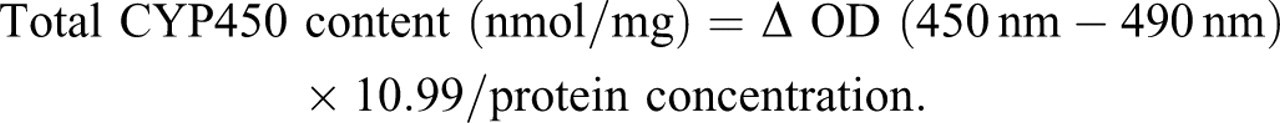Abstract
We aimed to examine the effect of an alkaloid extract of the roots of Rubus alceifolius Poir on liver damage and cytochrome enzymes, and underlying mechanism. Hepatotoxicity was induced in rats by treatment with carbon tetrachloride (CCl4). Rats were then treated with the hepatoprotective drug bifendate, or with low, medium, and high doses of an alkaloid extract from the roots of R alceifolius Poir. Both bifendate and alkaloid treatment decreased the increase in liver enzymes and cell damage caused by CCl4. Carbon tetrachloride treatment alone caused a decrease in total cytochrome P450 content, an increase in CYP2E1 and CYP3A1 messenger RNA (mRNA) levels, and an increase in CYP2E1 and a decrease in CYP3A1 enzymatic activity. Alkaloid treatment brought these concentrations and activities back toward normal. In summary, these results suggest that alkaloids from R alceifolius Poir may act to protect the liver through decreasing CYP2E1 enzymatic activity through decreasing its mRNA.
Introduction
Cytochrome P450 enzymes (CYP450) are a superfamily of mixed function oxidases. In the liver, they are found in the microsomal fraction and are the major enzymes in phase I drug metabolism. 1 Different isoforms of this enzyme family have different substrate specificities and different impacts on metabolism. CYP3A isoforms are the most important class and are involved in the metabolism of about 50% of drugs. CYP2E1, another isoform, is particularly important in toxicology because it converts a number of small molecular weight substances into toxins or carcinogens. 2 –4 These reactive and toxic intermediates formed during its action increase the formation of oxygen and hydroxyl free radicals 5 that damage cellular structure and cause hepatotoxicity. 6,7 In addition, liver damage has been shown in some studies to alter the activity of CYP450 enzymes. 8
Rubus alceifolius Poir belongs to the Rubus genus of the Rosaceae plant family. Its roots and leaves have a plain flavor and sweet taste, and it has been used as an herbal medicine in applications such as diuresis, detoxification, and bruise healing. It is also widely used to treat various types of hepatitis by the local population in Anxi, Southern Fujian. Previous studies by the present research group have shown R alceifolius Poir to have very good hepatoprotective effects. 9 –12 However, its mechanism of action and effect on drug metabolic enzyme concentrations in the liver is unknown. The study reported here aims to investigate the effect of total alkaloids extracted from R alceifolius Poir on both the messenger RNA (mRNA) expression and the enzyme activity of CYP2E1 and CYP3A1, and thus to provide insight into the mechanism for the hepatoprotective effects of these alkaloids. In this study, liver damage was produced with carbon tetrachloride (CCl4), and the effects of bifendate (a clinically used hepatoprotective drug) and low-, medium-, and high-dose alkaloid treatment on biological markers of liver damage, on total CYP450 enzymes, and on CYP2E1 and CYP3A1 mRNA and enzyme activity were compared.
Materials and Methods
Animals
In all, 60 Sprague Dawley rats 180 to 220 g (30 males and 30 females) were provided by Shanghai Experimental Animal Co Ltd, China, and allowed to acclimate in our animal facility for 5 days before use. The study was approved by the institutional review board of the Laboratory Animal Center of Fujian University of Traditional Chinese Medicine. All experiments were carried out under the guidelines of the Laboratory Protocol of Animal Handling of our college.
Preparation of Alkaloids and Assay of Total Alkaloid Content
The roots of R alceifolius Poir were collected from Anxi of Fujian Province, identified and authenticated by experts in our College, and extracted the alkaloids as follows. 13 The herb powder (1 g) was extracted with 50 mL chloroform:methanol:ammonia solution (15:4:3) for 2 hours in an ice bath, sonicated for 30 min, brought to room temperature and filtered. The filteed solution was collected and dessicated. The resultant residue was dissolved by 2 mL of 2% sulfuric acid solution and filtered. The filter paper and residue were re-washed with 2 mL of 2% sulfuric acid solution and with buffer solution (pH 3.6). Buffer was then added to make a final volume of 50 mL, and the solution saved for future use. Acid dye colorimetry was used to measure total alkaloid content. Total alkaloid content was 0.81 mg of alkaloid per gram of initial herb powder.
Reagents and Equipment
Reagents
Carbon tetrachloride was obtained from Chinese United Chemical Reagent Co, Ltd (Shanghai, China), erythromycin (a CYT3A1 substrate) from Shenggong Bio-Engineering Technology Co (Shanghai, China), aminopyrazolone morpholine (a CYT2E1 substrate) from Shanghai Reagent Factory (Shanghai, China), NADPH from Roche (Basel, Switzerland); and Trizol from Invitrogen (Carlsbad, California). Bifendate (dimethyl diphenyl bicarbonate) pills were purchased from IMC Pharmaceutical Co, Ltd (Zhejiang, China) and diluted to 1% concentration for application.
Alanine aminotransferase/Glutamic-pyruvic transaminase (ALT/GPT) and aspartate aminotransferase/Glutamic-oxaloacetic transaminase (AST/GOT) detection kits were obtained from Jiancheng Bio-engineering Institute (Nanjing, China), Bradford protein quantification and detection kit from Shenggong Bio-Engineering Technology Co, Ltd (Shanghai, China), deoxyribonucleoside triphosphate (dNTP) and reverse transcription (RT) Kit from MBI Fermentas Inc (Glen Burnie, Maryland), and Taq polymerase and RNAse inhibitor from TaKaRa Biotechnology (Otsu, Shiga, Japan). Primers were synthesized by Shanghai Handsome Bio Co, Ltd (Shanghai, China). Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining kits and related reagents were purchased from Maixin Ltd (Fujian, China).
Equipment
The following instruments were used: Du650 Spectrophotometer and 64R High-Speed Refrigerated Benchtop Centrifuge (Beckman, USA), BT 224 semiautomated spectrophotometer (Biotechnica, Italy), PCR-9600 (Perkon-Elmer, USA);GEL DOC 2000 gel imaging system (Bio-Rad), Sanheng multipurpose electrophoresis instrument (Beijing 61 Instrument Factory, China).
Animal Protocol, Drug Treatment, and Tissue Preparation
Animal protocol
In all, 60 rats were randomly divided into the following groups (5 males and 5 females in each group): saline, liver damage, and bifendate groups, and low-, medium-, and high-dose alkaloid treatment groups. Rats were given intraperitoneal (ip) injections of 0.2 mL saline (saline group), or 0.1 mL/100 g body weight of 99.5% CCl4 ip (liver damage, bifendate, and alkaloid groups) in order to induce liver damage. After a 24-hour period to allow the biochemical changes seen in CCl4-induced liver damage to appear, 8 rats were given intragastric administration of saline (saline and liver damage groups), 0.1g/kg body weight of 1% bifendate (bifendate group), 9,10 or alkaloid in a dose of 0.36 g/kg per d, 0.72 g/kg per d, or 1.44 g/kg per d (low-, medium-, and high-dose alkaloid groups, respectively). These doses were given once daily for 7 days.
Tissue and microsome preparation
After the last drug or saline treatment, rats were fasted (but given water) for 24 hours and then sacrificed. For measurement of indicators, 5 mL blood samples were collected, centrifuged, and the serum removed and stored at −20°C. The liver was removed, cleansed with cold saline, divided into 2 pieces, and stored at −80°C. A small piece of tissue from the central part of the right lobe (about 0.5 cm 3 ) was fixed in 10% formalin immediately for routine sectioning, hematoxylin and eosin staining, and histopathological examination.
Liver microsomes were prepared by differential centrifugation, suspended in Tris-HCl buffer (1 mL/g), and stored at −70°C until further use.
Measurement Methods
Determination of transaminase activity
Serum AST and ALT activities were measured with a
Histopathological examination of liver tissue
Morphological change in liver tissue was examined under a light microscope. Pathological changes in liver tissue were divided into 5 grades, as follows (Figure 1 ): normal cells, “−”; spotty necrosis, “+”; focal necrosis, “++”; necrosis in <one third of the cells in a lobule, “+++”; and necrosis in >one third of the cells in a lobule, “++++.”
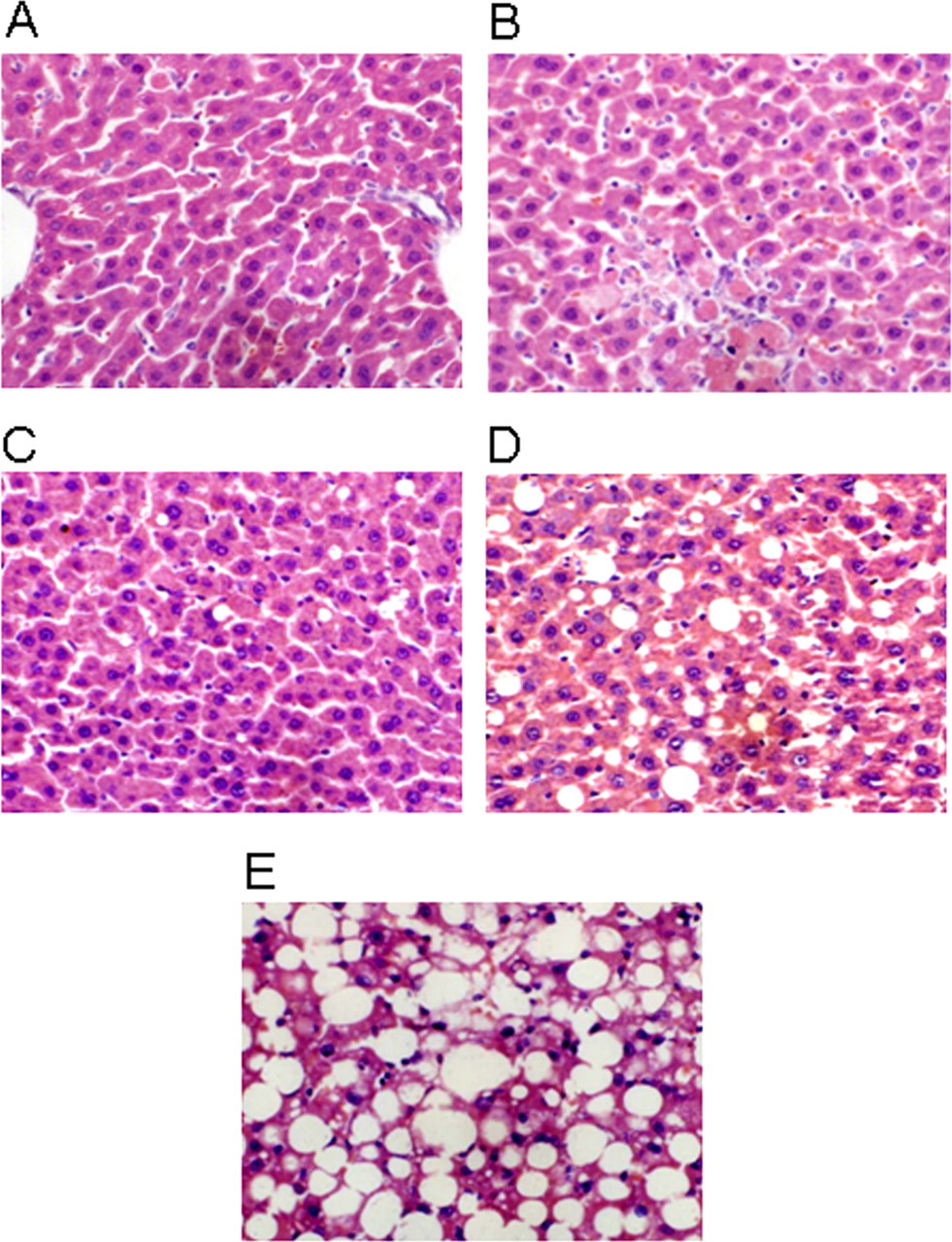
Grading system used to assess hepatic histopathological damage. (A) “−” normal liver cells; (B) “+” spotty necrosis; (C) “++” focal necrosis; (D) “+++” necrosis in less than one third of the cells in a lobule; (E) “++++” necrosis in more than one third of the cells in a lobule.
Measurement of microsomal protein concentration
Microsomal protein concentrations were measured with the Bradford Protein Assay Kit, according to the manufacturer’s instructions.
Quantification of total CYP450 enzyme levels
The concentration of total CYP450 enzymes was measured with a modified method of Zheng et al.
14
To 2 mL of buffer, 100 μL of microsome suspension and 0.5 mL of 10% sodium dithionite solution were added, mixed immediately, loaded into a colorimetric cup, and filled with CO for 30 seconds. A second 100 μL aliquot of the suspension was added to 2 mL of buffer, immediately mixed, and loaded into another colorimetric cup to serve as control. Optical density (OD) values at 450 nm and 490 nm wavelengths were recorded; and the CYP content calculated according to the following formula:
Measurement of CYP2E1 and CYP3A1 enzymatic activity
Aminopyrazolone morpholine-N-demethylase activity was used to measure CYP2E1 activity, 15 and erythromycin demethylase activity was used to measure CYP3A1 activity. To measure CYP2E1 activity, 20 mg/mL aminopyrazolone morpholine-N-demethylase was added to microsomes suspended in 0.02 mol/L Hepes buffer and the solution was kept at 37°C in a water bath for 3 minutes; 0.01 mL of 0.1 M NADPH was then added and the solution was kept in 37°C water bath for 30 minutes. The next steps were the same as those performed in making a formaldehyde standard curve. To measure erythromycin demethylase activity, the same procedure was used except for replacing aminopyrazolone morpholine-N-demethylase with 0.004 mol/L erythromycin demethylase. Both activities were calculated as formaldehyde production compared to a formaldehyde standard curve.
Expression of target gene mRNA
The expression of CYP3A1mRNA and CYP2E1mRNA was measured by reverse transcription ploymerase chain reaction (RT-PCR). The amplified product was separated by 1.5% agarose gel electrophoresis; and the band analyzed with a gel image processing system. β-actin was used as a housekeeping gene. The primers used are shown in Table 1 .
Primers for CYP3A1, CYP2E1, and β-Actin

Apoptosis index
The apoptosis index (AI) was determined by TUNEL staining (in situ end labeling, performed with the TUNEL detection kit, according to the kit instructions). The proportion of apoptotic cells in each group was recorded to determine the AI. After staining, 5 high-power fields (×400) were randomly selected in each slide and the average proportion of positive cells in each field counted and classified into 5 grades: grade 0: <2%, grade 1: <3% to 9%, grade 2: <10% to 19%, grade 3: <20% to 39%, and grade 4: >40%. 16
Statistical Analysis
For continuous variables, data are shown as median and interquartile range (IQR). The comparisons between the 6 groups were performed using the Kruskal-Wallis test, and the comparisons between 2 groups by the Mann-Whitney U test. For categorical variables, data are presented by number and percentage. The associations between categorical variables were tested using Fisher exact test. All statistical hypothesis tests were set with a significance level of .05. Statistical analyses were performed using Statistical Package for the Social Sciences (SPSS) 15.0 statistics software (SPSS Inc, Chicago, Illinois).
Results
Effect of CCl4, Bifendate, and Alkaloids on Biochemical and Histological Liver Damage
Table 2 shows weights, liver enzymes, and degree of liver pathology, and Figure 2A shows the AI, in the 6 groups of rats.
Summaries of Weights and Liver Function for the Rats in the 6 Groups
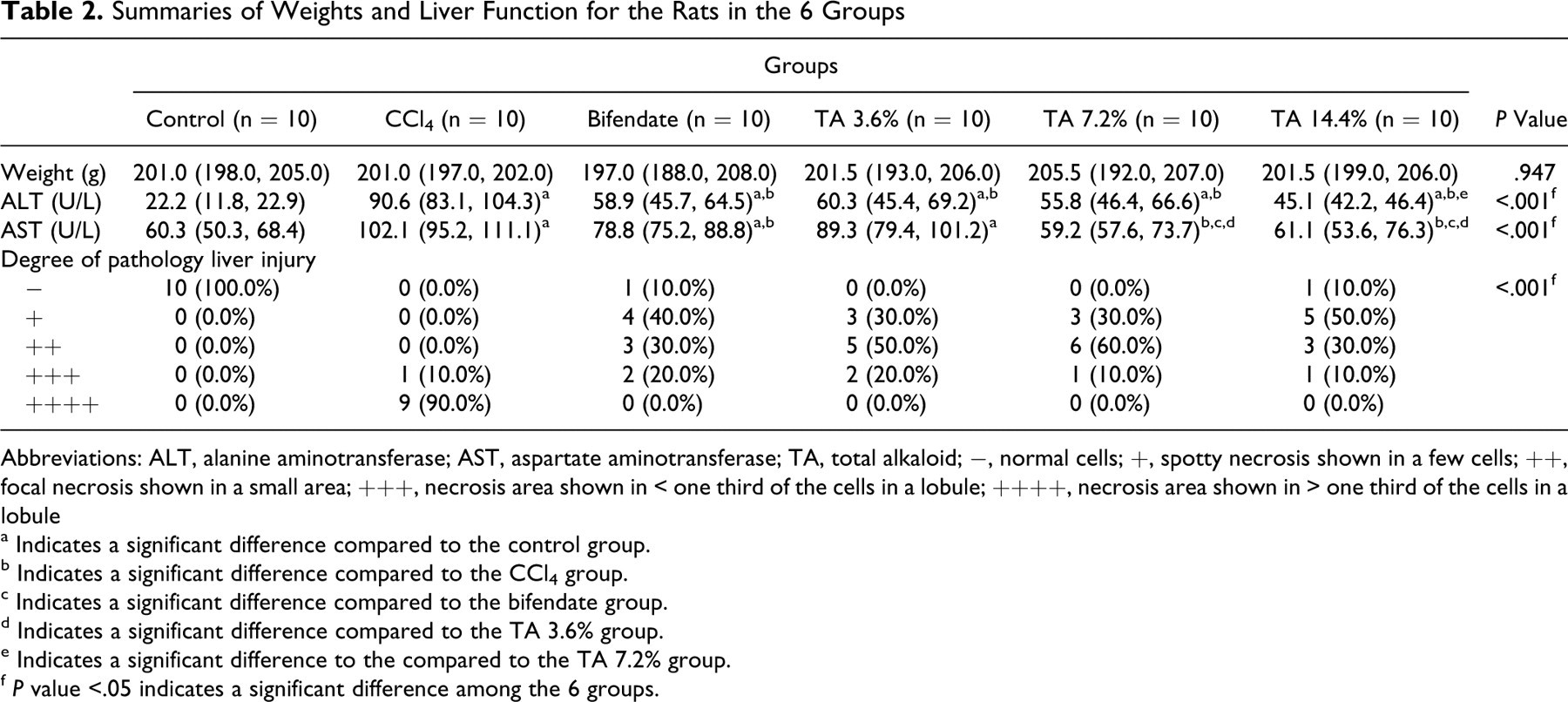
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; TA, total alkaloid; −, normal cells; +, spotty necrosis shown in a few cells; ++, focal necrosis shown in a small area; +++, necrosis area shown in < one third of the cells in a lobule; ++++, necrosis area shown in > one third of the cells in a lobule
a Indicates a significant difference compared to the control group.
b Indicates a significant difference compared to the CCl4 group.
c Indicates a significant difference compared to the bifendate group.
d Indicates a significant difference compared to the TA 3.6% group.
e Indicates a significant difference to the compared to the TA 7.2% group.
f P value <.05 indicates a significant difference among the 6 groups.
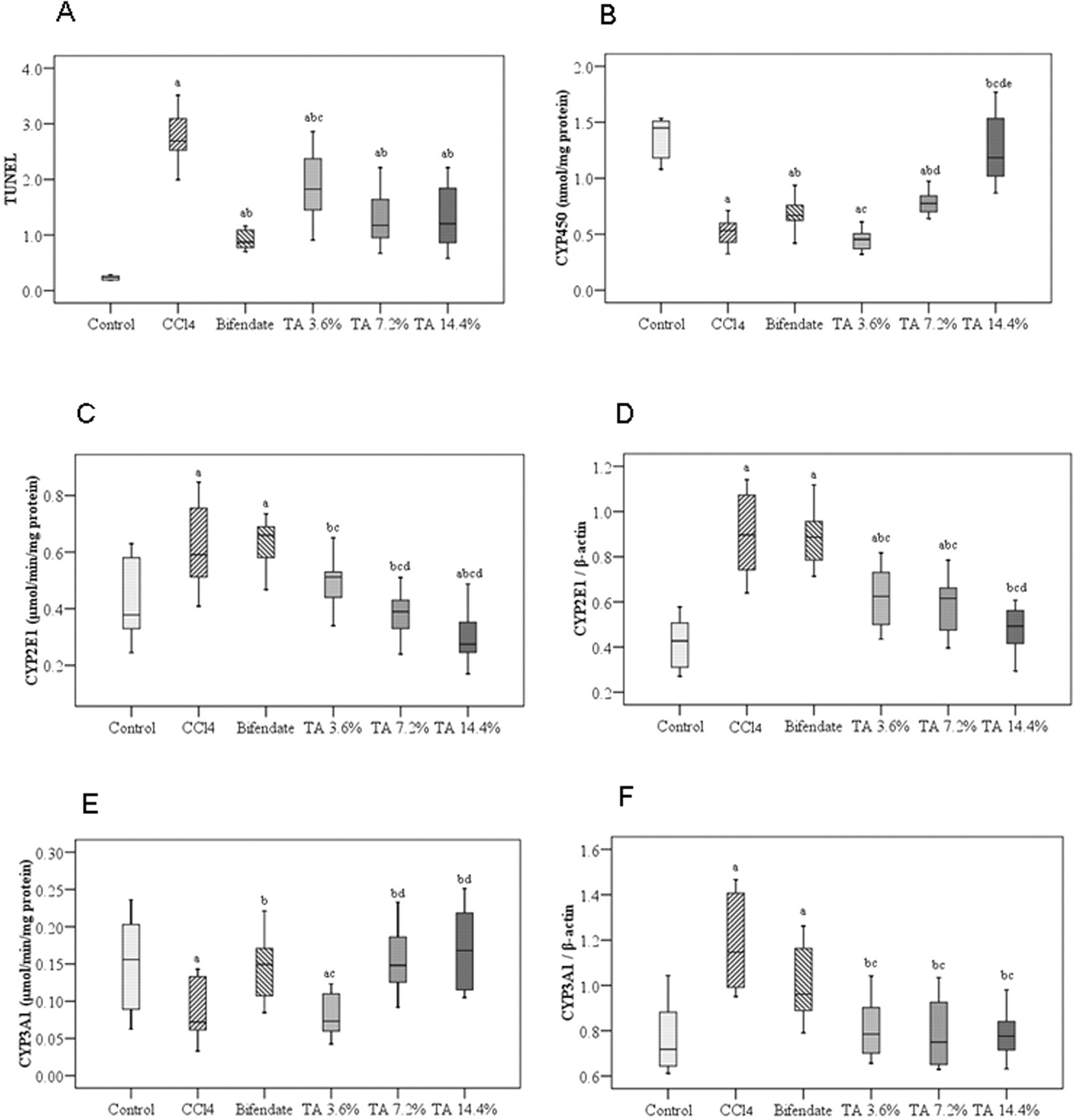
Box plots for apoptosis index (TUNEL assay), CYP450 concentration, CYP2E1 and CYP3A1 enzymatic activity, and mRNA expression in the 6 experimental groups. (A) apoptosis index (TUNEL assay); (B) CYP450 concentration; (C) CYP2E1 activity; (D) CYP2E1 mRNA expression; (E) CYP3A1 activity; (F) CYP3A1 mRNA expression. TA indicates total alkaloid; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling; a, significant difference compared to control group; b, significant difference compared to CCl4 group; c, significant difference compared to bifendate group; d, significant difference compared to TA 3.6% group; e, significant difference compared to TA 7.2% group.
Liver enzymes
Serum ALT and AST levels in the liver injury group (CCl4 treatment alone) were significantly elevated over those of the control group (saline treatment). When bifendate was given after CCl4 treatment, ALT and AST levels were significantly reduced compared to those in the liver injury group, but remained significantly higher than those of the control group. Alkaloid administration caused a dose-dependent decrease in both ALT and AST levels from those seen in the liver injury group. Aspartate aminotransferase levels were decreased to control levels by both medium- and high-dose alkaloid. However, ALT levels, although decreased from the levels in the liver damage group, were still twice those of the control group at the highest alkaloid dose used.
Liver histology
Histological examination showed CCl4 treatment to produce severe acute liver damage (Table 2). While no control rats showed histological signs of liver damage, 9 of the 10 rats in the CCl4-treated, liver damage group were in the highest damage category (> one third of the cells necrotic). No bifendate or alkaloid treated rat had liver damage of this severity. Both types of treatment lessened the liver damage caused by CCl4, and the effect of alkaloid treatment was dose-dependent.
Apoptosis
The TUNEL assay records DNA fragmentation, an indication of apoptosis and cell death. As expected, CCl4 treatment produced a significant increase in the AI obtained from this assay. Bifendate and alkaloid treatment both decreased the degree of DNA fragmentation caused by CCl4, but not to control levels. (Figure 2A)
Effect of CCl4, Bifendate, and Alkaloid on CYP450, CYP2E1, and CYP3A1
The results described above show CCl4 treatment to cause substantial acute liver damage and both bifendate and alkaloid treatment to alleviate this damage, although not completely. Figures 2 and 3 show the effects of CCl4, bifendate, and alkaloid on liver cytochromes.
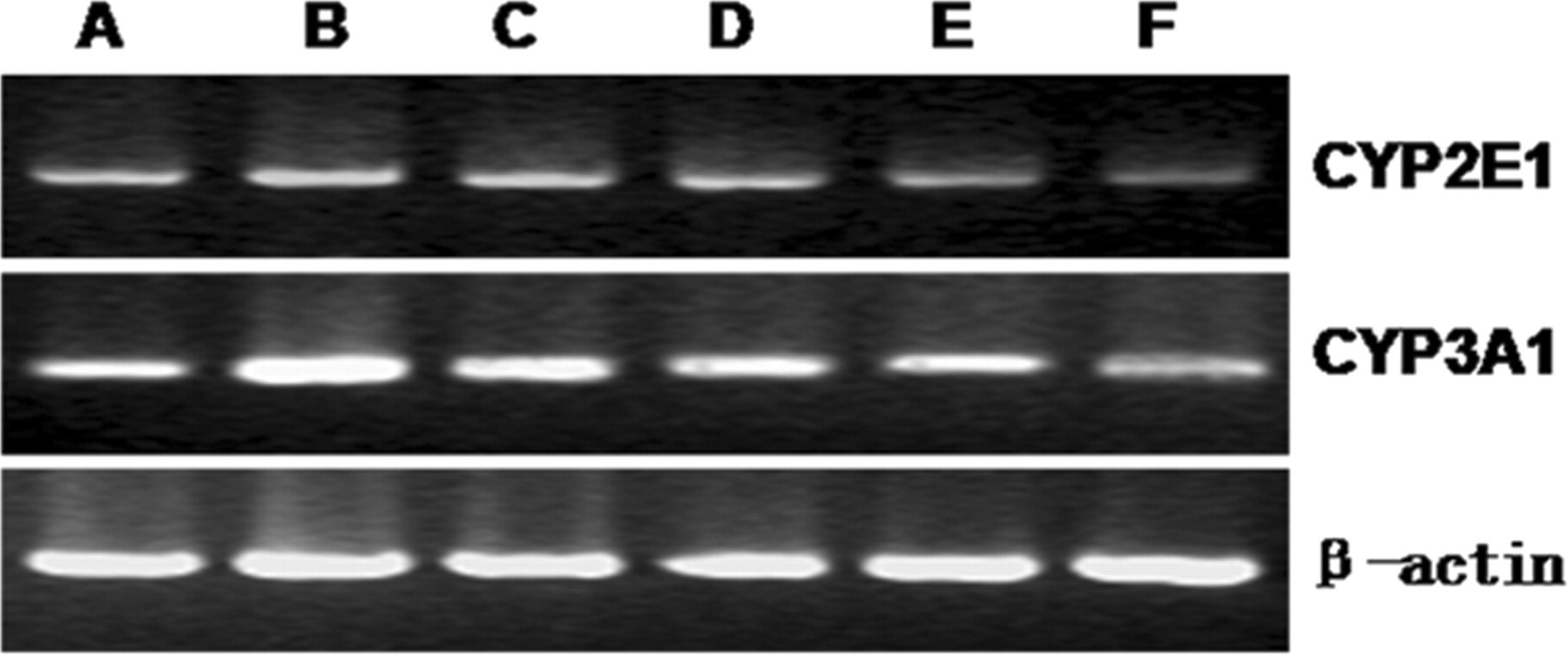
Expression of CYP2E1 and CYP3A1 mRNA, shown by electrophoresis, is increased in liver damage and decreased by alkaloid treatment. A indicates control group; B, liver damage group; C, biphendate group; D, alkaloid low-dose group; E, alkaloid medium-dose group; F, alkaloid high-dose group
CYP450 concentration
Carbon tetrachloride treatment decreased total CYP450 enzyme levels (Figure 2B). Bifendate increased the levels of this enzyme in CCl4-treated animals, but not completely back to control levels. Alkaloid treatment, although at the low dose did not change CYP450 levels over those seen with CCl4-treatment alone, at the high dose raised these levels completely back to those seen in normal, saline-treated rats.
CYP2E1 enzymatic activity and mRNA levels
Although CCl4 treatment decreased total CYP450 protein levels, this treatment increased the enzymatic activity of CYP2E1 (Figure 2C). Bifendate had no effect on this increase. Alkaloids, in contrast, reversed the increase in a dose-dependent manner to such an extent that CYP2E1 enzymatic activity was not only reduced to levels lower than those of the liver injury group, but at the highest dose, to levels significantly lower than those seen in the control group. CYP2E1 mRNA expression showed a pattern similar to that seen in enzymatic activity (Figure 2C and D, Figure 3). Carbon tetrachloride treatment significantly increased mRNA; bifendate had no effect; and alkaloid treatment caused a dose-dependent decrease to levels, at the highest dose, that were similar to control (Figures 2C, Figure 3).
CYP3A1 enzymatic activity and mRNA levels
Although CYP2E1 enzymatic activity and mRNA levels changed in a similar direction during CCl4, bifendate, and alkaloid treatment, CYP3A1 enzymatic activity and mRNA levels changed in opposite directions (Figure 2D and E, Figure 3). Carbon tetrachloride treatment significantly increased CYP3A1 mRNA levels, but significantly decreased its enzymatic activity. Alkaloid treatment completely reversed the changes made by CCl4, that is, alkaloid increased enzymatic activity and decreased mRNA, and in doing so brought both parameters back to normal levels. Bifendate caused a similar, but smaller, reversal of CCl4-induced changes in enzymatic activity and mRNA.
Discussion
We have shown in this study that an extract of alkaloids from R alceifolius Poir blocks the increase in CYP2E1 mRNA and enzyme activity that occurs in liver injury caused by CCl4, a classical model used for drug screening and the study of hepatitis. This action is likely to be the major mechanism by which these alkaloids produce their known hepatoprotective effects.
Reactive intermediates formed during the CYP2E1-induced metabolism of CCl4 are responsible for the increase in reactive oxygen species (ROS) that causes the liver damage. In the present study, we found, as have others, 4,17,18 that although total CYP450 protein was decreased during CCl4-induced liver damage, CYP2E1 activity was increased. Decreased antioxidant capacity also occurs in CCl4-induced liver injury. Thus, both an increased ability to generate ROS and a decreased ability to detoxify these species are involved.
Drug-induced liver injury is usually caused by excessive formation of ROS. These ROS cause apoptosis, an inflammatory response, and oxidative stress. Oxidative stress, among other actions, increases lipid peroxidation. 19,20 Our previous studies have shown that extracts of R alceifolius Poir effectively inhibit the formation of ROS, inflammatory cytokines, and lipid peroxidation. 9 –12,21
We have also reported previously that alkaloid extracts of this plant decrease both histological and biochemical signs of hepatic injury, 22 and more recently have reported that alkaloid extracts decrease the mRNA of 2 P450 isoforms, CYP 2E1 and CYP 3A1. 23 In the current study, we wished to examine the enzyme activity of CYP2E1 and CYP3A1 in addition to mRNA level and the evidence of alkaloid protective action against liver damage, and thus provide a comprehensive picture of Rubus alkaloid actions and the underlying mechanism responsible for them.
In searching for a causative mechanism, we found Rubus alkaloids to prevent both the increases in CYP2E1 mRNA and the increases in CYP2E1 activity seen in CCl4-induced liver damage, thus providing a likely explanation for their hepatoprotective action. An effect of Rubus alkaloids on CYP2E1 activity has not been previously reported. A recent report on treatment of rats with iridoid glucosides from Boschniakia rossica, an antioxidant and anti-inflammatory herbal medicine, stated that CCl4 treatment decreased CYP2E1 protein and activity and iridoid glucosides increased both. 24 And, negundeside, another herbal extract, has been reported to have no effect on CYP2E1, but instead, to increase antioxidant activity, and thus to act by an alternate mechanism for hepatoprotection. 25
Our data suggest that the Rubus alkaloids are hepatoprotective through their action in decreasing CYP2E1 mRNA and therefore CYP2E1 activity. But the actions of CCl4 and of these alkaloids on the other isoform studied, CYP3A1, are a puzzle, for CCl4 increased CYP3A1 mRNA but decreased its activity, and Rubus alkaloids decreased CYP3A1 mRNA and increased its activity. One possible explanation is that some toxic product from the increased CYP2E1 metabolic activity or perhaps from an initially increased CYP3A1 activity is an inhibitor of CYP3A1. This difference between the effect of CCl4 on CYP2E1 and CYP3A2 should be investigated further, keeping in mind that compounds that change specific CYP isoform activity in rats may not do so in humans, or may do so by different mechanisms.
In interpreting our results, one must remember that a change in the synthesis of cytochrome mRNA is not the only way that liver damage due to ROS is controlled. Stabilization of the existing mRNA can play a role. Alteration in 1 of the 2 pathways for cytochrome degradation (the slow lysosomal pathway or the faster proteosomal pathway) may also change the level of cytochrome activity. 26 In addition, changes in the levels of antioxidant substances are also involved in control of damage from ROS.
The results of the present study may be meaningful to guide proper drug treatment and to understand drug interactions and their metabolism inside the body. It helps to clarify the molecular mechanisms and targets for traditional Chinese medicine and provides new ideas and methods for the modernization of Chinese medicine study. We have recently performed a study to isolate and identify the active constituents of R alceifolius Poir extracts and found the constituent 1β-hydroxyeuscaphic acid to have the strongest effect against CCl4-induced hepatotoxicity.27 But no investigation as to the mechanism has yet been performed. And drug metabolism and processing of biological effects in the body is a very complex subject and requires much further study.
Footnotes
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The project was supported by the Nature Science Foundation of Fujian Province of China (No.2006J0109); The Project Sponsored by Open Fund of Fujian Key Laboratory of Integrative Medicine on Geriatrics (Fujian University of TCM)(No. 2008J1004-19 and 2008J1004-17); Developmental Fund of Chen Keji Integrative Medicine (CKJ2008054 and CKJ2008056