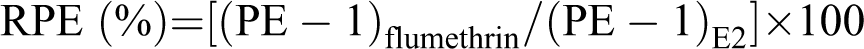Abstract
Pyrethroid pesticides are frequently used for household insect control of insects and in agriculture and livestock. Flumethrin is a pyrethroid that is used against ectoparasites in many animals. The goal of this study was to evaluate the cytotoxic, apoptotic, genotoxic, and estrogenic effects of flumethrin on the mammalian breast cancer cell line (MCF-7). Compared with control groups, a dose-dependent decrease was observed in cell viability at concentrations of 100 µM and higher. The cytotoxic and apoptotic effects detected by LDH assay and AO/EtBr staining increased significantly at a concentration of 1000 µM. The expression of BCL2, which is an anti-apoptotic gene, significantly decreased, whereas BAX, TP53, and P21 expression significantly increased. The results of a comet assay indicated that flumethrin significantly changed tail length, tail % DNA, tail moment, and Olive tail moment in concentrations above 1 and 10 µM. In addition, a 0.1 µM concentration of flumethrin affected ERα receptor mediated cell proliferation and increased transcription of estrogen-responsive pS2 (TFF1) and progesterone receptor (PGR) genes. As a result, flumethrin-induced apoptosis and cytotoxicity at a high concentration, while induced genotoxicity even at lower concentrations. Flumethrin is an endocrine disrupting insecticide with estrogenic effects at very low concentrations.
Introduction
Pesticides, the most commonly used chemicals, are categorized as organophosphates, carbamates, organochlorines, and pyrethroids. 1 Pyrethroids, which have been used since the 1980s due to their high efficacy and low environmental toxicity, are synthetically produced from natural pyrethrine. 2 Recent studies determined that pyrethroids comprise approximately 35% of the global insecticide market. 3 These increased use of synthetic pyrethroids poses a risk of chronic toxicity to humans and animals. 4 Moreover, pyrethroids are difficult to remove from the body due to their lipophilic properties and long-term chronic exposure causes toxic effects related to teratogenicity, carcinogenicity, and mutagenicity of non-target organisms. 2 Many pyrethroids have been shown to induce apoptosis, cause oxidative damage and have genotoxic effects. 5 Furthermore, some pyrethroids have endocrine disrupting effects, estrogenic or low anti-estrogenic activity, and anti-androgenic effects. 6
Flumethrin is a pyrethroid insecticide that has been used as an acaricide in animals since 1986. Its pharmacological action is through neural tissue sodium channels. 7 The structure of flumethrin, whose chemical name is (RS)-α-siyano-4-floro-3-fenoxybenzyl-3-(β,4-diklorositiril)-2,2-dimethylcyclopropanecarboxylate, is shown in Figure 1. It causes choreoathetosis and salivation syndrome (CS syndrome) because it contains a α-cyano group. 8 It has been synthesized against ectoparasites on animals and is widely used in livestock and domestic animals such as cats and dogs. 9 Flumethrin is widely used to control Varroa mites in apiculture. 9 Flumethrin was detected in Spain, Italy, and China in studies investigating pesticide residues in waxes and honey.10–12 However, flumethrin has toxic effects on various experimental animals. In in vivo studies, flumethrin exposure reduced hepatic enzyme levels and monooxygenase system activities as well as anti-pyrine metabolism. 13 Flumethrin increases malondialdehyde (MDA), aspartate transaminase (AST), and alanine aminotransferase (ALT) levels and decreases catalase and superoxide dismutase (SOD) levels, causing oxidative stress and hepatotoxic effects. 14

The chemical structure of flumethrin. Cas number: 69770-45-2.
However, there is insufficient information regarding the cytotoxic, genotoxic, and estrogenic effects of flumethrin in vitro. The objective of this study was to investigate the estrogenic effects and underlying mechanisms of the apoptotic and genotoxic effects of flumethrin on estrogen receptor positive MCF-7 breast cancer cells.
Materials and methods
Chemicals
Flumethrin Pestanal, analytical standard (Cas no. 69770-45-2), dimethyl sulfoxide (DMSO), cytochalasin B, and β-estradiol (E2) were purchased from Merck (Darmstadt, Germany). Phosphate-buffered saline (PBS), cell culture medium, and all other supplements were obtained from Multicell Wisent Bio Products (Saint-Bruno, Quebec, Canada) and sterile plastic materials were purchased from NEST (Science Co., Ltd., China).
Cell culture and treatment
The MCF-7 human breast carcinoma cell line is a well-established in vitro system characterized by its estrogen responsiveness through the expression of estrogen receptor (ER). 15 MCF-7 cells (American Type Culture Collection, Rockville, MD, USA) were grown in DMEM (high glucose with L-glutamine) supplemented with 1% penicillin-streptomycin and 10% heat-inactivated fetal bovine serum (FBS) Multicell Wisent Bio Products (Saint-Bruno, Quebec, Canada) at 37°C with 5% CO2 in a humidified incubator. Flumethrin was dissolved in DMSO to prepare 40 mM and 400 mM stock solutions. The solutions were filtered through 0.22 µm filters.
MCF-7 cells were exposed to flumethrin at concentrations of 10, 1, 0.1, 0.01, 0.001, and 0.0001 µM as described in previous studies to assess the estrogenic effects. 16 Based on the results of an E-screen assay, three concentrations of flumethrin (0.1, 1, and 10 µM) were used to ascertain the estogenic gene expression. To study the cytotoxic effects, eight different flumethrin concentrations (0.1–2000 µM) were evaluated based on prior research by Basci and Eraslan 17 (serum Cmax value for flumethrin) and assesment reports of the Joint FAO/WHO Meeting on Pesticide Residues (JMPR) and the European Medical Agency (EMAE 2000) (ADI levels) regarding the anticipated concentrations in serum after food intake. Four concentrations of flumethrin (1, 10, 100, and 1000 µM) were used to estimate the apoptotic and genotoxic effects.
Determination of estrogenic activity by E-screen assay
The E-screen assay was conducted according to the method described by Korner et al. 18 with slight modifications. The MCF-7 cells were seeded into 24-well plates at an initial density of 104 cells per well with phenol red-free DMEM supplemented with 10% charcoal dextran stripped (steroid-free) FBS and allowed to attach for 24 h. The medium was then replaced with experimental medium containing serial dilutions of flumethrin (from 0.0001 to 10 µM) and 17β-E2 (from 10−5 M to 10−10 M). A positive control was used to determine that E2-induced maximal proliferation at a concentration of 10−9 M. After 144 h of incubation (late exponential phase), the cells were fixed with methanol: acetic acid, stained for protein with sulforhodamine B, and the absorbance was measured (490 nm). The E-screen assay demonstrated the cell proliferation relative to the steroid-free control. The proliferative effect (PE) of flumethrin and E2 was the ratio of the maximum optical density (OD) value obtained with flumethrin- or estrogen-treated groups compared to the control group.
The estrogenic activity of flumethrin was evaluated by determining the relative proliferative effect (RPE, %) by comparing the maximum proliferation induced by flumethrin to that induced by E2. 19
The RPE classified E2 total agonists when they induced a relative proliferation between 80% and 100%. Partial and weak agonists induced a cell proliferation from 25% to 80% and from 10% to 25%, respectively. 20
Cell viability assay
The MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay was used to determine the effect of flumethrin treatments on MCF-7 cell viability, and the inhibitor concentration (IC50) value was evaluated. The MTT assay was conducted using a Cell Proliferation Kit I (Roche Diagnostics, Mannheim, Germany).
Briefly, MCF-7 cells (5 × 103 cells/well) were seeded into 96-well plates. After cell adhesion (approximately 24 h), the medium was replaced with fresh medium containing different concentrations (0.1–2000 µM) of flumethrin, and then the plates were incubated for 24 h. At the end of treatment, 10 µL of MTT I solution was added to each well and incubated for 4 h in the dark. Immediately after incubation, 100 µL of MTT II solution was added to each well to stop the MTT reduction and dissolve formazan crystals. The viable cells were read by an ELISA multi-well plate reader (Thermo Scientific Multiskan GO) at 570 nm. The untreated MCF-7 cells were removed for use as a control. The IC50 value (a concentration that yielded 50% fewer viable cells) was determined by analyzing the concentration and normalized response using GraphPad Prism v6.0 software.
LDH cytotoxicity assay
The possible cytotoxic effects of flumethrin exposure on MCF-7 cells was also evaluated by measuring LDH leakage into the extracellular fluid. LDH activity was spectrophotometrically measured using a Cytotoxicity Detection kit (Roche Diagnostics, Mannheim, Germany) according to the manufacturer’s instructions. In this assay, MCF-7 cells were seeded into 96-well plates (1 × 104 cells/well) and allowed to attach for 24 h. The medium was replaced with the experimental medium containing eight concentrations of flumethrin. After 24 h, the medium was transferred into new wells, 100 µl of the supernatant was transferred into new wells, and 100 µl of the kit mixture consisting of the strain and enzyme solutions was added to each well. The well plate was incubated for 30 min in the dark, read using a microplate reader at a wavelength of 492 nm, and calculated according to the absorbance values. Cells treated with 1% Triton X-100 were used as a high control (100% lysis).
Determination of lipid peroxides
The lipid peroxidation induced by flumethrin after 24 h of incubation was determined by the reaction of thiobarbituric acid (TBA) with malondialdehyde (MDA), a product formed by lipid peroxidation. The assay was conducted as previously described by Heath and Packer. 21 Briefly, 1 × 106 cells per well were seeded in six-well plates and incubated with different flumethrin concentrations, then collected in Tris-HCl buffer (pH: 7.4). The cells were homogenized with 2 ml of 15 mM Tris-HCl buffer (pH: 7.4) and centrifuged at 10,000 rpm for 10 min at 4°C. After centrifugation, 100 µl of supernatant was mixed with 1 ml of 0.05% thiobarbituric acid (TBA) in 20% TCA and incubated in boiling water for 30 min. The solution was cooled immediately on ice to stop the reaction and centrifuged at 10,000 rpm for 10 min. The supernatant’s absorbance was recorded using a microplate reader. The amount of MDA in each sample was calculated from a standard curve and the MDA concentration was determined as MDA nanomoles per milligram of protein based on the total protein content. The results were normalized using the protein content. 22
Quantification of apoptosis using acridine orange and ethidium bromide double staining
Nuclear morphology changes and types of cell death were determined by acridine orange and ethidium bromide (AO/EtBr) staining. Flumethrin-induced cell death in the MCF-7 cells was quantified using AO/EtBr double staining as previously described by Pajaniradje et al. 23 MCF-7 cells were seeded into 24-well plates at a density of 2 × 104 cells/well. Then the cells were exposed to different concentrations of flumethrin (1, 10, 100, and 1000 µM) for 24 h. The cells were then collected by centrifugation at 1000 rpm for 5 min and washed twice with PBS. A mixture of acridine orange and ethidium bromide (60 µg/ml of AO and 100 µg/ml EtBr) was added to the cells and they were incubated for 5 min at 37°C. The percentages of viable, early apoptotic, late apoptotic, and necrotic cells were determined in 200 cells. The cells were observed under a Zeiss 200 fluorescent microscope (Carl Zeiss, Germany).
Gene expression assessments
The mRNA expression of BCL-2, BAX, TP53, CDKN1, TFF1, ESR1, ESR2, and PGR in the MCF-7 cells was detected by real-time PCR. The primer sequences are shown in Table 1. Briefly, after flumethrin treatment, the MCF-7 cells (1 × 106 cells/well in a six-well plate) were lysed and the total RNA extracted using a Quick-RNA MiniPrep kit (Zymo Research, Irvine, CA, USA). The quantity (ng/µl) and quality (260/230) of the RNA obtained was measured using a spectrophotometer (Thermo Scientific Multiskan GO) with a µ drop plate. The first-strand cDNA was synthesized using a high-capacity cDNA reverse transcription kit (Applied Biosystems). Three duplicates were displayed for each sample. Real-time PCR was performed with a QuantStudio 5 Real-Time PCR (Applied Biosystems) using LightCycler 480 SYBR Green 1 Master (Roche Applied Science, Mannheim, Germany). The PCRs were displayed with a fluorescence quantitative thermocycler using the following parameters: first, predenaturation at 95°C for 10 min, then 45 circles of denaturation at 95°C for 10 s and annealing at 60°C for 30 s, followed by dissociation and cooling to 40°C for 30 s. The expression levels were normalized to GAPDH and subsequently normalized to an experimental control group (The 2−ΔΔCt method). 24
List of real-time qPCR primer sequences.

Micronucleus assay by cytokinesis block (CBMN assay)
A CBMN assay 27 was used to detect the genotoxic effects of flumethrin on MCF-7 cells. The cells were subcultured on 30 mm petri dishes containing 20 mm coverslips (NEST Science Co., Ltd., China). Twenty-four hours after seeding, the medium was removed, and cells were treated with 1, 10, 100, and 1000 µM of flumethrin for 24 h. The cells were then washed with PBS and incubated in medium including cytochalasin B (at a final concentration of 2 µg/ml) for 24 h. After these treatments, the cells were washed with PBS and fixed with glacial acetic acid/methanol (1:3 v/v) for 15 min at 4°C. Then the cells were washed twice with PBS, and slides were mounted. After air-drying, the slides were stained by 5% Giemsa for 10 min. The slides were analyzed under a light microscope at 200× and 400× magnification. The number of binucleated cells containing a micronucleus (MN), nuclear buds (NBUDs), and nucleoplasmic bridges (NPBs) was counted in cytokinesis blocked cells based on 1000 cells per sample. When 500 cells were randomly counted, the nuclear division index (NDI) was calculated according to the following formula: 28

Genotoxicity by single cell electrophoresis/comet assay
A comet assay to evaluate cell DNA damage was conducted according to the methodology previously described by Singh et al. 29 and Tice et al. 30 In this method, 5 × 104 cells were seeded per well in a 24-well plate. At the end of the exposure period, the MCF-7 cells were mixed with 150 µL low-melting agarose (0.5%) and added to microscope slides pre-coated with normal melting agarose (NMA). The slides were immersed in a cooled lysing solution (2.5 M NaCl, 100 mM EDTA, 10 mM Tris, 1% Triton X-100, and pH 10) for 12 h at 4°C. All of the steps after lysis were conducted in the dark to prevent additional DNA damage by other factors. The slides were submerged in electrophoresis buffer solution (300 mM NaOH plus 1 mM EDTA, pH>13) for 20 min to allow the DNA to unwind. The electrophoresis conditions were 25 V and 300 mA for 20 min. A buffer solution was used to neutralize the slides after electrophoresis (0.4 M Tris, pH 7.5). Each slide received EtBr (60 µL and 30 μg/ml) with a cover glass. The DNA damage was estimated using a Zeiss 200 fluorescence microscope (Carl Zeiss, Germany), and 100 cells were counted on each slide and confirmed with an image analysis system (TriTek CometScore 1.5, 2006).
Statistical analysis
Statistically significant values were compared via one-way ANOVA followed by Tukey’s post-hoc test using GraphPad Prism v6.0 software (StatSoft Inc.). To determine the significance, the data distribution was tested for normality using the Shapiro-Wilk test. The results are presented as mean ±standard error of the mean (SEM), and p < 0.05 was considered statistically significant.
Results
Estrogenic activity of flumethrin
The estrogenic activity of flumethrin was evaluated using the estrogen-dependent MCF-7 cell growth assay (E-screen assay). Figure 2(A) shows the proliferative effects (PE) after 144 h of incubation with different flumethrin concentrations. Flumethrin concentrations of 0.1 µM and 1 µM exhibited proliferative effects compared with 10−9 M E2, and the RPE values were 77% and 80%.

Determination of flumethrin’s estrogenic activity. Analysis of flumethrin’s estrogenic activity on MCF-7 cells using E-screen assay (A). Expression of TFF1 (B), ESR1 (C), ESR2 (D), and PGR (E) exposed to three flumethrin concentrations in MCF-7 cells. Each bar denotes the mean (±SEM) of three independent experiments conducted in triplicate. *p < 0.05, **p < 0.01, and ***p < 0.001 compared with the control group.
RT-PCR was used to analyze the expression of specific estrogen-responsive genes (TFF1, ESR1, ESR2, and PGR) in the MCF-7 cells at three concentrations of flumethrin exposure. Similar to estrogen, a 0.1 µM concentration of flumethrin resulted in a significant increase in the expression levels of TFF1, ESR1, and PGR genes compared to the control (p < 0.05 and p < 0.01) (Figure 2(B) to (D)). Otherwise, no significant change in the ERβ expression levels was observed in any of the groups except the positive control group (Figure 2(E)).
Effects of flumethrin on cell viability, cytotoxicity, and MDA levels
The effect of different concentrations of flumethrin on the survival of MCF-7 cells measured via MTT assay is shown in Figure 3(A). A 24 h incubation period with flumetrin at increasing concentrations (100–2000 µM) reduced cell viability in a concentration-dependent manner compared with untreated cells (p < 0.01 and p < 0.001). The IC50 value was 1671 µM for flumethrin incubated in MCF-7 cells at 24 h.

Flumethrin-induced cytotoxicity and oxidative stress. MCF-7 cells were treated with flumethrin for 24 h. The viability of the cells was measured using an MTT assay and are represented as the percent survival (A). LDH activity after MCF-7 cells were exposed to various flumethrin concentrations (B). Effect of flumethrin on MDA content in MCF-7 cells (C). Each bar denotes the mean (±SEM) of three independent experiments conducted in triplicate. Significance at *p < 0.05, **p < 0.01, and ***p < 0.001. (*) Compared with control.
As shown in Figure 3(B), the LDH activities of MCF-7 cells were induced at the different concentrations of flumethrin. When the groups were compared, as in the LDH activity of cells, a significant dose-dependent increase was observed in the 1000 and 2000 µM flumethrin treated groups (p < 0.01 and p < 0.001).
The amount of MDA in the MCF-7 cells using spectrophotometry after flumethrin exposure is shown in Figure 3(C). The MDA level significantly increased in the MCF-7 cells incubated with the highest concentrations of flumethrin (1000–2000 µM) compared with the control group (p < 0.05 and p < 0.001).
Effects of flumethrin-induced apoptosis in MCF-7 cells by mitochondrial pathway
Apoptotic, necrotic, and viable cells exposed to flumethrin were counted under a fluorescence microscope. Flumethrin triggered morphological features related to apoptosis in a concentration-dependent manner (Figure 4). A high concentration of flumethrin exposure markedly increased the proportion of apoptotic cells (p < 0.001) as indicated by the presence of condensed or fragmented nuclei, whereas the control cells had round, uniformly colored nuclei (Table 2).
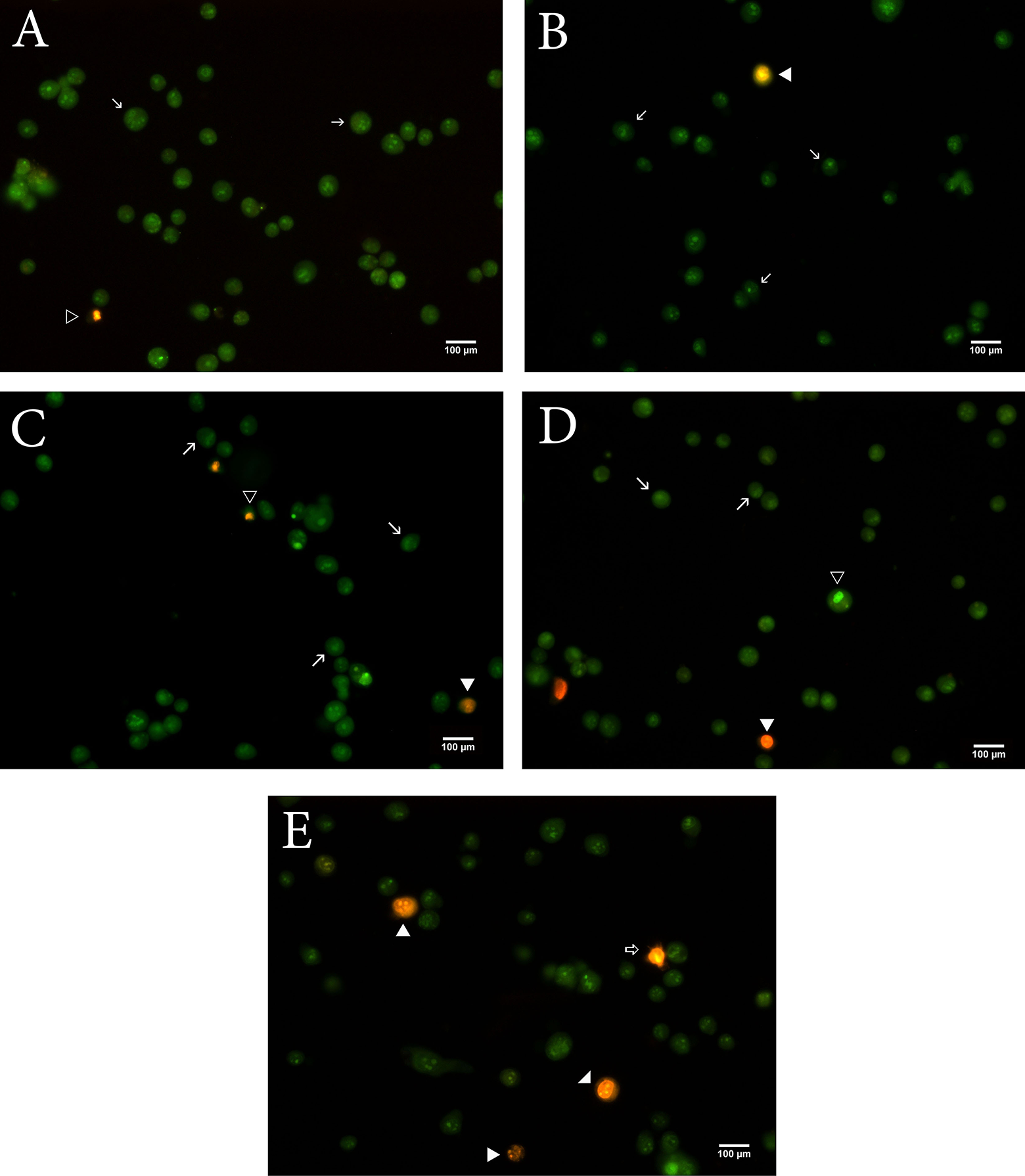
Images of fluorescent microscopic analysis of flumethrin-induced apoptosis in MCF-7 cells with AO/EtBr. Control cells are green (A). Flumethrin treated cells at concentrations of 1, 10, 100, and 1000 µM showing apoptotic cells, necrotic cells in orange/red; → viable cell, ▹ early apoptosis, ◂ late apoptosis, and ⇒ necrosis (B-D).
Percentages of viable, early apoptotic, late apoptotic, and necrotic cells after flumethrin treatment.

Data represent the mean ± standard error (SE) of three independent experiments conducted in triplicate for the five samples. *Indicates significant difference from the control, **p < 0.01, and ***p < 0.001.
After 24 h of flumethrin exposure to MCF-7 cells, the mRNA expression of selected genes was analyzed by quantitative real-time PCR (Figure 5(A) to (D)). In the cells exposed to flumethrin, the expression of BAX, TP53, and p21CDKN1 mRNA was significantly higher (p < 0.05 and p < 0.001) than in the untreated control cells only at the highest concentration (1000 µM), whereas the expression of BCL2 was significantly lower at the same concentration (p < 0.001).

Effects of flumethrin on BCL2 (A), BAX (B), TP53 (C), and p21CDKN1A (D) mRNA levels in MCF-7 cells after 24 h. Results are presented as the mean (±SEM). *p < 0.05, **p < 0.01, and ***p < 0.001 compared with control group.
Induction of micronucleus by flumethrin
The frequency of MN, NBUDs, and NPBs in the MCF-7 cells treated with increasing concentrations of flumethrin is shown in Table 3. Following flumethrin exposure, a concentration-dependent increase in MN was observed (p < 001). DNA damage was evident at all tested concentrations, and the NBUD frequency in the MCF-7 cells significantly increased in all of the experimental groups except the 1 µM concentration of flumethrin (p < 0.05 and p < 0.01). Moreover, higher concentrations (100 and 1000 µM) of flumethrin caused a significant increase in the NPB frequency in the MCF-7 cells compared with the control (p < 0.05 and p < 0.01). No significant differences were observed in the NDI values between all flumethrin treatments and the controls.
Effect of flumethrin on micronucleus, NBUDs, NPBs, and NDI in MCF-7 cells after 24 h.

Data represent the mean ± standard error (SE) of three independent experiments conducted in triplicate for the five samples. *Indicates significant difference from the control, *p < 0.05, **p < 0.01, and ***p < 0.001.
Effects of flumethrin on DNA strand breaks (comet assay)
DNA damage occurred in the MCF-7 cells treated with flumethrin for 24 h as indicated by the comet assay parameters Olive tail moment (arbitrary unit), tail moment (arbitrary unit), tail DNA (%), and tail length (µm) (Table 4). Flumethrin caused a statistically significant increase in the frequency of damage compared to the negative control sample at all concentrations except 1 µM (p < 0.01 and p < 0.001). This increase was similar in the positive control groups at a high concentration (1000 µM) after 12 h of treatment (Figure 6).
DNA damage by flumethrin in MCF-7 cells indicated by the various comet assay parameters.

Values represent mean ± S.E. of three independent experiments at each concentration.
Significance at *p < 0.05, **p < 0.01, and ***p < 0.001. Flu: flumethrin.

MCF-7 cells treated with different concentrations of flumethrin showing DNA damage (magnification 200×). Nuclei from untreated MCF-7 cells (negative control) (A). Nuclei from H2O2 (5 µM, positive control) treated MCF-7 cells showing DNA damage (B). Nuclei from 1, 10, 100, and 1000 µM flumethrin treated MCF-7 cells showing DNA damage (C-F).
Discussion
Pyrethroids including flumethrin are presently the most commonly used pesticides in agriculture and different veterinary applications, replacing pesticides that were more harmful to the environment.31,32 Although there are maximum residue limits (MRL) of flumethrin residue in ruminants, MRL have not been required for flumethrin residue in honey according to European Commission regulations. 33 The theoretical maximum daily flumethrin intake estimated by the EMEA 34 is 108 μg/day per person. Recent studies detected flumethrin residue on beeswax and honey.10–12 Although there are data on the animals it has not been investigated in humans after exposure to dietary absorption and elimination of flumethrin or metabolites. As stated in the JECFA evaluation report (2018), it has been shown that flumethrin is rapidly absorbed in rats, especially in the liver and plasma. Also, when applied topically, it has been reported to be present in all tissues including milk in dairy cows. There are few studies of flumethrin’s cytotoxic, apoptotic, or endocrine disrupting effects on mammalian cells.16,35–37 In addition, it has been shown by MTT and Neutral Red test that Unio Sp. have cytotoxic effects even at very low doses. 38 In this study, the estrogenic, cytotoxic, apoptotic, and genotoxic effects of flumethrin were determined on MCF-7 cell lines.
Recent studies show that pyrethroid group pesticides affect reproductive systems in many different pathways.39,40 Prior studies reported the estrogenic potential of pyrethroids via an E-screen assay using estrogen-dependent human breast cancer (MCF-7) cells.41,42 MCF-7 cells expressing estrogen receptors, endogenous estrogen-inducible markers such as progesterone receptor, ps2 protein, and the proliferative effects of estradiol and xenoestrogens on these cells were exerted via estrogen receptor-dependent and independent pathways.42,43 The estrogenic activity of various pyrethroids including flumethrin was also evaluated using transactivation in vitro assays. 16 Wielogorska et al. 16 tested 59 endocrine disrupting agents including pyrethroids for agonism and antagonism to human estrogen receptor via a reporter gene assay using a human mammary gland cell line. They found that flumethrin exhibited estrogenic activity with reported EC50 of 1.6 × 10−4 M. The present study showed that flumethrin could stimulate the proliferation of MCF-7 cells and upregulate the mRNA expression of PGR, ESR1, and TFF1. These results indicated that flumethrin has potential estrogenic activity. Flumethrin-induced TFF1, ESR1, and PGR gene expression and MCF-7 cell proliferation at nanomolar concentrations and maximally at 0.1 µM. The concentrations we used are similar to the amounts necessary to observe the estrogenic properties of various pyrethroids including permethrin, β-cypermethrin, and deltamethrin.41,44
This was in accordance with previous studies that described the cytotoxicity and genotoxicity of pyrethroids.45–50 Bifenthrin exhibited cytotoxic effect determined by MTT and LDH method and induced apoptosis in various cell lines.51,52 An in vitro study using MTT and a cytokinesis blocked micronucleus assay in lymphocytes showed that permethrin caused cytotoxicity and DNA damage. 46 Flumethrin has been reported to show toxic effects in a variety of experimental animals, causing oxidative stress, hepatotoxic effects, and inducing lipid peroxidation in rats.13,14,35 This study demonstrated that flumethrin exposure for 24 h increased cytotoxicity and lipid peroxidation in MCF-7 cells as shown by the significant induction of apoptosis. The effect of flumethrin’s cytotoxicity appeared at concentrations up to 100 µM. Concentrations from 0.1 to 100 µM had no effect on cell viability (MTT and LDH leakage) and lipid peroxidation. The present study also found that exposure to flumethrin-induced apoptosis significantly through a mitochondrial-dependent pathway in MCF-7 cells as indicated by the higher BAX, TP53, and p21 and lower BCL2 levels.
Since DNA is highly sensitive to chemicals, it is important to evaluate the effect of xenobiotics on DNA damage. The most commonly used techniques for demonstrating genotoxic damage are micronucleus and comet assays. 53 Prior studies investigated the genotoxic effect of pyrethroids. Pyrethroids exhibited different mechanisms of action for genotoxicity, such as induction of DNA strand breaks, chromosomal aberration (CA), and formation of micronuclei containing lagging chromosome fragments or whole chromosomes.46,53–55 However, with only one study assessing flumethrin, there are insufficient data on its genotoxic effects. Nakano et al. 36 found that flumethrin-induced chromosome and micronucleus aberrations in the bone marrow cells of mice. Flumethrin induced a reduction of the MI after the dermal treatment and the single i.p. exposure at all doses tested. 36 The present results revealed a significant induction of MN, NBUD, and NPB frequencies in MCF-7 cells exposed to flumethrin for 24 h, including low concentrations. The same treatment also caused DNA strand breaks.
However, some limitations should be noted. An important limitation of many in vitro toxicity methods is the lack of suitability for the in vivo situation related to the metabolism of xenobiotic chemicals. 56 Therefore, these findings with in vitro exposure cannot be considered as the entry of xenobiotics into the body. However, the results presented here are still important because they demonstrate the possible toxic effects of to flumethrin.
Conclusion
In conclusion, our study indicated that flumethrin demonstrated estrogenic activity and cytotoxic, apoptotic, and genotoxic effects in MCF-7 cell lines. The present results clearly suggested that flumethrin has estrogenic properties and may function as a xenoestrogen through a mechanism similar to that of E2. The results also showed that overall cell viability, LDH release, MDA activity, and apoptosis as endpoints of toxicity were strongly affected by the direct addition of up to 1000 µM of flumethrin in MCF-7 cells. Real-time PCR analyses demonstrated that flumethrin-induced apoptosis in MCF-7 cells at the highest 1000 µM concentration. This pesticide had the potential to induce genotoxicity even at lower concentrations. Flumethrin also has endocrine disrupting effects at concentrations far below estimated intake, genotoxic effects at non-cytotoxic levels, and toxic effects on mitochondrial activities, membrane integrity, and apoptosis. Considering the widespread use of flumethrin and its presence in the environment, more comprehensive studies should be conducted to understand the underlying environmental and human health risks.
Highlights
– Flumethrin reduced cell viability and proliferation in MCF-7 breast cancer cells.
– Flumethrin-induced apoptosis in MCF-7 cells by mitochondrial pathway.
– Flumethrin has estrogenic properties and may function as a xenoestrogen.
– Flumethrin had the potential to induce genotoxicity even at lower concentrations.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Istanbul University Scientific Research Projects (Project No. 23704).