Abstract
Background
The local renin-angiotensin system has been discovered in the eyes; thus, this study evaluates the Azilsartan effect in the retina and optic nerve toxicity induced by Cisplatin in vivo.
Methodology
Forty-eight male rats were randomly assigned into six groups of 8 animals. Group 1 was healthy control that received 0.5 mL/day of 0.5% carboxymethyl cellulose (CMC) orally (PO). Group 2 received a single dose of the 7.0 mg/kg CIS intraperitoneally with 0.5 mL/day of 0.5% CMC-PO. Groups 3 and 4 received 3.5 and 7.0 mg/kg/day of AZIL-PO, respectively. Groups 5 and 6 received 3.5 and 7.0 mg/kg/day of AZIL-PO, respectively together with a single dose of 7.0 mg/kg of CIS-IP. The ocular tissue and serum estimated the TNF-α, NF-kβ, and Casp-3. A complete blood count was also measured, and the eye was sent for histological examination.
Results
The administration of the 3.5 mg/kg AZIL significantly (p < 0.05) reduced the ocular tissue and serum TNF-α, NF-kB, and Casp-3 levels, when given to CIS treated group, while the 7.0 mg/kg AZIL does not. Additionally, azilsartan shows no negative impact on the CBC in rats. Finally, the eye histological examination showed a significant (p < 0.05) drop in the signs of inflammation and cellular degeneration, particularly after administration of the 3.5 mg/kg AZIL to the CIS-treated group.
Conclusion
A low dose of AZIL exerts an anti-inflammation and an anti-apoptotic effect through significant suppression of the pro-inflammatory mediators and an apoptotic biomarker by blocking the local angiotensin II type
Introduction
Cisplatin is a heavy metal chemotherapeutic agent. It effectively treats ovarian, testicular, head/neck, gastrointestinal, and lung cancers. 1 One of the considerable adverse effects of CIS is ocular toxicity which can cause vision loss at a therapeutic dose. 2 Clinical trials revealed that females with ovarian carcinoma who were treated with a therapeutic dose of CIS also suffered from visual abnormalities such as retinal toxicity that manifested in the form of cone dysfunction. 3
Treatment with CIS increased reactive oxygen species (ROS) and reactive nitrogen species (RNS) in retinal pigmented epithelium cells. Since CIS exerts its anticancer effect through the formation of DNA adducts and results in increased oxidative stress through the formation of free radicals and overwhelming the intrinsic antioxidant system that damages ocular system. 4 The pathogenesis of CIS-induced organ damage has been linked to several processes, including oxidative stress, apoptosis, and inflammation. 5 Moreover, CIS enhances the generation of ROS, which can activate p38 mitogen-activated protein kinases (MAPK), leading to apoptosis and enhanced upregulation of proinflammatory mediators that augment the cytotoxic effects of CIS. 6 Another possible mechanism of CIS neuro-toxicity is by blunting the protective effect of nuclear peroxisome proliferator-activated receptors-ϒ (PPAR-ϒ) against the free radicals. 7 The PPAR-ϒ expressed widely in retinal epithelial pigment, and its down regulation mediates many pathological consequences in the ocular tissue such as diabetic retinopathy and choroidal neovascularization. 8
Furthermore, vaso-occlusive lesion in the optic nerve have been reported with CIS administration that led to marked vision loss. Also, CIS elevated the inflammatory biomarkers such as nuclear factor kappa B (NF-kB) and tumor necrosis factor-α (TNF-α) and increased the level of oxidative stress markers such as malondialdehyde (MDA) and total oxidative stress levels in ocular tissue. 9 Several researches done to study the effectiveness of many anti-oxidants to counteract the toxicities induced by CIS and a significant reduction in inflammation and injury have been reported. Though, for some patients, CIS induced ocular toxicity is difficult to control and several patients continue to progress vision loss, indicating that further treatment modalities are required. 10
The classical circulating renin-angiotensin system (RAS) controls the blood pressure and electrolyte balance. Numerous studies revealed a pleiotropic that regulate inflammation, apoptosis, and tissue fibrosis. 11 Recently, the local RAS has been discovered the eyes. Multiple studies have demonstrated that RAS play a major role in the pathogenesis of many ocular diseases, including glaucoma, uveitis, diabetic retinopathy, and age-related macular degeneration (AMD). As such, the overexpression of angiotensin II type 1 receptor (AT1) receptor results in ocular pathogenesis through stimulation of oxidative stress and pro-inflammatory mediators, causing retinal ganglion cell dysfunction andoptic nerve ischemia. 12
Azilsartan (AZIL) Medoxomil is a prodrug which undergo hydrolysis in the gastrointestinal tract (GIT) and plasma to generate a more lipophilic and less acidic active metabolite than candesartan. 13 Moreover, AZIL is highly selective and has a 10,000-fold affinity for the AT1 receptor compared to AT2 receptor. AZIL is also an inverse agonist that suppress AT1 receptor activity in the lack of angiotensin II. 14 In this study, we aimed to evaluate the role of AZIL in CIS-induced ocular toxicity, thus, we determined tissue and serum TNF-α and NF-kβ levels as biomarkers of inflammation, and Casp-3 as a biomarker of apoptosis.
Materials and methods
Drugs and chemicals
CIS injection (50 mg/100 mL) was obtained from Kocak Pharmaceuticals, Turkey, and AZIL Medoxomil powder was obtained from Apollo Pharmaceuticals, Malaysia. Carboxymethyl cellulose (CMC) was obtained from Celotech company, China, while Ketamine 50 mg/mL was obtained from Bioveta company, Czech Republic, and Xylazine 2% was obtained from Intercheme company, Netherlands. Phosphate Buffer saline (PBS) was obtained from DNA biotech company, Ireland. The enzyme-linked immunosorbent assay (ELISA) kits that used for estimation of the TNF-α, NF-kB, and Casp-3 were obtained from Bioassay Technology company, UK.
Animal groups and study design
Forty-eight male Wister Albino rats weighing (270 ± 30 g) were obtained from the University of Tikrit, Iraq. Before the study, rats were acclimatized for 7 days and they were kept in a well-ventilated humidity-controlled room with 12 h light-dark cycle and at 24°C. The rats had free access to the standard pellets and drinking water. After that, the animals were randomly divided into six groups of eight rats each. The 1st group was kept as a healthy control and received 0.5 mL/day of 0.5% CMC by gavage tube. The 2nd group received a single dose of 7.0 mg/kg of CIS
15
intraperitoneally with 0.5 mL/day of 0.5% CMC orally. The 3rd and 4th (standard) groups were received 3.5 and 7.0 mg/kg/day of AZIL,
16
respectively that prepared in 0.5% CMC by gavage tube. The 5th and 6th groups were received 3.5 and 7.0 mg/kg/day of AZIL, respectively by gavage tube with a single dose of 7.0 mg/kg of CIS-IP (Figure 1). Treatments were continued for two consecutive weeks. Shows the schematic demonstration of the study design. CIS: Cisplatin, Hb: Hemoglobin, WBC: White blood cell, PLT: Platelet, TNF-α: Tumor necrosis factor α, and NF-kB: Nuclear factor kappa B.
Ethical approval
The research design and animal care protocol were approved by the Ethics Committee of the College of Pharmacy, University of Sulaimani, in accordance with generally accepted standards for the care and use of lab animals (No. PH35-21 on 14 November 2021).
Sample collection
On the 15th day of the study, rats were deeply anaesthetized using 50 mg/kg ketamine and 10 mg/kg xylazine, then sacrificed. Blood samples were obtained from heart, centrifuged at 3000 rpm for 10 min at room temperature; then, the serum was collected and stored at −20°C until the assay time. Simultaneously, the whole eyes were gently removed from the eyeball socket without any decoration.
Tissue preparation for enzyme-linked immunosorbent assay
Eyeballs were weighed, washed with ice-cold PBS (pH = 7.4), dissected, homogenized in the 9-fold volume of ice-cold PBS, then centrifuged at 5000 rpm for 20 min at 4°C. The supernatants were obtained and stored at −80°C.
Histopathological technique protocol
The excised whole eyeballs were fixed in 10% neutral buffered formalin for 3 days. Samples were dehydrated in a series of ascending concentration of ethanol (50%, 60%, 70%, 90%, and 100%). Then, samples were rinsed in xylene twice, embedded in paraffin, and left to solidify at room temperature. The blocks were sectioned into 5 μm using a semi-automated microtome (Leica-Germany), fixed on glass slides, and dried for 30 min. Next, sections were deparaffinized (xylene/3 times/5 min each) and then rehydrated with descending concentrations of ethanol (100%, 90%, 70%, 60% and 50%) for 5 min each. Finally, tissue sections were stained with Harris’s hematoxylin and eosin (H&E); and cover slipped. Furthermore, a semi-quantitative morphometric measure of the ocular tissues; retinal tissue, and optic nerve were examined using a light microscope (Olympus BX51, Japan) image analyzer (AmScope 3.7, for digital camera, MU300, 2019). Histopathological examination of lesion area, area of edema, inflammatory exudates, and vascular congestion within the ocular tissue were measured in μm. Additionally, the inflammatory and ocular decongested cells were counted in a total of 10 random fields under high power magnification (×1000). Finally, the lesions were scored as 0–10%: no lesions; 10–25%: mild; 25–50%: mild-moderate; 50–75%: moderate-severe; and 75–100%: marked-critical. 17
Statistical analysis
The data were executed with Graph Pad Prism version 8 software. Numerical variables were expressed as the mean ± standard deviation (SD). For the analysis of different groups, one-way variance analysis (ANOVA) was performed. Subsequently, to determine significance between the groups; Tukey multiple comparison, unpaired test was performed. The statistical significance level was set at p < 0.05.
Results
In this study, the 1st group was a healthy control and received 0.5 mL/day of 0.5% CMC, while the 2nd group received a single dose of 7.0 mg/kg of CIS intraperitoneally with 0.5 mL/day of 0.5% CMC orally. Then, 3rd and 4th groups were received 3.5 and 7.0 mg/kg/day of AZIL, respectively, while the 5th and 6th groups were received 3.5 and 7.0 mg/kg/day of AZIL, respectively with a single dose of 7.0 mg/kg of CIS intraperitoneally.
The effect of Azilsartan on pro-inflammatory cytokines in the ocular tissue and serum
Figure 2(a) showed that 3.5 mg/kg AZIL administration lowered the tissue TNF-α level significantly in CIS treated rats compared to CIS group (p = 0.03). In contrast, the 7.0 mg/kg AZIL administration to the CIS treated rats showed no significant effect on the tissue TNF-α level compared to CIS group (p = 0.13). The tissue TNF-α level was significantly higher in CIS group compared to the healthy control group (p = 0.005), the 3rd group (p = 0.005), and the 4th group (p = 0.03). On the other hand, there was a non-significance difference in the tissue TNF-α levels in the 3rd, 4th, 5th, and 6th experimental groups compared to healthy control group [(group 3 vs group1, p > 0.99), (group 4 vs group 1, p = 0.95), (group 5 vs group 1, p = 0.96), and (group 6 vs group 1, p = 0.68), respectively]. Interestingly, there was no significant difference in the tissue TNF-α levels between the groups that received two different doses of the AZIL (3rd vs 4th group; p = 0.96). Likewise, there was no significant change in the levels of the TNF-α in the ocular tissue between the combination groups that received two different doses of AZIL with CIS injection (5th group vs 6th group; p = 0.98). (a) Shows the effect of two different doses of the Azilsartan on the TNF-α levels of the experimental groups, and data are expressed as mean ± SD in the ocular tissues. *(p < 0.05), **(p < 0.01), ***(p < 0.001), ****(p < 0.0001) significantly different compared to the Cisplatin group. # (p < 0.05) significantly different compared to the healthy control group, using one-way ANOVA and unpaired t-test. (b). Shows the effect of two different doses of the Azilsartan on the TNF-α levels of the experimental groups, and data are expressed as mean ± SD in the blood serum. *(p < 0.05), **(p < 0.01), ***(p < 0.001), ****(p < 0.0001) significantly different compared to the Cisplatin group. # (p < 0.05) significantly different compared to the healthy control group, using one-way ANOVA and unpaired t-test.
Similarly, Figure 2(b) showed that administration a low dose of the AZIL to the CIS significantly decreased the serum TNF-α level compared to CIS group (p = 0.001), while co-administration a high dose of AZIL to CIS showed a non-significant change in the serum TNF-α level (p = 0.13). Furthermore, the serum TNF-α level was significantly higher in the group that received a single CIS injection compared to the healthy control group (p < 0.0001), the 3rd group (p < 0.0001), and the 4th group (p < 0.0001). Moreover, the serum TNF-α level showed no significant difference in the healthy control group compared to the 3rd group (p = 0.8254), the 4th group (p = 0.99), and the 5th group (p = 0.16), respectively. However, the serum TNF-α level was significantly higher in the co-therapy group of 7.0 mg/kg AZIL with CIS compared to the healthy control group (p = 0.002). However, the serum TNF-α levels showed no significant alteration between the 3rd and the 4th groups (p = 0.97). Also, there was no significant difference between the combination groups either regarding serum TNF-α levels (p = 0.41).
Figure 3(a) showed that CIS injection to the rats elevated the tissue NF-kB level significantly compared to the healthy control group (p < 0.0001). In this study, giving 3.5 mg/kg AZIL with the CIS group reduced tissue NF-kB level significantly compared to the CIS group (p = 0.04). While given 7.0 mg/kg AZIL with the CIS showed no significant impact on the tissue NF-kB level in comparison to the CIS group (p = 0.55). However, the tissue NF-kB level was significantly higher in the CIS group compared to the 3rd group (p = 0.0003) and the 4th group (p = 0.0016). On the other hand, the tissue NF-kB level in this research showed no significant difference among the healthy control group, the 3rd group (p = 0.98), and the 4th group (p = 0.74). Also, there was a significant difference in the tissue NF-kB level in the healthy control group compared to the co-therapy group of the 3.5 mg/kg AZIL with CIS (p = 0.0893), while the tissue NF-kB showed a significant increase in the combination group of 7.0 mg/kg AZIL with the CIS compared to the healthy control group (p = 0.0034). Moreover, the tissue NF-kB showed no significant change between the groups that received only low or high doses of AZIL (3rd group vs 4th group; p = 0.98). Similarly, the tissue NF-kB levels showed no significant difference between the combination groups (5th group vs 6th group; p = 0.70). (a) Shows the effect of two different doses of Azilsartan on the NF-kB levels of the experimental groups, and data are expressed as mean ± SD, in the ocular tissue. *(p < 0.05), **(p < 0.01), ***(p < 0.001), ****(p < 0.0001) significantly different compared to the Cisplatin group. #(p < 0.05) using one-way ANOVA and unpaired t-test. (b). Shows the effect of two different doses of Azilsartan on the NF-kB levels of the experimental groups, and data are expressed as mean ± SD, in the blood serum. *(p < 0.05), **(p < 0.01), ***(p < 0.001), ****(p < 0.0001) significantly different compared to the Cisplatin group. #(p < 0.05) using one-way ANOVA and unpaired t-test.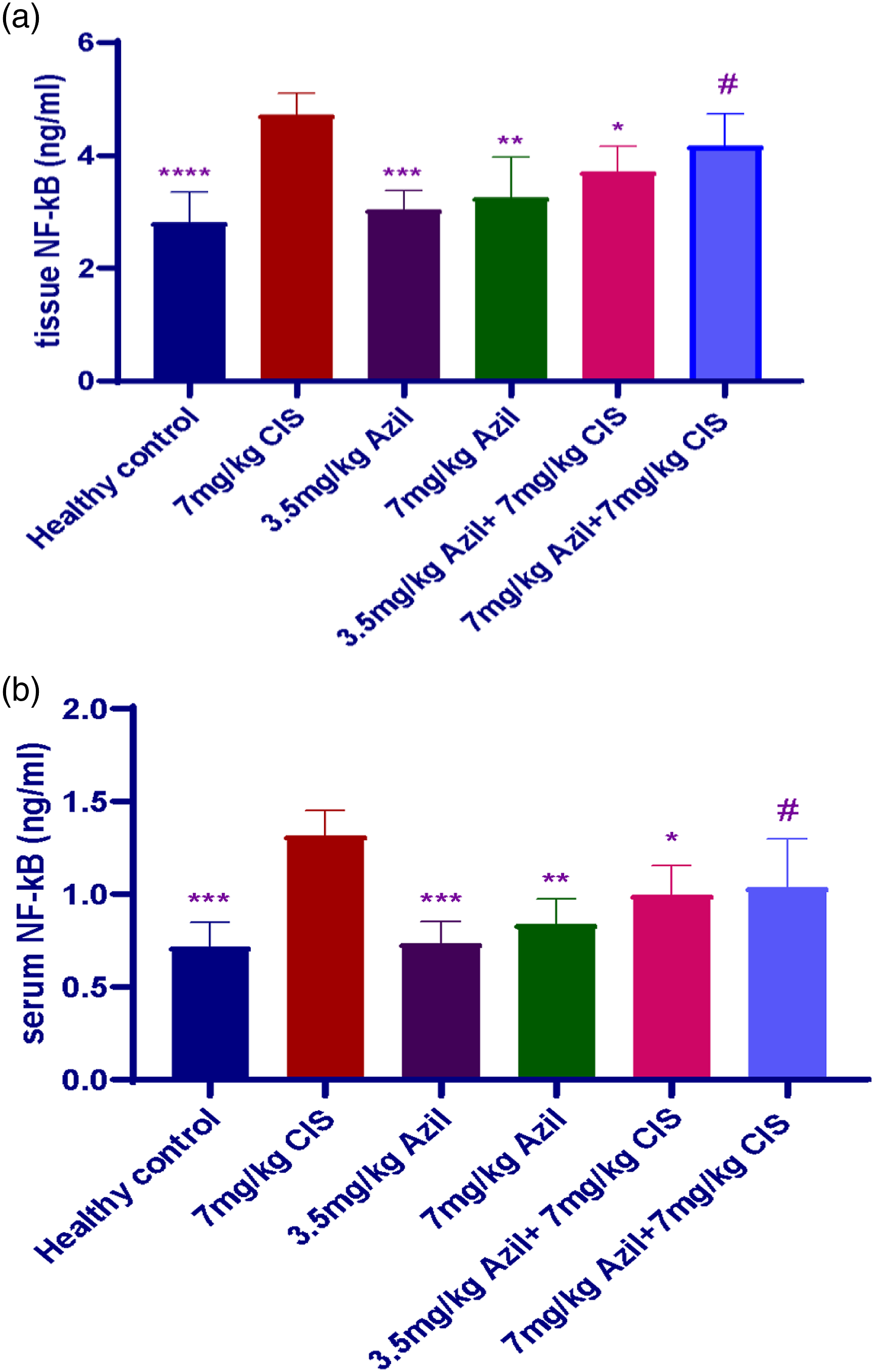
Figure 3(b) showed that the CIS significantly increased the serum NF-kB compared to the healthy control group (p < 0.0001), the 3rd group (p = 0.0001), the 4th group (p = 0.0012), and the 5th group (p = 0.04). In contrast, there was a non-significant change in the serum NF-kB in the group of 7.0 mg/kg AZIL with CIS compared to the CIS group (p = 0.10). Furthermore, there was no significant difference in the serum NF-kB levels in the healthy control group compared to the 3rd group (p > 0.99), the 4th group (p = 0.84), the 5th group (p = 0.10). In contrast, there was a significant increase in the serum NF-kB levels in the 6th group compared to the healthy control group (p = 0.04). However, there was no significant difference in the serum NF-kB levels between the groups that received only a low or a high AZIL doses (3rd vs 4th group; p = 92). Likewise, there was no significant change in the serum NF-kB levels between the combination groups (5th group vs 6th group; p = 0.99).
The effect of Azilsartan on Casp-3 levels in the ocular tissue and serum
Figure 4(a) showed a significant increase in the tissue Casp-3 levels after the CIS injection compared to the healthy control group (p < 0.0001). Similarly, the tissue Casp-3 levels increased significantly in the CIS group compared to the 3rd group and 4th group (2nd group vs 3rd group; p < 0.0001), (2nd group vs 4th group; p < 0.0001). Likewise, the tissue Casp-3 levels significantly decreased after co-administration of 3.5 mg/kg AZIL to the CIS compared to the CIS group (p = 0.002), while the tissue Casp-3 levels showed no significant difference in the combination of 7.0 mg/kg AZIL with CIS compared to the CIS group (p = 0.82). Furthermore, the tissue Casp-3 levels showed no significant change between the groups of rats that received only two different doses of the AZIL (3rd group vs 4th group; p = 0.96). Moreover, there was no significant change between the combination groups regarding the tissue Casp-3 levels were seen (5th group vs 6th group; p = 0.63). (a) Shows the effect of two different doses of Azilsartan on the Caspase-3 levels of the experimental groups, and data are expressed as mean ± SD, in the ocular tissue. *(p < 0.05), **(p < 0.01), ***(p < 0.001), ****(p < 0.0001) significantly different compared to the Cisplatin group. #(p < 0.05) significantly different compared to the healthy control group using one-way ANOVA and unpaired t-test. (b). Shows the effect of two different doses of Azilsartan on the Caspase-3 levels of the experimental groups, and data are expressed as means ± SD, in the blood serum. *(p < 0.05), **(p < 0.01), ***(p < 0.001), ****(p < 0.0001) significantly different compared to the Cisplatin group using one-way ANOVA and unpaired t-test.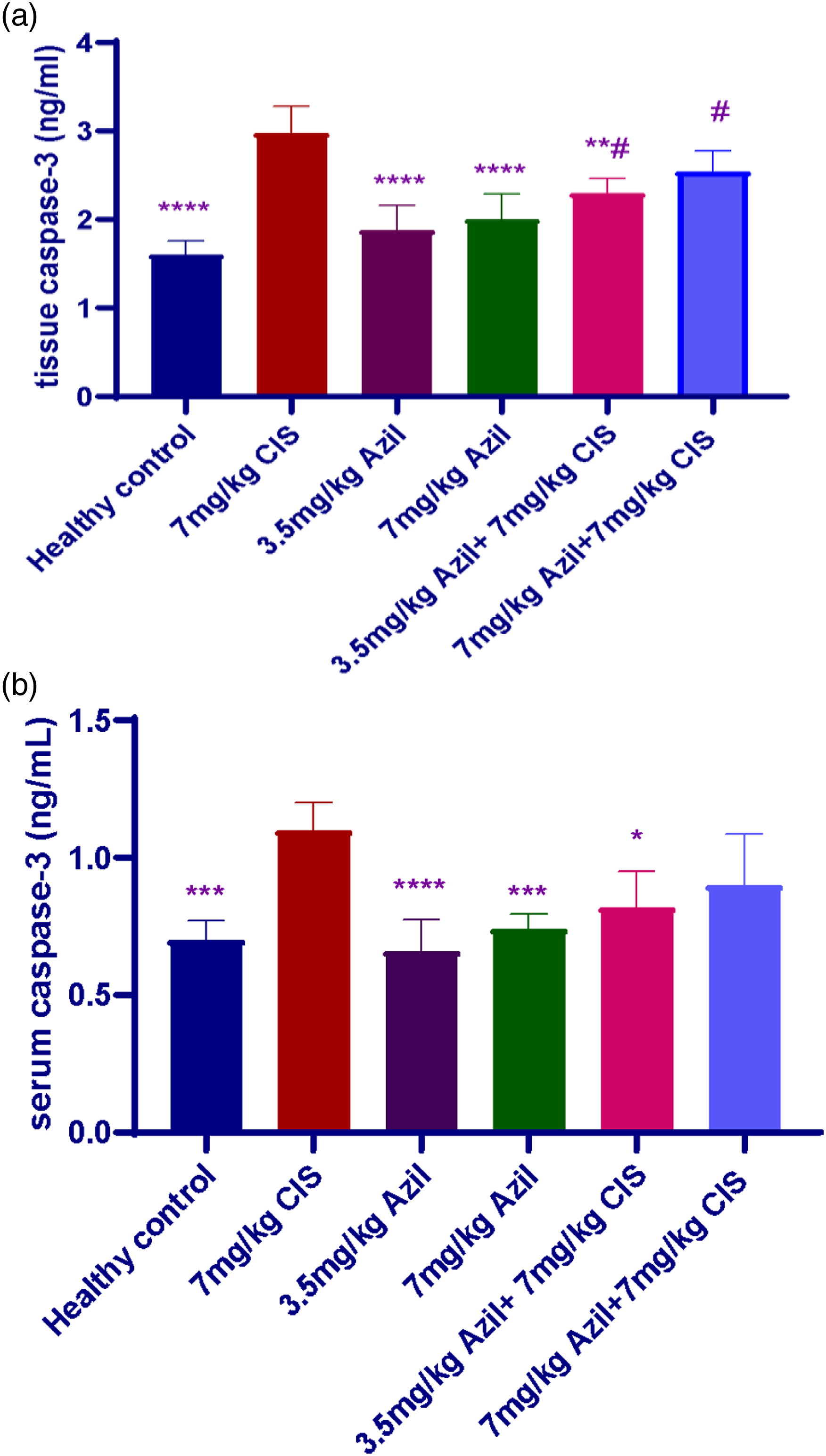
On the other hand, Figure 4(b) showed a significant elevation in the serum Casp-3 level after the CIS injection compared to the healthy control group (p = 0.0002), the 3rd group (p < 0.0001), and the 4th group (p = 0.000). Also, the co-administration of the 3.5 mg/kg AZIL to the CIS reduced the serum Casp-3 levels significantly compared to the CIS group (p = 0.01). While there was no significant difference in the serum Casp-3 after co-therapy of 7.0 mg/kg AZIL with the CIS compared to the CIS group (p = 0.11). In addition, there was no significant difference in the serum Casp-3 level of the healthy control group compared to the 3rd group (p = 0.99), the 4th group (p = 0.99), the 5th group (p = 0.59), and the 6th group (p = 0.11). Moreover, the serum Casp-3 showed no significant difference between the 3rd group and 4th group (p = 0.88), and the 5th group and 6th group (p = 0.88).
The effect of Azilsartan on the hemoglobin levels
Figure 5 showed a significant increase in the Hb levels after administration of high dose of the AZIL to the CIS-treated rats compared to the CIS only group (6th group, p = 0.000). In contrast, the AZIL low dose administration to the CIS treated rats showed no significant change in the Hb levels compared to the CIS only group (p = 0.75). CIS injection resulted in a significant decrease in Hb level compared to the healthy control group (p = 0.03) and the 4th group (p = 0.0003). There was no significant difference between the CIS only group and the 3rd group (p = 0.77). In contrast, there was no significant change in the Hb level in the healthy control group compared to the 3rd group (p = 0.99), the 4th group (p = 0.39), the 5th group (p = 0.41), and the 6th group (p = 0.51). Shows the effect of Azilsartan on the hemoglobin levels in the Cisplatin treated rats, data are expressed as means ± SD The values *(p < 0.5), ***(p < 0.001) significantly different compared to the Cisplatin group, using one-way ANOVA and unpaired t-test.
The Effect of Azilsartan on the platelets count in the CIS treated rats
Figure 6 showed a significant increase in the PLT levels after CIS injection to the rats compared to the healthy control group (p = 0.0103), the 3rd group (p < 0.0001), and the 4th group (p < 0.0001). On the other hand, co-administration of two different doses of AZIL to the CIS treated rats significantly reduced the PLT count compared to the CIS group (5th group vs the 2nd group; p = 0.004, 6th group vs the 2nd group; p = 0.001). In contrast, there was no significant difference in the PLT level in the healthy control group compared to the 3rd group (p = 0.33), the 4th group (p = 0.17), the 5th group (p = 0.99), and the 6th group (p = 0.97). Shows the effect of two different doses of Azilsartan on the platelet levels in the rats, data are expressed as means ± SD The values *(p < 0.05), **(p < 0.01), ***(p < 0.001), ****(p < 0.0001) significantly different compared to the Cisplatin group using one-way ANOVA and unpaired t-test.
The effect of Azilsartan on the white blood cells in the CIS treated rats
Figure 7 showed no significant alteration in the WBC levels among the experimental groups. In this study, CIS injection caused no significant decrease in the WBC count compared to the healthy control group (p = 0.53), the 3rd group (p = 0.14), the 4th group (p = 0.22), the 5th group (p = 0.57), and the 6th group (p = 0.99). Moreover, the WBC count showed no significant difference in the healthy control group compared to the 3rd (p = 0.95), the 4th group (p = 0.98), the 5th group (p > 0.99), and the 6th group (p = 0.79). Shows the effect of the two different doses of Azilsartan on the Cisplatin treated rats, data are expressed as mean ± SD The value ns represents non-significant difference among the experimental groups.
Histopathological results
Histological quantitative evaluation of eye lesions with different treatment values.
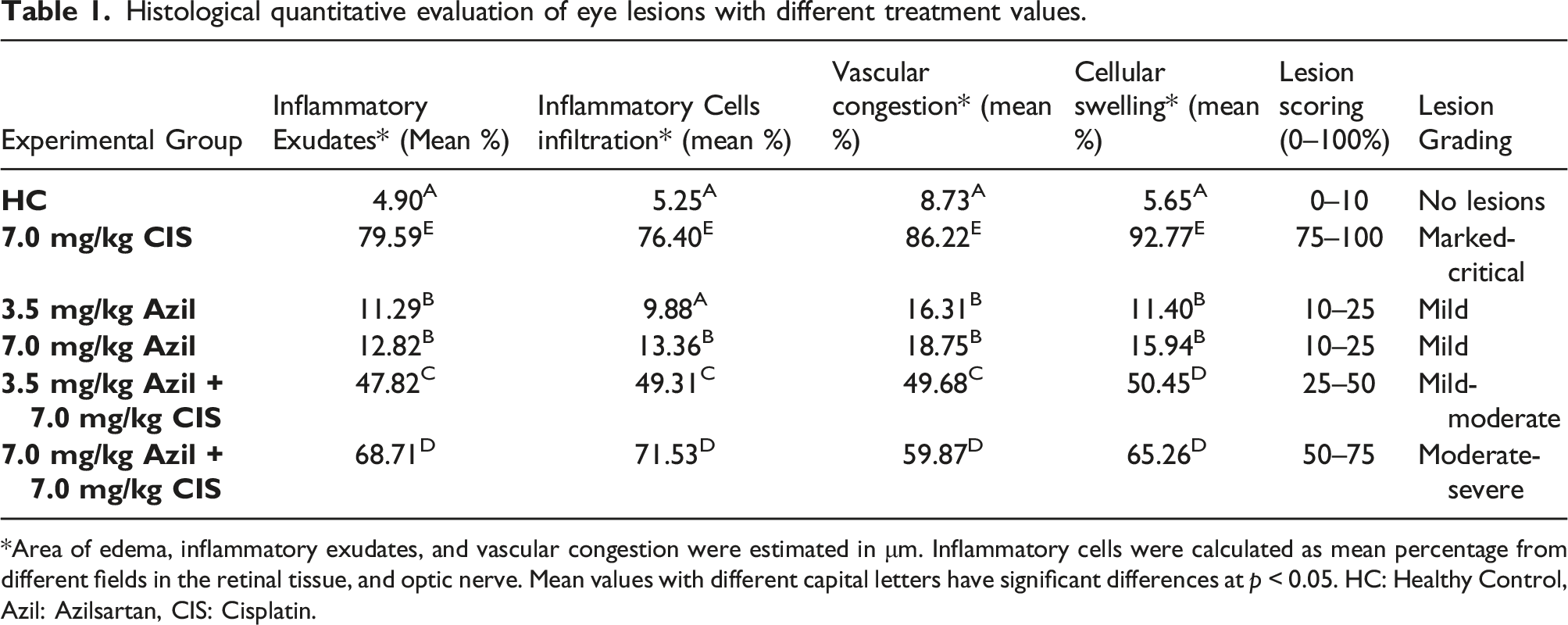
*Area of edema, inflammatory exudates, and vascular congestion were estimated in μm. Inflammatory cells were calculated as mean percentage from different fields in the retinal tissue, and optic nerve. Mean values with different capital letters have significant differences at p < 0.05. HC: Healthy Control, Azil: Azilsartan, CIS: Cisplatin.

Microscopic section of eye histological appearance. Normal histological arrangement and cell distribution of retina tissue, ganglionic cell layer (GCL), inner plexiform layer (IPL), inner nuclear layer (INL), outer plexiform layer (OPL), outer nuclear layer (ONL), and photoreceptor layer (PRL) in the healthy control group (a). Well-arranged nerve fiber, normal glial cellularity; oligodendrocytes (yellow arrows), vascular structures (black arrow), and perineurium (PN) in the healthy control group (b). In the 7.0 mg/kg Cisplatin group, the retina revealed severe-critical degeneration of photoreceptor cells (PRL), marked gliosis in the inner and outer nuclear layer (INL and ONL) (c). Marked ischemic optic nerves had a lot of inflammatory cells in the form of cell clusters (red arrows), multi vascular congestion (yellow arrows) with a severe-marked reduction in glial cells (d–f), (H &E stain).

Microscopic section of an eye in co-treatment groups. (a): The retina revealed mild vacuolation of photoreceptor cells (PRL), mild gliosis in the inner and outer nuclear layer (INL and ONL), and mild-moderate degeneration of ganglionic cells as indicated by black arrows in 3.5 mg/kg Azilsartan +7.0 mg/kg Cisplatin group. (b) and (c): Thinning and hemorrhage of epineurium (yellow arrows) of optic nerves with a mild reduction in glial cells 3.5 mg/kg Azilsartan +7.0 mg/kg Cisplatin group. (d): The retina revealed moderate vacuolar degeneration of photoreceptor cells (PRL), moderate gliosis, and vacuolation in the inner and outer nuclear layer (INL and ONL), and moderate vacuolar degeneration of ganglionic cells as indicated by black arrows in 7.0 mg/kg Azilsartan +7.0 mg/kg Cisplatin group. e and f: moderate ischemic optic nerves had a few inflammatory cells in the form of cell clusters (red arrows), vascular congestion (yellow arrows) with a moderate-severe reduction in glial cells in 7.0 mg/kg Azilsartan +7.0 mg/kg Cisplatin group, (H &E stain).
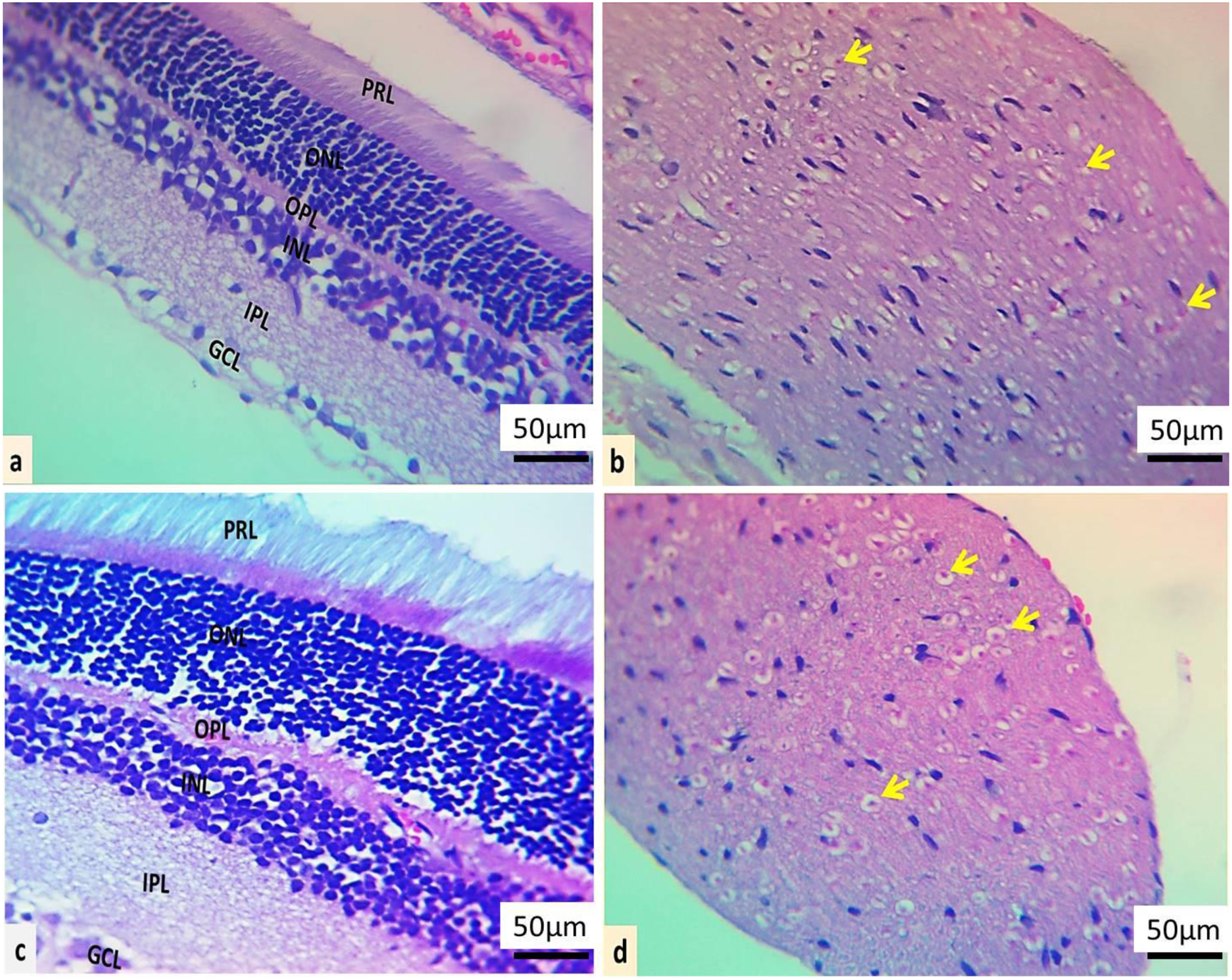
Microscopic section of the eye treated with the Azilsartan only groups. (a): The retina revealed normal cellularity in the 3.5 mg/kg Azilsartan group. (b): Mild vascular congestion (yellow arrows) in optic nerves in 3.5 mg/kg Azilsartan group. (c): Mild degeneration of photoreceptor cells, normal distribution of cells in other layers in the 7.0 mg/kg Azilsartan group. (d): Mild vascular congestion (yellow arrows) of optic nerves with a mild reduction in glial cell number as indicated by yellow arrows in 7.0 mg of Azilsartan group, (H &E stain).
Discussion
CIS’s cytotoxic nature can cause many ocular toxicities, including conjunctivitis, keratitis, optic neuritis, and impaired vision. CIS is associated with peripheral neuropathies that impact vision, such as corneal neuropathy, which could be directed to blindness. 18 Furthermore, CIS has various intracellular effects, including direct cytotoxicity associated with the production of ROS, the formation of covalent platinum-DNA adducts that cause DNA damage, mitochondrial dysfunction, activation of the MAPKs signaling pathway, induction of apoptosis, blunting the PPAR-γ and stimulation of inflammation. 19 The activation of the MAPK signaling pathway blunts the PPAR-γ protective effects, destabilizes the mitochondrial membrane potential, and causes the release of protein kinase-c, as well as stimulation of Casp-3 and Casp-induced cell death. Recently, many studies revealed that the RAS is associated with many diseases beyond its action on blood pressure and vasculatures. 20
In this regard, Kajiya et al., 2011 showed AZIL significantly improved adipogenesis in rats more than Valsartan through the overexpression of PPAR-γ. 21 Furthermore, many preclinical studies have revealed the protective effect of AZIL by inhibiting NADPH oxidase, a membrane-bound enzyme that generates free radicals and increases oxidative stress in the vasculature. 22 In addition, there are pieces of evidence of the inhibitory effect of AZIL on inflammatory cytokines such as TNF-α and suppression of NF-kB gene expression in vascular endothelial cells. However, the protective activity of AZIL is assumed by blocking the AT1 receptor and upregulating the angiotensin-converting enzyme 2 (ACE2)/angiotensin (1–7)/MAS receptor axis, as well as acting as a PPAR-γ agonist. 23 Our study measured TNF-α, NF-kB, and Casp-3 levels in ocular tissue to assess AZIL’s protective capability in overcoming CIS eye toxicity. Numerous studies found that CIS increased the mRNA expression of TNF-α in ocular tissue, which is involved in various inflammatory responses such as neutrophil differentiation, maturation, and activation. TNF-α, as a key link to intraocular inflammation, recruits’ leukocytes by mediating the production of intraocular chemokines that eventually destroy the blood-retinal barrier. 24 In this study, CIS elevated the TNF-α significantly compared to healthy control, causing inflammatory cell infiltration in the epithelial of the retina and optic nerve, which results in ocular inflammation. Whereas oral co-administration of the 3.5 mg/kg of AZIL to the CIS group significantly lowered TNF-α levels via reducing inflammation, minimizing ocular vessel congestion, and decreasing the retinal abnormality by ameliorating the signs of gliosis and preventing the ischemia of the optic nerve. The oxidative stress that was responsible for many pathogenesis mediators that led to AT1 upregulation in the retina was associated with an increase in TNF-α levels, which resulted in retinal microvascular abnormalities, as demonstrated in the previous study, and the condition was prevented by administering candesartan. 25 Also, the recent studies of AZIL application in mucositis and rheumatoid diseases demonstrated an anti-inflammatory action of AZIL by reducing the destructive effect of the proinflammatory cytokine TNF-α and preserving tissue integrity. 26 Additionally, NF-kB has been identified as a transcription factor that regulates the proinflammatory pathogenesis signaling pathway, which promotes the expression of inflammatory cytokines and chemokines. 27 Furthermore, rat models caused neurotoxicity and ocular inflammation by CIS injection, and the NF-kB expression levels were found to be high. 28 Moreover, the NF-kB activation in microglia is frequently neurotoxic due to the expression of additional proinflammatory cytokines and proapoptotic chemicals, such as TNF- α, Casp-3, and IL-1β, which result in photoreceptor apoptosis in the retina. 29 This study showed that NF-kB levels were significantly higher in rats that received CIS than in the healthy group. The NF-kB levels decreased significantly after administering 3.5 mg/kg of AZIL. These results are supported by a recent study, in which CIS elevated transcription factor NF-kB mRNA gene expression in the eye tissue by overproduction of free radical through upregulation of NADPH oxidase enzyme that causes mitochondrial dysfunction. 15 Previous research has shown that high levels of angiotensin II affect the activation of NF-kB through the AT1 receptor upregulation and the suppression of the PPAR-γ in the retina and the optic nerve. 30 Upregulation of oxidative stress status and activation of MAPK and NF-kB signaling pathways enhanced the neuronal damage in the incubated retina model, as stated by Palenski et al. 31 Furthermore, in vivo study demonstrated that NF-kB upregulation was associated with enhanced microglial activation that causes damage to the local retinal tissue and the release of proinflammatory cytokines. 32 AZIL is a highly selective AT1 receptor blocker. It acts as a PPAR-γ receptor agonist in the eye, downregulating cytokine production and NF-kB activation by suppressing the oxidative stress and MAPK signaling pathway. 33 This outcome parallels a recent in vivo study that showed AZIL has an inhibitory effect on the NF-kB level in cardiac tissue. 34 Casp-3 is a protein that provides information about the body’s apoptosis. Studies demonstrated an elevation in the Casp-3 expression in the ocular tissues post-CIS toxic dose administration. 15 In the present study, the tissue and serum Casp-3 increased significantly in the CIS group compared to the healthy group. At the same time, AZIL lowered the Casp-3 significantly and protected the retinal ganglion cells and optic nerve from apoptosis and degeneration. The ocular protection is significantly greater at the lower AZIL dose (3.5 mg/kg AZIL +7.0 mg/kg CIS) than at, the higher dose (7.0 mg/kg AZIL +7.0 mg/kg CIS). Similar results from other studies showed that lower doses of AZIL reduced inflammation and protected brain cells from injury in the cerebral ischemia pathological model. 35 Eventually, microscopic examination of the retina segments and optic nerve in our study indicates that AZIL at a low dose (3.5 mg/kg) given to the CIS group significantly reduced the cellular swelling, vascular congestion, and inflammatory cells. AZIL high dose has been linked to less favorable outcomes and profound decreases in blood pressure, which lowers blood circulation, increases vasodilation, and increases proinflammatory permeability to the eye vasculature. 36 In this experiment giving CIS to the rats caused optic nerve ischemia that led to degeneration of retinal layer and shrinkage in the photosensitive cells. AZIL co-administration with CIS in this study inhibited the optic nerve ischemia and showed a neuroprotection effect by blocking AT1R 37 which reduced the oxidative stress, mitochondrial dysfunction, and decreased apoptosis enzyme, caspase-3. Also, the biochemical analysis confirms the findings of the present study regarding the alleviation of inflammatory and apoptotic biomarkers that are parallel with lesion scoring findings which demonstrates the superiority of the low dose of AZIL in protecting the retina and the optic nerve in the CIS eye toxicity model. All these findings collectively support the protective effect of AZIL in minimizing the inflammation and degeneration in the retina and the optic nerve. The present study is the 1st to evaluate AZIL’s role in CIS-induced ocular toxicity. Finally, the two doses of AZIL given to the healthy rats in the current study showed no significant difference in the biomarker parameters and histological examination and without causing a significant effect on lesion scoring compared to the healthy control group. This outcome was revealed by the previous experimental study in a dementia rat model, which showed similarity in the overall outcome among the negative control group and non-induction groups. 16 Regarding the research on the ocular tissue, this study experiments with the effectiveness of 3.5 mg/kg and 7.0 mg/kg doses of AZIL for the 1st time, and such groups exhibit comparable findings with the healthy control group, which suggests the safety of such doses, particularly the 3.5 mg/kg AZIL dose on the retina and optic nerve. The treatment with an angiotensin receptor blocker for two weeks was associated with a decrease in the platelet count assessment in the fresh whole blood, and telmisartan minimized the platelet activation, as stated by Schäfer et al. 38 Generally, platelet main role is maintaining hemostasis and thrombosis in the body, however, platelets also actively involved in boosting inflammatory process by acting as immune cells, storing, and releasing multiple cytokines. 39 This study showed that AZIL-only groups reduced the platelet count significantly compared to the CIS group. Also, administration of AZIL to the CIS-treated rats decreased the PLT count; this result is the evenness with the aforementioned study, which revealing the antiplatelet effect of the AZIL is associated with diminishing the progression of inflammation. Moreover, AT1 receptor blockers elevated the blood hemoglobin levels in the anemic rats, 40 similar to the impact of the AZIL, particularly in the 6th group (7.0 mg/kg AZIL +7.0 mg/kg CIS) of this research on Hb levels. That suggests a good intervention by preventing the occurrence of anemia which may exacerbate the CIS toxicity. However, AZIL showed no significant impact on the WBCs. Likewise, AT1 receptor blockade by telmisartan and losartan showed similar results to the current results, preventing the immunity shut down by maintaining convenient WBCs.
Conclusions
AZIL shows anti-inflammatory and anti-apoptotic effects by down-regulation of pro-inflammatory TNF-α and NF-kB mediators and inhibiting the apoptotic mediator Casp-3 in the eyes of the CIS-treated rats through potently and selectively blocking local AT1 receptor in the retina and the optic nerve.
Footnotes
Acknowledgements
The authors appreciate the College of the Pharmacy, University of Sulaimani for its support and for providing instruments and tools for the practical procedures; also, a special thanks to the Biology Department, College of Science, University of Sulaimani for permission to use their animal house in this project.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The presented data were abstracted from a MSc thesis submitted by Noor Majid Raheem to the College of Pharmacy, University of Sulaimani.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
The data used to support the findings of this study are included within the article.
