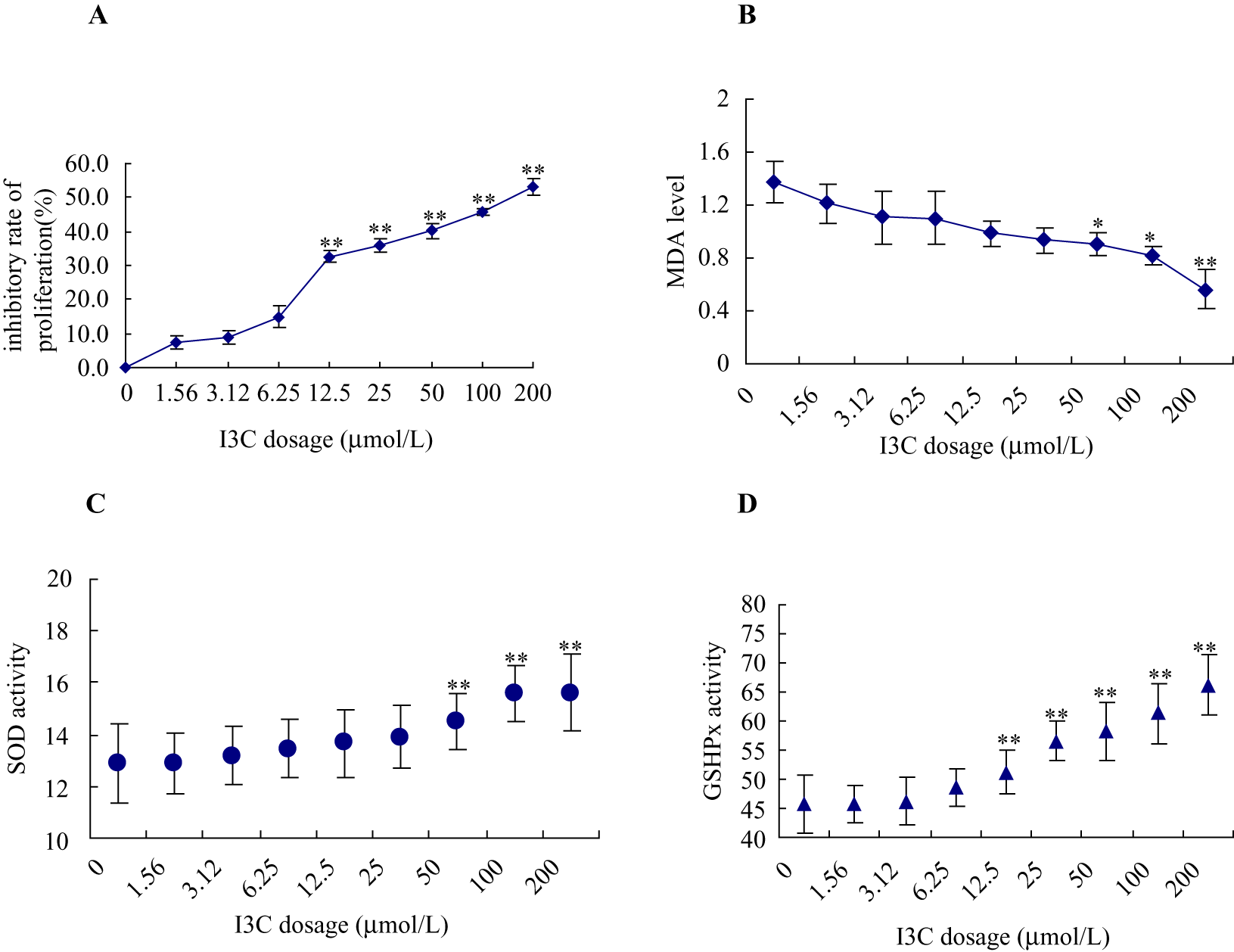
The effect of different doses of I3C on CNE1 cells. CNE1 cells were treated with 1.6, 3.2, 6.3, 12.5, 25, 50, 100, and 200 µmol/L I3C for 24 hours: (A) inhibitory rate of proliferation, (B) malondialdehyde (MDA) level, (C) superoxide dismutase (SOD) activity, and (D) glutathione peroxidase (GSHPx) activity were examined.
Effect of I3C on Cellular MDA Level and Activities of SOD and GSHPx
After I3C treatment, MDA level in cell lysates was decreased whereas the activities of SOD and GSHPx were gradually increased in every dose group (Figure 1, B-D). Statistical analysis showed that MDA levels in the cells treated with greater than 50 µmol/L I3C were significantly lower than those in the untreated cells (F = 58.074, P < .01). SOD activity in the cells treated with greater than 50 µmol/L of I3C and GSHPx activity in the cells treated with greater than 12.5 µmol/L I3C were significantly higher than those in untreated cells (F = 63.258, P < .01; F = 131.193, P < .01, respectively).
I3C Induced Apoptosis of CNE1 Cells In vitro
After treatment of I3C for 24 hours, CNE1 cells in every dose group from 1.6 to 200 µmol/L showed signs of apoptosis, demonstrated by the appearance of condensed blue fluorescent particles and significant nuclear morphological changes (Figures 2A and 2B ). TUNEL analysis showed that after treatment with I3C, CNE1 cells in every dose group showed signs of apoptosis, demonstrated by the staining of nucleus with different levels of yellow-brown color (Figures 2C and 2D).
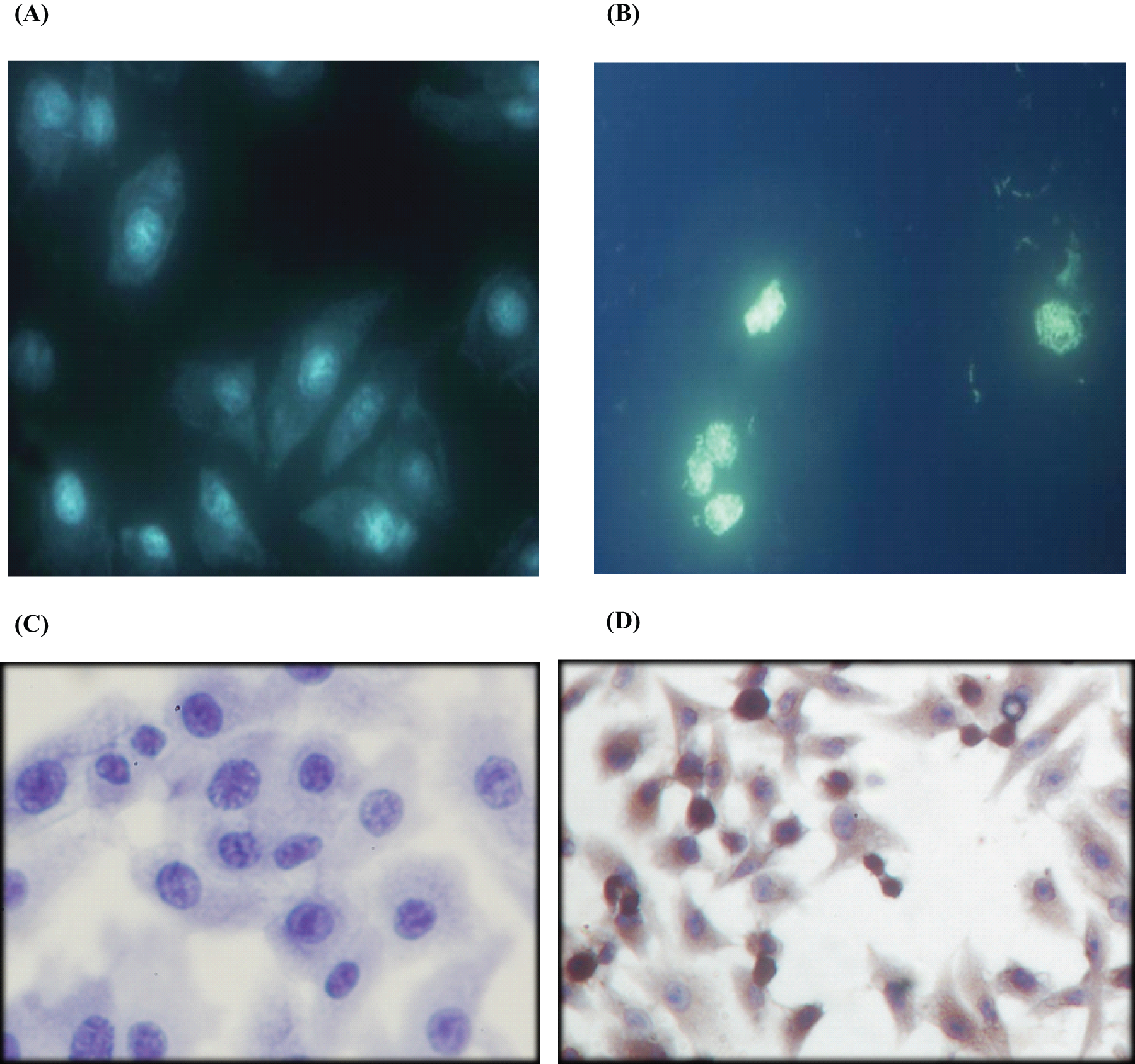
CNE1 cells displayed signs of apoptosis after I3C treatment (×400). (A) Hochest 33258 staining of cells in the control group; (B) Hochest 33258 staining of cells after I3C treatment; (C) TUNEL staining of cells as negative control; (D) TUNEL staining of cells after I3C treatment.
Expression of Cleaved Caspase-9 and Cleaved Caspase-3 In vitro
After I3C treatment, expression of cleaved caspase-9 and cleaved caspase-3 (17 KD and 19 KD, respectively) was significantly increased compared with the negative control and vehicle treatment groups. The highest expression was achieved in the cells treated with 80 µmol/L I3C. Further increases of I3C slightly decreased the expression of cleaved caspase-9 and cleaved caspase-3 (Figure 3). Image analysis results reveal that the IODs of cleaved caspase-9 of DMSO group and 0- to 200-µmol/L I3C groups were 0.03 ± 0.01, 0.09 ± 0.02, 0.12 ± 0.04, 0.24 ± 0.03, 0.51 ± 0.08, and 0.43 ± 0.06. The IODs of 19 KD cleaved caspase-3 of different groups were 0.11 ± 0.02, 0.18 ± 0.01, 0.19 ± 0.03, 0.38 ± 0.05, 0.39 ± 0.02, and 0.10 ± 0.04. The IODs of 17 KD cleaved caspase-3 of different groups were 0.11 ± 0.01, 0.19 ± 0.06, 0.23 ± 0.02, 0.42 ± 0.10, 0.58 ± 0.04, and 0.51 ± 0.09.
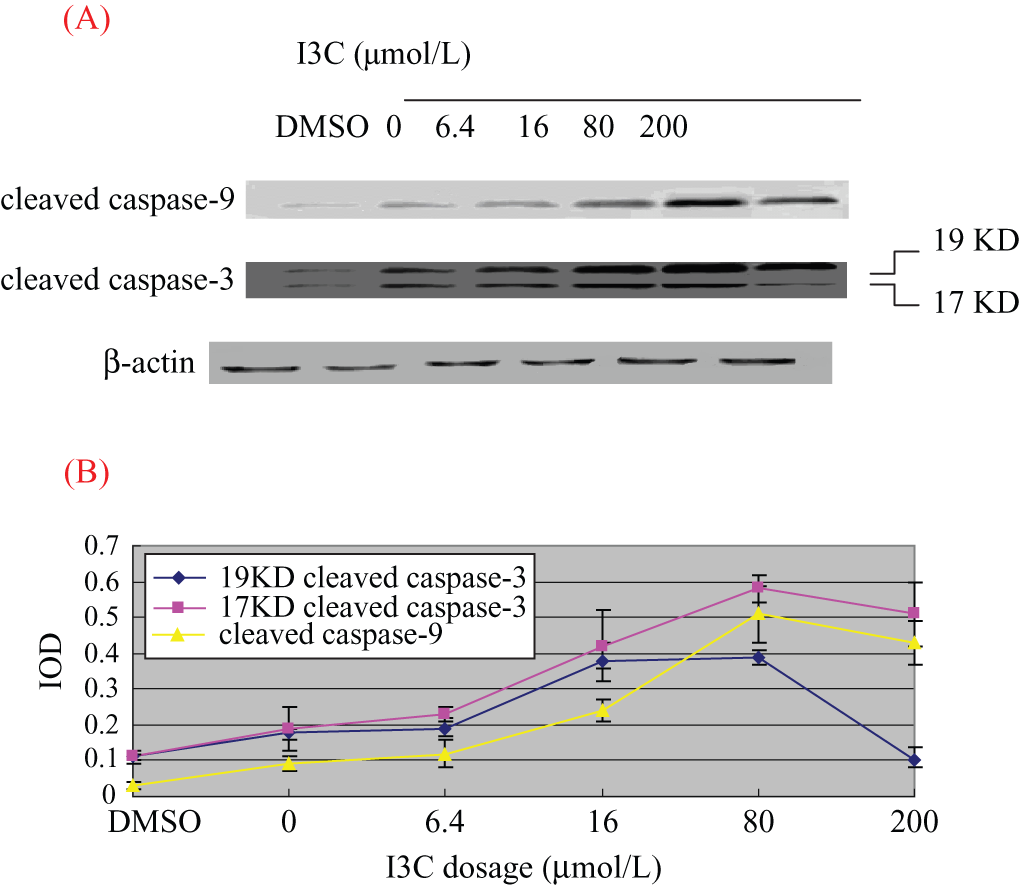
(A) Expressions of cleaved caspase-9 and cleaved caspase-3 in CNE1 cells after 24 hours of treatment with I3C and (B) image analysis results. (A) The expression of cleaved caspase-9 and cleaved caspase-3 tended to be elevated with the increase of I3C dosage from 0 to 80 µmol/L, whereas the protein expression slightly decreased at the dose of 200 µmol/L. (B) Image analysis results of the expression of cleaved caspase-9 and cleaved caspase-3 according to the increase of I3C dosage. The x-axis shows the I3C dosage, and the y-axis shows integral optical densities.
I3C Inhibited Growth of Tumors in Mice
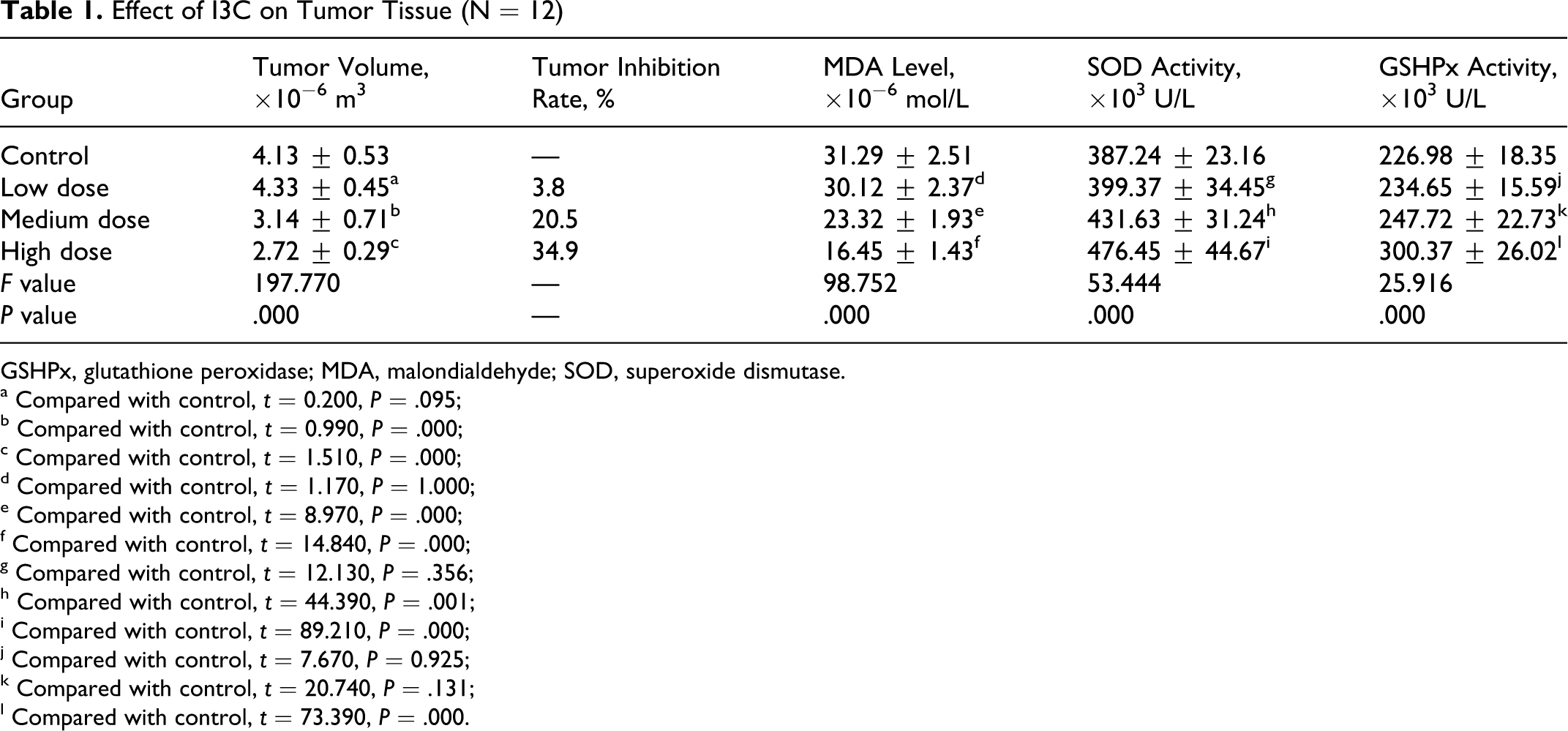
Different sizes of tumors were achieved in the CNE1 cell–transplanted nude mice that were treated with different doses of I3C. Statistically, the tumor sizes in the mice treated with medium or high doses of I3C were significantly smaller than those in the control mice (P < .01). The tumor growth inhibition rate was more than 20% in the mice treated with medium or high doses of I3C based on the calculation formula reported previously (Table 1 and Figure 4).
Effect of I3C on Tumor Tissue (N = 12)
GSHPx, glutathione peroxidase; MDA, malondialdehyde; SOD, superoxide dismutase.
a Compared with control, t = 0.200, P = .095;
b Compared with control, t = 0.990, P = .000;
c Compared with control, t = 1.510, P = .000;
d Compared with control, t = 1.170, P = 1.000;
e Compared with control, t = 8.970, P = .000;
f Compared with control, t = 14.840, P = .000;
g Compared with control, t = 12.130, P = .356;
h Compared with control, t = 44.390, P = .001;
i Compared with control, t = 89.210, P = .000;
j Compared with control, t = 7.670, P = 0.925;
k Compared with control, t = 20.740, P = .131;
l Compared with control, t = 73.390, P = .000.
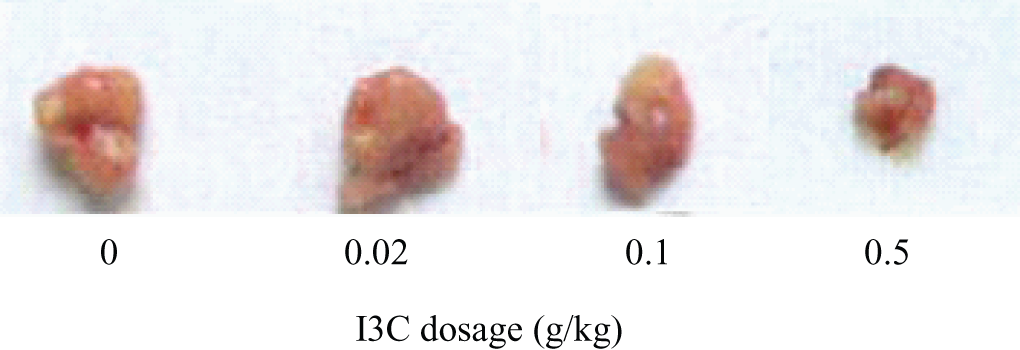
Tumor sizes in mice receiving different doses of I3C.
Effect of I3C on MDA Level, SOD and GSHPx Activities in Tumor Tissues
MDA levels in the tumor tissues of nude mice treated with medium or high doses of I3C were significantly lower than those in the control mice (t = 0.007 and 0.014, respectively, P < .01). SOD activity in the medium- and high-dose groups was significantly higher than that in the control group (t = 0.044 and 0.089, respectively, P < .01). GSHPx activity in the high-dose group was significantly higher than that in the control group (t = 0.074, P < .01) (Table 1).
I3C Increased the Expression of Cleaved Caspase-9 and Cleaved Caspase-3 in Tumor Tissues
With increasing dosage of I3C, expression of the cleaved caspase-9 (F = 89.632, P < .01) and cleaved caspase-3 (17 KD and 19 KD) was increased significantly (F = 39.864 and 56.629, respectively, P < .01) (Figure 5). Image analysis results reveal that the IODs of cleaved caspase-9 of 0- to 0.5-g/kg I3C groups were 0.18 ± 0.04, 1.96 ± 0.27, 1.81 ± 0.22, and 2.35 ± 0.03. The IODs of 19 KD cleaved caspase-3 of different groups were 0.89 ± 0.17, 0.96 ± 0.22, 1.26 ± 0.31, and 1.31 ± 0.39. The IODs of 17 KD cleaved caspase-3 of different groups were 0.01 ± 0.01, 0.17 ± 0.03, 0.21 ± 0.06, and 0.23 ± 0.09.
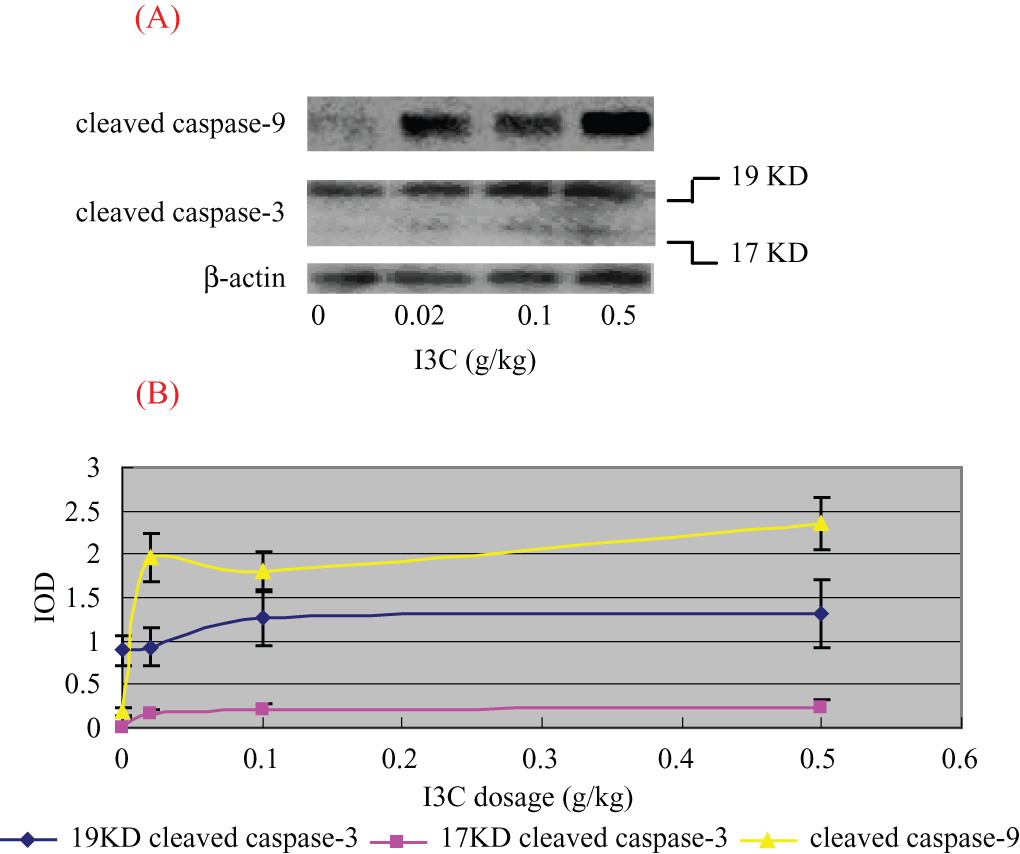
(A) Effect of I3C on the expressions of cleaved caspase-9 and cleaved caspase-3 in tumor tissues and (B) image analysis results. (A) The expression of cleaved caspase-9 and cleaved caspase-3 tended to be elevated with the increase of I3C dosage. (B) Image analysis results of the expression of cleaved caspase-9 and cleaved caspase-3 according to the increase of I3C dosage. The x-axis shows the I3C dosage, and the y-axis shows integral optical densities.
Correlation Analysis
After I3C treatment, reduction in tumor sizes was significantly correlated with the increase of cleaved caspase-9 expression (Pearson correlation coefficient r = –0.973, t = 10.157, P < .01). Reduction in tumor sizes was also significantly correlated with the increases of 19 KD (r = –0.732, t = 3.404, P < .01) and 17 KD (r = –0.901, t = 6.642, P < .01) cleaved caspase-3 expression.
Discussion
Nasopharyngeal carcinoma is endemic in China (eg, Canton, Guangxi, Taiwan) as well as in North Africa, 19,20 and it differs from other epithelial cancers of the head and neck in its sensitivity to radiotherapy and chemotherapy. The combination of cisplatin or 5-FU with radiotherapy has been widely used to treat nasopharyngeal cancer. 21,22 However, this regimen works poorly. 23,24 Furthermore, gastrointestinal toxicity and myelosuppression are severe side effects that often result in suspension of treatment. Consequently, new therapies with high efficacy and low toxicity for the treatment to nasopharyngeal cancer are urgently needed. Because I3C, one of the active ingredients in cruciferous vegetables, could inhibit a variety of malignant tumors (eg, rectal cancer, breast cancer, and lung cancer) through multiple mechanisms, with few or no side effects, 18 we postulated that I3C could also be used to treat nasopharyngeal cancer. In this study, we designed a series of experiments to investigate the effects of I3C, both in vivo and in vitro, on the induction of apoptosis and inhibition of cell proliferation or tumor growth. We also explored potential mechanisms by measuring the level of oxidative stress–related enzymes and expression of apoptosis-related molecules. The in vitro study shows that a certain dose of I3C inhibited the proliferation of human nasopharyngeal carcinoma cells and induced apoptosis as demonstrated by specific nuclear staining. In the apoptotic process, changes in the balance of oxidation and antioxidation suggested that oxidative stress may be one of the mechanisms involved in I3C-induced inhibition of CNE1 cell proliferation and induction of apoptosis. In vivo results showed that I3C treatment reduced the sizes of tumors. High tumor inhibition rates in the treatment groups suggested that maintaining certain levels of I3C in the body inhibited the growth of tumor cells.
Many studies have demonstrated that lipid peroxidation can affect biological macromolecules such as nucleic acid, protein, lipid, and carbohydrates by direct injury or damage to the composition and structure of cell membranes or can indirectly interfere with the redox state of cells, resulting in abnormal cell structure and function, thereby triggering tumor and tumor progression. 25 MDA is an end-product of lipid peroxidation and one of the indicators of imbalance of oxidation and antioxidation in the body. Antioxidant enzymes in the body, such as SOD and GSHPx, play an important role in the disruption of lipid peroxidation and removal and transformation of peroxide. 26 This study demonstrated that I3C significantly increased the activities of SOD and GSHPx and decreased MDA levels in the tumor tissues, suggesting that I3C improves the imbalance between oxidation and antioxidation, therefore inhibiting oxidative stress in tumor tissues.
The cysteinyl aspartate–specific protease (referred to as caspase) family, the most critical signaling molecules in the induction and execution of apoptosis, 27,28 has gained attention in molecular biological studies of apoptosis. In the caspase family, caspase-9 is the most upstream member of the caspase cascade. 29,30 Once the cell receives apoptosis stimulation, the mitochondria release cytochrome C and procaspase-9 simultaneously, which are then cleaved to generate the active caspase-9 in the presence of cytochrome C. Cleaved caspase-9 further cleaves other members of the caspases, including caspase-3, the most important executor of apoptosis. The formation of active caspase-3 initiates the caspase cascade, leading to programmed cell death. 30,31 In this study, we showed that expression of cleaved caspase-9 and caspase-3 was increased with increasing doses of I3C both in vivo and in vitro, suggesting that I3C can induce caspase-9 and caspase-3 activation. Furthermore, the expression level of cleaved caspase-9 and cleaved caspase-3 was negatively correlated with the tumor sizes. Further studies are needed to determine the relationship between caspase expressions and I3C-induced tumor inhibition, which could shed light on the role of I3C in chemoprevention of nasopharyngeal cancer.
