Abstract
Fibrosis is a common outcome of nearly all chronic diseases of liver that results in changes of its functions which requires medical attention. The current research aims to investigate the potential anti-fibrotic efficacy of Carvacrol against thioacetamide (TAA)-induced liver fibrosis in male rats using Ursodeoxycholic acid (UDCA) as a reference anti-fibrotic product. Carvacrol (25 and 50 mg/kg) markedly declined TAA-increased serum liver enzymes; alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP) and gamma-glutamyl transferase (GGT) as well as total bilirubin (TB) and direct bilirubin (DB) levels as well as increased levels of total protein (TP) and albumin. Carvacrol significantly reduced glutathione depletion (GSH), Nitric oxide (NOX) and malondialdehyde (MDA) accumulation in liver tissue. Additionally, its anti-oxidant effect brightened up via affecting markers of stress found in the cell as nuclear factor erythroid 2-related factor 2 (Nrf-2) where it still had high content and decreased Thioredoxin (Trx) level. The anti-inflammatory effect of Carvacrol was confirmed by decreasing nuclear factor kappa B (NF-κB), interleukin-1beta (IL-1β) and inducible nitric oxide synthase (iNOS) contents. Carvacrol showed anti-fibrotic effect clarified by turning down fibrosis-related markers; TGF-β1, matrix metalloproteinase-3 and 9 (MMP-3 and 9) and Autotaxin (ATX) contents. Furthermore, it decreased alpha smooth muscle actin (α-SMA) and caspase-3 immune-expression. The overall outcome of aforementioned markers results showed that Carvacrol suppresses the progression of liver fibrosis via its anti-oxidant, anti-inflammatory, anti-apoptotic effect and its ability in lowering Thioredoxin and Autotaxin; hence it can be categorized as a hepatoprotective natural substance.
Introduction
Therapies for liver fibrosis-related diseases have been progressed, such as those for viral hepatitis, but there are currently no approved fibrosis therapies available; as reversal of fibrosis is considered a lengthy and expensive process. 1 The past two decades have seen remarkable progress in understanding hepatic fibrosis, which has led to promising new advances in chronic liver disease diagnosis and treatment2,3
Scar formation and replacement of liver parenchymal tissue with fibrotic one is the leader mark of hepatic fibrosis that comes from chronic liver injury of any etiology.4,5 Fibrotic tissue consists of collagen accumulation after chronic inflammation that is powered by a cascade of events involving cytokines production by both circulating immune cells and hepatic stellate cells (HSCs) which are considered the main producers of that extracellular material of the fibrotic tissue.6,7 Activation and proliferation of HSCs are mediated by reactive oxygen species (ROS) and inflammatory cytokines. Activated HSCs promotes the expression of fibrotic biomarkers like (α-SMA) and (ATX) due to transformation of HSCs into myofibroblastic cells that secrete the large amounts of collagen, thereby producing liver fibrosis.2,8

The growing demand for natural and nutritious foods without chemical additives stimulates many researchers to investigate the hepatoprotective effects of natural compounds, such as naturally occurring antioxidants that cause a range of biological activities. 9 As ROS are activation candidate driver for HSCs and up-regulator of collagen, the anti-oxidant supplements can emerge as potentially anti-fibrotic agents either by protecting hepatocytes from ROS, or by inhibiting hepatic stellate cell activation.10,11
Carvacrol is a common monoterpene phenol that is present in several significant oil species. It has a distinctive hot, mild aroma of oregano and a pizza-like taste. 12 Carvacrol-rich essential oils are well-known to have strong antioxidant properties comparable to ascorbic acid, butyl hydroxyl toluene and vitamin E. It has been recognized for several therapeutic effects, including anti-inflammatory, anti-proliferative and anti-carcinogenic effects. Moreover, Carvacrol was reported to have a protective effect against hepatic cirrhosis induced by carbon tetrachloride in rats,13,14 and to attenuate hepatotoxicity, oxidative stress and, inflammation induced by thioacetamide in rats. 15
Although Silymarin has shown a prominent hepatoprotective effect, it has been reported that it may cause gastrointestinal disorders such as nausea and diarrhea. 16 Ursodeoxycholic acid (UDCA) is a secondary bile acid (metabolic byproduct of intestinal bacteria) and possesses anti-oxidant forces. UDCA is approved by the Food and Drug Administration for the treatment of fibrosis in chronic cholestatic disorders and treatment of biliary cirrhosis. 17 It improved liver function in many biliary disorders and preserved hepatocytes from amoxicillin-clavulanic acid-induced toxicity in rats, due to its anti-oxidant properties. 18 UDCA has been used as a standard drug to assess the anti-fibrotic effect of various therapeutics and natural products.19,20
Thioacetamide (TAA) is a well-established hepatotoxin. TAA undergoes bioactivation process which is catalyzed by CYP450 isoenzymes, with consequent production of sulphur dioxide leading to generalized hepatotoxicity, hepatic tissue necrosis and fibrosis. Induction of liver fibrosis in rats by TAA could be used as a reliable model, which mimics human chronic hepatic disease accompanied by toxic damage.21,22
Hence, in the current research we aimed to evaluate the probable anti-fibrotic effect of Carvacrol on TAA-induced hepatic fibrosis and the probable mechanistic pathway underlying such condition including autotaxin and thioredoxin modulation.
Materials and methods
Animals
Adult male Wistar albino rats, weighing 200–250 g, at age of 4 months were obtained from animal house colony of National Research Centre, Giza, Egypt. All animals were housed in metal cages at (22 ± 3°C, 55 ± 5 percent humidity and 12 h dark & light cycles) in a well-ventilated setting. Rats received commercial standard rat food pellets (≥21% proteins, ≥3.48% fats, ≤3.71% raw fiber and 1% multivitamins) and offered water ad libitum.
Ethical statement
In keeping with the guidelines of the National Institutes of Health (NIH publication No. 85-23, updated 2011) and in accordance with the Ethics Committee of the National Research Centre, Egypt (registration number 18/146), the study protocol and procedures were enforced.
Drugs
TAA (El-Gomhouria Company for drug and chemicals, Cairo, Egypt) was freshly prepared prior to administration. Carvacrol (Sigma Aldrich, Missouri, USA) was freshly prepared prior to administration by dissolving in 0.9% (w/v) saline solution. UDCA (MEPACO, Cairo, Egypt) as a fine powder and stored in an airtight container in a refrigerator below 10°C and was freshly prepared prior to administration.
Experimental design
Rats have been assigned to five groups of six rats each. Group 1 (control group); rats were intra-peritoneally (i.p) injected with 0.2 ml sterile saline twice weekly (days 2 and 6). Group 2 (TAA group); rats were i.p injected with freshly prepared TAA (200 mg/kg b.w.) in 0.2 ml sterile saline twice weekly (days 2 and 6) for 6 consecutive weeks 23 for liver fibrosis induction. Groups 3; rats received UDCA orally as 20 mg/kg b.w. twice weekly, 18 concurrently with TAA. Group 4 and 5; rats received daily oral dose of Carvacrol at doses of 25 and 50 mg/ kg/day, respectively, 15 concurrently with TAA twice weekly. Treatments in groups 3, 4 and 5 were administrated 1 h prior to TAA administration, at days 2 and 6 and continued for 6 weeks.
Blood and tissue samples
At the end of the experimental period, blood samples were collected from the retro-orbital venous plexus in clean test tubes under anesthesia using ketamine (Sigma-Aldrich, USA), 50 mg/kg i.p injected. Then rats were sacrificed by decapitation and their livers were immediately removed and divided into two parts, one part was preserved in 10% buffered neutral formalin and the other one was kept frozen at −80°C until further investigations.
The collected blood samples were allowed to clot, and then centrifuged at 3000 r.p.m. for 20 min, the obtained serum samples were held at −20°C for further use.
Biochemical analysis
Serum activities of ALT, AST, ALP and GGT were determined colorimetrically using kits of Spectrum® according to the method of Reitman and Frankel, 24 Bergmeyer et al, 25 Szasz. 26 Serum levels of; total and direct bilirubin, 27 total protein 28 and albumin 29 were determined using kits of Spectrum® for the colorimetric determination.
Determination of serum thioredoxin (Trx) level
Serum level of Trx was measured using ELISA kit (Cloud-Clone Corp., Houston, TX, USA) according to the manufacturer’s procedure.
Preparation of hepatic tissue homogenate
Liver specimens were weighed and homogenized in 0.15 M phosphate buffer. The aliquot was centrifuged using a cooling centrifuge (2k15; Sigma/Laborzentrifugen) at 1500×g at 4°C for 15 min and the supernatant was collected for direct assessment of biochemical parameters.
Determination of oxidative stress status and nitrosative stress in liver homogenate
Malondialdehyde (MDA), a lipid peroxidation marker, 30 Glutathione (GSH) content 31 and Nitric oxide (NOX), 32 were all determined colorimetrically in liver homogenate using kits of Bio Diagnostic Company, Egypt.
Determination of Nrf-2
Nuclear factor erythroid 2-related factor 2 (Nrf-2) was determined by using ELISA kits of Elabscience Biotechnology Co., Ltd, USA.
Determination of inflammatory markers
Liver contents of NF-κβ, IL-1β and iNOS were qualified using ELISA kits (Elabscience® Biotechnology Co., Ltd, USA), (cohesion Biosciences, Chinas) and (Cloud-Clone Corp., Houston, TX, USA), respectively according to the manufacturer’s instructions.
Determination of fibrosis markers
Hepatic contents of TGF-β1, MMP-3 and 9, and Autotaxin (ATX) were all determined in hepatic tissue homogenate using ELISA kits (Cell science, Canton, MA, USA), (Cloud-Clone Corp., Houston, TX, USA), respectively according to the manufacturer’s procedures.
Histopathological examination
After 24 h fixation in 10% buffered neutral formalin, autopsy specimens of liver of rats of different groups were routinely processed for obtaining paraffin sections. After washing in tap water, the specimens were dehydrated in serial dilutions of ethyl alcohol then they were cleared in xylene and finally embedded in paraffin. Paraffin blocks were sectioned at 4–5 µm thickness. The obtained tissue sections were collected on glass slides, de-paraffinized, and stained with hematoxylin and eosin (H&E) as well as Masson’s Trichrome (MTC) stains. 33 Metavir scoring system was used for semi-quantitative categorization of liver fibrosis extension, where, a scale of 0–4 of fibrosis was arranged as follows: F0: no fibrosis; F1: portal fibroplasia without portal septa; F2: portal fibroplasia with few septa extensions; F3: multiple septa without apparent cirrhosis; and F4: cirrhosis without apparent cirrhosis. 34
Immunohistochemical studies
Immunohistochemical studies were carried out for detection of caspase-3 and α-SMA expression on paraffin sections of liver of control and all treated groups using avidin-biotin peroxidase (DAB, Sigma Chemical Co.) according to method described by Ali et al. 35 Tissue sections were incubated with a monoclonal antibody for caspase-3 and α-SMA (1:100 and 1:200 dilutions respectively; (Abcam, Cambridge, MA, USA, ab4051 and ab7817), and reagents required for the avidin-biotin peroxidase (Vactastain ABC peroxidase kit, Vector Laboratories) method for detection of the antigen–antibody complex. Each marker expression was visualized by the chromagen 3,3-diaminobenzidine tetra hydrochloride (DAB, Sigma Chemical Co.). Quantification of the positive brown area of each marker’s expression was implemented as an optical density in 7 high power microscopic fields using image analysis software (Image J, 1.46a, NIH, USA).
Statistical analysis
All outcomes were expressed as mean ± SE. Data analysis was accomplished by one-way variance analysis (ANOVA) followed by a multiple comparison test by Tukey post-hoc test using the Graph Pad Prism software program (version 7.00). At p < 0.05, the difference was considered significant. The frequency data for nonparametric analysis was carried out via Kruskal wallis H test for comparing followed by the Mann-Whitney U test, and the nonparametric data were presented as median.
Results
Carvacrol ameliorated liver function markers; ALT, AST, ALP, GGT, total and direct bilirubin, albumin and total protein
Results in Table 1 revealed that TAA intoxicated group significantly elevated liver enzymes ALT, AST, ALP and GGT levels by 1.41, 1.47, 2.27, and 1.60 fold, respectively when compared to the control group. UDCA treatment significantly reduced serum ALT, AST, ALP, and GGT by 38.11%, 27.07%, 44.11%, and 33.22%, respectively as compared to TAA group. While treatment with Carvacrol at doses of 25 and 50 mg/kg b.wt showed a dose related significant decrease in serum levels of ALT, AST, ALP, and GGT levels by 39.97, 32.82%, 32.35, 25.28%, 50.27, 49.06%, and 30.91, 27.43%, respectively for the four enzymes as compared to the control TAA group.
Effect of Carvacrol on liver enzymes in TAA-induced liver fibrosis in rats.

One-way ANOVA was used to conduct statistical analysis, followed by Tukey’s multiple comparison studies. Mean ± SEM (n = 6) expressed the results. Using symbol * to express the significant difference from normal group and symbol # to express the significant difference from control group at P < 0.05.
Results in Table 2 revealed that TAA group showed significant increase in serum total and direct bilirubin levels by 1.73 and 1.82 fold, respectively and significant decreased in serum albumin and total protein levels by 1.48 and 1.4 fold, respectively when compared to the normal control group. While the UDCA treated group showed significant reduction in serum total and direct bilirubin levels by 37.25, 41.93%, respectively with significant increase in serum albumin and total protein levels by 32.26, 20.49%, respectively as compared to TAA group. Similarly, treatment with 25 and 50 mg/kg b. wt. Carvacrol induced a significant decrease in serum total bilirubin level by 39.21, 18.62%, a significant decrease in serum direct bilirubin level by 41.93, 25.81%, a significant increase in serum albumin level by 59.91, 51.15% and a significant increase in serum total protein level by 46.66, 36.29% as compared to control TAA group.
Effect of Carvacrol on liver bilirubin and proteins in TAA-induced liver fibrosis in rats.

One-way ANOVA was used to conduct statistical analysis, followed by Tukey’s multiple comparison studies. Mean ± SEM (n = 6) expressed the results. Using symbol * to express the significant difference from normal group and symbol # to express the significant difference from control group at P < 0.05.
Carvacrol improved oxidative stress markers
I/p injection of TAA twice weekly for 6 weeks significantly increased liver contents of MDA, NOx and serum Trx level by 2.28, 1.49 and 2.21 fold, respectively and significantly decreased in liver content of GSH by 2.43 fold when compared to the normal group. Ursodeoxycholic acid significantly reduced liver content of MDA, NOx and serum Trx level by 54.88, 30.1% and 45.2%, respectively and significantly increased liver content of GSH by 86.67% as compared to the corresponding TAA group. Treatment with 25 and 50 mg/kg b.wt. of Carvacrol showed a significant decrease in MDA liver content by (38.49, 43.03%), a significant decrease in NOx liver content by 32.1, 34.34%, a significant decrease in serum Trx level by 38.89, 59.26% and a significant increase in GSH liver content by 40.83, 56.66% respectively as compared to control TAA group (Table 3).
Effect of Carvacrol on oxidative stress markers in TAA and various treated groups.

One-way ANOVA was used to conduct statistical analysis, followed by Tukey’s multiple comparison studies. Mean ± SEM (n = 6) expressed the results. Using symbol * to express the significant difference from normal group and symbol # to express the significant difference from control group at P < 0.05.
Carvacrol suppressed Nrf-2 and inflammatory markers; NF-κβ, IL-1β and iNOS
A significant decrease in hepatic Nrf-2 content with a significant increase in hepatic contents of; NF-κβ, IL-1β and iNOS was noticed in TAA group compared to the control group. On contrary, UDCA administration, significantly (P < 0.05) increased Nrf-2 and significantly decreased the levels of NF-κβ, IL-1β and iNOS in hepatic tissue homogenate as compared to TAA group. While, Carvacrol treatment significantly (P < 0.05) elevated liver content of Nrf-2 as well as significantly lowered the hepatic contents of NF-κβ, IL-1β and iNOS, in a dose related response as compared to TAA group (Figure 1).
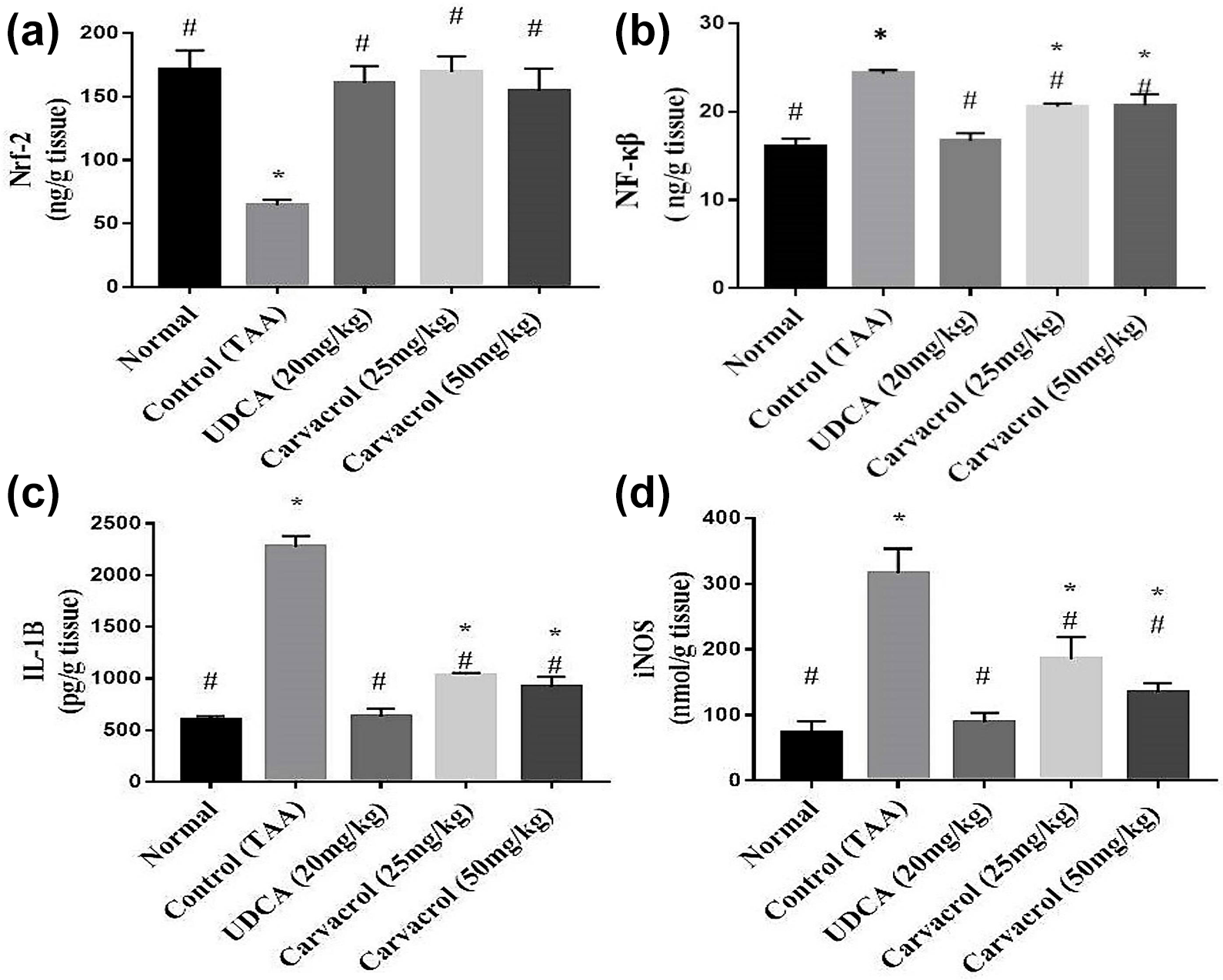
Effect of Carvacrol on Nrf-2, NF-κβ, IL-1B and iNOS contents in TAA-induced liver fibrosis in rats. One-way ANOVA was used to conduct statistical analysis, followed by Tukey’s multiple comparison studies. Mean ± SEM (n = 6) expressed the results. Where * represents the significant difference from control normal group and # represents the significant difference from control group at P < 0.05.
Carvacrol mitigated hepatic fibrosis markers; TGF-β1, MMP-3, MMP-9 and ATX contents
TAA administrated rats’ revealed significant (P < 0.05) increase in hepatic content of fibrosis markers; TGF-β1, MMP-3 MMP-9 and ATX when compared to control group. Treatment with UDCA showed a significant (P < 0.05) decrease in hepatic TGF-β1, MMP-3 MMP-9 and ATX contents. Similarly, treatment with Carvacrol at doses of 25 and 50 mg/kg. b.wt showed a dose related significant amendment decrease in liver content of TGF-β1, MMP-3 MMP-9 and ATX when compared to TAA group (Figure 2).
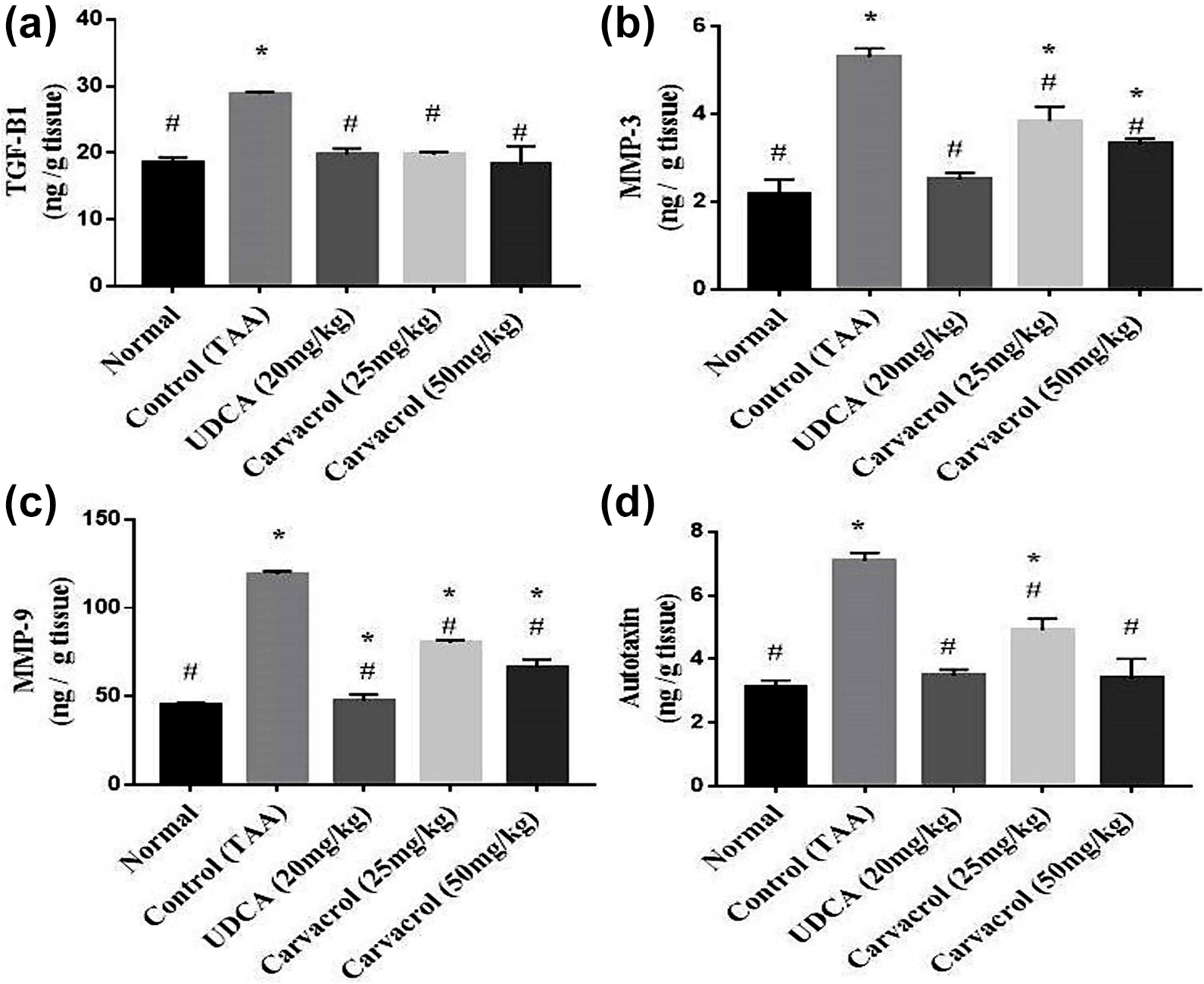
Effect of Carvacrol on hepatic contents of TGF-β1, MMP-3 MMP-9 and ATX in TAA-induced liver fibrosis in rats. One-way ANOVA was used to conduct statistical analysis, followed by Tukey’s multiple comparison studies. Mean ± SEM (n = 6) expressed the results. Where, * to express the significant difference from normal control group and # to express the significant difference from control group at P < 0.05.
Results of histopathological examination
Examination of liver of control rats showed normal histological structure of portal areas, central veins and hepatic parenchymal cells (Figure 3(a)), while livers of TAA administrated rats showed marked histological alterations. The portal tracts showed; vascular congestion, proliferation of bile duct epithelium, inflammatory cells infiltration and marked fibrous proliferation (Figure 3(b)) which showed peripheral extension toward the parenchyma as fibrous bands that resulted in parenchymal pseudolobulation (Figure 3(c)). The hepatic cells within those pseudolobules showed vacuolar degeneration with eccentric nuclei and sometimes cytoplasmic reticulation. Scattered apoptosis as well as inflammatory infiltrates along the fibrous septa were clearly seen (Figure 3(d)).

H&E stained liver sections. (a) Liver of control rat shows normal central vein and hepatic parenchymal cells. (b-) liver of TAA-administrated rat showing; (b) portal triad reaction as congestion (Co), inflammatory cells infiltration (arrow), fibrous proliferation and proliferated bile duct epithelium (dashed arrow), (c) peripheral extension of fibrous septa (arrow) toward the parenchyma with a resultant parenchymal pseudolobulation (PL). (d) Vacuolar degeneration of the hepatic cells within the pseudo-lobules (upper corner). (e and f) Liver of rats administrated UDCA showing moderate degree of fibrous proliferation within the portal areas with peripheral extension of incomplete septa (arrow) accompanied with mild inflammatory cells infiltration and proliferated bile duct epithelial cells. (g–i) Liver of rat administrated Carvacrol (25 mg/kg. b.wt) showing retraction of fibrotic reaction in the portal areas (arrow) without any parenchymal fibrosis as well as scars hepatocellular necrobiotic changes. (j–l) Liver of Carvacrol (50 mg/kg. b.wt) administrated rat showing mild fibroplasia in the portal areas and peripheral extension of thin incomplete septa (arrow) with mild hepatocellular degeneration.
Livers of rats administrated UDCA showed moderate degree of fibrous proliferation within the portal areas with peripheral extension of incomplete septa (Figure 3(e)) accompanied with mild inflammatory cells infiltration and scattered proliferated bile duct epithelial cells (Figure 3(f)). The hepatic cells showed mild degree of degenerative and necrotic changes. The administration of Carvacrol had more curative effect than that observed on using the standard drug particularly at the low dose, followed by the higher dose. In regards to, livers of rats administrated Carvacrol in a the low dose (25 mg/kg. b.wt) revealed near to normal appearance of the portal areas (Figure 3(g)) with minimal changes as; mild inflammatory reaction and bile duct proliferation without any evidence of fibrosis (Figure 3(h)) accompanied with mild degree of hepatocellular vacuolar degeneration (Figure 3(i)). while, livers of Carvacrol administrated rats at the high dose (50 mg/kg. b.wt), showed minimal portal tracts reaction (Figure 3(j)) with peripheral extension of thin incomplete septa toward the neighboring areas (Figure 3(k)). The hepatic cells in the vicinity showed moderate degree of vacuolar degeneration, scattered necrosis and apoptosis (Figure 3(l)). MTC-stained sections of various groups were used to evaluate fibrous tissue proliferation within the hepatic parenchyma and were quantified by image analysis software as an area percentage (Figure 4(a) to (f)). The highest significant area of fibrosis was observed in TAA group. While, administration of UDCA and Carvacrol showed significant decrease in fibroplasia extension compared with TAA group. The Metavir scoring (Figure 4(g)) of fibrous tissue extension was matching the results of the MTC-based area percent of fibrosis.

MTC-stained liver sections. (a) Control group, normal limited amount of fibrous tissue, (b) TAA group, extensive fibroplasia with bridging fibrosis, (c, d and f) UDCA and (25 and 50 mg/kg. b.wt) administered groups respectively, showing significant decreased parenchymal fibroplasia, (f) fibrosis extension expressed as area percent, (g) the Metavir scoring of fibous tissue extension in hepatic parenchyma of different groups. One-way ANOVA was used to conduct statistical analysis, followed by Tukey’s multiple comparison studies. Mean ± SEM expressed the results. Where * represents the significant difference from control normal group and # represents the significant difference from control group at P < 0.05.
Results of immunohistochemistry analysis
Livers of control rats showed mild normal α-SMA gestures around the portal and central veins where the existence of myofibroblasts (Figure 5(a)). Livers of TAA administrated rats revealed marked increased α-SMA expression in the portal areas, along the extended fibrous septa (Figure 5(b)), pericellular and in the perisinusoidal spaces. While, UDCA administration showed moderate immunoexpression of α-SMA in the portal areas and along the few incomplete septa (Figure 5(e)). The administration of Carvacrol revealed decreased expression of α-SMA particularly at the low dose level (Figure 5(d) and (e)). The quantitative analysis of the area percent of the positive brown color of α-SMA presented as the optical density revealed significant (P < 0.05) increased expression in TAA administrated group compared to that of the other treated groups (Figure 5(f)).
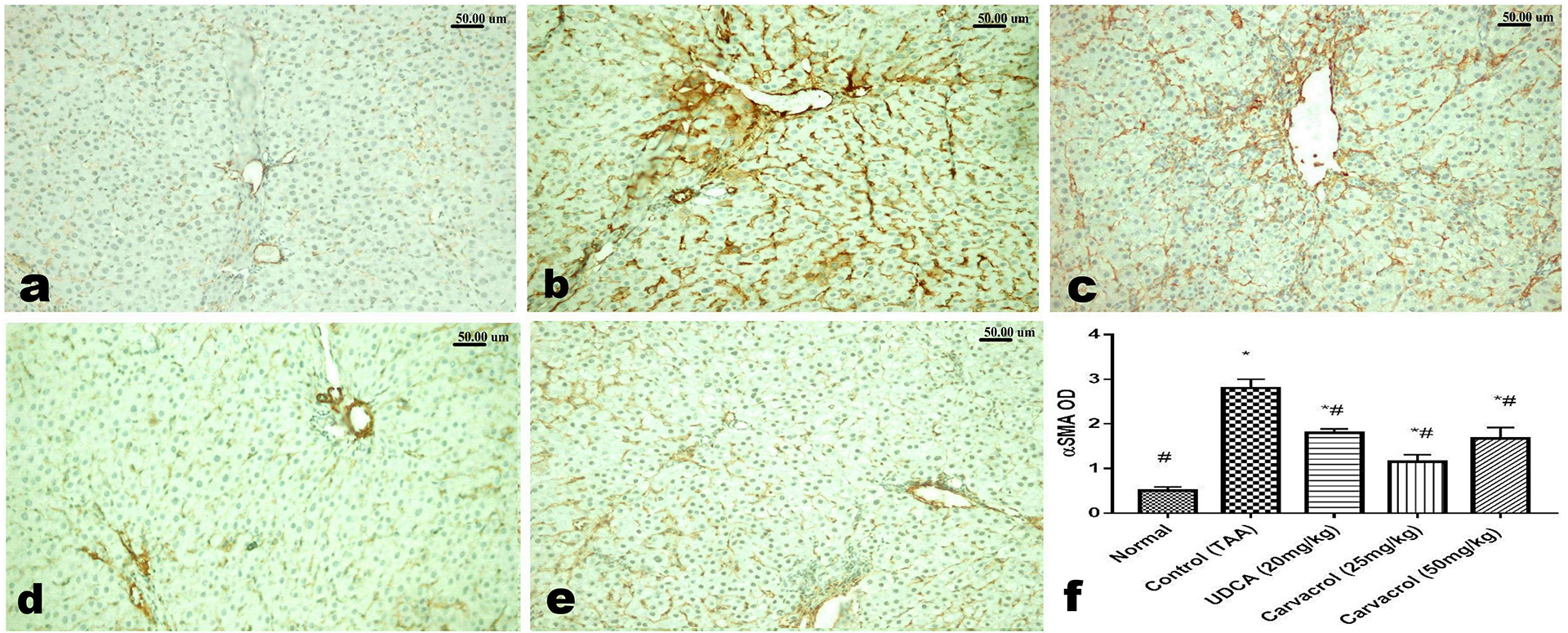
Immunohistochemical analysis of α-SMA. (a) normal mild expression of α-SMA around portal vein, (b) increased expression of α-SMA in the portal areas, along the fibrous septa, pericellular and perisinusoidal. (c) Moderate immunoexpression of α-SMA in the portal areas and along the few incomplete septa in liver of UDCA administer rat. (d and e) Decreased expression of α-SMA in livers of rats treated with low (d) and high (e) doses of Carvacrol. (f) Quantitative image analysis of the area percent of the positive brown color of α-SMA presented as the optical density. One-way ANOVA followed by Tukey’s multiple comparison tests were used for carrying out statistical analysis. Mean ± SEM (n = 6) expressed the results. Using symbol * to express the significant difference from normal group and symbol # to express the significant difference from control group at P < 0.05.
Regarding the caspase-3 stained sections, livers of control rats showed negative expression (Figure 6(a)) of caspase-3, while those of TAA-administrated rats showed widespread expression of caspase-3 in the hepatic cells (Figure 6(b)). Livers of UDCA administrated rats showed moderate expression of caspase-3 particularly among the myofibroblast cells (Figure 6(d)). Decreased caspase-3 expression was noticed in livers of rats treated with the low (Figure 6(d)) and the high (Figure 6(e)) doses of Carvacrol. The area percent of the positive brown color of caspase-3 presented as optical density revealed significant (P<0.05) increased expression in TAA administrated group compared to that of the other treated groups (Figure 6(f)).
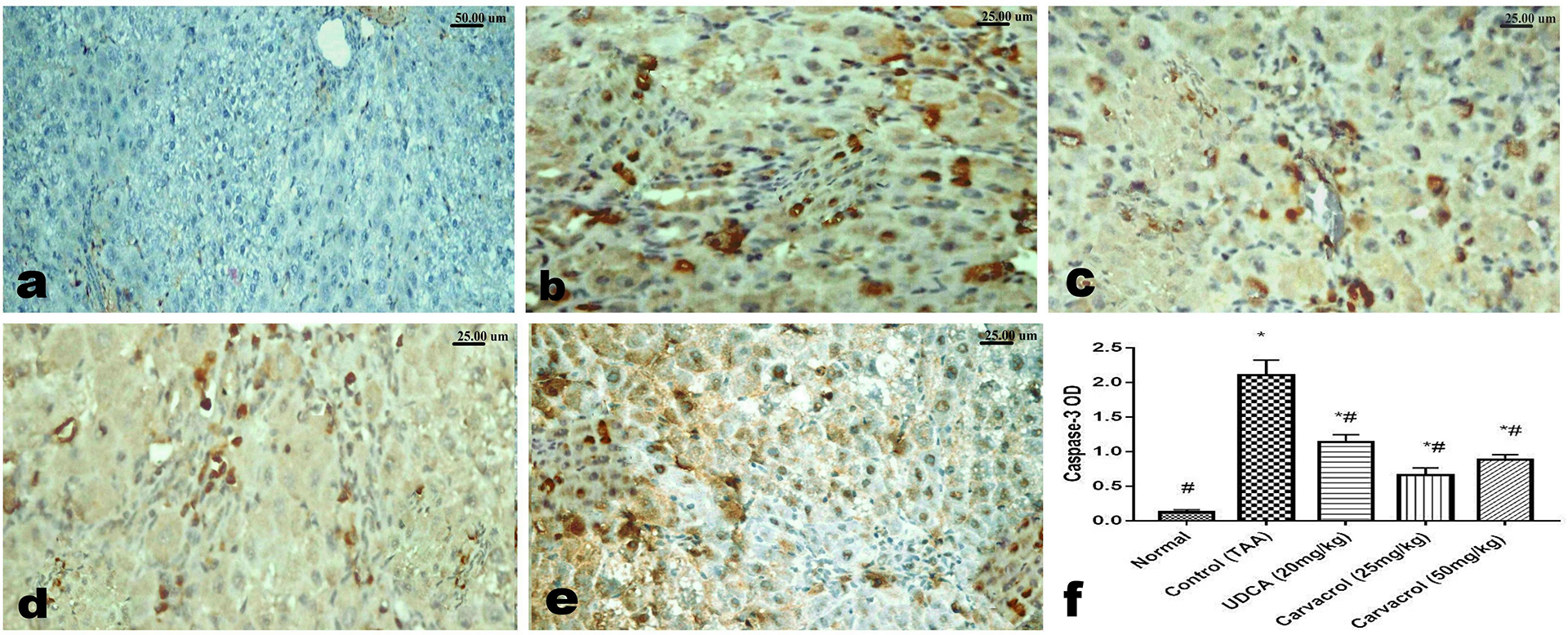
Immunohistochemical analysis of caspase-3. (a) liver of control rat showing negative caspase-3 expression (b) liver of TAA-administrated rat showing marked caspase-3 expression. (c) Moderate expression of caspase-3 in liver of UDCA administrated rat. (d and e) decreased caspase-3 expression in the in livers’ of rats treated with low (d) and high (e) doses of Carvacrol.(f) Quantitative image analysis of the area percent of the positive brown color of caspase-3 in 5 microscopic fields presented as optical density. One-way ANOVA followed by Tukey’s multiple comparison tests were used for carrying out statistical analysis. Mean ± SEM (n = 6) expressed the results. Using symbol * to express the significant difference from normal group and symbol # to express the significant difference from control group at P < 0.05.
Discussion
The current work offers a new viewpoint on the hepatoprotective effects of Carvacrol (25 and 50 mg/kg) on hepatic fibrosis induced by TAA in rats. To the best of the authors’ knowledge, this is the first study to investigate the role of liver enzymes, oxidative stress, inflammation, MMPS, apoptosis, fibrotic biomarkers and histopathological changes in the hepatoprotective role of carvacrol on TAA-induced liver fibrosis. Carvacrol exists as a component in many essential oils obtained from plants and is used in food industry as a preservative.
In the present investigation; TAA administration disrupted the cellular environment which directly led to a significant elevation in serum ALT, AST, ALP and GGT levels as compared to controls, indicating hepatocellular necrosis, mitochondrial damage and changes in hepatocyte membrane permeability, with leakage of hepatocellular enzymes into the circulation. This finding is in harmony with prior studies of Czechowska et al., 36 In addition, TAA administration resulted in a significant rise in serum bilirubin and significant decreased in both total protein and albumin levels as compared to control, which suggest hepatobiliary damage, increase in erythrocyte degeneration rate, and hypoproteinemia due to inflammatory reaction and impaired protein synthesis as a result of liver fibrosis Kadir et al., 37 The later results come in parallel with the histopathological observations which clearly demonstrated that the i.p. injection of TAA twice weekly resulted in distorted liver architecture, marked inflammatory cells infiltration and fibrous tissue proliferation, which is in line with prior study of Algandaby et al., 38 During TAA metabolism, the formation of reactive oxygen species result in marked oxidative stress, manifested by a substantial increase in the liver content of lipid peroxidation end product, MDA and NOx, which is in agreement with other studies.39,40
Carvacrol administration, significantly decreased the levels of the previous enzymes, substantially decreased serum total bilirubin and direct bilirubin levels, overcame the reduction in total protein and albumin levels and had a significant reduction effect on MDA and NOx contents, those findings are in agreement with those of Mohseni et al., 41 and reflect the hepatoprotective effect of Carvacrol.
TAA undergoes hepatic metabolism by cytochrome P450 (CYP450) to form a toxic metabolite, which is a free radical that binds to hepatic macromolecules leading to hepatic necrosis. 42 Previous investigations found that Carvacrol binds to the active site of CYP450 leading to its inhibition and blockage of TAA-induced hepatotoxicity. 43 This is inconsistence with the histopathological results and confirms the ability of Carvacrol to combat fibrosis induced by TAA administration, as hepatic tissues of Carvacrol administrated rats showed near to normal appearance of portal areas with minimal changes as; mild inflammatory reaction and minimal bile duct proliferation without any evidence of fibroplasia accompanied with mild degree of hepatocellular vacuolar degeneration as observed by Hussein et al. 44
Oxidative stress induced by TAA was manifested by decreased contents of the hepatic anti-oxidants; Nrf-2 and GSH. Such anti-oxidants play a key role in both the detoxification and scavenging of ROS. Those findings are inconsistence with the anti-oxidant properties previously recorded.40,45 In the present investigation, the down-regulation of Nrf-2 in the liver tissue was substantially prevented by Carvacrol with significant maintenance of GSH, This finding implies that the anti-oxidant ability of Carvacrol leads to inactivation of ROS through its radical scavenging effect. 12
On the other hand, Nrf-2 and NF-κB are two keys that regulate cellular responses to oxidative stress and inflammation, respectively. The absence of Nrf-2 can exacerbate NF-κB activity leading to an increase in pro-inflammatory cytokines production such as IL-1β and iNOS. 46 TAA intoxication is responsible for the drastic elevation of NF-κB and inflammatory cytokines IL-1β and iNOS which come in agreement with Kumar et al., 47 who recognized that NF-κB aggravates liver fibrosis through playing an important role in the regulation of inflammatory responses and pro-inflammatory cytokines IL-1β and iNOS production. Carvacrol reduced the elevated hepatic NF-κB content and also reduced hepatic inflammatory cytokines IL-1β and iNOS contents which is with in agreement with Khan et al., 43 stating that Carvacrol interrupts the phosphorylation of mitogen-activated protein kinase (MAPK), which plays a key role in regulating cellular growth through affecting proliferation, differentiation, and mechanism of cell death. The phosphorylation of this protein stimulates NF-κB activation, responsible for the secretion of IL-1β and iNOS inflammatory cytokines. Therefore, Carvacrol exerted hepatoprotective role possibly via its anti-inflammatory effect.
Moreover, Trx is a stress-inducible protein that acts on scavenging intracellular ROS, the expression of which is enhanced by various forms of stress, such as TAA which is known to induce hepatic fibrosis through the formation of free radicals in the liver, thus enhancing the expression of Trx in TAA-treated rat livers.40,48 Carvacrol administration reduced Trx level near to normal, which indicates that Carvacrol helps to prevent free radical development by inhibition of CYP450 and regression of TAA-induced hepatic fibrosis.
Furthermore, it was observed that the content of TGF-β1 was highly expressed in the liver of TAA-treated rats with elevated protein expression of α-SMA in hepatic tissues. 40 It is well mentioned that when oxidative stress and the resultant lipid peroxidation cause tissue necrosis and inflammation and hence promote the progression of tissue fibrogenesis leading to mobilization of inflammatory cells that produce pro-fibrotic mediators as TGF-β, which is responsible for activation and trans-differentiation of HSC to myofibroblast, a phenotype with strong expression of α-SMA proteins in liver tissues. 49 Carvacrol significantly decreased hepatic TGF-β1 content and showed reduced expression of α-SMA in liver compared to TAA group, revealing the anti-fibrotic activity of carvacrol against TAA-induced hepatic fibrosis.
TAA administration in the current work led to increase in hepatic contents of MMP-9 and MMP- which come in agreement with another former study of Bain et al., 39 The release of MMPs after liver intoxication is indicative of hepatic damage due to TAA. 43 Excessive accumulation of extracellular matrix (ECM) is a hallmark of fibrogenesis, that accumulation, is a result of imbalanced changes between matrix metalloproteinases (MMPs) and the tissue inhibitors of metalloproteinases (TIMPs), which leads to distortion of hepatic architecture and organ failure. 50 ' Carvacrol treatment in the current study showed a significant decrease in hepatic MMP-9 and MMP-3 contents that come in harmony with previous investigation, 43 and indicates the anti-fibrotic effect of Carvacrol via reducing alteration in MMPs.
TAA administration significantly elevated hepatic caspase-3 immune-expression in hepatic cells, this finding is inconsistence with previous studies,50,51 which stated that caspase-3 is a major enzyme in the apoptotic cascade and an indication of apoptotic cell death in TAA-induced liver fibrosis in rats. Carvacrol-treated groups exhibited decreased number of caspase-3 positive cells, implicating that Carvacrol may participate in suppressing hepatic fibrosis through decreasing apoptosis of hepatocytes and mitigated fibrogenesis.
Regarding, ATX, is a glycoprotein that converts lysophosphatidylcholine to the bioactive phospholipid lysophosphatidic acid. 39 ATX is secreted in response to inflammation, to stimulate the migration of fibroblasts, keratinocytes and leukocytes into the injured area. 52 It represents a more specific accurate biomarker for liver fibrosis estimation than type IV collagen, as any increase in ATX concentration is fairly specific for liver fibrosis. 39 This study displayed an elevated hepatic ATX content in TAA-administrated rats. It has been demonstrated that ATX is rapidly cleared through the liver sinusoidal endothelial cells. The phenotypic changes occurring in the liver during liver fibrosis may lead to reduced ATX clearance, thereby increasing its concentration. 39 Carvacrol treatment in this study significantly alleviated ATX content which denotes its effect in decreasing the phenotypic changes occurring in the liver and regression of hepatic fibrosis, which could be attributed to binding of Carvacrol to the active site of CYP450 leading to its inhibition and disruption of the TAA-induced hepatotoxicity. 43
The hepatoprotective effect of Carvacrol against TAA-induced liver fibrosis was similar to that of UDCA. As UDCA significantly reduced liver enzymes, exerted anti-oxidant and anti-inflammatory activities, reduced MMPs alterations, exerted anti-apoptotic and anti-fibrotic effects and attenuated the histopathological changes.
More studies are needed to understand the exact molecular mechanisms underlying the protective role of Carvacrol on TAA-induced hepatic fibrosis in rats.
Conclusion
In note, our findings strengthen the hepatoprotective effect of Carvacrol against TAA-induced liver fibrosis, promoting liver regeneration and down-regulating liver injury through featured mechanisms including; amelioration of liver function, attenuation of oxidative stress and inflammatory biomarkers, alleviation of MMPs activities, fibrotic Trx and ATX, apoptotic and histopathological changes.
Footnotes
Author contributions
All authors contributed to the reagents/materials/analysis tools, collected the material, analyzed the data and wrote and revised the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
