Abstract
Taraxasterol (TAR) is a kind of active compound extracted from dandelion and its molecular structure resembles steroid hormones. Recently, TAR has been reported to show an anti-tumor activity. However, the specific role of TAR in papillary thyroid cancer (PTC) has not been clarified. In this study, we investigated the effect of TAR on PTC cell migration, invasion and epithelial-to-mesenchymal transition (EMT) induced by TGF-β1. PTC cells were exposed to TGF-β1 (5 ng/mL) and then treated with different concentrations of TAR. We found that TAR showed no obvious cytotoxicity below 10 μg/mL but notably reduced migration and invasion of TGF-β1-treated PTC cells. Moreover, TAR treatment decreased MMP-2 and MMP-9 levels, and obviously affected the expression of EMT markers. We also observed that Wnt3a and β-catenin levels were significantly increased in TGF-β1-treated PTC cells while TAR inhibited these effects in a concentration-dependent manner. Additionally, activation of the Wnt pathway by LiCl attenuated the suppressive effect of TAR on TGF-β1-induced migration, invasion and EMT in PTC cells. Taken together, we highlighted that TAR could significantly suppress TGF-β1-regulated migration and invasion by reversing the EMT process via the Wnt/β-catenin pathway, suggesting that TAR may be a potential anti-cancer agent for PTC treatment.
Keywords
Introduction
Thyroid cancer is a prevalent endocrine malignancy and its most common type is papillary thyroid cancer (PTC) which accounts for over 80% of all thyroid cancer cases in the world.1–3 PTC is classified into conventional PTC (CPTC) and follicular variant PTC (FVPTC) and its diagnosis is generally based on nuclear features.4–7 Currently, multiple therapeutic methods have been available for PTC treatment and a majority of PTC patients are accompanied by a good prognosis.8–10 Unfortunately, there still are a small fraction of patients who develop local and distant metastasis and consequently suffer from a low survival rate.11,12 Therefore, it is desperately needed to have a better understanding of the mechanisms underlying PTC metastasis.
The epithelial-mesenchymal transition (EMT) is known as a crucial step in tumor metastasis and is characterized by the switch from epithelial markers to mesenchymal markers. 13 During the process, epithelial cells acquire mesenchymal features and their migratory and invasive properties are promoted.14–16 The EMT process can be triggered by activation of diverse transcription factors such as Snail and Twist.17,18 Furthermore, multiple signaling pathways have been identified as EMT regulators such as the Wnt/β-catenin signaling which plays an important role in progression of various cancers including PTC. 19
Dandelion is a kind of perennial herb and widely used for treatment of inflammatory conditions such as skin ulcers, malignant boils and acute conjunctivitis.20,21 Taraxasterol (TAR), a pentacyclic triterpene, is a main active constituent extracted from dandelion and has a molecular structure similar to steroid hormones. 22 Many studies have demonstrated its anti-inflammatory effects in vitro and in vivo. 23 Notably, a recent report showed its anti-tumor activity. 24 However, the specific role of TAR in PTC has not been clarified.
The purpose of this study was to investigate the effect of TAR on migration, invasion and EMT induced by TGF-β1 in PTC cells and to explore the underlying mechanisms, which provided evidence that TAR may be considered a promising agent for PTC treatment.
Materials and methods
Cell culture
Human PTC cell lines (TPC-1 and BCPAP) and normal thyroid cell line (Nthy-ori 3-1) were obtained from American Type Culture Collection (ATCC, Manassas, VA, USA) and cultured in RPMI 1640 medium (Gibco, Rockville, MD, USA) supplemented with 10% fetal bovine serum (FBS; Gibco) and 100 μg/mL penicillin/streptomycin at 37°C in a humidified atmosphere with 5% CO2. For TAR and TGF-β1 treatments, either TAR (2.5, 5, 10 or 20 μg/mL) or TGF-β1 (5 ng/mL) were added to the medium.
Cell viability assay
Cells were seeded in a 24-well plate at a density of 2 × 104 cells/well and cultured for 48 h. Thereafter, MTT (Sigma, St. Louis, MO, USA) was added to each well and cells were incubated for another 4 h. Subsequently, culture medium was discarded and DMSO (Sigma) was added. The absorbance at 570 nm was measured using a microplate reader.
Cell migration and invasion assays
Cell migration and invasion were detected using transwell assays. For the cell migration assay, cells (2 × 105) were suspended in serum-free medium and added to the upper chamber. Culture medium containing 10% FBS (Gibco) was added to the lower chamber. After 24 h incubation, cells migrating to the lower surface of the insert were fixed and stained with 0.1% crystal violet. The number of cells from five random fields was counted under a microscope. The cell invasion assay was performed according to the same procedure as that for the cell migration assay except that the inserts were pre-coated with Matrigel (BD Bioscience, San Diego, CA, USA).
Western blot
Total protein was isolated using RIPA buffer (Sigma), separated by 12% SDS-PAGE and transferred to PVDF membranes (Millipore, Billerica, MA, USA). After blocking with 5% non-fat milk, the membranes were incubated overnight at 4°C with primary antibodies against MMP-2 (sc-13595), MMP-9 (sc-13520), E-cadherin (sc-8426), α-catenin (sc-9988), N-cadherin (sc-8424), vimentin (sc-373717), Wnt3a (sc-136163), β-catenin (sc-7963) and GAPDH (sc-365062). Subsequently, the membranes were incubated at room temperature for 2 h with corresponding secondary antibodies. All antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Protein bands were visualized using an ECL kit (GE Healthcare, Piscataway, NJ, USA) and the intensity was quantified using the QuantityOne software (Bio-Rad, Hercules, CA, USA).
Statistical analysis
Data were presented as means ± standard deviation (SD) and analyzed using GraphPad Prism 5.0 software. The comparison between different groups was made using Student’s t-test or one-way ANOVA. P<0.05 was considered statistically significant.
Results
The effect of TAR on PTC cell viability
To determine the optimal concentration of TAR, we detected the viability of TPC-1 and BCPAP cells using the MTT assay after treatment with TAR at different concentrations (2.5 μg/mL, 5 μg/mL, 10 μg/mL and 20 μg/mL). As shown in Figure 1(A) and (B), TAR showed no obvious cytotoxicity below 10 μg/mL, but suppressed cell viability at 20 μg/mL. Therefore, the non-toxic concentrations of TAR were selected for subsequent experiments.

The Effect of TAR on PTC cell viability. TPC-1 (A) and BCPAP (B) cells were treated with different concentrations of TAR for 48 h and their viability was measured by the MTT assay. *p < 0.05 vs the control group.
TAR inhibits TGF-β1-induced migration and invasion of PTC cells
To examine the effects of TAR on PTC cell migration and invasion under TGF-β1 stimulation, we performed transwell assays. As shown in Figure 2(A), the number of migrating and invading TPC-1 cells was significantly increased after TGF-β1 treatment in comparison with corresponding control cells while TAR markedly inhibited TGF-β1-induced cell migration and invasion in a concentration-dependent manner. We obtained similar results for BCPAP cells (Figure 2(B)). Moreover, we investigated the effects of TAR on normal thyroid cell line Nthy-ori 3-1 and found no significant effect (Supplementary Figure S2).

TAR inhibits TGF-β1-induced migration and invasion of PTC cells. TPC-1 and BCPAP cells were exposed to TGF-β1 (5 ng/mL), followed by treatment with TAR (2.5, 5 and 10 μg/mL). TPC-1 (A) and BCPAP (B) cell migration and invasion were measured by transwell assays. *p < 0.05 vs the control group; #p < 0.05 vs the TGF-β1 group.
TAR inhibits TGF-β1-induced MMP-2 and MMP-9 in PTC cells
MMP-2 and MMP-9 play an important role in cancer cell migration and invasion,25–27 thus we detected the protein expression of MMP-2 and MMP-9 in PTC cells by western blot. As shown in Figure 3(A) and (B), MMP-2 and MMP-9 expression was obviously increased by TGF-β1 in TPC-1 and BCPAP cells while TAR concentration-dependently alleviated the effect.
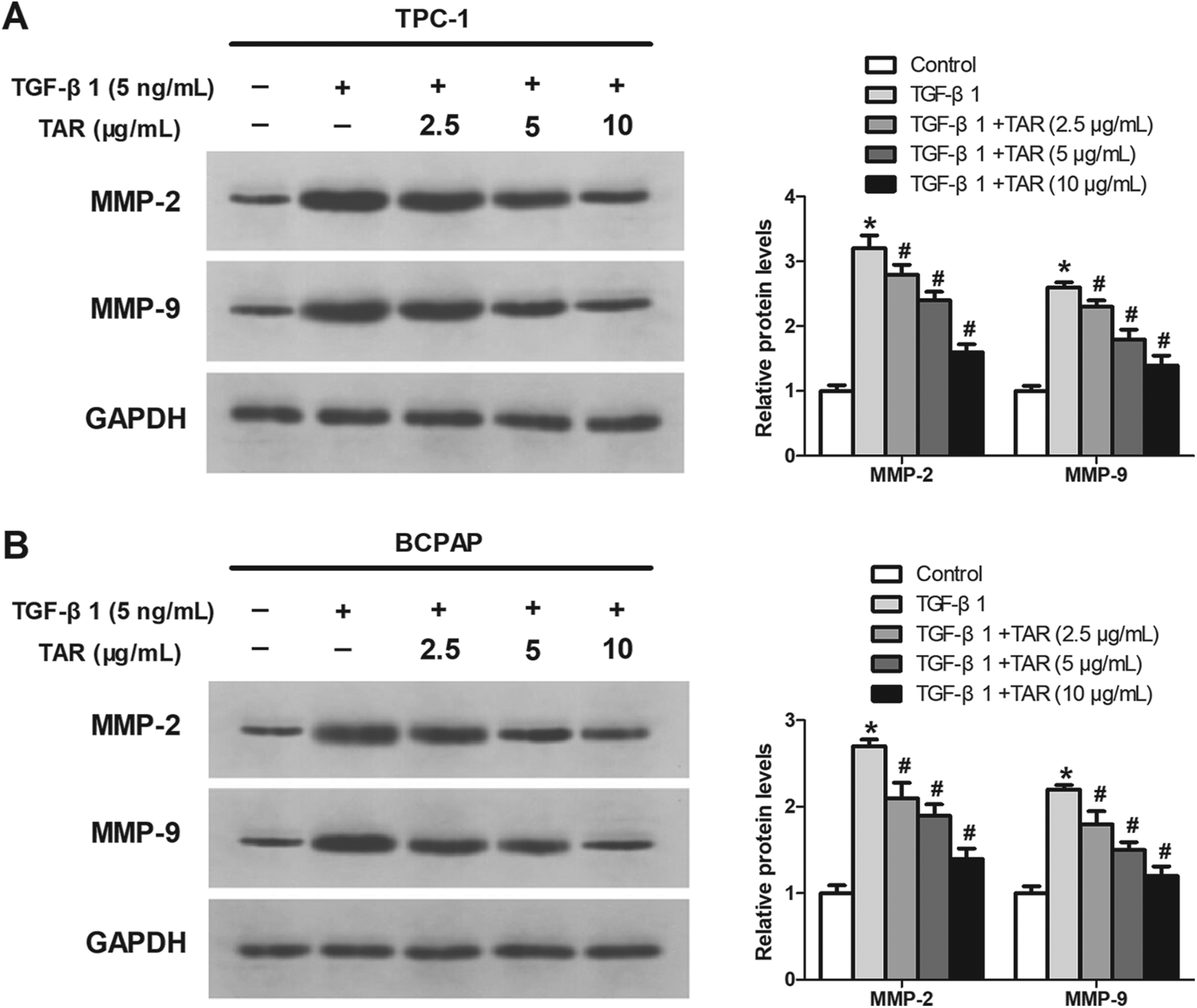
TAR inhibits TGF-β1-induced MMP-2 and MMP-9 in PTC cells. TPC-1 and BCPAP cells were pretreated with TGF-β1 (5 ng/mL) and then treated with different concentrations of TAR. The protein expression levels of MMP-2 and MMP-9 in TPC-1 (A) and BCPAP (B) cells were assessed by western blot. *p < 0.05 vs the control group; #p < 0.05 vs the TGF-β1 group.
TAR inhibits TGF-β1-induced EMT in PTC cells
We analyzed EMT markers in PTC cells by western blot. As shown in Figure 4(A) and (B), TGF-β1-inhibited expression of E-cadherin and α-catenin was significantly enhanced by TAR while TGF-β1-induced expression of N-cadherin and vimentin was obviously reduced by TAR in a concentration-dependent manner.
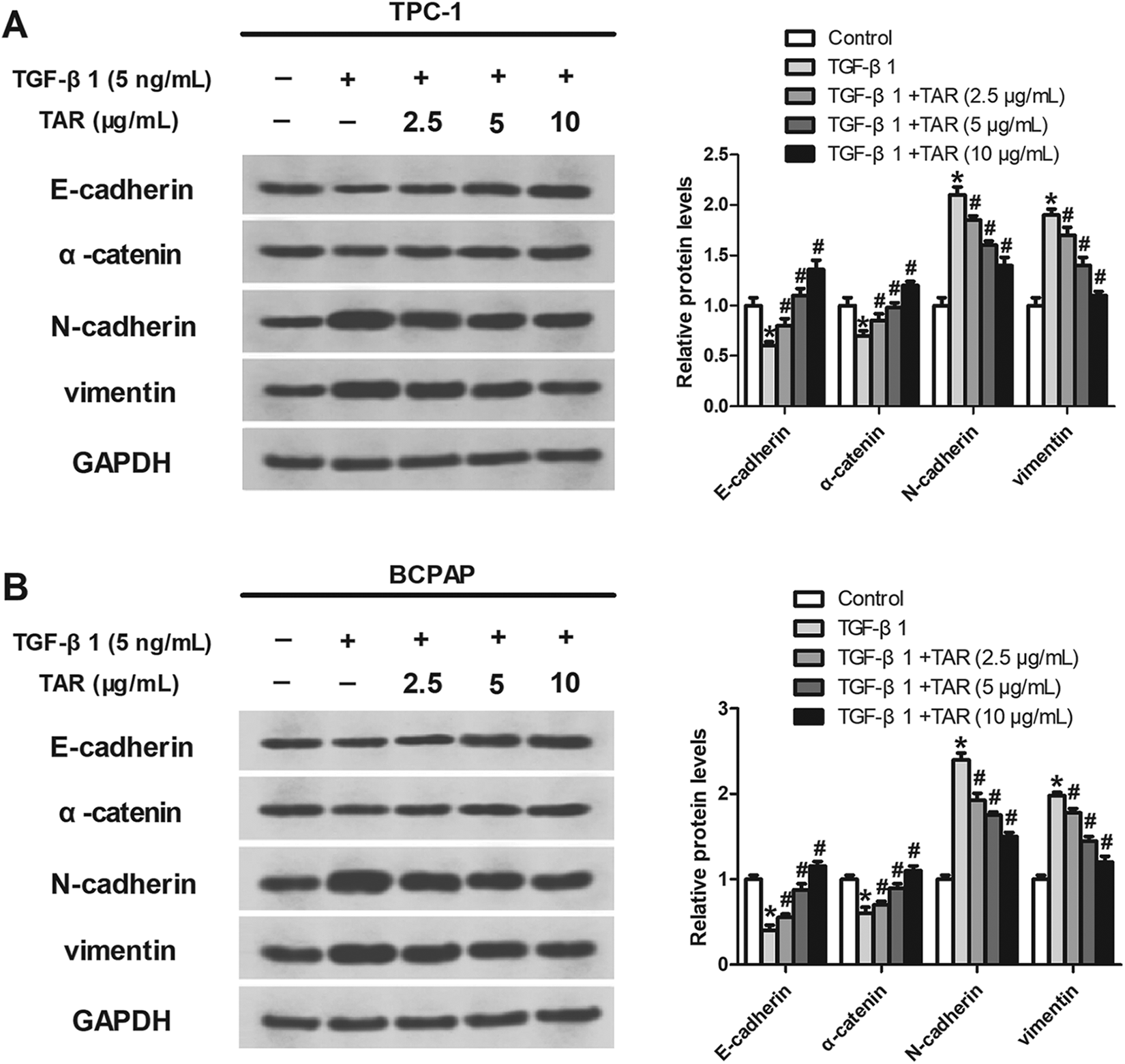
TAR inhibits TGF-β1-induced EMT in PTC cells. TPC-1 and BCPAP cells were treated with TGF-β1 (5 ng/mL), followed by addition of TAR (2.5, 5 and 10 μg/mL). The protein levels of E-cadherin, α-catenin, N-cadherin and vimentin in TPC-1 (A) and BCPAP (B) cells were detected by western blot. *p < 0.05 vs the control group; #p < 0.05 vs the TGF-β1 group.
TAR inhibits TGF-β1-induced activation of the Wnt/β-catenin pathway in PTC cells
The Wnt/β-catenin signaling pathway has been reported to be implicated in TGF-β1-induced cancer progression,28,29 therefore we investigated the effect of TAR on the Wnt/β-catenin pathway in TGF-β1-stimulated PTC cells. The results showed that the expression levels of Wnt3a and β-catenin were significantly increased in TGF-β1-treated TPC-1 cells while TAR inhibited these effects in a concentration-dependent manner (Figure 5(A) to (C)).

TAR inhibits TGF-β1-induced activation of the Wnt/β-catenin pathway in PTC cells. TPC-1 cells were exposed to 5 ng/mL TGF-β1, followed by treatment with different concentrations of TAR. (A) The protein levels of Wnt3a and β-catenin were determined by western blot. (B, C) Quantification of Wnt3a and β-catenin protein expression in TPC-1 cells. *p < 0.05 vs the control group; #p < 0.05 vs the TGF-β1 group.
Wnt pathway activator reverses the inhibitory effect of TAR on TGF-β1-induced migration and invasion of PTC cells
To further explore the functions of the Wnt/β-catenin pathway in TGF-β1-induced PTC progression, the Wnt pathway activator LiCl was used. 30 As shown in Figure 6(A) to (C), TAR inhibited TGF-β1-induced migration and invasion in TPC-1 cells while LiCl reversed these effects.

Wnt pathway activator reverses the inhibitory effect of TAR on TGF-β1-induced migration and invasion of PTC cells. (A–C) After treatment with or without LiCl (20 mmol/L), TPC-1 cell migration and invasion were detected using the transwell assays. *p < 0.05 vs the control group; #p < 0.05 vs the TGF-β1 group; &p < 0.05 vs the TGF-β1+TAR group.
Activation of the Wnt pathway attenuates the suppressive effect of TAR on TGF-β1-induced EMT in PTC cells
We also investigated the involvement of the Wnt pathway in TGF-β1-induced EMT of PTC cells. The results showed that TAR inhibited TGF-β1-induced down-regulation of E-cadherin and up-regulation of N-cadherin in TPC-1 cells while these inhibitory effects were attenuated after treatment with LiCl (Figure 7(A)–(C)).
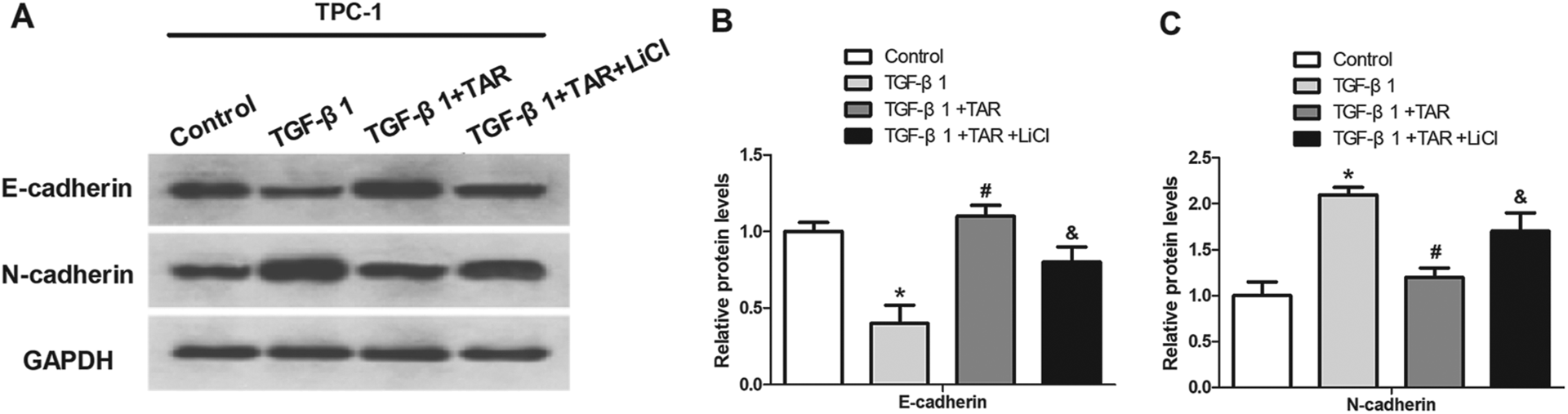
Activation of the Wnt pathway attenuates the suppressive effect of TAR on TGF-β1-induced EMT in PTC cells. (A–C) E-cadherin and N-cadherin expression in TPC-1 cells were measured by western blot, in the presence or absence of LiCl (20 mmol/L). *p < 0.05 vs the control group; #p < 0.05 vs the TGF-β1 group; &p < 0.05 vs the TGF-β1+TAR group.
Discussion
With an increasing incidence, PTC is considered a primary cause of thyroid cancer. 31 Its tendency to metastasis is responsible for poor prognosis of PTC patients and is also an important reason for the majority of patient deaths.11,12 Numerous studies have demonstrated that EMT is a crucial process during cancer progression and metastasis and could be promoted through multiple signaling pathways in cancer cells.32,33 Thus, exploration of novel agents targeting EMT-associated pathways is urgently needed for PTC treatment.
TGF-β1, a multifunctional cytokine, is an important regulator of diverse cellular functions such as embryogenesis and fibrosis.34–36 Accumulating evidence suggests that TGF-β1 could induce EMT in a variety of cancer cells and this process has some major shifts such as reduction in epithelial markers, acquirement of mesenchymal markers and changes in cytoskeleton rearrangement.37,38 For example, Liao et al. reported that breast cancer cells were sensitive to the EMT induction by TGF-β1 and subsequently their invasiveness was promoted. 39 In this study, we found that TGF-β1 obviously enhanced the migratory and invasive abilities of PTC cells. Furthermore, MMP-2 and MMP-9 expression were significantly increased by TGF-β1. On the other hand, TGF-β1 decreased the expression of E-cadherin and α-catenin and increased the expression of N-cadherin and vimentin, confirming induction of EMT by TGF-β1.
TAR, a kind of active compound extracted from dandelion, is structurally similar to steroid hormones which play a central role in cancer maintenance and progression, and therefore has been studied for its potential effects on hormone-dependent cancers. 22 Recently, TAR has been reported to show an anti-tumor activity. For example, Bao et al. suggested that TAR treatment significantly inhibited cell proliferation and induced cell apoptosis in liver cancer. 24 In our study, we found that TAR markedly suppressed TGF-β1-induced migration and invasion. Besides, TAR concentration-dependently alleviated TGF-β1-induced MMP-2 and MMP-9 expression. Our results also indicated that TAR partially restored TGF-β1-induced switch of E-cadherin to N-cadherin. These observations demonstrated that TAR inhibited TGF-β1-induced EMT and may have anti-metastatic effect in PTC cells.
The Wnt/β-catenin signaling pathway is known as a critical player in cancer progression.40–42 As an essential molecule in Wnt signaling, β-catenin is involved in pathogenesis of various cancers.43,44 In addition, nuclear translocation of β-catenin is frequently induced upon activation of Wnt signaling and subsequently initiates transcription of downstream genes and increases the expression of EMT regulators.45,46 More importantly, the Wnt/β-catenin pathway has been reported to be implicated in TGF-β1-induced cancer progression.28,29 For example, Chen et al. demonstrated that TGF-β1-induced EMT in colon cancer cells was mediated through activation of the Wnt/β catenin pathway. 47 In this study, we found that the expression levels of Wnt3a and β-catenin were significantly increased in TGF-β1-treated PTC cells, but TAR inhibited these effects in a concentration-dependent manner. We applied Wnt pathway activator LiCl to further explore the functions of the Wnt/β-catenin pathway in TGF-β1-induced PTC development. The results showed that activation of the Wnt pathway attenuated the suppressive effect of TAR on TGF-β1-induced migration, invasion and EMT in PTC cells, indicating the partial involvement of the Wnt pathway in the regulatory effect of TAR on TGF-β1-induced PTC progression.
In conclusion, our study provided compelling evidence that TAR exerted a modulatory effect on TGF-β1-induced migration, invasion and EMT in PTC and the Wnt/β-catenin pathway participated in the mechanism partially. Taken together, TAR may be considered a promising agent for PTC treatment.
Supplemental material
Supplemental Material, sj-pdf-1-het-10.1177_09603271211023792 - Taraxasterol inhibits TGF-β1-induced epithelial-to-mesenchymal transition in papillary thyroid cancer cells through regulating the Wnt/β-catenin signaling
Supplemental Material, sj-pdf-1-het-10.1177_09603271211023792 for Taraxasterol inhibits TGF-β1-induced epithelial-to-mesenchymal transition in papillary thyroid cancer cells through regulating the Wnt/β-catenin signaling by J Zhu, X Li, S Zhang, J Liu, X Yao, Q Zhao, B Kou, P Han, X Wang, Y Bai, Z Zheng and C Xu in Human & Experimental Toxicology
Footnotes
Authors’ note
Jinxiang Zhu, Xiang Li, and Shaoqiang Zhang authors contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hospital Fund of the First Affiliated Hospital of Xi’an Jiaotong University (No. 2019ZYTS-04), Xi’an Science and Technology Project (No. 2019114613YX001SF04(5)), the Hospital Clinical Research Project of the First Affiliated Hospital of Xi’an Jiaotong University (XJTU1AF-CRF-2020-020), the Special Reward Fund Project for Technological Transformation of Small and Medium-sized Enterprises in Shaanxi Province (2020), the Basic Natural Science Research Program of Shaanxi Province (2021JQ-386) and the Hospital Fund of the First Affiliated Hospital of Xi’an Jiaotong University (No.2019ZYTS-13).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
