Abstract
Hepatocellular carcinoma (HCC) is the most common form of liver malignancies worldwide. Alogliptin is an anti-diabetic that may have effective anticancer properties against many types of malignancies. Taxifolin is a flavonoid that has potent antioxidant, and anti-inflammatory properties. The objective of this study was to explore the impact of alogliptin and/or taxifolin on diethyl nitrosamine-induced HCC in rats. One hundred male Wistar rats were divided into five equal groups as follows: Control; HCC; HCC + Alogliptin; HCC + Taxifolin; and HCC + Alogliptin + Taxifolin group. The survival rate, liver function tests, tissue antioxidant enzymes, malondialdehyde (MDA), nuclear factor (erythroid derived 2)-like 2 (Nrf2), transforming growth factor beta 1 (TGF-β1), interleukin 1 alpha (IL-1α), and toll-like receptor 4 (TLR4) were measured. Also, hepatic caspase 3, caspase 9, beclin-1, and c-Jun NH2-terminal kinase (JNK) in addition to serum alpha-fetoprotein (AFP) and α-L-Fucosidase (AFU) were assessed. Specimens of the liver were subjected to histopathological examination. Alogliptin and/or taxifolin induced significant improvement of liver function tests with significant increase in the survival rate, tissue antioxidant enzymes, Nrf2, caspase 3, caspase 9, Beclin-1 and JNK activities associated with significant decrease in serum AFP and AFU, tissue MDA, TGF-β1, IL-1α and TLR4 expression compared to HCC group. These results were significant with taxifolin/alogliptin combination when compared to the use of each of these agents alone. In conclusion, taxifolin/alogliptin combination might be used as adjuvant therapy for attenuation of HCC.
Introduction
Hepatocellular carcinoma (HCC) represents the most common form of liver malignancies worldwide and is reported to be the second leading cause of death from diseases characterized by malignant transformation. 1 The exact etiological factors that contribute to the development of HCC are not yet fully elucidated. However, induction of formation of reactive oxygen species (ROS) was suggested to enhance the inflammatory cascade in the hepatic tissues leading to affection of the apoptotic pathways with subsequent malignant changes. 2 Surgical removal is the primary treatment of HCC, although this option had proven efficacy only in early non-metastasizing tumors. 1 As a result, the ongoing studies are directed towards the search for alternative therapeutic options with high efficacy and lower incidence of adverse events. 3
Dipeptidyl peptidase-4 (DPP-4) inhibitors are used widely nowadays for amelioration of hyperglycemia encountered in diabetic patients. 4 Recent reports suggested that DPP-4 inhibitors may possess a beneficial role in amelioration of the toxic effects created by the carcinogens on the different body cells. 5 In addition to the potent antioxidant and anti-inflammatory properties of DPP-4 inhibitors, they were reported to modulate the apoptotic pathways and hence confer a potential cytotoxic effect to the tumor cells. 6 Recent trials had proven that DPP-4 inhibitors suppress the degradation of glucagon-like peptide 2 which in turn may affect apoptosis in the different parts of the gastrointestinal tract. 7
Taxifolin is a flavonoid that is found in sufficient amounts in green leafy vegetables, fresh fruits, cocoa, and tea. 8 Recent studies reported that taxifolin has potent antioxidant and free radicals scavenging effects that might represent an interesting topic for research for management of diseases in which oxidative stress plays a fundamental role. 9 Also, taxifolin was suggested to modulate the expression of transforming growth factor beta 1 (TGF-β1) which plays a key role in regulation of the inflammatory events, autophagy and apoptosis. 10 In addition, taxifolin was reported to inhibit P-glycoprotein expression which is thought to be responsible for resistance of cancer cells to a wide range of chemotherapeutic agents. 11 Taken together, these properties might confer a potential role for taxifolin in amelioration of the malignant conditions. The aim of this work was to explore the possible effects of alogliptin and/or taxifolin on experimentally induced HCC in rats.
Methods and materials
Chemicals and drugs
Diethyl nitrosamine (DEN) and carbon tetrachloride (CCl4) were obtained from Sigma–Aldrich Co., Saint Louis, Missouri, USA (CAS # 55-18-5 and 56-23-5, respectively). Alogliptin and taxifolin were purchased from Cayman Chemical., Michigan, USA (CAS # 850649-62-6 and 24198-97-8, respectively). All other chemicals and reagents used in this study were purchased from Sigma–Aldrich Co., St. Louis, MO, USA and were of analytical grade.
Animals used
In the present study, 100 adult male Wistar rats weighing about 130–180 g were used. They were allowed to acclimatize for one week with free access to food and water ad libitum. All protocols used in this study complied with the ARRIVE guidelines and were executed according to the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines, EU Directive 2010/63/EU for animal experiments.
Induction of hepatocellular carcinoma
DEN was dissolved in corn oil and injected intraperitoneally in a single dose of 50 mg/kg body weight. After 2 weeks, the animals were injected intraperitoneally with a single dose of CCl4 (2 ml/kg) for enhancement of the carcinogenic effect of DEN. 12
Grouping of animals
Rats were randomly divided into five equal groups of 20 rat each as follows: a) Control group, received intraperitoneal injection of a single dose of corn oil; b) HCC group; c) HCC + Alogliptin group, received alogliptin by oral gavage in a dose of 3 mg/kg daily 13 ; d) HCC + Taxifolin group, received taxifolin by oral gavage in a dose of 50 mg/kg daily 14 ; and e) HCC + Alogliptin + Taxifolin group, received alogliptin concomitantly with taxifolin as formerly mentioned by oral gavage daily. Administration of alogliptin and taxifolin started from the first day of the experiment and continued for 6 months.
Assessment of the effect of alogliptin and/or taxifolin on the survival rate
The first day after injection of DEN was considered the zero point for determination of the survival rate by recording the number of the survived animals in each group at the end of each month for 6 months.
At the end of the experimental period, rats were anaesthetized with intraperitoneal injection of pentobarbital (30 mg/kg body weight), blood was withdrawn by cardiac puncture and animals were killed by cervical dislocation. Blood was centrifuged at 3000 rpm and serum was collected for determination of liver function tests and the tumor markers. Liver was excised, and parts of the hepatic tissues were homogenized in physiological saline (0.9% sodium chloride solution) using Teflon homogenizer and the homogenate was centrifuged at 3000 rpm for 10 min. The resulting supernatant was used for assessment of tissue biochemical parameters. Other parts of the hepatic tissues were processed for histopathological and immunohistochemical examination.
Assessment of liver function tests
Serum alanine transaminase (ALT) and aspartate transaminase (AST) were measured using kits supplied by Biomatik, USA, LLC (CAT # EKU02211 and EKE62019, respectively) according to the manufacturer’s instructions. Serum alkaline phosphatase (ALP) was assessed using kits supplied by Kamiya Biomedical Co., Seattle, WA, USA (CAT # KT-6158) according to the manufacturer’s guide. Serum gamma glutamyl transpeptidase (GGT) was determined using kits purchased from MyBioSource, Inc., San Diego, CA, USA (CAT # MBS9343646) according to the manufacturer’s protocol.
Determination of serum alpha-fetoprotein (AFP) and alpha-L-Fucosidase (AFU)
ELISA kits purchased from Kamiya Biomedical Co., Seattle, WA, USA were used for assay of serum AFP and AFU (CAT # KT-59172 and KT-67510, respectively) according to the manufacturer’s guide.
Assay of tissue oxidative stress parameters and nuclear factor (erythroid derived 2)-like 2 (Nrf2) content
Malondialdehyde (MDA) content of the hepatic tissues was assayed according to Ohkawa et al. 15 Tissue glutathione peroxidase (GPx) was assessed according to Chiu et al. 16 Tissue glutathione-S-transferase (GST) was measured according to Habig et al. 17 Tissue paraoxonase-1 (PON1) was determined according to McElveen et al. 18 Nrf2 content of the hepatic tissues was measured using kits supplied by Abcam, USA (CAT # ab207223) according to the manufacturer’s instructions.
Assay of the levels of transforming growth factor beta 1 (TGF-β1), interleukin 1 alpha (IL-1α) and toll-like receptor 4 (TLR4) in the hepatic tissues
ELISA kits purchased from R & D Systems, Minneapolis, MN, USA were used for assay of tissue TGF-β1 and IL-1α (CAT # DB100B and RRA00, respectively) according to the instructions of the manufacturer. TLR4 was determined in the hepatic tissues using kits obtained from Biomatik, Delaware, USA (CAT # EKF59022) according to the manufacturer’s guide.
Assay of beclin-1 levels and the c-Jun NH2-terminal kinase (JNK) levels in the hepatic tissues
ELISA kits purchased from Biomatik, Delaware, USA were used for assessment of tissue beclin-1 (CAT # EKU02685) according to the manufacturer’s guide. Tissue JNK level were measured using JNK 1/2 (pT183/Y185) ELISA kits obtained from Abcam, USA (CAT # ab176645) according to the protocol of the manufacturer.
Determination of caspase-3 and caspase-9 levels in the hepatic tissues
ELISA kits obtained from BioVision, Inc., CA, USA were used for assay of caspase-3 level in the hepatic tissues (CAT # E4592) according to the protocol of the manufacturer. Caspase-9 level was assessed using kits purchased from Elabscience, Texas, USA (CAT # E-EL-R0163) according to the manufacturer’s guide.
Histopathological examination
Specimens of the hepatic tissues were fixed in 10% formalin for 2 h and embedded in paraffin for 20 min. Then, paraffin blocks were cut into slices with thickness of 6 μm, stained with hematoxylin and eosin (H&E) and examined under light microscope.
Immunohistochemical examination of nuclear factor kappa B (NF-κB) p65 expression in the hepatic tissues
The de-paraffinized slides were incubated with citrate buffer (pH 6) for retrieval of the antigens. Then, they were put in 10% of normal goat serum obtained from SouthernBiotech, Birmingham, AL, USA (CAT # 0060-01) for 45 min in order to block binding of non-specific antibodies. After that, they were incubated overnight at 4°C with anti-NF-κB (p65) (Abcam, Cambridge, MA, USA, CAT # ab16502), USA), 1:100 dilution for assay of NF-κB (p65) expression. Then, the tissue sections were incubated with the secondary antibody, polyclonal goat anti-rat IgG immunoglobulin (SouthernBiotech, Birmingham, AL, USA (CAT # 3030-09). After that, tissue sections were counterstained with Mayer’s hematoxylin and examined under light microscope. The intensity of the immunohistochemical expression of NF-κB (p65) was quantified as follows: the largest number of cells showing positive NF-κB (p65) nuclear expression (+++); the intermediate number of cells showing positive NF-κB (p65) nuclear expression (++); and (+) refers to the lowest number of cells showing NF-κB (p65)-positively stained nuclei.
Statistical analysis
Data obtained from this study were analyzed using Minitab statistical software version 19 and presented as mean ± standard deviation (SD). Parametric results were statistically analyzed using one-way analysis of variance (ANOVA) test followed by Tukey-Kramer test. Non-parametric results were analyzed using Kruskal–Wallis test followed by post hoc Dunn’s test. The differences between the studied groups were considered statistically significant when the level of p-value was < 0.05.
Results
Effect of alogliptin and/or taxifolin on the survival rate
The survival rate was significantly increased with administration of alogliptin and/or taxifolin compared to HCC group. The improvement of the survival rate was significant with alogliptin/taxifolin combination compared to rats treated with each of these agents alone (Table 1).
The survival rate in the different groups at the end point of the study.

a Significant compared to the control group; bSignificant compared to HCC group; cSignificant compared to HCC + Alogliptin group; dSignificant compared to HCC + Taxifolin group.
Effect of alogliptin and/or taxifolin on liver function tests
The serum levels of AST, ALT, ALP and GGT were significantly reduced in rats treated with alogliptin and/or taxifolin compared to HCC group. Alogliptin/taxifolin combination had the upper hand in reduction of the levels of these parameters compared to rats treated with each of these agents alone (Figure 1).
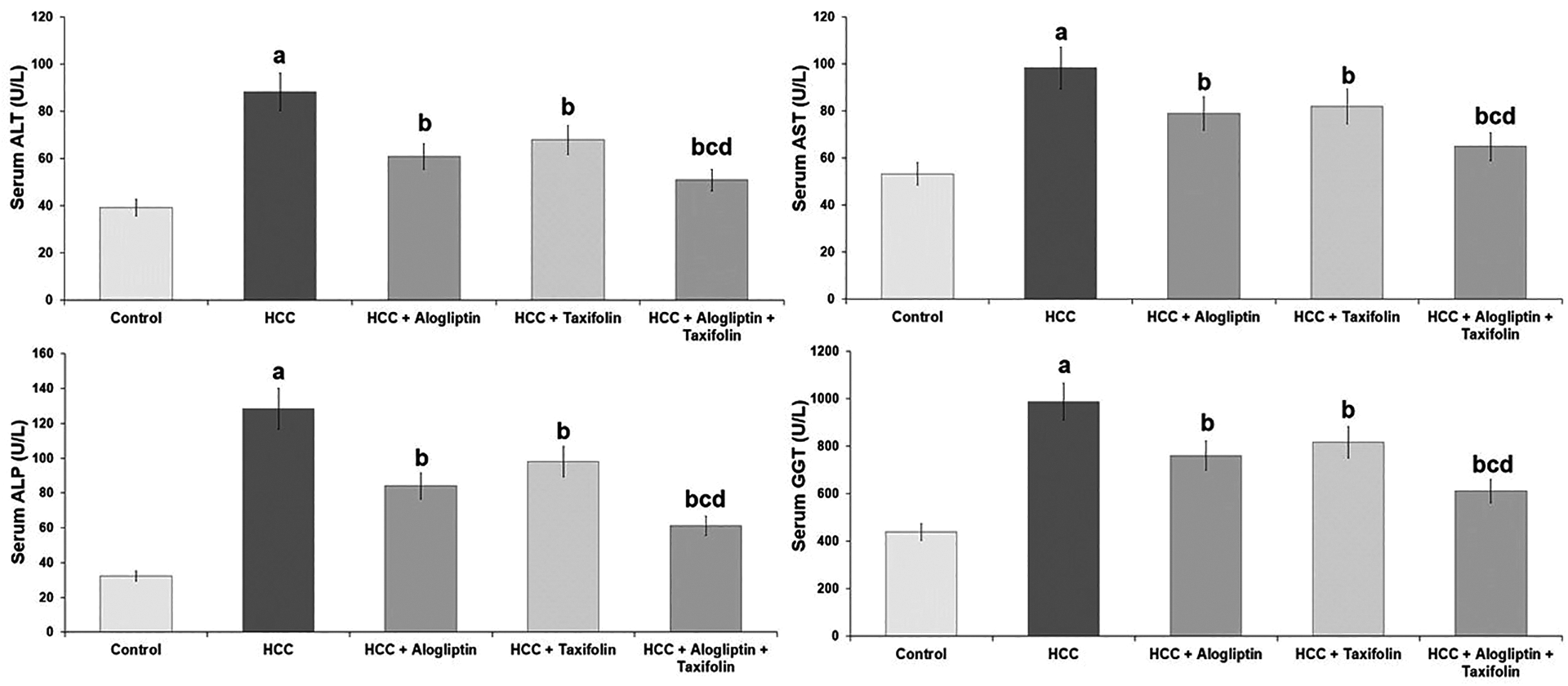
Effect of alogliptin and/or taxifolin on liver function tests (Mean ± SD). aSignificant compared to the control group. bSignificant compared to HCC group. cSignificant compared to HCC + Alogliptin group. dSignificant compared to HCC + Taxifolin group.
Effect of alogliptin and/or taxifolin on tissue antioxidant status and Nrf2 content
Administration of each of alogliptin or taxifolin induced significant increase in tissue GPx, GST, PON1 and Nrf2 content associated with significant decrease in tissue MDA compared to HCC group. The increase in tissue antioxidant enzymes and Nrf2 content and the decrease in MDA content were significant in rats treated with alogliptin/taxifolin combination compared to rats treated with each of these agents alone (Table 2).
Effect of administration of alogliptin and/or taxifolin to HCC rats on tissue malondialdehyde (MDA), glutathione peroxidase (GPx), glutathione-S-transferase (GST), paraoxonase1 (PON1) and nuclear factor (erythroid derived 2)-like 2 (Nrf2) content.
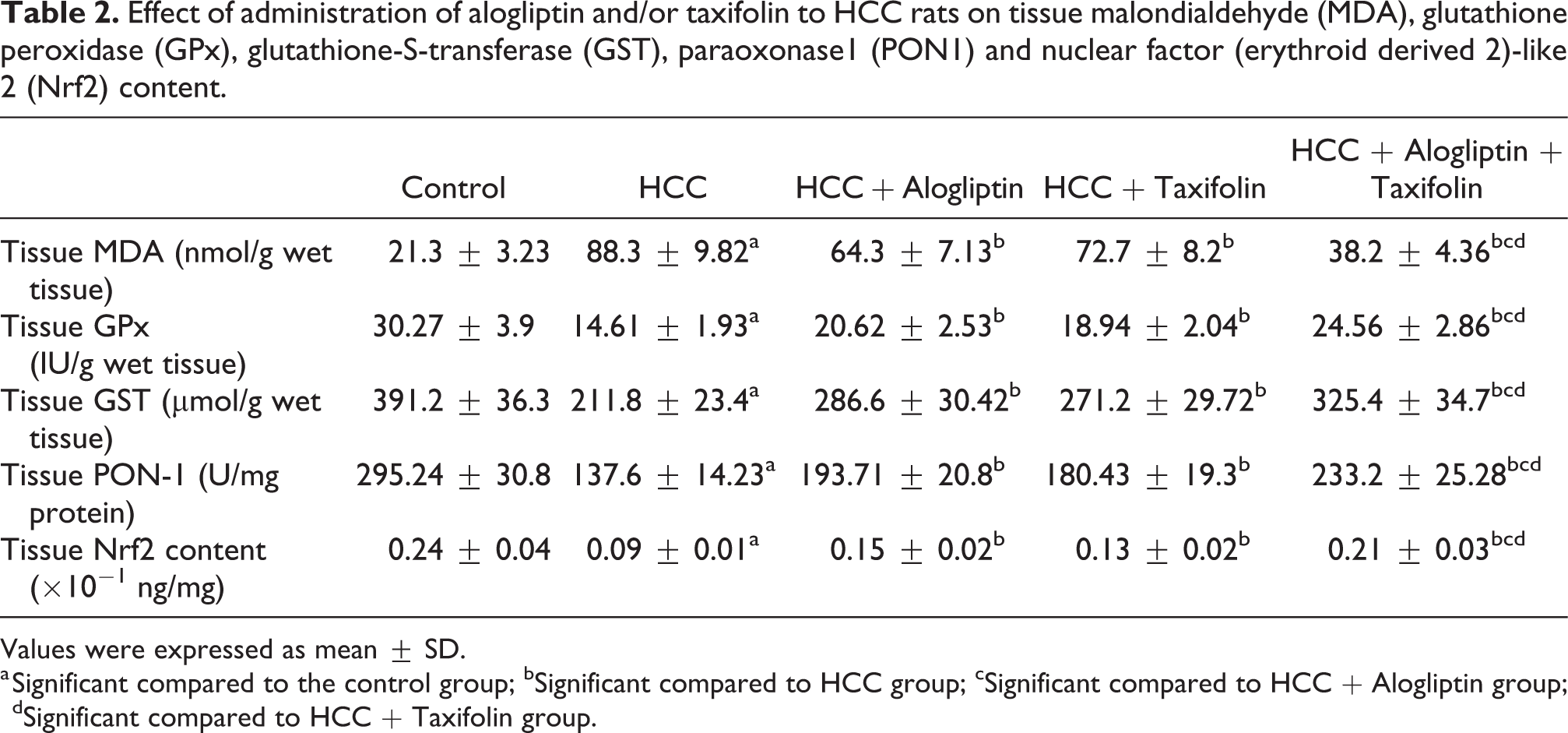
Values were expressed as mean ± SD.
a Significant compared to the control group; bSignificant compared to HCC group; cSignificant compared to HCC + Alogliptin group; dSignificant compared to HCC + Taxifolin group.
Effect of alogliptin and/or taxifolin on tumor tissue TGF-β1, IL-1α and TLR4
Tissue levels of TGF-β1, IL-1α and TLR4 were significantly reduced with administration of alogliptin and/or taxifolin compared to HCC group. This decrease was significant with alogliptin/taxifolin combination when compared to rats treated with each of these agents alone (Table 3).
Effect of administration of linagliptin and/or taxifolin to HCC rats on tissue transforming growth factor beta 1 (TGF-β1), interleukin-1-alpha (IL-1α) and toll-like receptor 4 (TLR4).

Values were expressed as mean ± SD.
a Significant compared to the control group; bSignificant compared to HCC group; cSignificant compared to HCC + Alogliptin group; dSignificant compared to HCC + Taxifolin group.
Effect of alogliptin and/or taxifolin on serum AFP and AFU
Serum levels of AFP and AFU were significantly decreased with administration of alogliptin and/or taxifolin compared to HCC group. The decrease in the levels of these tumor markers was significant in rats treated with alogliptin/taxifolin combination compared to those treated with each of these agents alone (Figure 2).

Effect of alogliptin and/or taxifolin on serum alpha-fetoprotein (AFP) and alpha-L-Fucosidase (AFU) (Mean ± SD). aSignificant compared to the control group. bSignificant compared to HCC group. cSignificant compared to HCC + Alogliptin group. dSignificant compared to HCC + Taxifolin group.
Effect of alogliptin and/or taxifolin on tissue beclin-1, JNK, caspase-3 and caspase-9 levels
Significant increase in tissue beclin-1, JNK, caspase-3 and caspase-9 levels were observed in rats treated with alogliptin and/or taxifolin compared to HCC group but alogliptin/taxifolin combination was superior to the use of each of these agents alone (Table 4).
Effect of administration of alogliptin and/or taxifolin to HCC rats on hepatic tissue beclin-1, caspase-3, caspase-9 and the c-Jun NH2-terminal kinase (JNK) levels.

Values were expressed as mean ± SD.
a Significant compared to the control group; bSignificant compared to HCC group; cSignificant compared to HCC + Alogliptin group; dSignificant compared to HCC + Taxifolin group.
Effect of alogliptin and/or taxifolin on the histopathological and immunohistochemical picture
HCC group showed groups of the well differentiated tumor cells with loss of the normal hepatic architecture and marked congestion of the hepatic sinusoids (Figure 3(b)) with markedly positive immunostaining for NF-κB (p65) (Figure 4(b)). This picture was significantly improved in rats treated with either alogliptin or taxifolin evidenced by increasing areas of necrosis, increased deposition of collagen fibers around the portal tracts and decreased congestion of the hepatic sinusoids (Figure 3(c) and (d)) with decreased expression of NF-κB (p65) (Figure 4(c) and (d)). The improvement in the histopathological picture and the decrease in NF-κB (p65) immunostaining were significant in rats treated with alogliptin/taxifolin combination compared to those treated with each of these agents alone (Figures 3(e) and 4(e)).

Hematoxylin & eosin stained sections from (

A photomicrograph of immunostaining of NF-κB (p65) of liver sections from (
Discussion
HCC represents about 90% of cases of primary liver carcinoma and is considered as the sixth most common malignancy all over the world. 19 In the present study, injection of DEN followed 2 weeks later by CCl4 administration induced the development of HCC manifested by significant deterioration of liver function tests with significant increase in serum AFP and AFU and malignant transformation of the hepatocytes on histopathological examination. This was in agreement with Mao et al. 20 who reported that DEN causes DNA damage by reacting with nucleophiles in the hepatocytes such as DNA-bases. These DNA changes render the liver cells more susceptible to the toxic effects of ROS leading to induction of carcinogenesis. Moreover, DNA mutations induced by DEN administration may lead to increased expression of the oncogenes, induction of the inflammatory cascade and modulation of the apoptotic pathways. 21
The role of Nrf2 in initiation and propagation of HCC represents an attractive item of research in many studies. 22 Raghunath et al. 23 reported that decreased Nrf2 content in the hepatic tissues was associated with increased production of ROS which are incriminated on a wide scale in the pathogenesis of HCC. Sun et al. 24 had proven that enhanced Nrf2 production in the hepatic tissues was associated with increased activity of the antioxidant enzymes leading to amelioration of the toxic effects of free radicals on the hepatocytes. In addition, Lampiasi and Montana 25 stated that there is an interplay between Nrf2 signaling and NF-κB expression which may represent the basis of the cellular response to the carcinogens. These findings were in agreement with the results of our study where the significant increase in the hepatic tissues content of Nrf2 induced by alogliptin and/or taxifolin was accompanied with significant increase in the activity of the antioxidant enzymes and significant decrease in tissue MDA and NF-κB (p65) expression compared to HCC group. Bishnoi et al. 26 reported that DPP-4 inhibitors had the ability to enhance Nrf2 expression with significant increase in the survival rates of cancer patients. Also, Wang et al. 27 attributed the possible antitumor effects of taxifolin to increased expression of Nrf2 genes through epigenetic modifications resulting in amelioration of oxidative stress and inflammation.
Accumulating evidences had proven the hypothesis that TGF-β1 creates a favorable microenvironment for growth and metastatic spread of HCC. 28 Giannelli et al. 29 reported that TGF-β1 facilitates epithelial-to-mesenchymal transition and enhances fibrogenesis which may contribute to the propagation of HCC. In addition, TGF-β1 was proven to promote phosphorylation of Smad2/3 leading to increased expression of the proinflammatory cytokines which have a major role in the development and prognosis of HCC. 30 This was in the same line with our results where administration of DEN followed by CCl4 promoted the expression of TGF-β1 and was associated with increased levels of IL-1α and intense immunostaining of NF-κB compared to the control group.
Coinciding with our results, alogliptin was reported to have potent anti-inflammatory effects, possibly via affecting TGF-β1/Smad3 signaling pathway with subsequent inhibition of the generation and release of the proinflammatory cytokines, including IL-1α and tumor necrosis factor alpha. 31 Moreover, El-Sahar et al. 32 attributed the anti-inflammatory effects of alogliptin to modulation of the gene expression of TLR4/MYD88/NF-κB signaling pathway. Also, administration of taxifolin in the present study was associated with decreased expression of TGF-β1 and IL-1α and moderate NF-κB immunostaining compared to rats injected with DEN alone. The explanation for this finding was proposed by Salama and Kabel 9 who suggested that taxifolin may exert anti-inflammatory effects through modulation of PI3K/AKT and p38 MAPK signaling with subsequent affection of TGF- β1-mediated inflammatory response.
TLR4 are pattern-recognition receptors that can recognize intracellular abnormalities leading to enhancement of adaptive immune responses. 33 Emerging data reported that TLR4 are highly expressed in certain types of malignancies including HCC, and they can promote tumor growth, proliferation and release. 34 Abnormal activation of TLR4 may precipitate several inflammatory responses and autoimmune reactions that may even reach to malignant transformation as an end result. 35 This was in agreement with our results where DEN injection induced significant increase in TLR4 expression associated with increased IL-1α and NF-κB levels compared to the control group. This response was ameliorated with administration of alogliptin which supported the hypothesis that the anti-inflammatory effects of DPP-4 inhibitors can be attributed to affection of TLR4/TGF-β1/Smad3 signaling pathway. 32 Also, Manigandan et al. 36 attributed the decrease in TLR4 levels observed with taxifolin administration in our study to its effect on NF-κB expression with subsequent inhibition of chemotaxis and enhancement of the activity of caspases resulting in increased apoptosis of the tumor cells.
JNK belongs to a group of serine/threonine (Ser/Thr) protein kinases referred to as the mitogen-activated protein kinase (MAPK) family. 37 JNK is largely involved in cell differentiation, proliferation, survival, invasion, and apoptosis. In addition, recent studies had proven its key role in the pathogenesis of metabolic disturbances, inflammatory disorders, epithelial-to-mesenchymal transition, and malignant transformation. 38 Recent trials reported that JNK may enhance the expression of a group of the pro-apoptotic genes and affect TGF-β1/Smad signaling pathway leading to affection of apoptosis. 39 Moreover, inhibition of JNK expression may be associated with significant decrease of beclin-1 levels leading to subsequent modulation of autophagy which is the main defense mechanism that hinders cancer development in the human body. 40 Accordingly, down-regulation of JNK expression was proven to be strongly associated with development of HCC. 41 This was in the same line with our results where the untreated HCC rats showed significant decrease in tissue JNK, beclin-1, caspase-3 and caspase-9 level in comparison to the control group. Alogliptin in our study was able to ameliorate these changes which was in agreement with Murase et al. 42 who reported that DPP-4 inhibitors may decrease the expression of the antiapoptotic proteins and increase JNK activity with subsequent activation of the apoptotic pathways. Moreover, alogliptin was proven to increase the activity of caspases which directly modulate the pathways of apoptosis and autophagy. 43 In addition, the effect of taxifolin encountered in the present study on beclin-1, caspase-3 and caspase-9 level may be attributed to its effect on JNK/TGF-β1 signaling with resultant modulation of autophagy and apoptosis. 44
In our study, alogliptin/taxifolin combination induced significant improvement of liver function tests associated with significant increase in the survival rate, tissue antioxidant enzymes and Nrf2 with induction of apoptosis. Moreover, this combination significantly reduced the levels of tumor markers with significant inhibition of TGF-β1/TLR4 signaling pathway. In addition, this combination had improved the histopathological and the immunohistopathological observations compared to rats treated with each of these drugs alone. The basis that may underlie these results might be the fact that each of these agents could significantly affect Nrf2 and TGF-β1/TLR4 signaling resulting in amelioration of the toxic effects of oxidative stress and the inflammatory markers induced by the carcinogens on liver cells. Moreover, both agents were proven to synergistically modulate the activity of several mediators involved in autophagy and apoptosis. Binkhathlan and Lavasanifar 45 stated that increased expression of P-glycoprotein may underlie not only the resistance of liver cells to DPP-4 inhibitors, but also the resistance of the malignant cells to the chemotherapeutic agents. Dewanjee et al. 46 reported that taxifolin is a strong inhibitor of P-glycoprotein expression at both the cytoplasmic and the nuclear levels. As a result, taxifolin may significantly enhance the cellular uptake of alogliptin by the hepatic tissues and decrease resistance of HCC cells to its effect.
Conclusion
Taxifolin/Alogliptin combination had an ameliorative effect on HCC in rats better than the use of each of these drugs alone. This may be attributed to their synergistic antioxidant and anti-inflammatory effects together with their ability to modulate apoptosis and autophagy of HCC cells. Moreover, taxifolin may inhibit P-glycoprotein expression resulting in enhancement of the cellular uptake of alogliptin. Further studies are needed to evaluate the possibility of clinical implication of these findings as adjuvant tool for treatment of HCC.
Footnotes
Acknowledgment
The current work was supported by Taif University Researchers Supporting Project number (TURSP-2020/29), Taif University, Taif, Saudi Arabia.
Disclosure statement
All authors of this manuscript confirm their compliance with the ICMJE authorship criteria.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Taif University Researchers Supporting Project number (TURSP-2020/29), Taif University, Taif, Saudi Arabia.
