Abstract
Secondhand smoke (SHS) is an important health issue worldwide. Inhaling SHS during pregnancy could cause abnormalities in the internal tissues of newborns, which may then impair fetal development and even cause severe intrauterine damage and perinatal death. However, the understanding of cytopathic mechanisms of SHS by maternal passive smoking on fetus liver during pregnancy is still limited. This study analyzed the effects of high-dose SHS (SHSH) on fetus liver using a maternal passive smoking animal model. Experiments showed that hepatic matrix metalloproteinase-9 activity and terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick-end labeling-positive cells were significantly increased in livers from fetuses of hamsters treated with SHSH. Similarly, expressions of both extrinsic and intrinsic apoptotic molecules were significantly higher in livers from fetuses of hamsters exposed to SHSH. Additionally, significantly increased inflammatory proteins, including transforming growth factor β, inducible nitric oxide synthase, and interleukin 1β, and fibrotic signaling molecules, including phosphorylated Smad2/3, SP1, and α-smooth muscle actin, were observed in the fetus livers from hamsters treated with SHSH. This study revealed that SHSH not only increased apoptosis through intrinsic and extrinsic pathways in the livers of fetuses from hamsters exposed to SHSH but also augmented hepatic fibrosis via Smad2/3 signaling.
Introduction
Although the total consumption of cigarettes in the United States decreased by 33% in the last decade, the use of paraphernalia such as hookahs (water pipes) has increased and the consumption of alternative tobacco products such as cigarillos, cigars, bidis, kreteks, and smokeless tobacco (snuff, dip, snus, and chewing tobacco) increased by 123%. 1 Therefore, secondhand smoke (SHS) is still considered as a serious health issue worldwide. SHS contains more than 5000 chemical constituents, including acetaldehyde, benzene, tobacco-specific nitrosamines, aromatic amines, and dioxins. 2,3 Accordingly, chronic exposure to SHS by nonsmokers has been associated with various diseases such as asthma, lung cancer, renal malfunction, cardiovascular disorders, and liver diseases. 4 –9 Evidences have indicated the growth-suppressing effects of maternal passive smoking on fetus. Inhalation of SHS upon pregnancy causes abnormalities in the internal tissues of newborns, including increased apoptosis, mesenchymal changes, hyperplasia of bronchial muscles, abnormal proliferation in hematopoiesis and bile duct cells, immature glomeruli of kidney, epithelial hypoplasia of stomach, and hypoplasia of intestinal villi. 10 Exposure to SHS during pregnancy also impairs fetal development and causes intrauterine growth retardation, prematurity, low birth weight, congenital anomalies, and, in cases that are more severe, intrauterine or perinatal death. 10,11
Apoptosis is a recognized mechanism of the elimination of redundant or impaired cells. Several studies have shown that mitochondria may play an important role in apoptosis by releasing cytochrome c and by activating caspase 9, which then activates caspase 3 that is responsible for DNA-cleavage action. 12,13 Mitochondria have also been targeted in studies of reactive oxygen species-mediated effects of tobacco smoke exposure, which induces mitochondrial depolarization along with apoptosis in normal human monocytes. 14 Another study reported that chronic exposure to cigarette smoke induces apoptosis in the rat testis, which may be a pathogenic mechanism of defective spermatogenesis. 15 Additionally, lung tissues of newborn rats exposed to cigarette smoke have a higher rate of apoptosis compared to controls. 10 These findings reveal a strong association between maternal passive smoking and fetal tissue injuries induced by apoptosis.
Although various studies have documented the harmful effects of maternal passive smoking on fetus, understanding of the effects and mechanisms of SHS on fetus liver during pregnancy is still limited. Therefore, this study investigated the effects of SHS, especially apoptosis and fibrosis, on fetus liver using a maternal passive smoking animal model and verifies the possible harmful effects on hamster fetus.
Methods
Animals and SHS exposure
Syrian hamster is a well-established and common animal model for investigating the effects of maternal smoking on fetus development in recent decades. 16,17 Syrian hamsters at 5 weeks of age were purchased from the National Center for Experimental Animals (National Science Council, Taiwan, Republic of China). All hamsters were kept in an animal room at 22°C with a 12-h light/12-h dark cycle. Animals were allowed to adapt to the environment for 1 week after their arrival before the experiment started. All protocols were approved by the Institutional Animal Care and Use Committee of Chung Shan Medical University, Taichung, Taiwan (approval number: 1452). The principles of laboratory animal care (NIH publications) were followed. Pregnant female hamsters were confirmed at fifth day after mating and then randomly divided into three groups (four pregnant hamsters for each group) as control, SHSL, and SHSH. For experiments, the hamsters were placed in whole-body exposure chambers (40 × 40 × 39 cm 3 ; CH Technologies, Westwood, New Jersey, USA) and daily exposed to 0, 5, or 15 cigarettes (Long Life Mild Slims, Taiwan), representing control, low-dose secondhand smoke (SHSL), and high-dose secondhand smoke (SHSH), respectively. The inhalation dosage of SHS is according to our previous study. 13 Filtered air was introduced into the chamber at a low rate of 200 L/min. Hamsters were exposed to cigarette smoke for 30 min, twice a day for 7 days. The hamsters were then killed by carbon dioxide asphyxiation. Experiments were repeated on three separate occasions. The fetus quantities for three independent experiments were 43 (13, 19, and 11 fetuses for control, SHSL, and SHSH groups), 29 (9, 13, and 7 fetuses for control, SHSL, and SHSH groups), and 37 (16, 12, and 9 fetuses for control, SHSL, and SHSH groups), respectively. The liver samples were harvested and stored at −80°C for further analysis.
Gel zymography
Matrix metalloproteinase (MMP)-9 and MMP-2 activities were analyzed by gelatin zymography assays as previously described. 18 Ten microliters of 10-fold diluted serum or 20 μg protein lysates of livers from hamster fetus were separated by an 8% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels containing 0.1% gelatin. Gels were washed for 30 min in 2.5% Triton X-100 to remove the SDS and then soaked in the reaction buffer (40 mM tris(hydroxymethyl)aminomethane -hydrochloride, pH 8.0, 10 mM calcium chloride, and 0.02% sodium azide at 37°C for 24 h. Gelatinolytic activity was visualized by staining the gels with 0.5% Coomassie brilliant blue R-250 and relative MMP levels were quantitated by a gel documentation and analysis system (Appraise, Beckman-Coulter, Brea, California, USA).
TUNEL assay
Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) is an established method for detecting the fragmented DNA, which is known as a characteristic hallmark of apoptosis.
19
The livers from hamster fetus were embedded into optimal cutting temperature (OCT) compound (Tissue-Tek, Miles Inc., Elkhart, Indiana, USA
Masson’s trichrome staining
To identify collagen deposition in livers of hamster fetuses, Masson’s trichrome staining was performed as described in our previous study. 21 Briefly, a small piece of liver tissue was collected and fixed in 10% buffered formalin. The sample was then embedded in paraffin and sliced into 5-μm-thick sections. The sections were then deparaffinized, hydrated in a descending alcohol series, and boiled thrice for 5 min in 1 mM ethylenediaminetetraacetic acid, pH 8.0 (antigen retrieval), prior to staining. Masson’s trichrome staining was performed by staining collagen with blue and myocytes with red. Images were obtained using Zeiss Axiophot microscopes.
Protein extraction
All procedures were performed at 4°C in a cold room. Liver tissues from hamster fetus were homogenized in PRO-PREP solution (iNtRON Biotech, Korea) at a concentration of 1 mg tissue/10 µL PBS. The homogenates were placed on ice for 10 min and then centrifuged at 15,600g for 10 min. Supernatant of the homogenates was collected and stored at −80°C for further experiments. Protein concentration of tissue extracts was determined according to the method described elsewhere 22 using bovine serum albumin (BSA) as standard.
Western blotting
Protein samples were separated in 10% or 12.5% SDS-PAGE and electrophoretically transferred to a nitrocellulose membrane (Amersham Biosciences, Piscataway, New Jersey, USA), as described elsewhere. 23 After blocking with 5% nonfat dry milk in PBS, antibodies against Fas, Fas-associated protein with death domain (FADD), caspase 8, cytochrome c, apoptotic protease activating factor 1 (Apaf-1), caspase 9, caspase 3, transforming growth factor β (TGF)-β, inducible nitric oxide synthase (iNOS), interleukin (IL)-1β, phosphorylated Smad2/3 (p-Smad2/3), Smad2/3, SP1, α-smooth muscle actin (α-SMA) (Santa Cruz Biotechnology, California, USA) and β-Actin (Upstates, Charlottesville, Virginia, USA) were diluted in PBS with 2.5% BSA and incubated for 1.5 h with gentle agitation at room temperature. The membranes were washed twice with PBS-Tween for 1 h and secondary antibody that was conjugated with horseradish peroxidase (HRP) (Santa Cruz Biotechnology) was added for binding the secondary antibody. Pierce’s Supersignal West Dura HRP Detection Kit (Pierce Biotechnology Inc., Rockford, Illinois, USA) was used to detect antigen–antibody complexes, which were then quantified by densitometry (Appraise, Beckman-Coulter, Brea, California, USA).
Statistical analysis
All statistical analyses were performed using SPSS 10.0 software (SPSS Inc., Chicago, Illinois, USA). Experiments were performed in triplicate. Data are reported as means ± standard deviation (SD) of three independent experiments and evaluated by one-way analysis of variance with a post hoc Dunnett’s test for multi-comparison. Significant differences were established at p < 0.05 and indicated by a symbol (*) in the figures.
Results
SHS increases hepatic MMP-9 activity and apoptosis in hamster fetus
Gelatin zymography and TUNEL assay were performed to study the cytopathic effects of SHS on the livers of hamster fetuses. The activity of MMP-9 relative to MMP-2 was significantly higher in the livers of the hamster fetus treated with SHSH than the control group (p = 0.014). Conversely, Figure 1(a) shows that MMP-9 activity in the fetal livers did not significantly differ between the control group and the SHSL group (p = 0.562). The ratio of MMP-9 activity to MMP-2 activity was calculated and shown in the lower panel of Figure 1(a). The number of TUNEL-positive hepatic cells was also significantly larger in the hamster fetus from the SHSH group (p = 0.010). Conversely, significantly fewer TUNEL-positive hepatic cells were observed in the hamster fetus from both control and SHSL groups (Figure 1(b)). The mean percentages of TUNEL-positive hepatic cells in the hamsters from control, SHSL, and SHSH groups were 1.68 ± 0.37, 1.92 ± 0.58, and 5.98 ± 1.19, respectively (Figure 1(c)). Since no significantly increased MMP-9 activity (p = 0.562) and TUNEL-positive cells (p = 0.441) were detected in the fetus livers from the hamsters of SHSL group, the following advanced analyses were merely performed on the fetus livers from hamsters treated with SHSH.
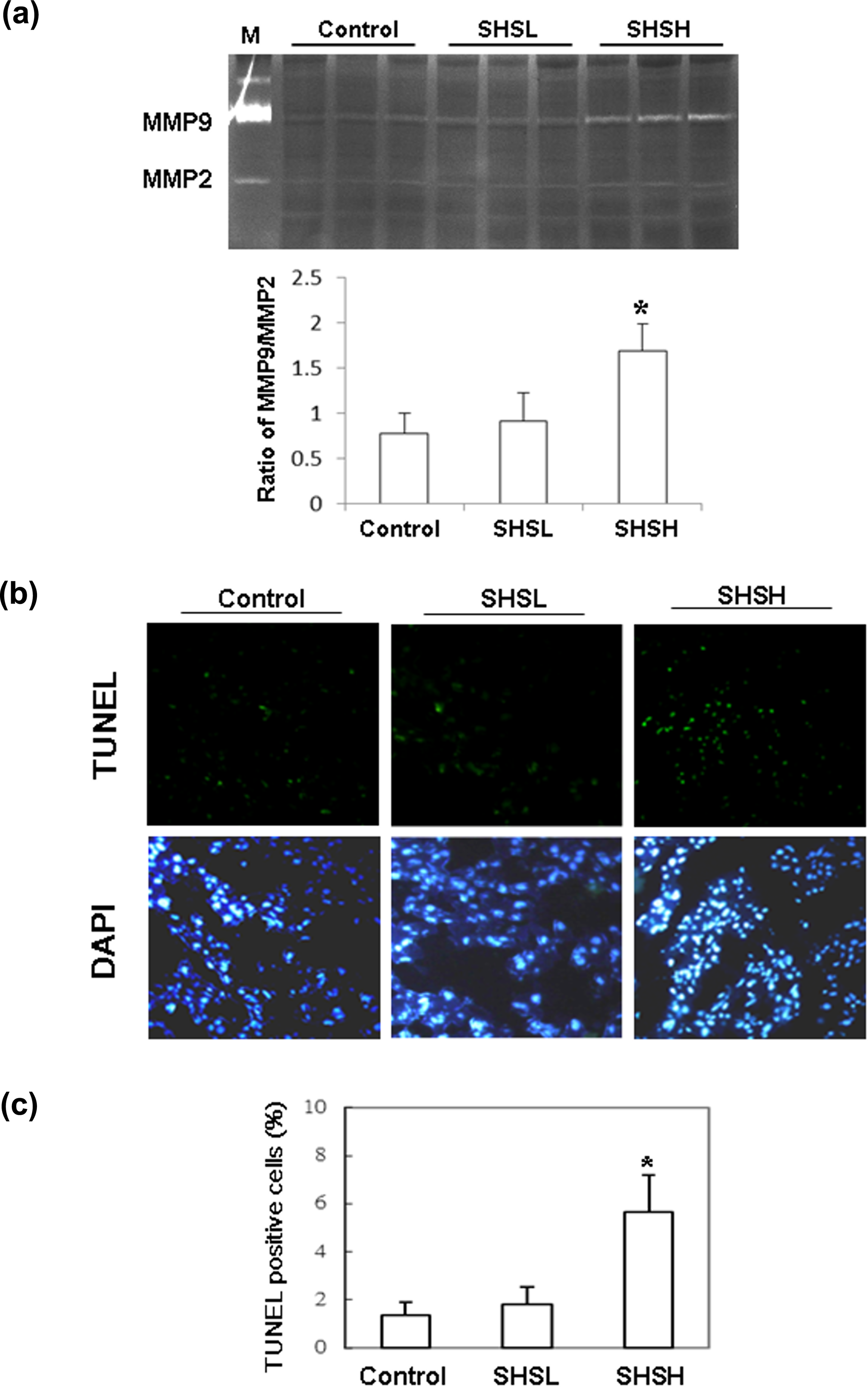
The MMP activity and TUNEL-positive cells in fetus livers. (a) The ratio of MMP9/MMP2 activity (control: n = 8; SHSL: n = 10; SHSH: n = 6) and (b) representative stained apoptotic cells of liver sections with TUNEL assay in hamster fetus were magnified by ×200. (c) The percentages of apoptotic cells were calculated. Bars present the percentage of TUNEL-positive cells relative to total cells (10 mice × 10 scope field count in each group) and indicate mean values ± SD (control: n = 10; SHSL: n = 9; SHSH: n = 6). *Significant differences as compared to the control. Similar results were obtained in three independent experiments. MMP: matrix metalloproteinase; TUNEL: terminal deoxynucleotidyl transferase dUTP nick-end labeling; SD: standard deviation.
SHS induces apoptotic signaling in the livers of hamster fetus
For further confirmation that SHS induced hepatic apoptosis in the hamster fetus, Fas-related and mitochondrial-dependent components were detected by Western blotting. Figure 2(a) shows that the amounts of Fas-dependent apoptotic components, including Fas, FADD, and cleaved caspase 8, were significantly larger in the fetal livers of hamsters treated with SHSH than in those from the control group. The p values of Fas, FADD, and cleaved caspase 8 were 0.008, 0.011, and 0.042, respectively. Figure 2(b) to (d) shows the ratios of Fas, FADD, and cleaved caspase 8 to β-actin, respectively. Additionally, expressions of mitochondrial-dependent apoptotic components, including cytochrome c, Apaf-1, and cleaved caspase 9, were also detected. Significantly increased amounts of cytochrome c, Apaf-1, and cleaved caspase 9 were detected in the livers of the hamster fetus that were treated with SHSH than in those from the control group (Figure 3(a)). The amounts of cytochrome c, Apaf-1, and cleaved caspase 9 proteins relative to the quantity of β-actin were calculated and shown in the lower panel of Figure 3(a). The p values of cytochrome c, Apaf-1, and cleaved caspase 9 were 0.004, 0.034, and 0.012, respectively. Furthermore, the amount of cleaved caspase 3, the downstream apoptotic molecules, was significantly higher in the fetus livers from the SHSH group than in those from the control group (Figure 3(b); p = 0.013). The amount of cleaved caspase 3 protein relative to that of β-actin is shown in the lower panel of Figure 3(b).

Expression of (a) Fas, FADD, and cleaved caspase 8 in fetus livers. Quantified results of (b) Fas, (c) FADD, and (d) cleaved caspase 8 are shown on the basis of β-actin (control: n = 13; SHSH: n = 9). *Significant differences as compared to the control. Similar results were obtained in three independent experiments. FADD: Fas-associated protein with death domain.
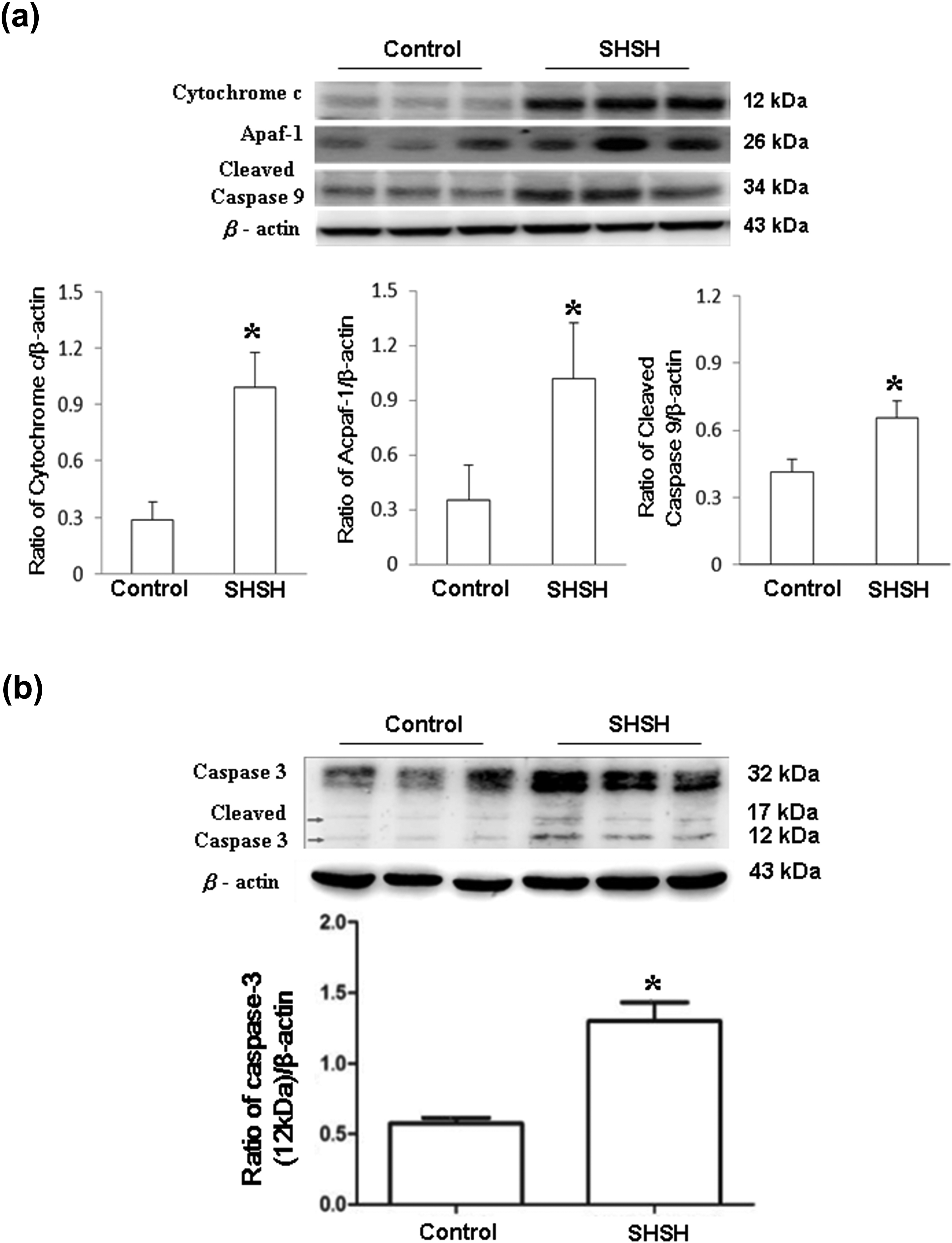
Expression of (a) cytochrome c, Apaf-1, and cleaved caspase 9 in fetus livers. Quantified results of cytochrome c, Apaf-1, and cleaved caspase 9 are shown on the basis of β-actin. (b) caspase 3 and its cleaved form were detected by immunoblotting. Quantified results are shown in the lower panel (control: n = 13; SHSH: n=9). *Significant differences as compared to the control. Similar results were obtained in three independent experiments. Apaf-1: apoptotic protease activating factor 1; SHSH: high-dose of secondhand smoke.
SHS induces hepatic inflammation and fibrosis in hamster fetus
To study the effects of SHS on inflammatory proteins in the livers of hamster fetuses, expressions of TGF-β, iNOS, and IL-1β were detected by Western blotting. Figure 4(a) to (c) shows that the amounts of TGF-β, iNOS, and IL-1β proteins in the fetus livers were significantly higher in the SHSH group than in the control group. The ratios of TGF-β, iNOS, and IL-1β relative to β-actin were calculated and shown in the lower panel of Figure 4(a) to (c)). The p values of TGF-β, iNOS, and IL-1β were 0.002, 0.041, and 0.037, respectively. To further confirm the effects of SHS on hepatic fibrosis, expressions of p-Smad2/3, Smad2/3, SP1, and α-SMA were also examined (Figure 5). Significantly increased p-Smad2/3, SP1, and α-SMA relative to the quantities of their controls were detected in the fetus livers from hamster treated with SHSH than in those from the control group (Figure 5(a)). Quantified results are shown in the lower panel of Figure 5(a) and the p values of p-Smad2/3, SP1, and α-SMA were 0.009, 0.006, and 0.017, respectively. Additionally, markedly increased collagen deposition (blue area) was also detected in the liver sections from the hamster fetus that were treated with SHSH as compared to those from the control group (Figure 5(b)).

Expression of (a) TGF-β, (b) iNOS, and (c) IL-1β proteins in fetus livers. Quantified results are shown on the basis of β-actin (control: n = 13; SHSH: n = 9). *Significant differences as compared to the control. Similar results were obtained in three independent experiments. TGF: tumor growth factor; iNOS: inducible nitric oxide synthase; IL-1β: interleukin 1β; SHSH: high-dose of secondhand smoke.

Expression of (a) p-Smad2/3, Smad2/3, SP1, and α-SMA proteins in fetus livers and (b) representative photographs of liver cross sections with Masson’s trichrome staining (control: n = 7; SHSH: n = 6) were magnified by ×200. Quantified results are shown in the lower panel on the basis of Smad2/3 and β-actin, respectively (control: n = 13; SHSH: n = 9). *Significant differences as compared to the control. Similar results were obtained in three independent experiments. α-SMA: α-smooth muscle actin; SHSH: high-dose of secondhand smoke; p-Smad2/3: phosphorylated Smad2/3.
Discussion
SHS is known to cause liver injuries by inducing inflammatory cytokine expression, apoptosis, and fibrosis. 24,25 Livers from fetuses of hamsters exposed to SHSH showed significantly increased MMP-9 activity and TUNEL-positive cells as compared to the controls. Expressions of both extrinsic and intrinsic apoptotic molecules were also significantly higher in livers from fetuses of pregnant hamsters exposed to SHSH. Accordingly, inflammatory molecules, including TGF-β, iNOS, and IL-1β, were significantly increased in livers from fetuses of hamsters exposed to SHSH. Meanwhile, significantly augmented fibrosis signaling, including increased p-Smad2/3, SP1, and α-SMA, and collagen deposition were also observed in livers of fetuses from hamsters exposed to SHSH. Conversely, these phenomena were not detected in the livers of fetuses in hamsters treated with SHSL or in those from the control group. This study firstly revealed that high dose of maternal passive smoking not only increases apoptosis through both intrinsic and extrinsic pathways in the livers of hamster fetuses but also augments hepatic fibrosis via Smad2/3 signaling.
Accumulating evidence indicates that maternal passive smoking has growth-suppressing effects on the fetus. 26,27 Passive smoking upon pregnancy causes abnormal morphological changes in internal tissues of newborns, including lung, liver, stomach, kidney, and intestinal tissues. Notably, pronounced abnormalities in hematopoiesis and proliferation of bile duct cells are the most common variations observed in liver tissues of pups exposed to SHSH. 10,28 Another study of complementary DNA array indicated that environmental cigarette smoke stimulated pro-apoptotic genes and downregulated the genes of cell cycle, which then impair development of the fetus. 29 Maternal passive smoking also induces hypoxia-related genes and several oncogenes and receptors involved in proliferation and differentiation of leukocytes in the fetal liver, which bear hematopoietic functions. 29 A very recent study of the human fetal liver proteome reported that maternal smoking affects proteins with roles in posttranslational protein processing and secretion, stress responses, and detoxification and homeostasis, which involve in necrosis, apoptosis, and cancer development. 30 Apoptosis is caused by caspase activation, mainly death receptor and mitochondrial pathways. These two pathways are sometimes referred to as the extrinsic and intrinsic apoptosis pathways, respectively. 31 Similarly, TUNEL-positive cells were significantly increased in the livers of fetuses from hamsters exposed to SHSH. The fetus livers also showed significantly increased intrinsic (Fas, FADD, and cleaved caspase 8) and extrinsic (cytochrome c, Apaf-1, and cleaved caspase 9) apoptotic molecules, which indicated a cytopathic effect of maternal passive smoking on fetus livers. However, the chemical substances yielded by smoking, which induce apoptosis, are still needed to be verified.
Besides the hazardous effects reported in previous studies, smoking causes various adverse effects on organs that have no direct contact with the smoke itself such as liver. Smoking is known to have three major adverse effects on the liver, including toxic effects, immunological effects, and oncogenic effects. 24 The liver has an important role in processing drugs, alcohol, and other toxins so that they can be removed from the body. 32 Heavy smoking yields toxins that induce necro-inflammation and are associated with various hepatic lesions such as fibrosis and hepatocellular carcinoma. 33,34 Many studies have indicated that inhalation of SHS during pregnancy causes abnormalities in the internal tissues of newborns. 10,26,27 However, the cytopathic mechanisms of SHS by material passive smoking on fibrosis of fetus liver are still unclear. In the current study, significantly increased inflammatory proteins, including TGF-β, iNOS, and IL-1β, and collagen deposition were firstly observed in the fetus livers from hamsters treated with SHSH. Accordingly, augmented expressions of p-Smad2/3, SP1, and α-SMA were also detected in the fetus livers that were treated with SHSH. These findings show that maternal passive smoking induces fetal liver fibrosis by activating Smad2/3 fibrotic signaling.
Altogether, this study revealed that maternal passive smoking is associated with induction of inflammatory responses and apoptosis in fetus livers as well the augmented hepatic fibrosis by activating Smad/SP1 signaling. However, there is still a compelling need on efforts to ensure the possible pollutants of SHS for understanding their precise mechanism on fetus hepatic injury during embryonic and early postnatal development.
Footnotes
Acknowledgments
The authors thank Ted Knoy for his editorial assistance.
Author contribution
Both the authors CWH and CTH contributed equally.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by clinical research grant from Kaohsiung Armed Force General Hospital, Kaohsiung, Taiwan (No. 104-08) and partially supported by Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW104-TDU-B-212-113002).
