Abstract
Tacrolimus (TAC) is the drug of choice in immunosuppressive therapy for organ transplantation; however, adverse effects are still a major concern. The current study aims to decipher the short-term exposure of TAC on rat hepatocytes in relation to activation of hedgehog (HH) signaling pathway. Time dependent study was conducted using primary rat hepatocytes treated with TAC (36 µM) for 6, 12, 24 and 48 h. Western blot analysis was performed using cell lysate in order to analyze the regulation of HH pathway proteins including HHIP, SMO, PTCH, IHH, SHH, and GLI transcription factors. The study revealed change in protein expression of HH signaling molecules with activation of HH pathway, due to downregulation of HHIP, and enrichment of HH ligands with activation of SMO and GLI transcription factors. It is therefore, concluded that short term TAC exposure leads to upregulation of HH pathway in liver, which may initially act to repair the liver damage but can worsen the condition in case of prolonged immunosuppressive therapy. This insight could lead to understand association of off target effects of immunosuppressive drugs and occurrence of other liver diseases in transplant patients when it comes to long term immunosuppressive therapy. These findings also illuminate a novel direction that use of HH inhibitor might provide a therapeutic strategy for immune suppression related liver disorders.
Keywords
Introduction
Tacrolimus (TAC) is one of the extensively used immunosuppressive drug used for prophylaxis of transplant patients. 1 It is the fermentation product of Streptomyces and belongs to the family of calcineurin inhibitor. Compared to other immunosuppressive drugs, use of TAC as an immunosuppressive agent has markedly reduced the risk of rejection but adverse effects of life-long immunosuppression are still a major concern. 2 Owing to narrow therapeutic index and clinical outcomes in transplant patients, myriads of adverse effects and contraindications of TAC has been reported which include hepatotoxicity, encephalopathy, diabetes mellitus, nephropathy and increased susceptibility to COVID-19, along with other infectious diseases like cancer, because of immunosuppression. However, paucity of information about the underlying molecular mechanisms in tandem with meager data is an obstacle to conclude the risks associated with adverse effects of this drug in case of prolonged course of treatment.3–7 Immunosuppressive drugs may also have off target effects due to impaired mitochondrial and T-cell functions which lead to production of reactive oxygen species and induction of apoptotic cell death. 8 Patients undergoing immunosuppressive therapy after organ transplantation often display obesity and hyperlipidemia owing to accumulation of triglycerides in liver, therefore, liver targeted management should be taken into account to avoid liver complications.9,10 Molecular explorations of signaling pathways involved in liver injury are paramount to inhibit the inflammatory response. Various signaling pathways have been studied to examine liver complications ascribed to lipid accumulation in hepatocytes, including TLR-4/MyD88/NF-κB and Nrf2/HO-1 pathway, TLR4/NF-κB and MAPK pathway and Hedgehog (HH) signaling pathway.11–13 HH pathway has a pivotal role in tissue development and growth but its deregulation can lead to injury related disorders and cancers, growing evidence from in vitro and in vivo studies have shown regulation of HH signaling in liver injury. 14 This pathway functions through ligands, namely; sonic hedgehog (SHH) and Indian hedgehog (IHH)), with some membrane receptors patched (PTCH) and smoothened (SMO), and glioblastoma (GLI) transcription factors. 15 HH pathway has a critical role in liver fibrosis and liver regeneration and the extent of liver injury is governed by the expression of HH ligands (SHH and IHH) and GLI transcription factors. 16 Healthy liver has low level of HH ligands and high level of hedgehog interacting protein (HHIP), which is a potent HH antagonist. 17 In liver injury, liver epithelial cells produce HH ligands which accumulate and negatively regulate production of HHIP. In the absence of HHIP, these ligands activate inflammatory response by triggering hepatic stellate cells and other progenitor cells through ligand-receptor interactions and activate downstream transcription factors, as a result, HH pathway is turned on. GLI proteins are a family of GLI-1, GLI-2 and GLI-3 transcription factors and have an important role in development, but, if deregulated may lead to defects. 18 Activation of SHH-GLI components have been reported in growth, apoptosis, proliferation and liver inflammation. 19 Till date, there is no published data reporting dysregulation of HH signaling pathway by the use of TAC or any other immunosuppressive drug in animal model. Therefore, this study was aimed to report activation of HH pathway due to short term exposure of TAC.
Materials and methods
Drug used
Tacrolimus monohydrate (Y0001926), molecular formula C44H71NO1 was purchased from Sigma Aldrich Co., Saint Louis, Missouri, USA (CAS. No. 109581-93-3). The drug was of European Pharmacopoeia (EP) standards category.
Experimental animal care
Male Sprague Dawley rats (250 ± 25 g) were raised and caged under standard conditions of light and dark cycles and fed normal rat chow & water ad libitum. Animal experiments were carried out with consent of Institutional Animal Care and Use Committee (IACUC protocol #2012A00000126-R1) and were duly in compliance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No.8023, revised 1978).
Isolation and culturing of rat hepatocytes for in vitro model of TAC exposure
Primary rat hepatocytes were isolated by ex vivo normothermic liver perfusion method, as described previously. 20 Number of hepatocytes yielded per liver were approximately 100–150 million with >95% viability by trypan blue exclusion. Primary rat hepatocytes were cultured in media (DMEM (Gibco) with 10% (v/v) penicillin-streptomycin (Invitrogen) and 10% FBS (Gibco)). The concentration of cells used was 2.5 × 105 cells in 6-well collagen (0.1 mg/mL of collagen type 1 solution (Sigma)) coated plate. After culturing for 24 h, hepatocytes were treated with 36 µM dose of TAC (Sigma, Cat. No. Y0001926) dissolved in culture media, followed by incubation of plates at 37°C in a humidified chamber containing 5% CO2 for 6, 12, 24 and 48 h time points. No drug was added in the control group.
Quantitation of protein from cell lysates
After the said experimental time points, cells from experimental groups were lysed using 1X RIPA buffer (Thermo Scientific, USA) supplemented with Protease inhibitor cocktail (Complete Mini, Roche) for 10 min on ice. The same protocol was followed for the control group. Protein quantification was carried out by Bicinchoninic assay (BCA assay) using Pierce® BCA Protein assay kit (Thermo Scientific, USA) as per the manufacturer’s instructions.
Western blot analysis for in vitro model of TAC exposure
Western blotting was performed to evaluate relative protein expression of HH pathway proteins including HHIP, SMO, PTCH, IHH, SHH, GLI-1, GLI-2, and GLI-3. 20 µg of protein from each group was used to run SDS-PAGE. Gel was transferred onto PVDF membrane (0.45 µM, Millipore, MA, USA) and western blot was performed. Membrane blocking was done in 5% skimmed milk followed by incubation with primary antibodies (List of antibodies used are provided in supplemental file). The blots were developed using horseradish peroxidase conjugated secondary antibody (Cell Signaling Technology, Beverly, MA, USA) for 3 h. Immobilon Western chemiluminescent HRP substrate (Millipore, Massachusetts, USA) was used to develop membranes and protein bands were visualized using the FluorChem™ HD-2 (Alpha Innotech, CA, USA) and quantified by Image J software after normalization with loading control, beta actin (β-actin).
Statistical analysis
Data analysis was done using one-way ANOVA followed by Tukey’s Post Hoc test by Graph Pad Prism 5 (San Diego, CA) and p > 0.05 was considered as statistically significant.
Results
Tacrolimus exposure modulates protein expression of hedgehog signaling pathway
Short term exposure of TAC resulted in dysregulation of HH signaling molecules with significant downregulation of HH antagonist and upregulation of ligands and transcription factors (Figure 1, 2). Overall, the results highlighted a significant upregulation of HH signaling molecules at 6, 12, 24 and 48 h time points.

Effect of short term tacrolimus exposure on protein expression level of HH components. Blot images for protein expression of HHIP, IHH, SHH, SMO, PTCH, GLI-1, GLI-2, GLI-3 and β-actin in control, 6, 12, 24 and 48 h treatment groups.

Effect of short term tacrolimus exposure on protein expression level of components of HH signaling pathway. Graphical representation for protein expression of HH components in control, 6, 12, 24 and 48 hours treatment groups, a. HHIP, b. IHH, c. SHH, d. SMO, e. PTCH, f. GLI-1, g. GLI-2, h. GLI-3. Data was normalized with the expression of a housekeeping gene, β-actin. Data is represented as Mean ± SEM (n = 6). *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001.
Short term exposure of TAC downregulates HHIP
Protein expression analysis revealed a significant downregulation of HHIP in TAC treated cells as compared to control group (Figure 2a). A significant difference was also observed among all treatment groups. However a marked significant downregulation was evident at 6 h treatment group.
Short term TAC exposure results in increased expression of HH ligands
Protein expression of HH ligands; SHH and IHH, profoundly increased in all treatment groups in case of IHH (Figure 2b) and at 6, 24 & 48 h treatment groups in case of SHH (Figure 2c). Also significantly increased expression was observed in between treatment groups. The maximum significantly increased protein expression was observed at 6 and 24 h in case SHH and IHH respectively.
Short term TAC exposure modulates differential expression of receptors SMO and PTCH
Receptor SMO showed an augmented expression in all treatment groups with maximum expression in 6 h treatment group as compared to the control group. When compared between the treatment groups, a significant change in protein expression was observed (Figure 2d). On the contrary, downregulation of receptor PTCH was observed in all treatment groups when compared to the control group (Figure 2e). Also, a significant change was evident among treatment groups with maximum significant downregulation at 6 h treatment group.
Short term TAC exposure activates GLI factors
Results highlighted marked upregulation of GLI-1-3 in all treatment groups, as compared to the control group. A maximum significant expression of GLI factors was observed at 24 h. Also, a significantly different expression profile was observed among all treatment groups (Figure 2f, g, h). Overall, data revealed a significant upregulation of GLI factors in experimental groups.
Discussion
With the use of immunosuppressive agents, although the risk of rejection has been reduced but adverse effects of life-long immunosuppression are still a major concern. 2 Growing evidences from in vitro and in vivo retrospective studies have shown regulation of HH signaling in liver injury. 14 Moreover, activation of SHH-GLI components have been reported in growth, apoptosis, proliferation and liver inflammation. 19 We hypothesized that use of TAC as an immunosuppressive therapy may lead to activation of HH signaling pathway. HH pathway is primarily activated during embryonic development, but recently, several studies support the fact that this pathway is reactivated in injured liver to repair or regenerate. Adult healthy liver has high level of HHIP and low level of HH ligands, accumulation of HH ligands cause SHH and IHH to bind with receptor PTCH and eventuate the release of SMO, which then activates downstream GLI transcription factors. 21
The current study revealed that TAC treatment induced upregulation of ligands SHH and IHH due to downregulation of HHIP, which in turn triggered activation of signaling events by ligand-receptor interactions and transcription of GLI factors (Figure 3). HHIP overexpression has been described in terms of decreased apoptotic proliferation in human liver 22 likewise, in the current study, high levels of HHIP in control group was notable as compared to TAC treated cells. Decreased expression of HHIP in 6 hours treatment group might be attributed to half-life and terminal elimination of TAC. Despite limited available information about the pharmacokinetics of this drug, it has been previously reported that the absorption of TAC is maximum within 1 to 2 hours following oral administration whereas, terminal elimination from blood takes about 12 hours. 23 Consistent to our findings, previous studies have demonstrated that enrichment of SHH and IHH ligand molecules perpetuate liver damage by upregulation of receptors and activation of SMO dependent GLI proteins. 18 SMO receptors are considered more potent in activation of GLI by SHH pathway. 16 Once the signaling cascade is activated, accumulation of SMO tend to degrade receptor PTCH, 24 which justifies downregulation of PTCH in current study. These evidences support the notion that downregulation of HHIP and enrichment of SHH and IHH in treatment groups are responsible for activation of GLI factors and may be a hallmark of liver injury. TAC induced activation of HH pathway may indicate initiation of liver repair associated with injury. GLI are the key players of HH pathway. Enhanced expression of GLI transcription factors have been previously reported to play a significant role in various diseases such as acute liver injury 13 which is in line with findings of this study. As GLI factors are known to maintain intrahepatic population and pro-fibrotic agents, therefore, their overexpression might lead to liver diseases. 25 Rao et al. has further shown a significant contribution of GLI-1 overexpression in CCL4 induced liver fibrosis mice model due to accumulation of extracellular matrix and proliferation of hepatic stellate cells. 26 Likewise, GLI-1 overexpression due to stress and activation of SHH signaling pathway in other models of liver injury induced by alcohol and diabetes have been reported in previous studies.27,28 This increased expression of GLI-2 has been attributed to augmented rates of proliferation and angiogenesis. 29 Similarly, SMO, PTCH and GLI-3 overexpression has been described in pathogenesis of acute liver injury followed by administration of D-galactosamine (D-GalN) and lipopolysaccharide (LPS) via HH and NFkB65 signaling pathway. 30
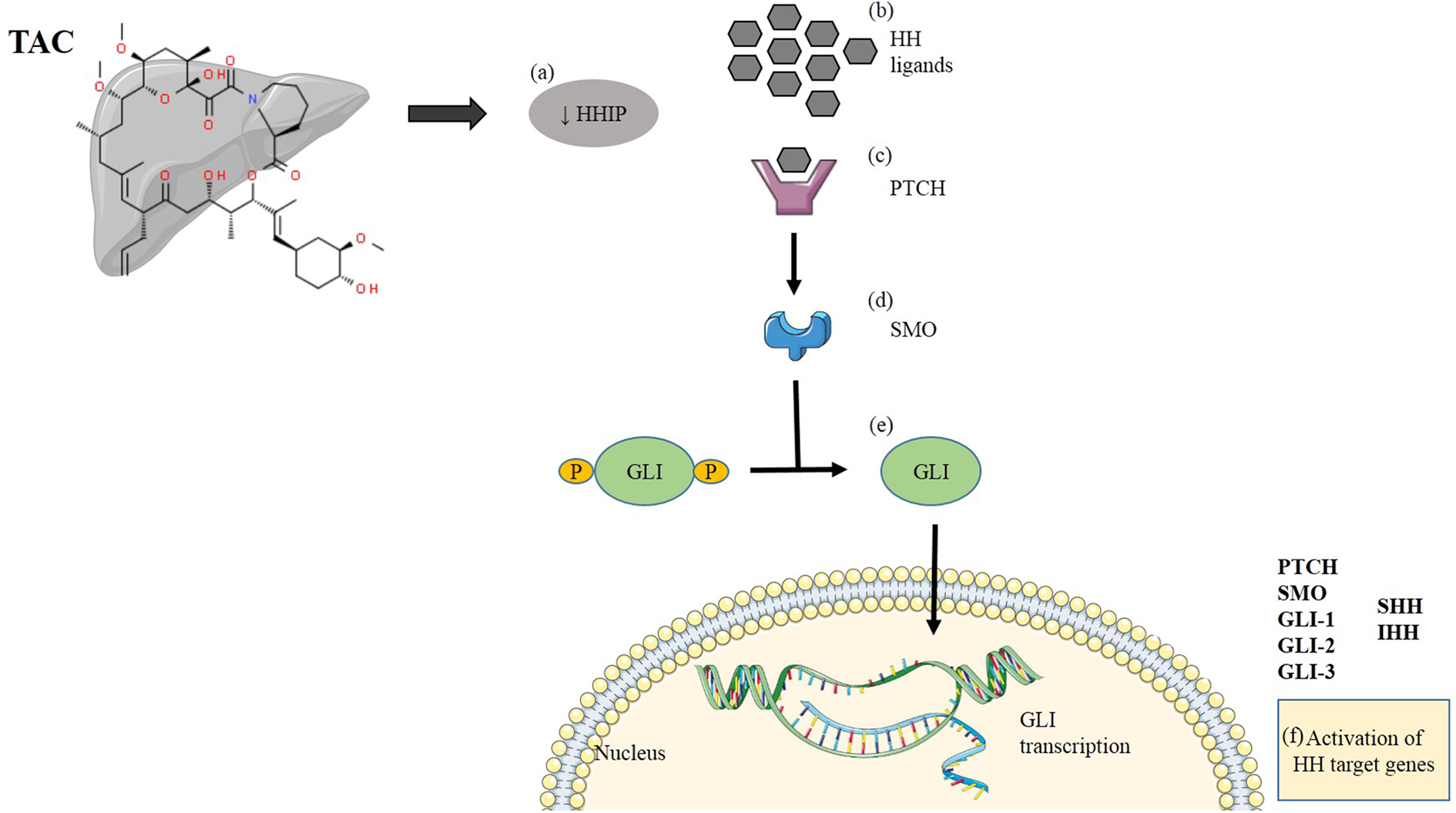
Interaction of TAC with HH signaling pathway: a, b) Short term exposure of TAC downregulates HHIP which causes enrichment of HH ligands. c, d) Overexpression of ligands cause binding of HH ligands with receptor PTCH, as a result of which receptor SMO is relieved, and proteolytic degradation of GLI protein takes place. e) GLI protein translocates into the nucleus where it is actively transcribed into GLI transcription factors. f) These GLI factors are responsible for activation of HH target genes.
Taken together, current study revealed effect of TAC on regulation of HH pathway and SHH/SMO dependent GLI activation. Thus, it can be inferred that short term exposure of TAC has a significant potential to activate HH pathway through upregulation of GLI factors. It is, therefore, proposed to take into account factors that mitigate adverse effects of immunosuppressive drugs. These findings may open new avenues in risk management of prolonged immunosuppressive therapy accompanying liver complications, concerning increased life expectancy and survival of transplant patients.
However, further research is needed to investigate other molecular mechanisms responsible for accumulation of HH components in relation to lifelong TAC treatment so as to minimize transplant related issues with improved therapeutic approaches, which is limitation of this study.
Supplemental material
Supplemental Material, sj-pdf-1-het-10.1177_09603271211017313 - Interaction of tacrolimus through hedgehog signaling pathway: An in vitro evaluation using rat hepatocytes
Supplemental Material, sj-pdf-1-het-10.1177_09603271211017313 for Interaction of tacrolimus through hedgehog signaling pathway: An in vitro evaluation using rat hepatocytes by N Fatima, N Sheikh, AR Satoskar, BK Jha, T Akhtar, A Tayyeb and I Ashfaq in Human & Experimental Toxicology
Footnotes
Acknowledgment
We highly appreciate the sincere cooperation of Dr. Sylvester Black, The Ohio State University, Columbus-OH and his lab members for providing rat hepatocytes, Higher Education Commission, Pakistan for providing an opportunity to accomplish the research work, and other colleagues for reading manuscript and critical discussions.
Data availability statement
The data used to support the findings of this study can be available upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
