Abstract
Almonertinib, a new third-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor, is highly selective to EGFR T790M-mutant non-small cell lung cancer (NSCLC). However, there is no available information on the form and molecular mechanism of Almonertinib-induced death in NSCLC cells. Herein, CCK-8 and colony formation assays, flow cytometry, electron microscopy, and western blots assay showed that Almonertinib inhibited NSCLC cells growth and proliferation by inducing apoptosis and autophagy which can be inhibited by a broad spectrum of caspase inhibitor Z-VAD-fmk or autophagy inhibitor chloroquine. Importantly, Almonertinib-induced autophagy was cytoprotective in NSCLC cells, and the blockade of autophagy improved cell apoptosis. In addition, Almonertinib increased reactive oxygen species (ROS) generation and clearance of ROS through pretreatment with N-acetyl-L-cysteine (NAC) inhibited the decrease of cell viability, apoptosis and increase of LC3-II induced by Almonertinib. The results of Western blot showed that both EGFR activity and downstream signaling pathways were inhibited by Almonertinib. Taken together, these findings indicated that Almonertinib induced apoptosis and autophagy by promoting ROS production in NSCLC cells.
Introduction
Lung cancer is a malignant tumor with the highest morbidity and mortality in the world. According to the 2018 global cancer statistics, the incidence of lung cancer accounted for 11.6% of the total cases and the mortality rate accounted for 18.4% of the total cancer deaths. 1 Non-small cell lung cancer (NSCLC) accounts for 85% of lung cancers, and the 5-year survival rate for NSCLC patients with conventional therapies such as chemotherapy, radiation, and surgery is only 15%. 2 Dose-limiting toxicity and acquired drug resistance are still dilemmas that needs to be solved.3,4 Therefore, it is urgent to find and develop safer and more effective drugs to treat NSCLC.
Epidermal growth factor receptor (EGFR) plays a critical role in cell survival, proliferation,5,6 differentiation, 7 motility, 8 tumorigenesis, 9 metastasis, 10 and drug resistance. 11 EGFR signaling has been shown to be abnormal in a variety of human tumors,12,13 including overexpression and gene mutations, which are associated with poor tumor prognosis.14,15 Therefore, EGFR is an important target for the treatment of tumors. There are also clinical benefit on NSCLC patients treated with EGFR tyrosine kinase inhibitor (TKI) which consist of two types: (1) small-molecule EGFR-TKIs that inhibit the tyrosine kinase activity of the EGFR intracellular domain; and (2) EGFR monoclonal antibodies that inhibit the activation of the EGFR ligand-binding domain. 16 Almonertinib (HS-10296) is a mutant-selective, and irreversible third-generation EGFR-TKI, which has strong inhibitory effect on EGFR T790M mutation than wild-type EGFR. Almonertinib was approved by the China Food and Drug Administration in 2020 for the treatment of advanced NSCLC patients with T790M-mutant who were resistant to the first- and second-generation EGFR-TKIs, such as gefitinib and afatinib.
Autophagy and apoptosis play important roles during lung cancer progression, both of them are important physiological activities of cells, which help to maintain the homeostasis of the intracellular microenvironment. 17 Apoptosis has been widely studied in the past three decades and is considered to be the main mechanism of cell development and programmed death. However, the function and molecular mechanism of autophagy have not been studied as deeply as apoptosis. Autophagy is a highly conservative evolutionary process that induces the degradation of unnecessary or damaged organelles or cytoplasmic contents in a lysosome-dependent manner. 18 And it can be activated by extracellular or intracellular stress, such as starvation, reactive oxygen species (ROS), hypoxia, etc.18–20 We previously identified that some compounds play an anti-cancer role by inducing autophagy in colon cancer cells. 21 Moreover, gefitinib and erlotinib have also been reported to induce autophagy in various cancer cell lines,22,23 and some studies have investigated the anticancer drug AZD9291-induced autophagy.24,25 However, whether Almonertinib could induce autophagy or apoptosis and the potential effects and mechanisms have no in-depth study.
In this study, we demonstrated the inhibitory effect of Almonertinib on various lung cancer cells, investigated the effects of Almonertinib on apoptosis and autophagy of H1975 and HCC827 cells, and the potential effects and mechanisms of the Almonertinib-induced apoptosis and autophagy in the NSCLC NCI-H1975 cells were studied.
Materials and methods
Reagents and antibodies
Almonertinib (Figure 1) was from Jiangsu Haosen Pharmaceutical Co., Ltd (Jiangsu, China) and dissolved in dimethylsulfoxide (DMSO) at a concentration of 20 mM for storage at −20°C. CCK-8 was purchased from Biosharp (Hefei, China). Annexin V FITC/PI apoptosis detection kit was purchased from Keygen Biotech (Nanjing, China). Z-VAD, chloroquine (CQ) and N-acetyl-L-cysteine (NAC) were from MedChemExpress (Monmouth Junction, NJ, USA). Dulbecco’s modified eagle’s medium (DMEM) medium and roswell park memorial institute (RPMI) 1640 medium were purchased from Gibco Life Technologies (Grand Island, NY, USA). Penicillin, streptomycin, and phosphate-buffered saline (PBS) were purchased from Biosharp (Hefei, China). Primary antibodies against ERK (#4695)/p-ERK (#4370), p-Akt (#4060), Caspase-3 (#9662), PARP (#9532), and SQSTM1/p62 (#8025) were purchased from Cell Signaling Technology (Beverly, MA, USA). Antibodies against EGFR (ab52894)/p-EGFR (ab40815), Akt (ab8805), and LC3B (ab192890) were purchased from Abcam (Cambridge, UK). The effective working concentration for the above was 1:1000. And the secondary antibodies were from Abbkine (California, USA), diluted to 1:5000.

The chemical structure of Almonertinib (HS-10296).
Cell lines and cell culture
NCI-H1975 was obtained from Keygen Biotech (Nanjing, China), HCC827 and A549 were purchased from BeNa Cultule Collection (Beijing, China). H1975 and HCC827 cells were cultured in RPMI 1640 medium. A549 cell was cultured in a DMEM medium. All cultured mediums were supplemented with 10% fetal bovine serum, 100 units/mL penicillin and 100 μg/mL streptomycin. Cells were grown in a 5% CO2 incubator at 37°C.
CCK-8 assay
The cells were inoculated in 96-well plates at 7 × 103 cells/well, and placed in a constant temperature 37°C, 5% CO2 incubator. After the cells were attached, the culture medium in the well was replaced by different concentration of Almonertinib. After a certain period of time, 10 µL of CCK-8 solution was added to each well and incubated in the incubator for 2 h. The absorbance of each well at this wavelength was measured at 450 nm using a plate reader (Bio-Rad Laboratories, Hercules, CA, USA).
Colony formation assay
Cells were seeded in 6-well plates at 6000 cells per well. After 24 h, the medium was removed and replaced with different concentration of Almonertinib. The cells were then placed in an incubator for another 5 days. At the certain time, the medium was removed and the cells were washed twice with PBS and fixed with paraformaldehyde at −20°C for 10 min. Then the crystal violet was added and left at room temperature for 10 min, washed with double-distilled water and dried at room temperature, before being counted.
Annexin V-FITC/PI staining assay
Cells were seeded in 12-well plates at 1.5 × 105 cells/well and treated with Almonertinib. After 24 h incubation, NCI-H1975 cells were harvested, washed, and re-suspended in 300 µL of binding buffer containing 3 µL of Annexin V-FITC and 5 µL of PI for 30 min. The cells were collected and analyzed by using a flow cytometer (BD Biosciences, State of New Jersey, USA).
Electron microscope study
All cells were gently scraped off when they reach a certain level of growth. Then the cells were centrifuged at 12 000 g for 5 min at 4°C and washed for the three times with PBS. Next, they were collected and fixed with 3% glutaraldehyde and 2% paraformaldehyde in 0.1 M PBS buffer (pH 7.4) for 2 days. Finally, they were analyzed by an electron microscope of the First Affiliated Hospital of University of Science and Technology of China.
RNA interference
Small interfering RNA (siRNA) targeting ATG5 was designed and synthesized by GenePharma (Shanghai, China). Cells were seeded in 6-well plates at 3 × 105 cells per well for 24 h. H1975 cells were transfected with ATG5 siRNA according to the manufacturer’s instructions. The total protein was extracted 48 h after transfection and detected by Western blot.
Mitochondrial membrane potential
Cells were seeded in 6-well plates at 3 × 105 cells per well for 24 h to reach exponential growth before treatment. Changes in mitochondrial membrane potential were evaluated using a mitochondrial membrane potential assay kit (Beyotime Biotechnology, Wuhan, China) according to the instructions of manufacturers. Stained cells were visualized using a fluorescence microscope (Olympus, Tokyo, Japan).
Reactive oxygen species (ROS) generation assay
The generation of intracellular ROS was measured using DCFH2-DA probe. NCI-H1975 cells were seeded in 6-well plates at 2 × 105 cells per well. Cells were treated with 6 μM Almonertinib for 6 h in the presence or absence of NAC (5 mM, 1 h), and followed by incubation with DCFH2-DA probe for 30 min. Cells were collected and washed with PBS. The generation of ROS was observed using a fluorescence microscope (Olympus, Tokyo, Japan).
Western blot analysis
Cells were collected by the centrifuge and lysed on ice for 30 min using RIPA lysate. Then the lysates were centrifuged at 12 000 g at 4°C for 30 min. The protein was separated by SDS-PAGE and transferred to the PVDF membrane after using the bicinchoninic acid (BCA) assay to detect protein concentrations. Next, the membranes were blocked with 5% skim milk in PBS with 0.1% Tween 20 and incubated with primary antibodies overnight at 4°C, followed by incubation with the corresponding secondary antibodies. Finally, the gel imaging system (Bio-Rad, USA) was used to get the images.
Statistical analysis
Statistical analysis was performed with one-way analysis of variance and Tukey’s test. The difference was considered statistically significant at *P < 0.05 and **P < 0.01. Values are expressed as the mean ± SEM of three experiments.
Results
Almonertinib reduced the survival of NSCLC cells
To assess the effects of Almonertinib on the growth of NSCLC cells, in this study, we used various NSCLC cell lines. NCI-H1975 cells (EGFR-resistant mutation: L858R/T790M mutation), HCC827 cells (EGFR-sensitive mutation: E746-A750 deletion) and A549 cells (wild-type EGFR) were treated with various concentrations of Almonertinib for 24, 48 or 72 h. Cell viability was determined by the CCK-8 assay. As shown in Figure 2(a), Almonertinib remarkably (P < 0.05) reduced the viability of all NSCLC cell lines in a dose-dependent manner, and the sensitivity of H1975 and HCC827 cells to Almonertinib was higher than that of A549 cells. Furthermore, we determined the effect of Almonertinib on clonogenic growth and found that Almonertinib could significantly inhibit H1975 and HCC827 cells colony formation rather than A549 cells (Figure 2(b)). These results suggested that Almonertinib inhibited NSCLC cells proliferation and reduced cell survival. H1975 and HCC827 cells were more sensitive to Almonertinib than A549 cell.

Almonertinib reduced the survival of NSCLC cells. (a) A549, H1975 and HCC827 were treated with HS-10296 (2, 4, 6, 8, 10 and 12 μM) or vehicle control (DMSO, 1‰) for 24, 48, 72 h, respectively. The cell viabilities were determined using CCK-8 assay. Data were shown as mean ± SEM (n = 3 independent experiments). (b) A549, H1975 and HCC827 were treated with Almonertinib (0.1 μM) for 24 h, and medium was replaced and colonies growth was noticed at Day 7. (1) control group; (2) 0.1 μM Almonertinib group.
Almonertinib-induced apoptosis in H1975 and HCC827 cells
To confirm the pro-apoptotic effect of Almonertininb on NSCLC cells, apoptosis in H1975 and HCC827 cells was determined by flow cytometry following staining with Annexin V/PI. The results (Figure 3(a) and (b)) showed that Almonertinib significantly (P < 0.05) induced the apoptosis in H1975 and HCC827 cells in a dose-dependent manner rather than A549 cells. To further verify the pro-apoptotic effect of Almonertinib, we performed Western blot analysis and found that Almonertinib upregulated cleaved caspase-3 and PARP (Figure 3(c)). Moreover, we further used Z-VAD-fmk, a broad spectrum of caspase inhibitor to illustrate the in intrinsic mechanism of Almonertinib. As shown in Figure 3(d), CCK-8 assay result showed that co-incubation with Z-VAD-fmk partly (P < 0.05) reversed the inhibitory effect of Almonertinib on the cell viability of H1975 and HCC827 cells. These data confirmed that Almonertininb can induce caspase-dependent apoptosis in NSCLC H1975 and HCC827 cells (Table 1).

Almonertinib-induced apoptosis in H1975 and HCC827 cells. (a) Induction of apoptosis in NSCLC cells was determined using flow cytometry following staining with Annexin V/PI. (b) Effects of Almonertinib on the expression of caspase-3 and PARP were analyzed by Western blotting in H1975 and HCC827 cells. (c) After pre-incubation with 10 μM z-VAD-fmk for 1 h, cells were treated with 6 μM HS-10296 for 24 h. Then, relative cell viability was assessed with CCK-8 assay. All the experiments are carried out three times. Data were shown as mean ± SEM (n = 3), * P < 0.05, ** P < 0.01, compared to the control group.
The apoptosis rate of NSCLC cells induced by HS-10296.a

a The data in the table are the percentage of cell population in different times of the Figure 3(a).
Almonertinib-induced autophagy in H1975 and HCC827 cells
Autophagy is a double-edged sword in tumors because it can cause tumor cells survival or death. A number of studies have reported that the mechanism of TKI resistance is closely related to autophagy. Therefore, we subsequently explored whether Almonertinib causes autophagy in NSCLC cells. Interestingly, when we observed the inside characteristics of cells resulting from Almonertinib treatment, we found that both autophagosomes and autophagolysosomes were accumulated markedly in the H1975 and HCC827 cells. Furthermore, the expression of p62 and LC3-II, protein markers of autophagy, were determined by Western blot. As shown in Figure 4(b) and (c), Almonertinib significantly (P < 0.05) promoted the expression of LC3-II and decreased the expression of p62 in a concentration-dependent manner in the H1975 and HCC827 cells. Finally, cells were co-incubated with Almonertinib and autophagy inhibitor chloroquine (CQ), however, CQ increased the cytotoxic effect of Almonertinib on lung cancer cells (Figure 4(d)). These results indicated that Almonertinib significantly induced autophagy in H1975 and HCC827 cells.

Almonertinib induced autophagy in H1975 and HCC827 cells. (a) Electron microscopy of cells treated for 24 h with DMSO or 6 μM Almonertinib for 24 h. Scale bar is 2 μm. (b) Western blot analysis was performed to measure the autophagy-related protein expression levels. (c) Quantitative analysis of proteins in (b). (d) After pre-incubation with 20 μM CQ for 1 h, cells were treated with 6 μM Almonertinib for 24 h. The relative cell viability was assessed with CCK-8 assay. All the above data are mean ± SEM. ** P < 0.01 versus control group.
Inhibition of autophagy enhances Almonertinib-induced apoptosis in H1975 cells
CQ inhibits the final stage of autophagy, preventing the degradation of autophagosomes by increasing lysosomal pH. As shown in Figure 5(a), the cotreated groups showed increased autophagic vacuole formation compared with the Almonertinib-treated groups in H1975 cells. To explore the role of Almonertinib-induced autophagy in H1975 cells, we treated H1975 cells with DMSO, Almonertinib, CQ, or both Almonertinib and CQ and found that combination treatment was more susceptible to induce apoptosis, which was evidenced by the significantly (P < 0.05) higher ratio of apoptotic cells than the Almonertinib-treated group (Figure 5(b)). As shown in Figure 5(c), In Almonertinib-treated cells, LC3-II expression increased, however, in the cotreatment group, LC3-II expression levels were further increased. Autophagy was inhibited in the cotreated group, which exhibited increased cleaved Caspase-3. These results indicated that Almonertinib-induced autophagy was cytoprotective and that inhibiting autophagy could increase apoptosis.
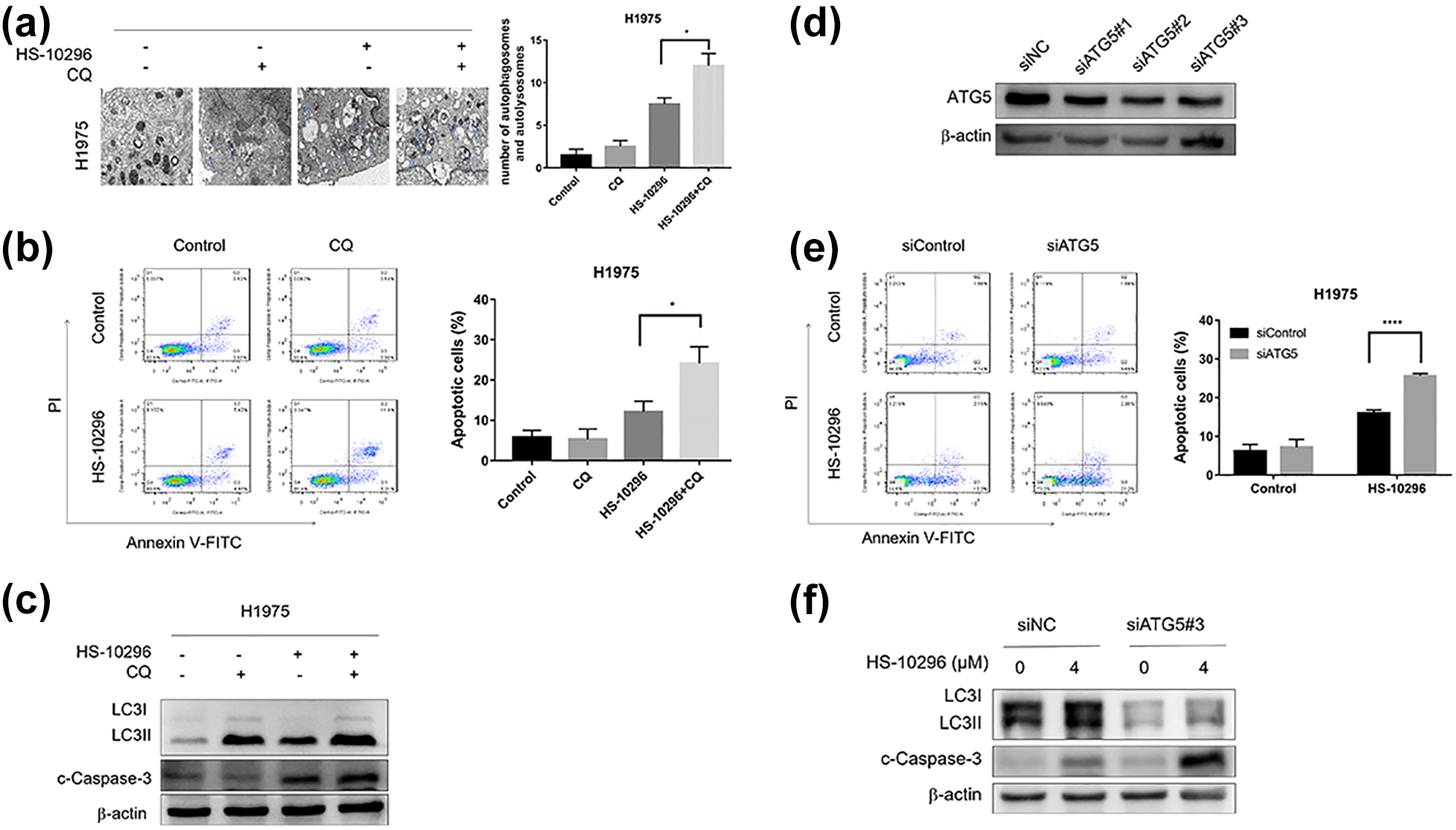
Inhibition of autophagy enhances Almonertinib-induced apoptosis in H1975 cells. Representative transition electron microscopy images of H1975 cells treated with DMSO, CQ, Almonertinib, or a combination of Almonertinib and CQ. Arrows: autophagosomes and autolysosomes. (b) Inhibition of autophagy with CQ enhanced the apoptosis of Almonertinib-treated H1975 cells. (c) Western blotting was performed to analyze the expression of autophagy- and apoptosis-related proteins after the treatment of H1975 cells with Almonertinib with or without CQ. (d) H1975 cells were transfected with ATG5 siRNA, the protein expression levels of ATG5. (e) Inhibition of autophagy by ATG5 downregulation enhanced the apoptosis of Almonertinib-treated H1975 cells. (f) After ATG5 downregulation, the expression levels of cleaved Caspase-3, and LC3 in H1975 cells with or without Almonertinib treatment were determined by western blotting. Data are expressed as the mean ± SEM (*P < 0.05, ****P < 0.0001).
To further confirm the role of Almonertinib-induced autophagy, we inhibited autophagy using genetic tools. H1975 cells were transfected with ATG5 siRNA for 24 h and then treated with or without Almonertinib for another 24 h. As shown in Figure 5(d) and (f), the expression of ATG5 and LC3B were inhibited by ATG5 siRNA, suggesting that autophagy had been inhibited. The group treated with Almonertinib and ATG5 siRNA showed significant significantly (P < 0.05) increased apoptotic cells and cleaved Caspase-3 levels, compared with the group treated with negative control siRNA (Figure 5(e) and (f)). Overall, these results demonstrated that inhibition of autophagy enhance the efficacy of Almonertinib in H1975 cells (Tables 2 and 3).
The apoptosis rate of NSCLC cells induced by different treatment.a

a The data in the table are the percentage of cell population in different times of the Figure 5(b). CQ: chloroquine.
The apoptosis rate of NSCLC H1975 cells induced by different treatment.a
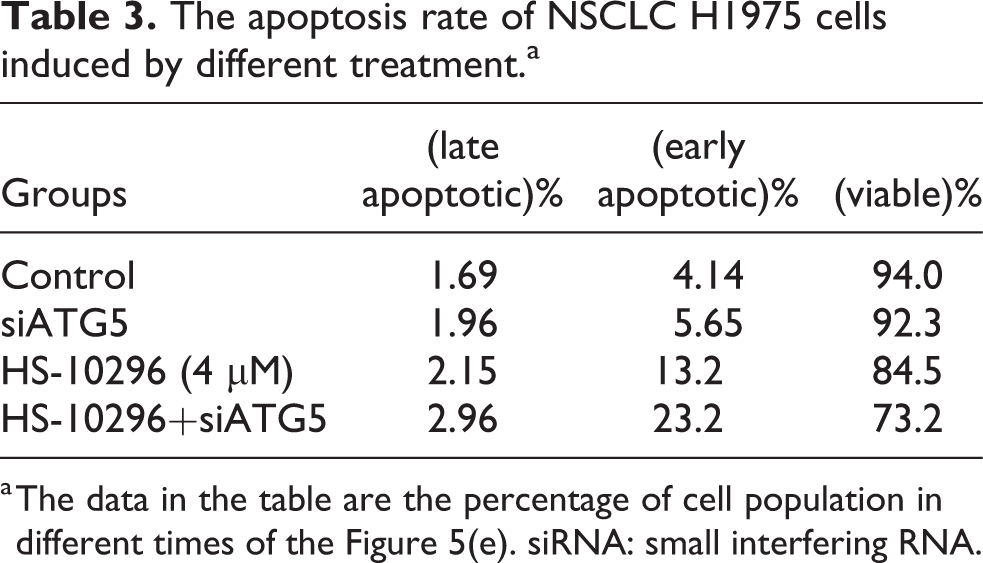
a The data in the table are the percentage of cell population in different times of the Figure 5(e). siRNA: small interfering RNA.
ROS-mediated Almonertinib-induced apoptosis and autophagy in NSCLC cells
To further explore the molecular mechanism of Almonertinib-induced apoptosis and autophagy, we detected the mitochondrial membrane potential (MMP) and intracellular ROS generation in H1975 and HCC827 cells after treatment with Almonertinib using JC-1 and DCFH2-DA probe. As shown in Figure 6(a), with the increase of Almonertinib concentration, red fluorescence gradually converted to green fluorescence, which indicated the decrease of mitochondrial membrane potential, an indicator of ROS generation. And in Figure 6(b), Almonertinib promoted the ROS levels in H1975 and HCC827 cells more intuitively. In addition, ROS generation induced by Almonertinib was reversed partly after pretreatment with NAC (5 mM, 1 h).
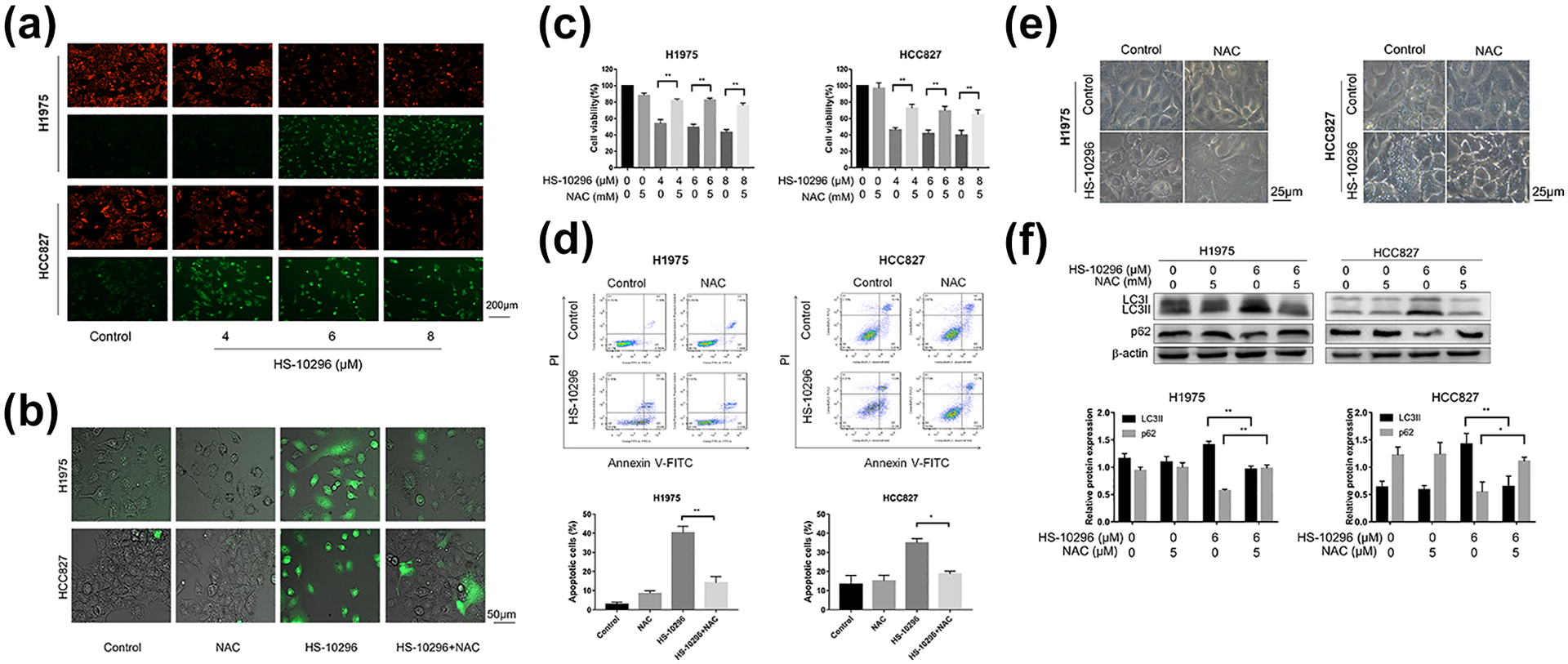
ROS-mediated Almonertinib-induced apoptosis and autophagy in NSCLC cells. (a) H1975 and HCC827 cells were examined by fluorescence microscopy following staining with JC-1. (b) ROS production was monitored using 10 μM DCFH2-DA and then detected by fluorescence microscopy after treatment of cells with Almonertinib (6 μM) and/or NAC (5 mM) for 6 h. (c) After pre-incubation with 5 mM NAC for 1 h, cells were treated with Almonertinib (4, 6, 8 μM) for 24 h. The relative cell viability was assessed with CCK-8 assay. (d) After pre-incubation with 5 mM NAC for 1 h, cells were treated with Almonertinib (6 μM) and/or NAC (5 mM), then stained with Annexin V-FITC/PI and measured using flow cytometry. (e) Cells were incubated with 6 μM Almonertinib for 24 h with or without pretreatment with NAC (5 mM, 1 h), and the formation of cytoplasmic vacuoles was observed by an axiovert 200 invertedmicroscope. (f) The protein expressions of LC3 and p62 were detected by western blot assay and quantitative analysis of proteins. Data are expressed as the mean ± SEM. **P < 0.01 versus control group.
Also, as shown in Figure 6(c) to (f), we found that the Almonertinib induced decrease in cell viability and apoptotic effects in H1975 and HCC827 cells were partly significantly (P < 0.05) reversed after pretreatment with NAC (5 mM, 1 h). In addition, NAC reversed the promotion effect of Almonertinib in cytoplasmic vacuoles formation and LC3-II expression. Taken together, Almonertinib-induced apoptosis and autophagy in NSCLC cells were mediated by ROS (Tables 4 and 5).
The apoptosis rate of NSCLC H1975 cells induced by different treatment.a

a The data in the table are the percentage of cell population in different times of the Figure 6(d). NAC: N-acetyl-L-cysteine.
The apoptosis rate of NSCLC HCC827 cells induced by different treatment.a
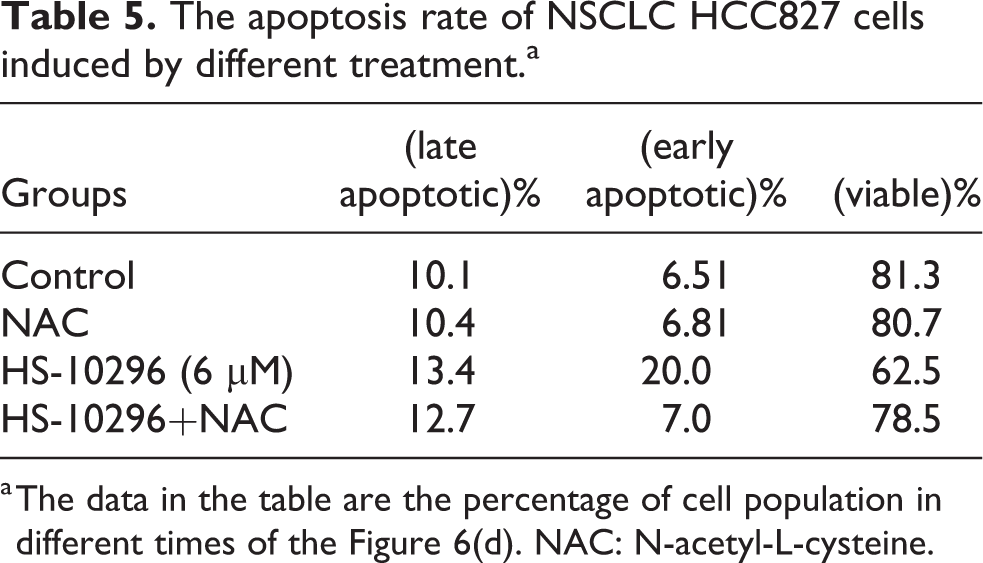
a The data in the table are the percentage of cell population in different times of the Figure 6(d). NAC: N-acetyl-L-cysteine.
Almonertinib markedly depressed EGFR signaling pathways in NSCLC cells
EGFR signaling pathway plays an important role in tumor tumorigenesis and progression, and has been regarded as a critical target for the treatment of NSCLC. In our study, it was apparent that phosphorylated EGFR levels were reduced. The PI3K/Akt and ERK pathway play important roles in promoting the cell survival and were recognized as downstream effector of the EGFR signaling pathway. Therefore, we investigated the effects of Almonertinib on the PI3K/AKT and ERK pathway. As we expected, Almonertinib significantly (P < 0.05) downregulated the expression of phosphorylated Akt and ERK in NSCLC cells (Figure 7).
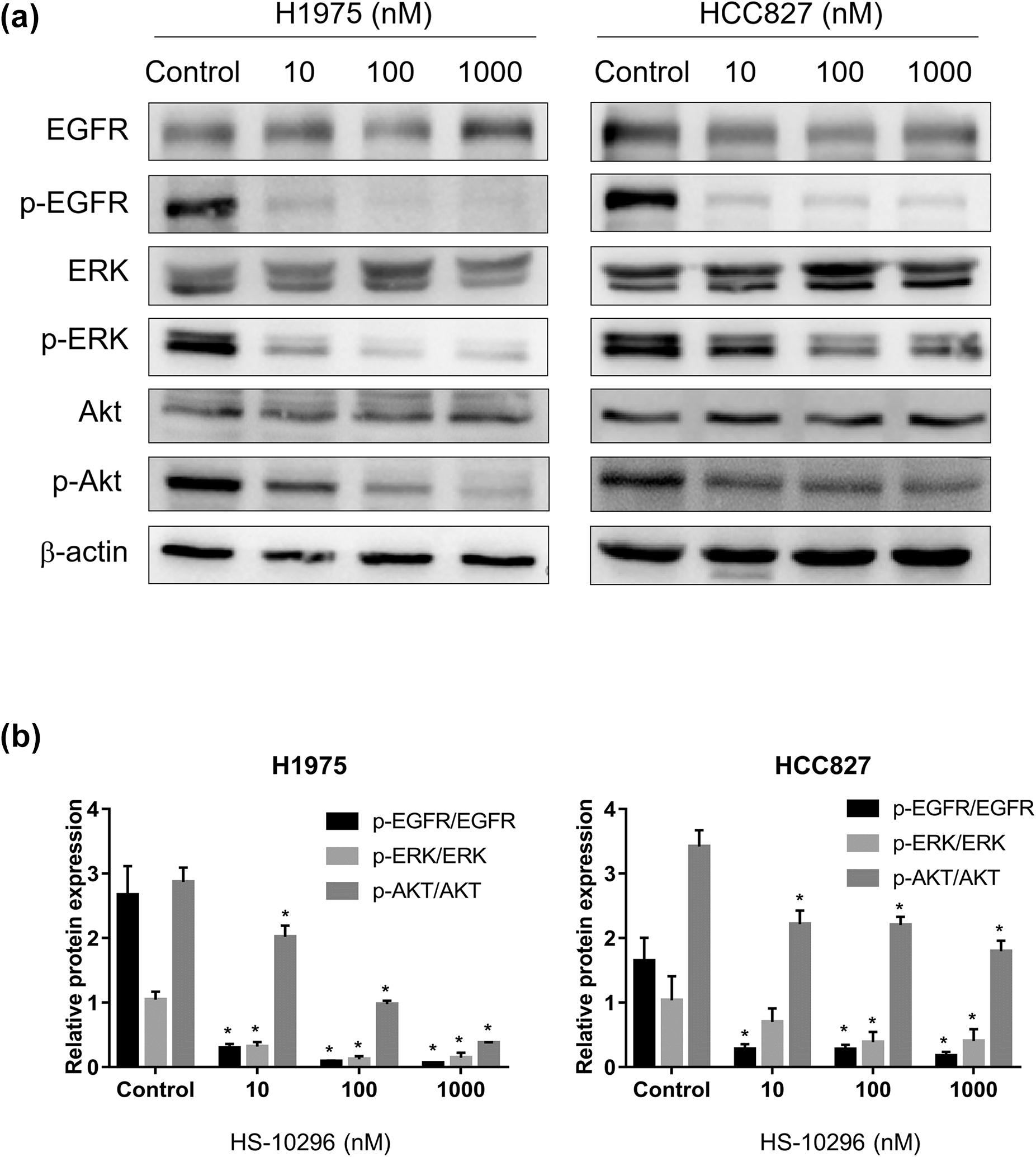
Almonertinib markedly depressed EGFR signaling pathways in NSCLC cells. (a) Effects of Almonertinib on the EGFR signaling pathway were analyzed by Western blotting. (b) Quantitative analysis of proteins. *P < 0.05 versus control group.
Discussion
In recent years, traditional therapies have failed to meet the needs of NSCLC patients and the prognosis of NSCLC remains poor. 26 Targeted therapy provides a new option for NSCLC patients, which possesses good efficacy and low side effects. Targeted drugs for EGFR, such as gefitinib and erlotinib, are used to treat NSCLC patients with EGFR-sensitive mutations.27,28 However, the emergence of primary and acquired resistance limited their efficacy greatly. Among them, the occurrence of acquired drug resistance may be related to the selective secondary mutation of EGFR, and the EGFR mutation p.T790M in exon 20 represents the most frequent mechanisms. 29 Almonertinib is a third-generation EGFR-TKI that has high selectivity on EGFR T790M-mutant NSCLC. Herein, we indicated for the first time that Almonertinib exhibited anti-tumor effects via inducing apoptosis and autophagy dependent on promoting ROS production.
Autophagy is an evolutionary highly conserved cellular process of selfdigestion, which can be regulated by the activation of multiple downstream signaling pathways of EGFR.30,31 Meanwhile, whether EGFR-TKIs-induced autophagy via inhibition of EGFR was controversial. 23 Previously studies have indicated that autophagy inhibition by CQ could enhance the sensitivity of NSCLC cells to erlotinib 32 and gefitinib, 33 and in this report, we had demonstrated that Almonertinib significantly induces apoptosis and autophagy in NSCLC NCI-H1975 and HCC827 cells. And we found that Almonertinib-induced decrease in cell viability and apoptotic effects could be partly reversed after inhibition of autophagy. Therefore, the Almonertinib-induced autophagy contribute to cell survival and the blockade of autophagy could enhance the sensitivity of NSCLC to Almonertinib (Figure 8).

Schematic diagram of the mechanism by which EGFR tyrosine kinase inhibitor Almonertinib induces apoptosis and autophagy via generation of reactive oxygen species in non-small cell lung cancer cells. Almonertinib inhibits the activity of EGFR, leading to the excessive production of intracellular ROS, inducing apoptosis and autophagy, which, however, plays a role in promoting cell survival.
ROS is a series of oxygen-containing and active species that plays a critical effect on a series of cellular programs, including autophagy and apoptosis, etc. 19 However, previous studies showed that EGFR-TKIs such as gefitinib and erlotinib-induced G1 phase cell cycle arrest through inhibition of EGFR pathway.34,35 Our work demonstrated that Almonertinib significantly elicited ROS generation while inhibition of ROS production reversed the Almonertinib-induced apoptosis and autophagy in the NSCLC NCI-H1975 cells. These results indicated that apoptosis and autophagy were triggered by ROS that induced by Almonertinib. Almonertinib, as a EGFR-TKI, inhibited the growth and proliferation of cells by inhibiting the activity of EGFR, which should be the most ideal effect. Previous report indicated the generation of ROS might be the “off-target” effect of Almonertinib with high concentration. 24 However, ROS generation might be a stimulus response caused by inhibition of EGFR downstream signal transduction. 36 Therefore, Almonertinib-induced generation of ROS was still the result of targeting EGFR. The accumulation of intracellular ROS may be due to the increase of ROS production from mitochondrial respiration, nicotinamide adenine dinucleotide phosphate oxidase and ER stress, etc. or decreasing scavenging capacity of ROS in cancer cells. 37 Thus, further studies need to determine the sources and functions of ROS.
We also demonstrated a significant decrease in EGFR activity and its downstream signaling pathways such as ERK and AKT following treatment with Almonertinib. The ERK and PI3K/AKT cascades were major signaling networks triggered via EGFR-activation, which involved genetic alterations in regulatory proteins and were closely related with tumorigenesis, cell survival, proliferation, etc. 38 However, more researches are needed to discover its effect on apoptosis and autophagy induced by Almonertinib.
In summary, the results from our studies indicated that Almonertinib effectively inhibit the growth of NSCLC cells harboring EGFR T790M mutation and EGFR-sensitive mutation, and induced apoptosis as well as autophagy while the generation of ROS plays an important role. Importantly, Almonertinib-induced autophagy was cytoprotective in NSCLC cells, and the blockade of autophagy improved apoptosis. All of that indicated that autophagy was a new molecular therapeutic target in NSCLC. Combination treatment with Almonertinib and an autophagy inhibitor may be a useful therapeutic strategy for NSCLC treated with EGFR-TKI. Our findings indicated that Almonertinib was an effective anticancer agent for the treatment of NSCLC patients with EGFR T790M mutation. These findings may contribute to design novel strategies for NSCLC patients who are resistant to first- and second-generation EGFR-TKIs.
Supplemental material
Supplemental Material, sj-pptx-1-het-10.1177_09603271211030554 - EGFR tyrosine kinase inhibitor Almonertinib induces apoptosis and autophagy mediated by reactive oxygen species in non-small cell lung cancer cells
Supplemental Material, sj-pptx-1-het-10.1177_09603271211030554 for EGFR tyrosine kinase inhibitor Almonertinib induces apoptosis and autophagy mediated by reactive oxygen species in non-small cell lung cancer cells by X Ge, Y Zhang, F Huang, Y Wu, J Pang, X Li, F Fan, H Liu and S Li in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Anhui Science and Technology Major Project (201903a07020029), National Major New Drug creation Science and Technology Project (2019ZX09303001), Anhui Innovation and Entrepreneurship leading talents Special support Program Project (2016) and Anhui Education Department key Project (KJ2018A0238), Anhui Natural Science Foundation (1908085QH373). Anhui Provincial Key Research and Development Project (202104g01020017)
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
