Abstract
Background:
In aluminum phosphide (AlP) poisoning, death is mainly due to cardiovascular failure and refractory acute heart failure. There is a lot of evidence showing thyroid hormones have cardioprotective effects.
Objective:
The purpose of this study was to evaluate the effect of oral liothyronine in the treatment of AlP poisoning.
Methods:
Twenty-four patients from intensive care unit of Baharloo Hospital, Tehran, Iran, were included based on the inclusion and exclusion criteria. They were randomly divided into two parallel groups of 12 cases and 12 controls. Intervention in the case group was administration of 50 µg liothyronine via nasogastric tube after gastric lavage, in the first 6 h of poisoning. In both groups, the routine treatment of AlP poisoning was performed. Blood samples were prepared at the beginning of the study and after 12 h. Patients were followed up till discharge from the hospital or death.
Results:
The findings demonstrated that oral liothyronine was able to significantly improve systolic blood pressure, arterial blood pH, and total thiol molecules and also could decrease lipid peroxidation, increase catalase activity, and prevent further decline in total antioxidant capacity.
Conclusion:
Liothyronine administration is effective in controlling AlP poisoning and can improve patients’ outcome.
Introduction
Aluminum phosphide (AlP) is an extremely toxic compound, with chemical formula AlP, used as a pesticide for storing cereal grains. The toxicity of AlP is due to releasing phosphine (PH3). AlP reacts with water or acids to release PH3. PH3 is a colorless, flammable, toxic gas. 1 In some countries, AlP tablets have been used for suicide in recent years. 2 Cardiomyocytes are one of the main targets for PH3. 3 Death is mainly due to refractory acute heart failure and profound shock. 2 The frequency of AlP poisoning varies from 0.3% to 11.7% of all poisoned patients admitting per year in different countries and regions. 2,4 The incidence seems higher in some areas of India. 4 Although the incidence of AlP poisoning is low worldwide, the mortality rate is high (40–80%). 5 The toxicity of AlP is due to cytotoxic effects of PH3 which may lead to increase free radicals, inhibition of cellular enzymes, and disturbance in the electron transport chain. PH3 is able to enter the cell where the mitochondria is one of the main targets for PH3. 6 The exact mechanism for AlP poisoning is not clear, although cardiomyocyte toxicity happens due to PH3’s two main mechanisms: (1) inhibition of mitochondrial complexes followed by disturbance in electron transport chain and (2) increased free radicals specially reactive oxygen species (ROS) and oxidative stress which may result in lipid peroxidation. 2,7 In other side, PH3 can also inhibit some antioxidant enzymes such as catalase and peroxidase. 8,9 Due to the increased incidence of AlP poisoning in the recent years, numerous studies have been done in this regard specially to find an efficient therapeutic strategy. N-acetylcysteine, 10,11 coconut oil, 12 hydroxyethylstarch, 13 glutathione, vitamin C, vitamin E, beta carotene, melatonin, 14 vasopressin, milrinone, 15 iron sucrose, 16 and acetyl-L-carnitine 17 were used and reported previously.
One of the compounds which seem to be effective in controlling AlP poisoning is triiodothyronine (T3). The effectiveness of T3 in AlP poisoning was evaluated for the first time in an animal study. 18 The use of thyroid hormones in the control and treatment of AlP poisoning is mainly because of the cytoprotective effects of these compounds. There is even the possibility that in the AlP poisoning, a tissue hypothyroidism is happening. Thyroid hormones have two types of effects on mitochondria. One is rapid that occurs within minutes/hours after hormone treatment. This type of thyroid hormone effects is induced by nonnuclear pathways. The other one occurs in one to several days after hormone treatment and is mediated by nuclear pathways. 19 Thyroid hormones’ rapid cytoprotective effects occur by nonnuclear pathways. Antioxidant effects of thyroid hormones play the main role in the cytoprotective effects of these hormones. 20 These antioxidant effects may be due to (1) increased activity and expression of uncoupling proteins in the mitochondria and mild uncoupling by thyroid hormones that can lead to a reduction in ROS amounts without decreasing adenosine triphosphate (ATP) synthesis, 21,22 (2) increased activity of mitoKATP, 23 –26 and (3) increased activity and expression of antioxidant hormones. 27,28 Thyroid hormones have also some antiapoptotic roles in the cell. 29 –35
With regard to the above points, it is hypothesized that thyroid hormones can be effective in controlling AlP poisoning. In this study, we are trying to answer the following questions. May liothyronine administration reduce the complications of AlP poisoning and improve this condition? What is the effect of liothyronine administration in the early hours of the poisoning on mortality, clinical parameters, laboratory findings, and oxidative stress parameters?
Methods
Ethics
This study was approved by the Ethics Committee of Tehran University of Medical Sciences with reference number 9111296004-143516 and has been registered in the Iranian Registry of Clinical Trials with code number IRCT2015041521785N1. The informed consent was taken from the patients or from their relatives after the intervention.
Chemicals
The compounds used in this study are as follows: 25 µg of liothyronine tablets were obtained from Iran Hormone Co. (Tehran, Iran). Catalase and superoxide dismutase (SOD) kits were purchased from Cayman Chemical Co. (Ann Arbor, Michigan, USA). Free T3 (FT3) and thyroid-stimulating hormone (TSH) kits were obtained from Pishtaz Teb Co. (Tehran, Iran). Malondialdehyde (MDA), sulfuric acid, n-butanol, thiobarbituric acid (TBA), bovine serum albumin, magnesium chloride, sodium carbonate, ferrous sulfate, Tris-hydrochloric acid buffer, trichloroaceticacid,2,4,6-tripyridyl-s-triazine (TPTZ), dithionitrobenzoic acid (DTNB), and thiazolyl blue tetrazolium bromide (98%) were purchased from Sigma Chemical Co. (St Louis, Missouri, USA).
Study subject and treatment design
This study is a randomized, not blinded, case-controlled clinical trial on acute AlP-poisoned patients who admitted during the first 6 h after onset of poisoning to the intensive care unit (ICU) of the Baharloo Hospital, Tehran, Iran, from April to September 2015. Acute AlP-poisoned patients were included in this study with the inclusion measures of acidosis, systolic blood pressure (SBP) less than 100 mmHg, refractory cardiogenic shock, and persistent coma. The exclusion criteria consisted hypersensitivity to liothyronine, acute myocardial infarction, thyroid diseases, adrenal insufficiency, and medical treatment for AlP poisoning in any other medical centers before admission to Baharloo center. Diagnosis of AlP poisoning was through history of exposure and clinical findings. In suspected cases, silver nitrate test for PH3 detection was done on stomach contents.
In both groups, the standard and routine therapy was performed immediately after admission as follows: gastric decontamination with potassium permanganate (1:10,000, orally) and sodium bicarbonate (HCO3; 44 mEq, orally); inhalation of 100% oxygen; normal saline solution intravenous (IV), 19–20 mL/kg; hydrocortisone IV, 100 mg qid; pantoprazole IV, 40 mg bid; magnesium sulfate IV infusion 1 g bid; metoclopramide IV 10 mg qid; and calcium gluconate 10%, IV qid. In the case group, drug intervention was performed with 50 µg liothyronine (single dose) administered by nasogastric tube immediately after gastric lavage. All of the patients were followed up till discharge from hospital or death.
Blood sampling
Venous blood sampling
Venous blood samples were prepared from both groups for assessment of the study variables on admission (baseline) and 12 h after admission. Venous blood samples (5–7 mL) were collected in simple blood collection tubes (containing no anticoagulant) and then the tubes were incubated in an upright position at room temperature for 30–45 min to allow clotting. After clotting, the tubes were centrifuged for 15 min at 1000 relative centrifugal force. The supernatants (serum) were carefully aspirated and collected into especial tubes. These tubes were adequately labeled with the relevant information and stored at −80°C.
Arterial blood sampling
One milliliter of arterial blood was collected with a needle and preheparinized syringe from the radial artery on admission (baseline) and 12 h after admission in both groups.
Measurement of variables
In this study, the mortality rate was primary dependent variable (primary outcome) and SBP, FT3, TSH, arterial blood pH, lipid peroxidation, total antioxidant capacity (TAC), total thiol molecules (TTM), SOD, and catalase were the secondary dependent variables (secondary outcomes). All of the secondary outcomes were measured two times on admission and 12 h after admission.
Mortality rate
The mortality rate has been just the primary dependent variable (primary outcome) in this study. It was evaluated at the end of the study.
Systolic blood pressure
SBP was measured with a mercury sphygmomanometer in supine position. 36 The unit of measurement of blood pressure in this study is millimeter of mercury.
Arterial blood pH
Arterial blood pH was measured with a blood gas analyzer device. Normal arterial pH range is 7.38–7.42 (7.40).
Free triiodothyronine
FT3 levels were measured in patients’ serum with chemiluminescence immunoassay (CLIA) under the routine hospital laboratory conditions. The reference range of FT3 in adults is 1.4–4.2 pg/mL.
Thyroid-stimulating hormone
TSH levels were measured in patients’ serum with CLIA under the routine hospital laboratory conditions. The reference range of TSH in adults is 0.2–5 mIU/L.
Lipid peroxidation
Lipid peroxidation was assayed with TBA reactive substances (TBARS) test as described previously. 37 MDA is an end product of lipid peroxidation that reacts with TBA to produce MDA-TBA complex, which can be determined with a spectrophotometer at 532 nm. The results were calculated as micromole MDA per liter (µmol/L).
Total antioxidant capacity
TAC of serum was measured by ferric reducing ability of plasma (FRAP) assay according to the method described previously. 38 The ability of plasma in reducing of ferric tripyridyltriazine (Fe3+-TPTZ) complex to ferrous tripyridyltriazine (Fe2+-TPTZ) complex is the base of this test. Fe2+-TPTZ complex has a blue color which can be read at 593 nm. The results were calculated as millimole per liter. 38
Total thiol molecules
Thiols are endogenous molecules, which contain the sulfhydryl group (–SH) attached to a carbon atom. TTMs were determined in serum with the method of Hu (1994) by using DTNB and measuring the absorbance at 412 nm. 39 TTM concentrations were expressed as micromole per liter.
Superoxide dismutase
Serum SOD activity was evaluated by Cayman’s SOD assay kit (Ann Arbor, Michigan, USA) according to the manufacturer’s protocol. The absorbance of prepared samples was read at 440–460 nm and the activities were reported as unit per milliliter.
Catalase
Catalase activity was evaluated by Cayman’s catalase assay kit. In this case, after preparing samples according to the protocol, the absorbance was read at 540 nm. Catalase activities were expressed as unit per milliliter (U/mL).
Evaluation of illness severity
In this study, severity of illness in patients admitted to ICU was evaluated within the 24 h after admission by new Simplified Acute Physiology Score II (SAPS II) and the mean of the predicted mortality rate was compared in both the case and control groups. SAPS II is a severity of disease classification system. In SAPS II, the illness severity for patients admitted to ICU is measured 24 h after admission according to 15 clinical and paraclinical parameters. The parameters consist of age, type of admission, chronic diseases, heart rate, temperature, SBP, Glasgow Coma Scale (GCS), White Blood Cell (WBC), serum urea or Blood Urea Nitrogen (BUN), sodium, potassium, HCO3, bilirubin, urine output, Partial pressure of O2 in arterial blood (PaO2)/Fraction of inspired oxygen (FIO2). 40
Statistical analysis
The data were analyzed with StatsDirect Ver. 3.0.131. Differences in SBP, FT3, TSH, arterial blood pH, lipid peroxidation, TAC, TTM, SOD, and catalase before and after liothyronine treatment were analyzed and compared with t-tests. The results were expressed as mean ± standard error of the mean (SEM). All data were found to be normally distributed. p Values of 0.05 or less were considered to be statistically significant.
Results
In this study, there were 33 cases of AlP poisoning referred to the hospital from April to September 2015. One of them died before admission, three expired before 12 h, in two cases, the AlP tablets had been expired, one case had hypothyroidism and was already taking levothyroxine, and two cases were completely asymptomatic. Because of the above, 9 cases were excluded and finally 24 patients were included and randomly divided into two parallel groups by randomization sheet, case (intervention) group (n = 12; 11 men and 1 woman) and control group (n = 12; 8 men and 4 women). All of the patients had attempted suicide. There was only one suspected case of AlP poisoning in which the diagnosis of AlP poisoning was confirmed by silver nitrate test. The results are sorted in three categories that include (1) the results of mortality rate and clinical variables evaluation consisting of SBP, FT3, TSH, and arterial blood pH; (2) the results of oxidative stress markers consisting of lipid peroxidation, TAC, TTM, SOD, and catalase; and (3) apart from the evaluation of the study variables, some additional findings were also obtained which were related to age, sex, ingested dose of AlP, time interval between AlP ingestion and admission, time interval between AlP ingestion and liothyronine treatment, severity of illness/predicted mortality, duration of hospitalization in ICU, time interval between AlP ingestion and death, amount of IV therapy, and dose of vasopressor drug (norepinephrine).
Clinical findings detected in both the case and control groups were as follows: nausea, vomiting, epigastric pain, hematemesis, headache, dizziness, fatigue, agitation, chest discomfort, hypotension, tachycardia, arrhythmias, tachypnea, dyspnea, rales, rhonchi, and hematochezia.
Mortality rate and clinical/paraclinical findings
Mortality rate
The number of patients died in the control group was four, while three patients in the case group died. In other words, mortality rate is 33.3% in the control group and 25% in the case group.
Systolic blood pressure
The results of SBP in both the case and control groups at the admission (baseline) and after 12 h are as follows: case group/baseline 92 ± 1.5, control group/baseline 93.5 ± 1.9, case group after 12 h 103.08 ± 4, and control group after 12 h 85.91 ± 6.7. Data are shown as mean ± SEM and based on millimeter of mercury. There is a statistically significant difference between SBP in the case and control groups after 12 h (p < 0.05). SBP in the case group after 12 h was more than SBP in the case group at the admission (p < 0.05). The results are shown in Figure 1(a).

Changes in (a) SBP, (b) arterial blood pH, (c) FT3, and (d) TSH in various groups. Data are shown as mean ± SEM.
Arterial blood pH
The results of arterial blood pH in both the case and control groups at the admission (baseline) and after 12 h are case group/baseline 7.33 ± 0.016, control group/baseline 7.32 ± 0.051, case group after 12 h 7.40 ± 0.018, and control group after 12 h 7.33 ± 0.21. Data are shown as mean ± SEM. Arterial blood pH in the case group after 12 h is significantly different from pH in the control group after 12 h (p < 0.05). There is a statistically significant difference between baseline and post 12 h in the case group (p < 0.05). The results are shown in Figure 1(b).
Free triiodothyronine
The results of FT3 in both the case and control groups at the admission (baseline) and after 12 h include case group/baseline 2.73 ± 0.175, control group/baseline 3.59 ± 0.157, case group after 12 h 3.4 ± 0.213, and control group after 12 h 2.64 ± 0.105. Data are shown as mean ± SEM and based on picogram per milliliter. There is a statistically significant difference between FT3 in the case group after 12 h and the control group after 12 h (p < 0.05). FT3 in the case group after 12 h is also significantly more than FT3 in the case group at the admission (p < 0.05). The results are shown in Figure 1(c).
Thyroid-stimulating hormone
The results of TSH in both the case and control groups at the admission (baseline) and after 12 h are as follows: case group/baseline 1.92 ± 0.334, control group/baseline 1.18 ± 0.207, case group after 12 h 0.55 ± 0.076, and control group after 12 h 1.38 ± 0.132. Data are shown as mean ± SEM and based on milli-international units per liter. TSH in the case group after 12 h is significantly different from TSH in the control group after 12 h (p < 0.05). There is a statistically significant difference between TSH in the case group after 12 h and baseline TSH in the case group (p < 0.05). The results are shown in Figure 1(d).
Oxidative stress markers
Lipid peroxidation
MDA as an end product of lipid peroxidation was evaluated by TBARS and the results in both the case and control groups at the admission (baseline) and after 12 h are as follows: case group/baseline 1.509 ± 0.145, control group/baseline 1.469 ± 0.168, case group after 12 h 1.556 ± 0.171, and control group after 12 h 1.865 ± 0.196. Data are shown as mean ± SEM and based on micromole per liter. There is no statistically significant difference between MDA in the case and control groups after 12 h. The results are shown in Figure 2(a).
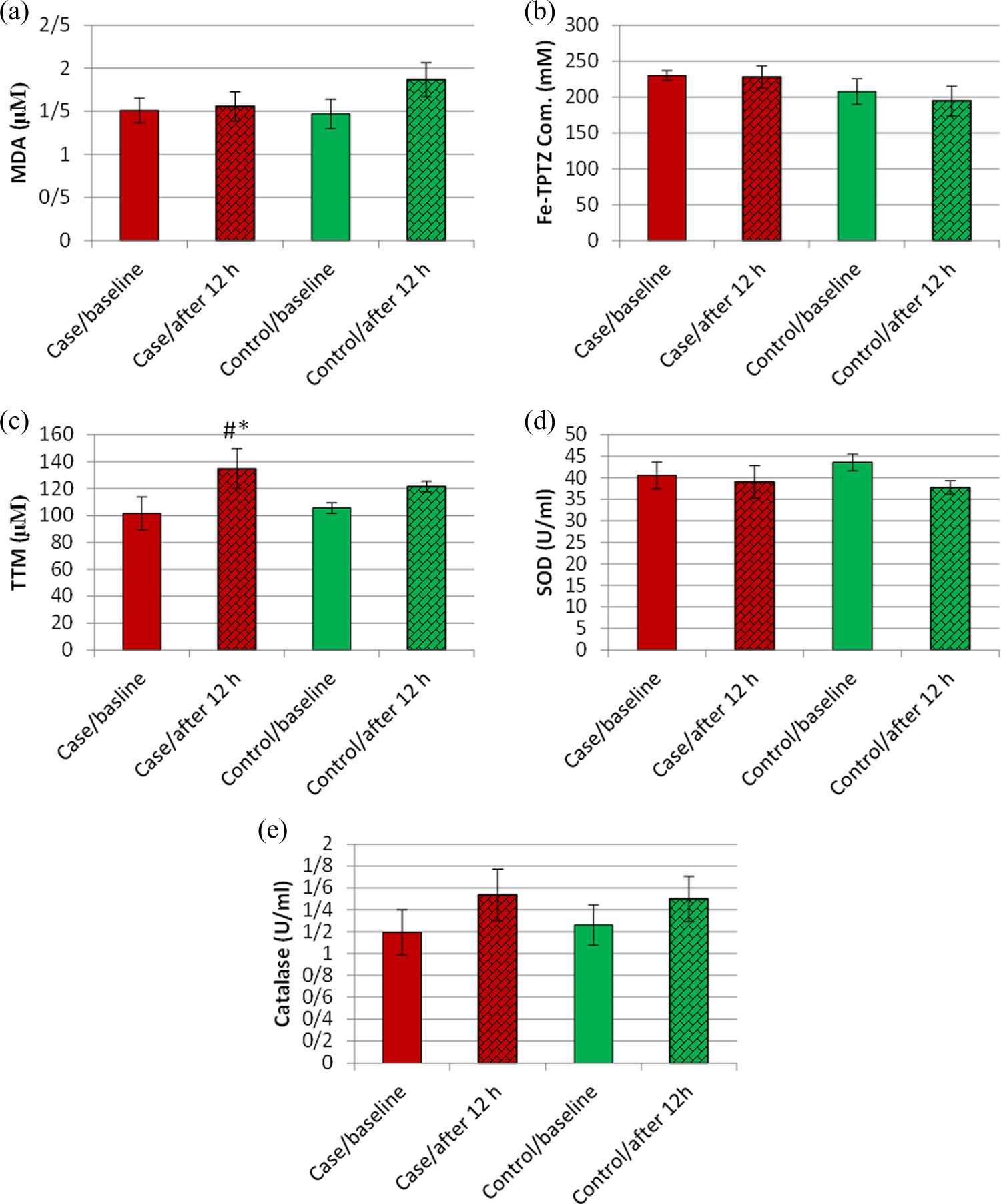
Changes in (a) MDA (lipid peroxidation), (b) Fe-TPTZ com. (TAC), (c) TTM, (d) SOD activity, and (e) catalase activity in various groups. Data are shown as mean ± SEM.
Total antioxidant capacity
TAC was evaluated by FRAP assay by measuring Fe2+-TPTZ complex, the results in both the case and control groups at the admission (baseline) and after 12 h are as follows: case group/baseline 229.75 ± 6.757, control group/baseline 207.39 ± 17.705, case group after 12 h 228.64 ± 15.29, and control group after 12 h 194.33 ± 20.841. Data are shown as mean ± SEM and based on millimolar. There is no statistically significant difference between Fe2+-TPTZ complex in the case and control groups after 12 h. The results are shown in Figure 2(b).
Total thiol molecules
The results of TTM measurement in both the case and control groups at the admission (baseline) and after 12 h are as follows: case group/baseline 101.54 ± 12.295, control group/baseline 105.5 ± 14.781, case group after 12 h 134.75 ± 3.891, and control group after 12 h 121.45 ± 3.939. Data are shown as mean ± SEM and based on micromole per liter. There is a statistically significant difference between TTM in the case and control group after 12 h (p < 0.05). TTM in the case group after 12 h is also significantly more than TTM in the case group at the admission (p < 0.05). The results are shown in Figure 2(c).
Superoxide dismutase
The results of SOD activity in both the case and control groups at the admission (baseline) and after 12 h are as follows: case group/baseline 40.54 ± 3.138, control group/baseline 43.58 ± 3.799, case group after 12 h 39.04 ± 3.923, and control group after 12 h 37.443 ± 1.601. Data are shown as mean ± SEM and based on unit per milliliter. There is no statistically significant difference between SOD activity in the case and control groups after 12 h. The results are shown in Figure 2(d).
Catalase
The results of catalase activity in both the case and control groups at the admission (baseline) and after 12 h are as follows: case group/baseline 1.195 ± 0.206, control group/baseline 1.26 ± 0.235, case group after 12 h 1.543 ± 0.184, and control group after 12 h 1.499 ± 0.207. The data are shown as mean ± SEM and based on unit per milliliter. There is no statistically significant difference between catalase activity in the case and control groups after 12 h. The results are shown in Figure 2(e).
Demographics of patients and additional findings
Table 1 summarized the results of demographics of patients and additional findings.
Demographics of patients and additional findings.

AlP: aluminum phosphide; SAPS II: Simplified Acute Physiology Score II; ICU: intensive care unit; IV: intravenous; SEM: standard error of the mean.
Discussion
The main mechanism for AlP poisoning is not exactly known, and for that reason, a special antidote for AlP poison is not available yet. 1,2 In numerous studies, it was demonstrated that disturbance in the electron transfer chain and oxidative stress has the main role in AlP poisoning pathogenesis. 2
Mortality rate is the primary outcome of this study. As mentioned, mortality rate is 33.3% in the control group and 25% in the case group. Although mortality rate was improved with liothyronine administration, the difference is not statistically significant. However, when compared to mortality rates predicted with SAPS II, this seems an acceptable result. In the control group, predicted mortality rate was 50.62 ± 4.66 and in the case group, it was 49.77 ± 4.172. It shows there was a successful management in both the groups, but it is more evident in the case group with liothyronine administration.
Refractory cardiogenic shock is the most cause of death in patients poisoned with AlP. 2,41 The exact mechanism for refractory cardiogenic shock is not known. It may be occurring due to arrhythmia, electric conduction disturbance, and myocardial damage. Cardiogenic shock can present with profound hypotension which is not responsive to conventional treatment. 42 Our clinical findings show that SBP has been improved 12 h after liothyronine administration in the case group and there are statistically significant differences in the mean SBP between the case group after 12 h and baseline as well as the control group after 12 h. In other words, liothyronine administration could be able to maintain SBP in the higher range and it was effective to control cardiogenic shock. Previously, effectiveness of T3 administration in improving SBP was shown in an animal study by Abdolghafari et al. 18 In a case report, Mehrpour et al. showed digoxin is useful in AlP-poisoned cardiogenic shock, but it needs to be confirmed in further studies. 43 Glucagon is an inotrope agent, which was suggested for the treatment of AlP poisoning. The inotropic effect of glucagon is due to increased intracellular cAMP and calcium. 44 There is a case report showing glucagon is effective in this regard. 45 Hydroxyethyl starch is a colloid volume expander that can increase intravascular volume and blood pressure. Intravascular administration of hydroxyethyl starch was suggested for the treatment of severe hypotension in AlP poisoning. 13 In other words, both digoxin and glucagon have inotropic effect and can increase blood pressure; although ATP demand is increased and may finally lead to ATP depletion in AlP poisoning. On the other hand, PH3 reduces cardiac energy reserves because of oxidative phosphorylation disturbance and ATP depletion, 6,46,47 whereas thyroid hormone administration enhances ATP generation. 18,48 There is a similar problem regarding hydroxyethyl starch, which may increase preload and ATP demand.
Metabolic acidosis is one of the serious complications of AlP poisoning. It is due to circulatory failure and tissue hypoperfusion. 6 Patients with AlP poisoning may develop metabolic acidosis which may be refractory and not responding to the standard therapy. 2,5,48 The standard treatment for metabolic acidosis is IV HCO3, whereas in severe and refractory metabolic acidosis, patients may need hemodialysis. 20 As shown, arterial blood pH was maintained in the normal range in the case group 12 h after liothyronine administration. The mean arterial blood pH in the case group 12 h after liothyronine administration has a statistically significant difference with the mean of arterial blood pH in the baseline of the case group and the mean control group after 12 h. This finding is strongly in favor of the effectiveness of liothyronine administration in AlP poisoning. It may be secondary to the improvement of tissue perfusion and hemodynamic status of patients. 6
In this study, serum FT3 was evaluated for two aims including to check the level of FT3 in the serum of patients with AlP poisoning and to assess the serum level of FT3 after liothyronine administration. The findings demonstrate that mean FT3 in the case group after 12 h is significantly more than baseline and also more than the controls after 12 h. It means that liothyronine has had a good absorption from the gastrointestinal tract and could maintain serum FT3 level in an appropriate normal range more than baseline. On the other side, the mean serum FT3 in the control group after 12 h is in the normal range but obviously less than baseline. It appears that the patients may be more prone to have a probable tissue hypothyroidism in AlP poisoning. In this regard, our findings are somewhat different from the findings of Abdolghafari et al. In their study, there was no significant change in serum T3 concentration in AlP-poisoned group in comparison to controls, although treatment with T3 increased the serum T3 concentration in AlP-poisoned group. 18
TSH is one of the important variables in this study. TSH was evaluated to assess the adequacy of serum FT3 after liothyronine administration. As shown, the mean serum TSH is significantly less in the case group after 12 h in comparison to baseline and the control group after 12 h. It demonstrates serum FT3 adequacy to suppress TSH secretion after liothyronine administration in the line to achieve the aim of this study.
PH3 increases the production of free radicals through a disturbance of oxidative phosphorylation, inhibiting antioxidant enzymes and reduction of glutathione level. 2,6 Lipid peroxidation as a result of oxidative stress is one of the main causes of cardiomyocytes damage in AlP poisoning.2 In our study, MDA as one of the end products of lipid peroxidation has been increased in the both groups after AlP poisoning, although this rising was more in the control group after 12 h in comparison with the case group. It seems that liothyronine administration is able to reduce free radical harmful effects as well as oxidative stress. These findings are similar to the Abdolghafari et al. findings with regard to the lipid peroxidation. 18 N-acetylcysteine as an antioxidant agent can be effective in AlP poisoning. 9 –11 N-acetylcysteine infusion significantly decreases the plasma MDA level and the duration of hospitalization in AlP poisoning. 11 It seems N-acetylcysteine antioxidant effects are more than T3 antioxidant effects in AlP poisoning. In addition, magnesium nanoparticle has some effects in the reduction of lipid peroxidation. 46
TAC is one of the parameters which is reduced in AlP poisoning. 49 Our results show that TAC was decreased in both the case and control groups after AlP poisoning, although this reduction is more obvious in the control group. It appears that liothyronine administration could be effective in this regard. Abdolghafari et al. reported similar findings related to TAC. 18
Thiols are molecules which contain the –SH attached to a carbon atom. Thiols are endogenous compounds and found in all body cells. Thiols are the major part of total body antioxidants and have a significant function against free radicals. Decreased levels of thiol have been reported in oxidative stress and AlP poisoning. 49,50 In this study, to evaluate the effectiveness of thyroid hormones against oxidative stress, TTM was measured. The findings showed in spite of increase in the serum level of TTM in both the case and control groups after 12 h in comparison to the baseline, this rise in the case group is statistically significant. Our finding regarding the TTM is strongly in favor of the useful effect of liothyronine administration against ROS. The useful effect of T3 in improving and elevating the thiol level had been demonstrated by Abdolghafari et al. 18
Increased activity and expression of the antioxidant enzymes is one of the critical mechanisms for reducing the amounts of free radicals. The dismutation reaction of superoxide (O2 −) to oxygen or hydrogen peroxide is catalyzed by SOD enzymes. Superoxide anion is a byproduct of oxygen metabolism, which may be harmful to cells in oxidative stress. 20 Thus, SOD plays an important role in reduction of the harmful effects of oxidative stress and its activity should be increased in oxidative stress condition. Our findings showed that the activity of SOD was decreased in both the case and control groups. Similar findings were also achieved in the previous study, 18 though some studies have shown a raised SOD activity after T3 administration. 28 It seems that the effects of thyroid hormones on SOD activity in oxidative stress should be evaluated in further studies.
Catalase is one of the critical enzymes in all living cells exposed to oxygen. Catalase acts as an antioxidant agent in oxidative stress and catalyzes oxygen peroxide to oxygen and water. 20 In this study, the findings showed that catalase activity increased in both the case and control groups. Increased catalase activity is more in the case group in comparison with the one of the control group after 12 h, although this raising is not much. Some studies showed that the activity of catalase was obviously raised by thyroid hormones in both the hypothyroid and euthyroid state. 27,28
In favor of the effectiveness of liothyronine administration in controlling AlP poisoning, there are some additional findings which may be important. First, the mean ICU hospitalization duration in the case group is less than that of the control group. This difference is statistically significant (p < 0.05). This suggests that liothyronine administration was able to reduce the time of ICU hospitalization. Second, the mean vasopressor drug dose in the case group is less than that of the control group and this difference is statistically significant (p < 0.05). In other words, liothyronine administration could improve patients’ hemodynamic, so that they required smaller doses of vasopressor drugs.
Conclusion
As the conclusion from the above, regardless of clinical interventions, it appears that thyroid function test may have a significant and decisive change during AlP poisoning, which has to be assessed more in further researches. Liothyronine administration has an efficient effect on improving the hemodynamic status of patients. Even though, mortality rate in the case group was decreased in comparison with the control group, with regard to the sample size, this parameter should be evaluated in the future studies with larger sample size. Although oral administration of liothyronine is somewhat effective in AlP poisoning, liothyronine IV administration acts faster and more effectively. Therefore, it is necessary to conduct a similar study with liothyronine IV administration. According to the results of this study, liothyronine has been able to play an effective role in maintaining SBP as well as improving hemodynamic status, maintaining blood pH within the normal range and the improvement of the oxidative stress parameters, particularly serum levels of TTM. So, this compound might play some roles in controlling AlP poisoning, but multicenter studies with liothyronine IV form are required.
Footnotes
Acknowledgment
The authors would like to thank the staff of the emergency room, ICU-A, ICU-C, and lab of Baharloo Hospital and the staff of the Pharmaceutical Sciences Research Center, Tehran University of Medical ciences. Authors also thank Iran National Science Foundation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by a grant from TUMS coded 93-04-33-27628.
