Abstract
Background
Intoxication with cadmium (Cd) ions leads to hippocampal damage and cognitive impairment. However, omega-3 polyunsaturated fatty acids (n-3 PUFAs) exert neuroprotective effects in different animal models of neurodegeneration.
Purpose
This study compared the neuroprotective effect of the n-3 PUFA, docosahexaenoic acid (DHA), and its downstream metabolite, resolvin D1 (RVD1), on hippocampal damage and memory deficits in cadmium chloride (CdCl2)-treated rats.
Research design
Control or CdCl2 (0.5 mg/kg)-treated rats were subdivided into three groups (n = 18/each) and treated for 6 weeks as follows: (1) fed control diet, (2) fed DHA-rich diets (0.7 g/100 g), or (3) treated with RVD1 (0.2 μg/kg, i.p).
Results
Treatment with a DHA-rich diet or RVD1 significantly increased the levels of docosahexaenoic acid and RVD1, respectively, in the hippocampal of CdCl2-treated rats without affecting the reduction in the expression of the 15-lipooxygenase-1 (ALOX15). These effects were associated with improvements in rats’ memory function and hippocampal structure, as well as a redction in the hippocampal levels of reactive oxygen species (ROS), malondialdehyde (MDA), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), nuclear localization of the nuclear factor-kappa beta p65 (NF-κB p65), and expression of cleaved caspase-3. Concomitantly, hippocampi of both groups of rats showed significantly higher levels of Bcl-2, superoxide dismutase (SOD), and glutathione (GSH), as well as enhanced nuclear levels of the nuclear factor erythroid 2–related factor 2 (Nrf-2). The effects of RVD1 on all these markers in the CdCl2-induced rats were more profound than those of DHA. Also, the increase in the nuclear protein levels of Nrf-2 and the decrease in the levels of Bax and nuclear protein levels of NF-κB p65 were only seen in the hippocampal of CdCl2 + RVD1-treated rats.
Conclusion
RVD1 is more powerful than DHA in preventing CdCl2-induced memory loss and hippocampal damage in rats.
Introduction
The environmental exposure to the heavy metal, cadmium (Cd), has increased during the last decades due to its wide distribution in foods, vegetables, soil, smoking, and daily consumable products such as batteries. 1 Currently, the neurotoxicity of Cd ions is well-reported in both humans and experimental studies and was shown to be associated with poor learning, low attention, encephalopathy, peripheral neuropathy, cerebral hemorrhage, olfactory dysfunction, hyperactivity, poor concentration, and cognitive deficits.2-6 Besides, chronic exposure to Cd ion is a serious risk factor for several neurodegenerative disorders such as Parkinson’s disease (PD) and Alzheimer’s disease (AD).7-9
The hippocampus is a part of the limbic system that is located in the medial temporal lobe of the brain. 10 Currently, the role of the hippocampus in learning and memory is well-established and well-explained in excellent studies and reviews.10-12 During the last decades, extensive research has been conducted to reveal the precise mechanisms that underlie the pathogenesis of Cd ions-induced hippocampal damage. Overproduction of reactive oxygen species (ROS) and oxidative stress remains the major-known mechanism by which Cd ions induce neuroinflammation and neural damage in the hippocampus and other areas of the brain, which subsequently leads to memory loss and cognitive impairment.1-3 Indeed, although Cd ions are inactive, they can cross the blood–brain barrier (BBB) and induce ROS via several mechanisms including scavenging endogenous antioxidant enzymes, displacement of other metals needed for antioxidant activities (e.g., Zn), oxidation, degradation of thiol-containing proteins (e.g., glutathione (GSH)), inhibition of the mitochondrial oxidative phosphorylation, promoting mitochondrial damage, and elevating the intracellular Ca2+ concentration due to oxidation of Ca+2 handling proteins.1-7,13,14 Subsequently, Cd ions lead to activation of intrinsic (mitochondria-mediated) and extrinsic (Fas/FasL-mediated) cell death in the cholinergic and non-cholinergic neurons which leads to a reduction in the neural synaptic activity, impairment of the cholinergic system, depletion in the neurotransmitters (e.g., acetylcholine/Ach) levels, and impaired neurotransmission by activating numerous apoptotic pathways such as mitogen-activated protein kinases (MAPKs), mammalian target of rapamycin (mTOR), and protein phosphatase 2A (PP2A) and suppression of other survival pathways such as 5’ AMP-activated protein kinase (AMPK) and sirtuin-1 (SIRT1) .2-7,13-17
Nonetheless, the brain is considered one of the organs that is very rich in lipids and polyunsaturated fatty acids (PUFAs). 18 Generally, PUFAs compose cell membranes of most neurons and exist as omega-3 (n-3 PUFAs) and omega-6 (n-6 PUFAs). Currently, it is well-accepted that a higher n6-PUFAs or n-6/n-3 PUFA ratio is associated with neuroinflammation, neural apoptosis, and adverse mental health.19,20 On the other hand, administration of n-3 PUFAs or increasing the n-3/n-6 PUFA ratio is a neuroprotective strategy to alleviate the brain oxidative stress, neuroinflammation, and apoptosis and preserve the memory function in several neurodegenerative disorders.18,21-24 However, n-3 PUFAs dominate in the neural cell membranes as either docosahexaenoic acid (DHA) or eicosapentaenoic acids (EPA), both of which have diverse actions.25,26 Currently, the important direct effect of EPA and DHA in suppressing the neural oxidative stress, inflammation, and apoptosis, as well as acting as antidepressing and cell signaling molecules is well-established.27-32 However, other studies have shown that both EPA and DHA can act to suppress neural oxidative stress, inflammation, and apoptosis by generating special pro-resolving mediators (SPMs) including resolvins, neuroprotectins (NPs), and maresins.33-35
Resolvins D1 and D2 (RVD1 and RVD2) are derived from the metabolism of DHA through the action of special enzymes, namely, 5- lipooxygenase-1 (ALOX5) and 15-lipooxygenase-1 (ALOX15). 33 However, RVD1 is the most common antioxidant and anti-inflammatory SPM derived from DHA. In most cells including the neurons, RVD1 acts through two receptors, namely, lipoxin A4 receptor/formyl peptide receptor 2 (ALX/FPR2) and the orphan, GPR32. 36 The activation of ALX receptors by different agonists such as methyl esters, lipoxin A4, RVD1, or other agonists or exogenous administration of RVD1 was demonstrated to have potent anti-inflammatory, antioxidant, and pro-resolving effects in several inflammatory conditions in the brain, liver, kidney, skin, and lung-induced injury.37-40 Within this view, several lines of evidence have shown that the RVD1 protective effect is mainly mediated by reducing the generation of ROS, inhibiting oxidative stress, and suppressing inflammatory cytokines production through upregulating the nuclear factor erythroid 2–related factor 2 (Nrf-2) and inhibiting the nuclear factor-kappa beta (NF-κB), two master transcription factors responsible for the synthesis of antioxidants and inflammatory cytokines, respectively.41-47 In a very recent elegant study, it has been demonstrated that chronic administration of RVD1 improved memory function and protected the hippocampal cells of high fat diet-induced oxidative stress, inflammation, and apoptosis by upregulation of Nrf-2 and suppression of P66Sch. 39
In a single study, it has been shown that n-3 PUFAs can protect against cadmium chloride (CdCl2)-induced cortical injury and alleviate the reduction in antioxidant enzymes. 18 However, the authors showed that the co-treatment with n-3 PUFA has more beneficial effects than post-treatment. Up-to-date, the effect of Cd ions on the hippocampal metabolism and levels of DHA and EPA, as well as the protective effects of DHA and RVD1 against Cd ions-induced hippocampal damage and memory loss is never investigated before.
Therefore, in this study, we assumed that CdCl2 may induce hippocampal injury in rats by reducing the content of DHA and/or suppressing their conversion to RVD1 through impairing the expression of ALOX15. Also, we hypothesized that administration of either DHA or RVD1 may attenuate CdCl2-induced hippocampal damage by suppressing oxidative stress, neuroinflammation, and apoptosis and compared their net effects.
Materials and methods
Animals
Adult male Wistar rats aged 8 weeks and weighing 160–180 g were supplied from the animal house at the College of Science at King Khalid University (KKU), Abha, Kingdom of Saudi Arabia. During all the treatment periods, all animals were housed under controlled temperature (23 ± 1°C), humidity (59–61%), and a 12 h dark/light cycle. Male rats were housed in plastic cages of four rats per cage. Two female rats were added to each cage to reduce the sex stress. All procedures included in this study were approved by the ethical committee at King Khalid University which is as per those published by the US National Institutes of Health animal (NIH publication No. 85-23, revised 1996).
Control and docosahexaenoic acid-rich diets
Composition of both control and DHA diet used in the experimental study.

DHA: docosahexaenoic acid.
Experimental procedure Rats were divided into six groups (n = 18/group) as follows: (1) Control rats: fed normal diet and administered an equivalent volume of 1% ethanol solution (prepared in phosphate buffer saline (PBS)/pH = 7.4) (as a vehicle); (2) Control + DHA: fed DHA-rich diet and received the equivalent volume of 1% ethanol solution; (3) Control + RVD1: fed normal diet and received an intraperitoneal dose of RVD1 (0.2 μg/kg) (dissolved in 1% ethanol solution); (4) CdCl2-model rats: fed control diet and were intragastrically administered CdCl2 solution (prepared in distilled water) at a final dose of 0.5 mg/kg. (5) CdCl2 + DHA: fed DHA-rich diet and received CdCl2 (0.5 mg/kg); (6) CdCl2 + RVD1: fed control diet and treated with RVD1 (0.2 μg/kg, i.p). All treatments were given daily to desired groups for a total period of 6 weeks which was shown previously required to induce hippocampal damage using this CdCl2 dose .
14
The dose of RVD1 and its route of administration have been previously used by other authors who have shown potent protective effects on memory function, inflammation, and neural apoptosis in animal models of HFD and PD.39,49 In our preliminary experiments, administration of 0.1% ethanol for control rats for six consecutive weeks did not affect the hippocampus structure and oxidative stress markers, as well rats’ memory of rats as compared to control rats received no treatments (data not shown). For this reason, the control group that received no treatment was omitted for simplicity.
Passive avoidance learning test (PALT)
The assessment of PALT was done 12 h after the last treatment in all rats of all groups (n = 18/each) as previously described by us. 50 This test determines the ability of the rat to remember a previously exposed foot electrical shock when entering a small dark room. In brief, each rat was placed in the study wooden apparatus (50 × 50 × 35 cm) that is composed of one large illuminated room and a smaller darkroom which are separated by a small door. The floor of the small room is attached to an electrical shock source. With an open door, each rat was placed in a large room and was allowed to freely explore the apparatus by entering into the small room (exploration phase). This exploration phase was repeated every 30 min for three trials each of 5 min. The rooms were cleaned between trials. During the last trial, once the rat entered the small room, the door was closed and the rat was exposed to an electrical shock (50 Hz, 1.5 mA, for 1 s) and left in dark for an extra 10 s. Then, the animal was returned to its cage, and the apparatus was cleaned. Two hours later, the protocol was repeated (testing phase), and the time needed for each animal to enter the darkroom was recorded. Animals with intact memory will avoid entering the darkroom during the testing phase. The time that is needed for each rat to enter the darkroom was recorded. Then, all rats were retained in their cages for the next 24 h.
Morris water maze (MWM)
Two days after the assessment of PALT, memory function was reassessed for all rats (n = 18/group) by the MWT as previously described in our laboratories. 51 In brief, the maze (diameter of 1.5 m/depth of 50 cm) was filled with water (23 ± 1°C). The pool was divided hypnotically into four quadrants (e.g., west (W), south (S), east (E), and north (N)), and a scape platform was placed in the southern-west (SW) quadrant and submerge 2 cm below the water. Besides, milk was added to the water to hide the escape platform. During the trials, the tested rat was placed at one quadrant (e.g., northern-east (NE), E, and N) (to equalize the distance) and released to find the platform. Accordingly, each rat was given three trails per quadrant per day which were separated by 5 min intervals. This procedure was repeated over the next 5 days. Each trial was given a total period of 90 s. If the rat failed to find the platform, it was directed to it. Both the escape latency and escape length were recorded and analyzed for each trail per day. On day 6, an extra trial (probe trial), where the escape platform was removed, was conducted and the number of crossing time over the hidden platform position was recorded.
Brain and hippocampi collections and processing
Rats were anesthetized by using an intraperitoneal dose of sodium pentobarbital (65 mg/kg) and killed by cervical dislocation. The skull of each rat was opened, and the brain of each rat was rapidly placed on ice. Some brains (n = 6/group) were rapidly fixed with 10% buffered formalin and forwarded to the histology laboratories. The other brains (n = 12) were dissected under a dissecting microscope to separate the hippocampi which were snap-frozen in liquid nitrogen and preserved at −80°C for further biochemical analysis. Accordingly, parts of these hippocampi were homogenized in 9 volumes of ice-cold phosphate-buffered saline (PBS, pH of 7.4) and then centrifuged at 11,000 x g for 10 min at 4°C. The supernatants of these total cell homogenates were isolated and used for the biochemical analysis. Other parts of the hippocampi were homogenized in nine volumes radioimmunoprecipitation (RIPA) assay buffer (Cat. No. Ab156034, Abcam, Cambridge, UK) plus protease inhibitor cocktail (Cat. No. P8340; Sigma Aldrich, St Louis, MO, USA) and centrifuged as above to isolate the supernatants which were used for the western blotting protocol. In other samples, lipids were freshly extracted using the chloroform/methanol mixture (2:1, v/v) as previously described by Folch et al. (1957) and used for determining the levels of DHA. On the other hand, the nuclear/cytoplasmic fractions were prepared from other parts of the hippocampi using a commercially available isolation kit as per the manufacturer’s instructions (Cat. No K256, BioVision Inc., USA). The purity of the nuclear and cytoplasmic fractions was determined by the detection of lamin A/C and α-tubulin, nuclear, and cytoplasmic markers. In all isolated fractions, protein concentrations were determined using a commercially available assay kit (Cat. No. 23,200, ThermoFisher).
Biochemical analysis
Hippocampal levels of RVD1 were measured using a special ELISA kit (Cat. No. MBS047677, MyBioSource, CA, USA). Hippocampal levels of total ROS were analyzed using a fluorometric kit (Cat. No. STA-347, Cell Biolabs, CA, USA). Levels of reduced GSH and malondialdehyde (MDA) in the rats’ hippocampi were measured using assay kits (Cat. No. 7511-100-K, R&D systems, Mn, USA, and Cat. No. KA3736, Abnova, Taiwan). Levels of superoxide dismutase (SOD), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α) were measured by ELISA kits (Cat. No. MBS036924, Cat. No. MBS2507393, and Cat. No. MBS175908, respectively, MyBioSource, CA, USA). The nuclear activities of NF-kβ p65 and Nrf-2 were measured using special assay kits (Cat. No. 40069 and Cat. No. 50296; Active Motif, Tokyo, Japan). M2 Spectramax plate reader (Molecular Devices, USA) was used to measure the absorbance while the fluorometric signal was measured using an FL600Bio fluorescent reader (Tek Instruments, Inc., USA). All procedures were done following the manufacturer’s instruction for n = 6/group.
Analysis of docosahexaenoic acid in the hippocampal tissues
Total lipids were extracted from the frozen hippocampal tissues of all groups of rats. The samples were transmethylated by BF3/methanol after saponification. Fatty acid methyl esters were analyzed by gas–liquid Hewlett-Packard chromatography (model 5890/series II) that is equipped with a FAMEWAX capillary column (30 m × 0.25 mm × 0.25 µm, Restek Corporation, Bellefonte, PA, USA) with the helium as the carrier gas. The analysis conditions were an initial temperature at 130°C for 1 min, increased to 150°C at a rate of 30°C/min, and then to 215°C at a rate of 3°C/min. DHA was identified using a known standard, and its level was evaluated by comparing the retention time.
Western blotting
This was done according to our previous studies. 51 Total and nuclear protein levels were prepared in the loading buffer and boiled for 5 min. Equal concentrations of proteins from each sample of similar fractions (40 µg) were separated by SDS-PAGE gels and transferred onto nitrocellulose membranes. Membranes were blocked with skimmed milk, washed in TBST buffer, and then incubated with the primary antibodies against nuclear factor-kappa beta p65 (NF-κB p65) (Cat. No. 3034, 65 kDa, 1:1000), Nrf-2 (4399, 120 kDa), ALOX15 (Ab119774, 75 kDa, 1:1000), cleaved caspase-3 (Asp175) (Cat. No. 9664, 17:19 kDa, 1:1000), Bax (Cat. No. 2744, 20 kDa, 1:1000), Bcl-2 (Cat. No. 2876, 28 kDa, 1:1000), β-actin (Cat. No. 4970, 45 kDa, 1:3000) (Cell Signalling Technology, USA), and Lamin A (Cat. No. sc-518,013, 69 kDa, 1:2000) (Santa Cruz Biotechnology, USA). Then, the membranes were washed again with the tris-buffered saline plus Tween 20 (TBST) buffer and incubated with the corresponding horse radish-peroxidase-conducted secondary antibodies. After washing, the signal was developed using the super signal west Enhanced Chemiluminescence (ECL) substrate (Thermo Scientific). The membranes were then photographed, and the signal intensity was analyzed by C-DiGit Blot Scanner (LI-COR, USA) and its associated software. Internal control was run between the different gels for normalization. The relative expression of the total proteins was normalized with the corresponding expression of β-actin where the nuclear proteins were normalized to the expression of Lamin A. This procedure was done for n = 6/group.
Histology evaluation
Buffered formalin-fixed brains were dehydrated in increasing concentrations of ethanol (70–100%). The tissues were then cleared with xylene and embedded in paraffin wax. After that, the tissues were cut 5 µM using the Reichert’s rotatory microtome, stained with hematoxylin and eosin (H & E), and examined and photographed under a light microscope system with a camera (Model no. Olympus-PM 10 SP automatic micrographic system, Japan). Histological evaluation was done for n = 6 rats/group.
Statistical Analysis
All data will be analyzed using GraphPad Prism statistical software package (version 6) using Student’s two-way analysis of variance (2-way ANOVA) followed by Tukey’s test for multiple comparisons. Values were considered significantly different at p > 0.05 and presented as mean ± SD.
Results
Effect of exogenous administration of resolvin D1 and docosahexaenoic acid on rats’ memory function of the control and cadmium chloride-treated rats
The assessment of the memory function in all experimental rats was conducted by two tests, namely, MWM and PALT. During the MWM, the memory function was assessed over 5 days where both the escape latency and path length were measured. Animals with intact memory are expected to show a progressive reduction in the values of these parameters. Besides, a probe trial was conducted where the escape platform was removed and the ability of the rat to remember the location of this platform was measured as the number of crossing time. During PALT, the ability of the rat to remember a previous electric foot shock when entering a dark room was measured by the time required to decide to enter this room. Normal values of all these parameters which were not significantly different from those measured in the control rats over all periods of the study were observed in the control + DHA and control + RVD1-treated rats. However, CdCl2-treated rats showed a significant increase in the escape latency and escape path length over all five testing days of the MWM as compared to control rats (Figure 1(A) and (B)). Also, they showed a reduction in the number of crossing above the removed escape platform (Figure 1(C)) and required less time to enter the dark area during the PALT (Figure 1(D)). On the other hand, a significant reduction in the escape latency and escape path length with a concomitant increase in the number of crossing and the time required to enter the dark area were seen in both CdCl2 + DHA and CdCl2 + RVD1 as compared to their corresponding values measured in the CdCl2-treated rats. However, while no significant variation in all these parameters was seen between the control and CdCl2 + RVD1, the values remained significantly varied in CdCl2 + DHA as compared to control rats (Figure 1(A)–(D)). Escape latency (time to find the hidden platform), escape path lengths, the number of crossing over the location of the removed platform during the MWM test (A–C), and time required to enter the darkroom during the passive avoidance learning test (D) in all experimental groups of rats. Data were presented as mean ± SD of 18 rats/group, (a) significantly different as compared to control rats, (b) significantly different as compared to control + DHA-treated rats, (c) significantly different as compared to control + resolvin D1, (d) significantly different as compared to CdCl2-treated rats, and (e) significantly different as compared to CdCl2 + DHA-treated rats. CdCl2: cadmium chloride; DHA: docosahexaenoic acid.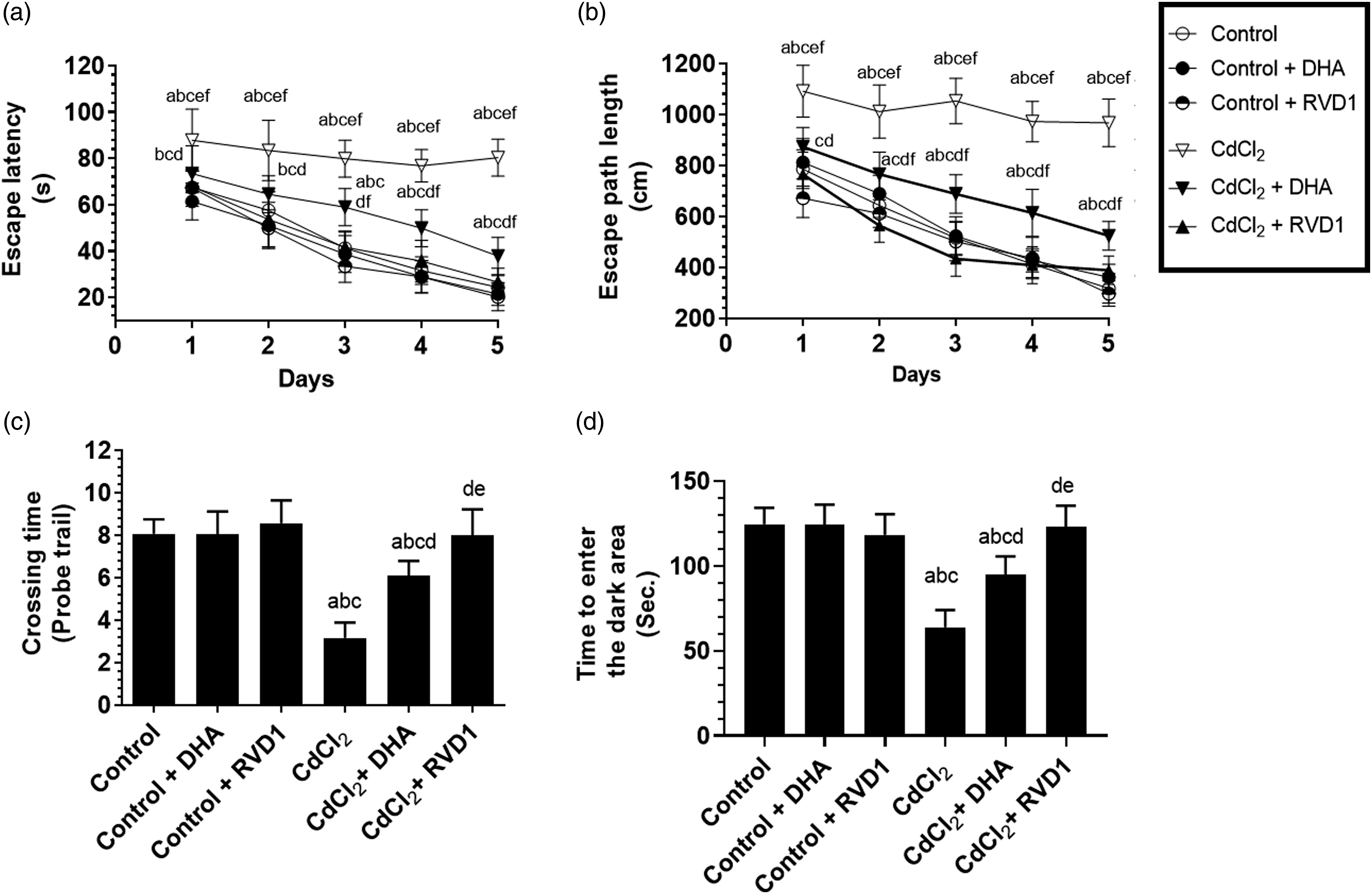
Effect of exogenous administration of resolvin D1 and docosahexaenoic acid on rats’ hippocampi structure of the control and cadmium chloride-treated rats
Control, control + DHA, and control + RVD1-treated rats showed an intact structure of the dental gyrus area of their hippocampi, the three layers including the granular cell layer (GCL), molecular layer (ML), and polymorphic layers (PL) were seen intact (Figure 2(A)–(C)). The cells of the GC layer of all these rats looked large and contained intact vesicular nuclei (Figure 2(A)–(C)). However, the administration of CdCl2 to rats reduced the number of cells in the GC layer (Figure 2(D)). This was associated with an increased number of necrotic and karyolytic cells, as well as perineuronal edema with a reduction in the number of neurons composing the PL (Figure 2(D)). On the other hand, CdCl2 + DHA-treated rats showed an improvement in the number of cell layers composing the GC layer. However, an increased number of necrotic and apoptotic cells with some degeneration is still seen (Figure 2(E)). On the contrary, almost normal architectures with a normal number of cell layers and almost no necrotic or apoptotic cells in the glandular cells forming the GC layer (Figure 2(F)). However, an increased number of neural cells was seen in the PL of both the CdCl2 + DHA and CdCl2 + RVD1-treated rats (Figure 2(E) and (F)). Photomicrographs of the dental gyrus area of the hippocampi of all groups of rats stained with hematoxylin and eosin staining. 200X. (A–C) were taken from control, control + DHA, and control + resolvin D1 (RVD1)-treated rats, respectively. All these sections, the three layers of the dental gyrus including granular cell layer, molecular layer, and polymorphic layers, were seen intact. The GC layer contained intact larger round cells with intact vesicular nuclei (thin black arrow). Note the presence of the oligodendrocytes at the sub-glandular layer (thick black arrow), astrocytes in both the ML and PL (yellow arrow), and the abundant neurons in the PL layer (thick red arrow), (D) was taken from a CdCl2-treated rat and showed a significant reduction in the number of layers forming the GCL (long black arrow) with an evidence of degeneration/necrosis (red arrow) and karyolytic nuclei (small black arrow). Perineuronal edema in the sub-GCL (curved arrow) with a reduction in the number of neurons was also seen in the PL, (E) was taken from a CdCl2 + DHA-treated rat and showed an increase in the number of GCL (thin black arrow). However, necrotic (thin red arrow) and karyolitic cells (short black arrow) with some degeneration were still seen abundantly in the GCL, and (F) was taken from a CdCl2 + RVD1-treated rat and showed almost normal architectures of GCL with normal intact cells. However, neural cells were seen in the PL of both CdCl2 + DHA- and CdCl2 + RVD1-treated rats (thick red arrow). CdCl2: cadmium chloride; DHA: docosahexaenoic acid; RVD1: resolvin D1.
Effect of exogenous administration of resolvin D1 and docosahexaenoic acid on the hippocampal levels of DVD1 and docosahexaenoic acid, and the expression of 15-lipooxygenase-1 in the control and cadmium chloride-treated rats
Cadmium chloride significantly reduced the levels of DHA and RVD1 and the protein expression of ALOX15 in the hippocampi of the treated rats (Figure 3(A)–(C)). On the other hand, a significant increase in the levels of DHA and RVD1 with no alterations in the protein levels of ALOX15 was observed in the hippocampi of both the control and CdCl2-treated rats which were fed either DHA or RVD1 as compared to control rats or CdCl2-treated rats, respectively (Figure 3(A)–(C)). Levels of DHA (A) and resolvin D1 (RVD1) (B) in the total brain homogenates, as well as total protein level of 15-lipooxygenase-1 (C) in the hippocampal of rat of all experimental groups. Data were presented as mean ± SD of six rats/group, (a) significantly different as compared to control rats, (b) significantly different as compared to control + DHA-treated rats, (c) significantly different as compared to control + resolvin D1 (RVD1), (d) significantly different as compared to CdCl2-treated rats, and (e) significantly different as compared to CdCl2 + DHA-treated rats. CdCl2; cadmium chloride; DHA: docosahexaenoic acid; RVD1: resolvin D1.
Effect of exogenous administration of resolvin D1 and docosahexaenoic acid on the markers of oxidative stress and inflammation, as well as antioxidants in the hippocampi of the control and cadmium chloride-treated rats
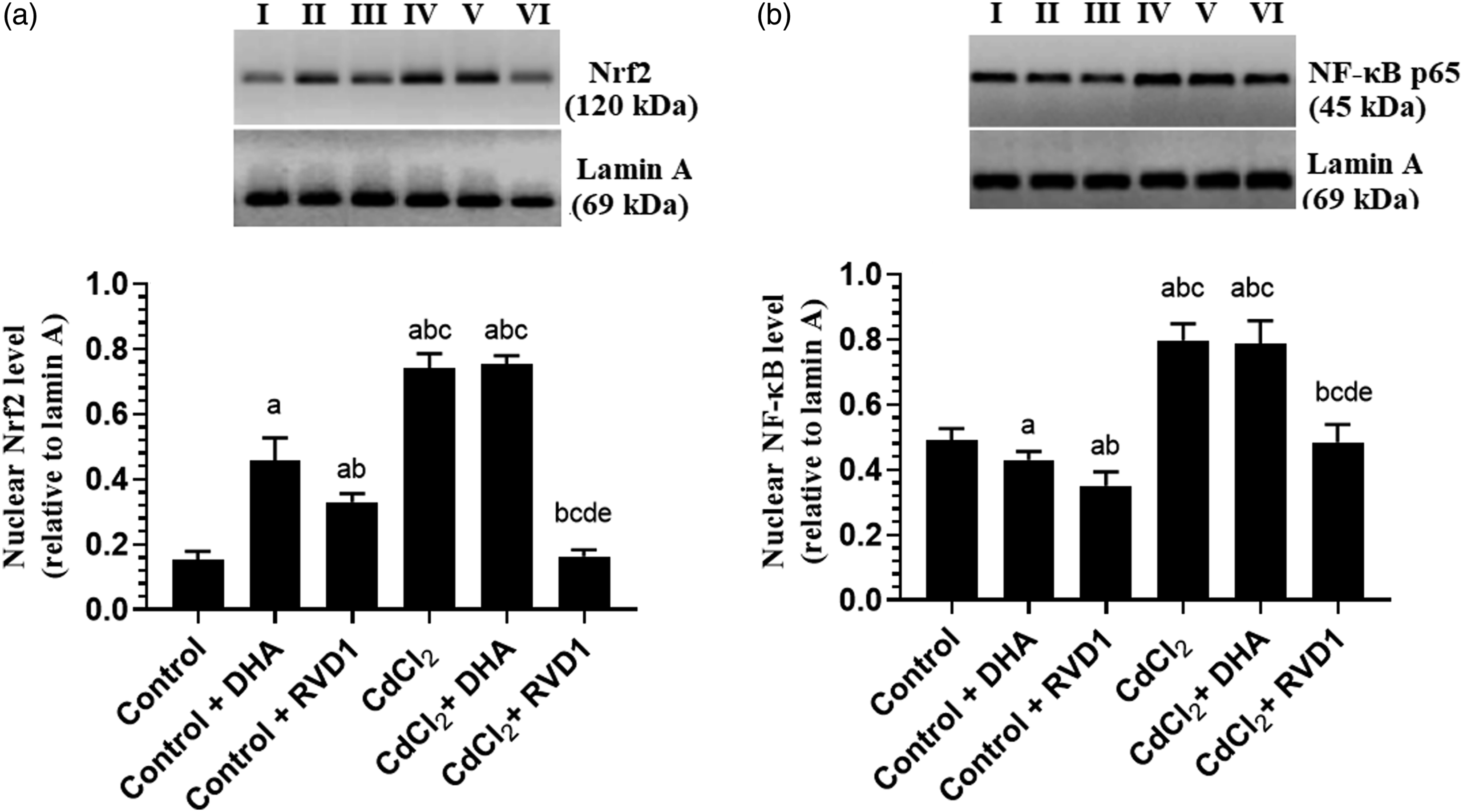
Control + DHA and control + RVD1-treated rats showed a significant increase in the levels of GSH and SOD, as well as the nuclear activity of Nrf-2 with a concomitant reduction in the levels of ROS, MDA, TNF-α, and IL-6 and the nuclear activity of NF-κB p65 as compared to control rats (Figure 4(A)–(D) and Figure 5(A)–(D)). At the same time, the nuclear levels of Nrf-2 were significantly increased, whereas the nuclear levels of NF-κB p65 were significantly reduced in the hippocampi of both control + DHA and control + RVD1 as compared to control rats (Figure 6(A) and (B)). However, the levels of ROS, MDA, TNF-α, and IL-6, as well as the nuclear activity and protein levels of both Nrf-2 and NF-κB p65 were significantly increased, whereas the levels of SOD and GSH were significantly decreased in the hippocampal homogenates of CdCl2-treated rats as compared to control rats (Figure 4(A)–(D), Figure 5(A)–(D), and Figure 6(A) and (B)). However, the levels of all these markers were significantly reversed in both the CdCl2 + DHA and CdCl2 + RVD1 as compared to CdCl2-treated rats. Of note, the levels of all these measured biochemical markers which were measured in the hippocampi of CdCl2 + RVD1 were not significantly different from their corresponding levels measured in the control rats (Figure 4(A)–(D), Figure 5(A)–(D), and Figure 6(A) and (B)). Besides, the levels of ROS, MDA, TNF-α, and IL-6, as well as the nuclear activity and protein level of both NF-κB and Nrf-2 were significantly lower, whereas the levels of SOD and GSH were significantly higher in CdCl2 + RVD1 rats as compared to CdCl2 + DHA-treated rats and were not significantly different as compared to their levels depicted in the hippocampi of the control rats (Figure 4(A)–(D), Figure 5(A)–(D), and Figure 6(A) and (B)). Levels of reactive oxygen species (A), malondialdehyde (B), superoxide dismutase (C), and reduced glutathione (D) in the hippocampi of rat of all experimental groups. Data were presented as mean ± SD of six rats/group, (a) significantly different as compared to control rats, (b) significantly different as compared to control + DHA-treated rats, (c) significantly different as compared to control + resolvin D1, (d) significantly different as compared to CdCl2-treated rats, and (e) significantly different as compared to CdCl2 + DHA-treated rats. CdCl2: cadmium chloride; DHA: docosahexaenoic acid. Nuclear activity of the nuclear factor erythroid 2–related factor 2Nrf-2 (A) and nuclear factor kappa beta (NF-κB) (B), as well as levels of tumor necrosis factor-α (C) and interleukin-6 (D) in the hippocampi of rat of all experimental groups. Data were presented as mean ± SD of six rats/group, (a) significantly different as compared to control rats, (b) significantly different as compared to control + DHA-treated rats, (c) significantly different as compared to control + resolvin D1, (d) significantly different as compared to CdCl2-treated rats, and (e) significantly different as compared to CdCl2 + DHA-treated rats. CdCl2: cadmium chloride; DHA: docosahexaenoic acid. Protein levels of the nuclear factor erythroid 2–related factor 2Nrf-2 (A) and nuclear factor kappa beta (NF-κB) (B) in the hippocampi of rat of all experimental groups. Data were presented as mean ± SD of six rats/group, (a) significantly different as compared to control rats, (b) significantly different as compared to control + DHA-treated rats, (c) significantly different as compared to control + resolvin D1, (d) significantly different as compared to CdCl2-treated rats, and (e) significantly different as compared to CdCl2 + DHA-treated rats. CdCl2: cadmium chloride; DHA: docosahexaenoic acid.

Effect of exogenous administration of resolvin D1 and docosahexaenoic acid on the markers of intrinsic cell death in the hippocampi of the control and cadmium chloride-treated rats
With no change in protein levels of Bax and cleaved caspase-3, the administration of both DHA and RVD1 to control rats significantly increased the hippocampal protein levels of Bcl-2 as compared to their levels detected in the hippocampi of the control rats which received the vehicle (Figure 7(A)–(C)). However, the protein levels of Bax and cleaved caspase-3 were significantly increased, whereas protein levels of Bcl-2 were significantly reduced in the hippocampi of CdCl2-treated rats as compared to control rats (Figure 7(A)–(C)). A significant increase in the levels of Bcl-2 with a coincided decrease in the protein levels of Bax and cleaved caspase-3 with a parallel increase in the protein levels of Bcl-2 were seen in the hippocampi of both the CdCl2 + DHA and CdCl2 + RVD1 as compared to CdCl2-treated rats (Figure 7(A)–(C)). However, when compared with each other, the reduction in the protein levels of Bax and cleaved caspase-3 as well the increase in the protein levels of Bcl-2 were more profound in the hippocampi of CdCl2 + RVD1 as compared to their levels measured in CdCl2 + DHA-treated rats (Figure 7(A)–(C)). Protein levels of cytochrome-c (A), Bax (B), and Bcl-2 (C) in the hippocampi of rats of all experimental groups. Data were presented as mean ± SD of six rats/group, (a) significantly different as compared to control rats, (b) significantly different as compared to control + DHA-treated rats, (c) significantly different as compared to control + resolvin D1, (d) significantly different as compared to CdCl2-treated rats, and (e) significantly different as compared to CdCl2 + DHA-treated rats. CdCl2: cadmium chloride; DHA: docosahexaenoic acid.
Discussion
The molecular mechanisms of CdCl2-induced hippocampal and brain damage have been extensively investigated, the effect of CdCl2 on the brain metabolism of n-3 PUFAs was poorly studied. In this study, we are showing that the administration of CdCl2 to rats is associated with a significant reduction in the hippocampal levels of both DHA and RVD1 with concomitant downregulation of ALOX15. On the other hand, the administration of both DHA and RVD1 not only attenuated the hippocampi structure and improved the rats’ memory function but also suppressed CdCl2-induced generation of ROS, lipid peroxidation, inflammation, and apoptosis. Interestingly, the protective effects of RVD1 on rat’s memory function, hippocampi structure, and all other measured biochemical endpoints were significantly more profound than those observed after the treatment with DHA, where complete protection was seen with RVD1. A graphical abstract demonstrating these mechansims is shown in Figure 8. A graphical abstract describing the damaging effect of CdCl2 on hippocampal neurons in relation to its effect on DHA metabolism and the protective roles of DHA and resolving D1. Under normal conditions, the high levels of DHA in the brain are broken down to other metabolites such as RVD1/2, mediated by the enzyme 15-lipooxygenase. DHA and RVD1 prevent neural oxidative stress, inflammation, and neurodegeneration (apoptosis) by (a) reducing the production of ROS, (b) activating the nuclear factor erythroid 2–related factor 2Nrf-2 which upregulates the cellular antioxidants such as superoxide dismutase and glutathione, and (c) suppression of the nuclear factor kappa beta and subsequent reduction in the production of the inflammatory cytokines. CdCl2 induces hippocampal neural by increasing ROS, overwhelming cellular antioxidant, activating NF-Kβ, and in the hippocampal cells while reducing Bcl-2/Bax ratio. However, CdCl2 also fosters neural damage by reducing the brain levels of DHA and RVD1 and suppressing the 15-LOX. Administration of both DHA and RVD1 can prevent these events by reversing these pathways without affecting the expression of 15-LOX with a more powerful and profound effect on RVD1. CdCl2: cadmium chloride; DHA: docosahexaenoic acid; ROS: reactive oxygen species; RVD1: resolvin D1.
The hippocampus is the hot spot of the brain that is responsible for declarative, spatial, spatial, and long-term episodic memories.52,53 Damage of the hippocampus in humans and animals is associated with cognitive deficits and memory loss. 53 The neurotoxicity of Cd ions is mediated by overproduction of ROS and subsequent inflammation and apoptosis of both the cholinergic and non-cholinergic neurons of the hippocampus and cerebral cortex.13,52,54 In this regard, Cd ions can deplete the intracellular GSH stores, overwhelm the cellular antioxidant enzymes, displacement of redox-active metals (e.g., Fe2+), and impairing of the mitochondria oxidative phosphorylation process.55-58 Subsequently, Cd-induced ROS can stimulate neuroinflammation and apoptosis by activating the transcription factors, NF-κB, and other numerous intrinsic and extrinsic apoptotic pathways such as p53/Bax, JNK, and p38 MAPK), both of which may, in turn, foster the production of the ROS.14,50,51,59,60 However, scavenging ROS or overexpression of antioxidant enzymes effectively prevented hippocampal apoptosis and inflammation and preserved the rodent’s memory after exposure to CdCl2.61-64
In the same line as these pieces of evidence, CdCl2-treated rats of this study showed impaired memory function as tested by PALT and WMT and severe loss of the pyramidal cells of the dental gyrus of their hippocampi. Also, the exclusive role of oxidative stress, inflammation, and apoptosis was confirmed by the obvious damage and loss of the hippocampal cells with the increased ROS and MDA levels; reduced GSH, SOD, and Bcl-2 levels; transactivation of NF-κB; and higher levels of TNF and IL-6 and protein expression of Bax and cleaved caspase-3. However, and surprisingly, CdCl2 significantly increased the nuclear activation and translocation of Nrf-2, master antioxidant transcription factor synthesis of phase II antioxidant enzymes (e.g., SOD, γ-GCS, GPx, and HO-1) in most cells antioxidant genes such as in the hippocampi of intoxicated rats.65,66 These findings contradict the observed hippocampal reduction in the levels of GSH and SOD after CdCl2 administration.
Supporting our data, the expression of Nrf-2 was significantly induced in different brain areas including the hippocampus, cerebral cortex, striatum, and cerebellum of the adult rat’s brain after intoxication with CdCl2. 67 In the same line, CdCl2 stimulates Nrf-2 in other tissues including the kidneys, hearts, and livers of experimental animals by overproduction of ROS. 68 Besides, Cd ions can activate the Nrf-2 through multiple mechanisms. 69 Despite this, ROS induced by CdCl2 can overwhelm the cellular antioxidant enzymes and reduce their levels in the cerebral cortex, hippocampus, liver, and kidney of rats and mice.70-73 Therefore, it seems reasonable that the reduction in hippocampus antioxidants is due to the scavenging of antioxidants, and the increase in Nrf-2 is an adaptive cellular mechanism to alleviate the oxidative injury induced by CdCl2 73 . However, ROS induces NF-κB, but Nrf-2 can inhibit it. 74 Like our data, CdCl2 can stimulate the activity of NF-κB in the brain of rats. 48 Therefore, it could be possible that the observed transactivation of NF-κB in the hippocampi of CdCl2 is mainly inducing in response to the higher levels of ROS where the increase in Nrf-2 is a protective mechanism to suppress this activation which remained high.
On the other hand, DHA and EPA are major n-3 PUFAs in the brain and substantial levels are required for normal neural development and brain function. 75 DHA is abundantly available in the brain of humans and rodents.76,77 However, reduced brain levels of DHA are associated with impaired neurogenesis, impaired neurotransmitter signaling, altered expression of membrane receptors, and memory deficits, where oxidative stress and neuroinflammation were shown to be the major contributing factors for the development of AD, PD, Huntington’s disease, as well as to depression, schizophrenia, and bipolar disease.67,78-81 Up-to-date, the effect of CdCl2 on brain levels of n-3 PUFAs is largely unknown. A very interesting observation reported in this study is the significant reduction in DHA content in the hippocampi of CdCl2-treated rats. Such a mechanism was never shown which may participate indirectly in the pro-oxidant, pro-inflammatory, and memory disturbing effects of CdCl2. However, DHA in the brain is responsive to the amount of dietary DHA. 82
The question raised here is how does CdCl2 reduce the brain levels of DHA despite the abundant quantities of α-linolenic acid (α-LNA, 18:3 n-3) and linoleic acid (LA, 18:2 n-6), two dietary precursors that can be converted (by desaturation and elongation) to DHA in the mammalian’s livers. 82 Interestingly, even in the absence of dietary DHA, the brain usually maintains normal levels of DHA through the hepatic conversion of both α-LNA and LA and increasing the half-life of DHA by suppressing its metabolizing enzymes (Ca2+-independent phospholipase A2 (iPLA2) and COX-1). 82 Given the well-reported hepatic toxic effect of Cd ions,83,84 it could be possible that this heavy metal reduces the brain content of DHA by suppressing the conversion of these precursors. Besides, Cd ions can activate COX1/2 and PLA2 in several organs such as the testes.85,86 Therefore, it could be also possible that CdCl2 increased the metabolism of DHA in rats’ brains by activating these enzymes.
On the other hand, the administration of DHA to CdCl2-treated rats restored normal hippocampal DHA content but partially improved the rat’s memory function and the structure of the dental gyrus of their hippocampi. The hippocampi of CdCl2-treated rats which were received DHA also showed no significant alterations in the nuclear accumulation Nrf-2 nor NF-κB, but had a partial reduction in their activities which suggests that DHA may interfere with the activity of these transcription factors possibly by some nuclear post-translation modifications such as phosphorylation and acetylation. However, a partial increase in the levels of GSH, SOD, and Bcl-2, as well as a partial decrease in the levels of ROS, MDA, TNF-α, and IL-6, and protein levels of caspase-3 and Bax were observed in the hippocampi of these rats. Of note, control rats that also fed the DHA-rich diet showed a significant increase in the nuclear levels and activation of Nrf-2 and levels of SOD and GSH with a concomitant decrease in the levels of MDA, TNF-α, and IL-6, as well as the transactivation of NF-κB in their hippocampi. These data may suggest that the supplementation of DHA to CdCl2-treated rats affords partial protection on the cognitive function and the hippocampal oxidative damage by suppressing oxidative stress and inflammation through modulating the activities of NF-κB and Nrf-2.
Supporting these data, previous studies have shown that supplementation of DHA stimulated neurogenesis and protected the hippocampus and improved memory in different animal models of brain injury as well as in individuals with dementia and cardiovascular disorders.79,81,87-91 Also, the protective effects of DHA on various tissues were largely attributed to their potent antioxidant and anti-inflammatory effect mediated by direct scavenging of ROS, upregulation of antioxidants, activation of Nrf-2/HO-1 axis, suppressing the production of inflammatory eicosanoids through competing n-6 PUFA, increasing the synthesis of anti-inflammatory docosanoids, inhibiting NF-κB, and stimulating the resolution of inflammation.92-97
However, it is well-accepted now that DHA acts by producing more effective metabolites such as PLMs such resolvins (RVDs), NPs, and maresins.33-35 The protective effect of these PLMs is well-reported in several organs and was shown to be mediated by mechanisms that are very similar to those exhibited by DHA.28,33 Among all, we were very interested to investigate the role of RVD1 in the hippocampal damaging effect of CdCl2 and its potential therapeutic effect, given the previous literature which has shown its crucial role in memory function and brain health.39,98 The exclusive observation in this study is that we have found a significant reduction in the levels of RVD1 and the expression of ALOX15 in the hippocampi of CdCl2-treated rats which could be explained by the parallel decrease in the hippocampal content of DHA. However, the administration of DHA to these rats did not correct this impairment. The sustained decrease in the levels of RVD1 may explain why the DHA produced only a partial effect on hippocampal health and memory function.
Surprisingly, the administration of RVD1 to CdCl2-treated rats completely restored the normal memory function of the treated rats and completely prevented CdCl2-induced oxidative stress, inflammation, and apoptosis in their hippocampi. However, like DHA, RVD1 did not affect the expression of ALOX15, but significantly normalized hippocampal RVD1, completely attenuated the impairment in the memory function, and restored the normal levels of all measured biochemical markers in the hippocampi of CdCl2-treated rats. Besides, RVD1 significantly increased GSH, SOD, and Bcl-2 levels, suppressed the production of TNF-α and IL-6, as well as the activity of NF-κB, and significantly the activation of Nrf-2 in the hippocampi of control rats, thus confirming its potent antioxidant and anti-inflammatory effect within the CNS. However, the amelioration in memory function and markers of hippocampal oxidative stress, inflammation, and apoptosis was more profound in the RVD1-treated rats than those exerted by DHA. This leads us to another conclusion where CdCl2 induces hippocampal damage by lowering the levels of DHA and RVD1 where it also limits the therapeutic potential of DHA by downregulating/inhibiting ALOX15. Therefore, it seems reasonable that treatment with RVD1 yields more neural benefits than DHA in this animal model.
Of note, the presence of ROS and inflammatory cytokines that are generated by stressful stimuli can induce a rapid conversion of DHA to these resolvins by activating the ALOX15 enzyme. 99 Also, the activity of the ALOX15 is largely depended and determined by the activation of cytosolic PLA2 and a special protein named 5-lipoxygenase activating protein or FLAP. 99 Despite the observed antioxidant and anti-inflammatory effect of DHA and RVD1 in the brain of CdCl2-treated rats, our data may suggest that CdCl2 inhibits ALOX15 in a mechanism(s) that is not related to oxidative stress and inflammation.
Similar to the protective effect of RVD1 reported here, aspirin-triggered resolvin D1 (AT-RVD1) prevented orthopedic surgery-induced cognitive decline.39,98 Also, exogenous administration of RvD1 protected against traumatic brain injury (TBI)-induced cognitive impairment in mice by preserving the mitochondria of the astrocytes and reducing the generation of inflammatory cytokines and ROS. 98 RVD1 effectively prevented brain, lung, and hepatic injury in several animal models by suppressing the production of the inflammatory cytokines, reducing the generation of ROS, and upregulation of endogenous antioxidants mediated by inhibiting NF-κB and upregulation of Nrf-2.34,41,42,44-46 In a very recent elegant study, it has been demonstrated that chronic administration of RVD1 can improve memory function and protect the hippocampal cells of high fat diet-induced oxidative stress, inflammation, and apoptosis by upregulation of Nrf-2 and suppression of P66Sch. 39
Despite these findings, most importantly, further analysis methods to measure levels of RVD1 and DHA such as gas chromatography will add more accurate data to this study. Besides, our data are still observational and needs further studies in transgenic animals to validate them, Besides, the lack of measurements and examining the roles of on the RVD1 receptors, as well as on the hippocampal levels of other RMPs such as RVD2, NPs, and the expression/activity of their still a valid limitation in this study. Furthermore, longer periods of treatment with DHA are also needed.
In conclusion, our data are the first to show chronic exposure to CdCl2 impairs the hippocampal levels of ALOX15 and subsequently impairs the metabolism and levels of DHA and RVD1. Based on the data on our hands, and given the well-known antioxidant and anti-inflammatory effects afforded by DHA and RVD1, we currently believe that such reductions in the hippocampal levels of DHA and RVD1 are major causes of CdCl2-induced hippocampal damage and the associated impairment in the cognitive function. Besides, our data are novel to show that the replacement therapy with either DHA or RVD1 is a protective strategy to alleviate CdCl2-induced neurotoxicity and memory deficits by silencing oxidative stress, neuroinflammation, and apoptosis, independent of modulating ALOX15 expression/activity, but mainly through suppression of NF-κB and upregulation of Nrf-2. However, these data also showed that the effect of RVD1 on all these damaging pathways is much profound than the effects afforded by DHA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the deanship of Scientific Research at King Khalid University, Abha, KSA for supporting this work under grant number (R.G.P.1/88/41).
Data availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
