Abstract
Aim:
A high cholesterol diet (HCD) is known to cause metabolic dysregulation, oxidative stress, cardiovascular diseases and atherogenesis. Zingerone is a pharmacologically active component of dry ginger. Zingerone has been shown to have a wide range of pharmacological properties, including scavenging free radicals, high antioxidant activity, suppressing lipid peroxidation and anti-inflammatory. This study aimed to investigate the effects of Zingerone on HCD-induced atherosclerosis in rats.
Methods:
Animals were divided into four categories (n = 6). Group I: normal control, Group II: zingerone control (20 mg/kg b.wt.), group III: HCD-induced atherosclerosis, Group IV: HCD + zingerone, respectively, for 8 weeks.
Results:
The HCD-fed rats resulted in a significant increase in an atherosclerotic lesion, lipid peroxidation, lipid profile, high-density lipoprotein concentration, cardiac markers, body weight, reduced antioxidant status, and displayed atherosclerosis. These findings were conventional by up-regulated expression of lipid regulatory genes like sterol-regulatory-element-binding protein-c (SREBP-c), fatty acid synthase (FAS), acetyl-CoA carboxylase (ACC), acetyl-CoA synthetase (ACS), liver X receptor–alpha (LXR-α), and down-regulated expression of acetyl-CoA oxidase (ACO), peroxisome proliferator-activated receptor-alpha (PPAR-α) and carnitine palmitoyl transferase-1 (CPT-1) in HCD-fed rats. These significant changes were observed in the zingerone-treated rats for the last 4 weeks.
Conclusion:
These findings suggest that zingerone reduced atherosclerosis by modulated the atherosclerotic lesion, lipid profile, antioxidant status and lipid regulatory gene expression in HCD-fed rats.
Introduction
Atherosclerosis (AS) is the leading underlying cause of coronary artery disease and is a complex, chronic inflammatory condition marked by lipid accumulation, resulting in lesions of an artery wall. Several metabolic and environmental factors are closely involved in the formation of atherosclerotic plaque. Population studies have demonstrated that elevated total cholesterol (TC) and low-density lipoprotein cholesterol (LDL), 1 lipoprotein are the main reasons for atherosclerotic cardiovascular diseases.2,3 Atherogenesis is a complex course that concerns some mechanisms, including endothelial dysfunction, vascular proliferation, matrix degradation, oxidative stress, inflammation, thrombosis and apoptosis.
Lipid metabolic abnormalities are manifested by an increase in plasma lipid and lipoprotein fractions, which confers an increased risk of cardiovascular diseases (CVDs). Lipoprotein degradation and oxidative mechanisms, in general, play a significant role in the pathogenesis of atherosclerosis. High fat or cholesterol-rich diet may lead to increased oxidative stress via free radical production (ROS) accompanied by a free radical release that may pose a risk of diabetes and CVD. Oxidative stress arises from an imbalance between free radicals formation and their elimination by the antioxidant defense system. 4 Increase production of hydroxyl radicals cause lipid peroxidation to lead to the destruction of articular tissues and malondialdehyde (MDA) formation. 5
Hypercholesterolemia has recently been linked to elevated oxidative stress, and lipid peroxidation may cause atherosclerosis and myocardial infarction. The increased level of total cholesterol, oxidized LDL and lipoprotein is a significant factor in vascular damage. Thus, modulation of oxidative stress under hypercholesterolemic conditions is a rational therapeutic approach, and attempts were made to recognize medicinal plants antioxidant properties against atherosclerosis. 6
Natural products have been proposed as potential therapeutic agents in the prevention and treatment of cardiovascular diseases. Herbal medicines have long been used to treat many diseases worldwide; most herbs mechanisms have not been established. Many conventional plant treatments are still used for atherosclerosis, but most evidence of their beneficial effects is anecdotal. 7 Aware of the increasing prevalence of atherosclerosis and coronary heart diseases, it is imperative to test and identify zingerone (4 (4-hydroxy-3-methoxyphenyl)-2-butanone) possible therapeutic role that could be added to our diet. Zingerone is an ingredient of ginger (Zingiber officinale) and is formed by a retro-aldol reaction from gingerol on reaction via cooking or thermal degradation. 8 Zingerone is a phenolic alkanones, with exact pharmacological properties mainly focused on antioxidants, 9 anti-inflammatory 10 properties. In the streptozocin (STZ) induced diabetic nephropathy in the animal model, zingerone plays a vital role in considering its metabolic complications.11,12 In an experimental study, zingerone has to lower the effect of oxidative stress and inflammation in cisplatin-induced nephrotoxicity. 13 Besides, zingerone an anti-atherosclerosis and attenuate lipogenesis effects have not yet been revealed in HCD-induced rats. Although zingerone has undergone such vast research by many scientists, its impact on atherosclerosis and coronary heart disease in HCD-fed rats have not yet been investigated. Therefore, this study was designed to assess the protective effect of zingerone on HCD-induced atherosclerosis in the rat by evaluating lipid profile, lipid regulation, hepatic function indicators, and cardiac markers.
Materials and methods
Chemicals and kits
Zingerone (Sigma Aldrich, St Louis, Missouri, USA), PCR array kits (Qiagen, USA) were acquired from. All other biochemical kits for this analysis procured from the Yehua Biological Technology Company (Shanghai, China).
Experimental rats
Male albino Wistar rats (150–180 g) were used for this experiment. All animals were placed within appropriate conditions in a clean, air-conditioned room at a temperature of (25 ± 2°C) with 50% humidity and 12:12 h dark/light duration. Animals were acclimatized for 1 week before experimentation and provided standard diet and water ad libitum to the laboratory conditions. The experimental methodology was permitted by the Institute Animal Ethics Committee of Tangshan Gongren Hospital (Ethic number: TSSGRYY20200825), Tangshan, China.
At the start of the experiment, control rats were fed a standard pellet diet; the other groups were fed high cholesterol and water ad libitum. Group I: Control and were supplemented with standard rat chow and water for 8 weeks. Group II: Zingerone (20 mg/kg b.wt.) administered rats for the last 4 weeks. Group III: HCD rats received 5% cholesterol and 0.5% cholic acid mixed with standard pellet diet (powder form) for 8 weeks. Groups IV: HCD (8 weeks) + zingerone (20 mg/kg b.wt.) oral administration in the last 4 weeks. Zingerone was administered by mixing 0.05% dimethyl sulfoxide with the vehicle directly into the stomach using a gastric tube in the morning.
Assessment of blood pressure
Blood pressure development was measured by the tail-cuff plethysmography method in the conscious restrained rat at ambient temperature routinely with an IITC Electro-Sphygmomanometer. All the blood pressure values were recorded by using a computerized data procurement system and software.
We measured the body weight and blood pressure of rats weekly once. After cervical dislocation, blood was immediately collected in chilled centrifuge tubes and centrifuged 600 g at 10°C for 10 min to get the serum. The blood, collected in EDTA coated centrifuge tube, was centrifuged at 600 g for 10 min, and the plasma was separated by aspiration.
After the separation of plasma, the buffy coat enriched in white cells was removed and the remaining erythrocytes were washed three times with physiological saline. A known volume of erythrocyte was lysed with hypotonic phosphate buffer at pH 7.4. The hemolysate was separated by centrifugation at 700 g for 10 min and the supernatant was used to estimate enzymatic antioxidants.
Histopathology (H&E)
The aorta was fixed in 4% paraformaldehyde and dehydrated in graded ethanol and embedded in paraffin. Paraffin sections were sliced using a slide microtome and dewaxed using xylene. The slides were kept in hemotoxylin and eosin (H&E) and examined under a light microscope.
Estimation of plasma lipids
Total cholesterol (TC), triglyceride (TG) and high-density lipoprotein cholesterol (HDL-C) levels in plasma were determined using commercially available kits according to the manufacturer’s protocol from Yeshua Biological Technology Company (Shanghai, China) with a semi-autoanalyzer. Low-density lipoprotein cholesterol (LDL-C) and very-low-density lipoprotein cholesterol (VLDL-C) were calculated from TG, TC and HDL-C concentrations following Friedewald formula (VLDL cholesterol = TG/5: LDL-C = TC − [HDL-C+VLDL-C]). 14 Apolipoprotein B (APO B) was measured as per the manufacturer’s instructions by ELISA kit (Abcam Scientific company, Shanghai, China).
Estimation of lipids heart tissue
We extracted myocardial tissue lipids as per the protocol described by Folch et al. 15 Then, we analyzed the levels of TC, TG, FFA, and PL by the protocols of Allain et al., 16 McGowan et al., 17 Falholt et al., 18 and Zilversmit and Davis, 19 respectively.
Measurement of cardiac markers
In this study, we measured the activities of creatine kinase-MB (CK-MB) and creatine kinase (CK) during HCD and/or zingerone by the methods described by Okinaka et al. 20 The serum levels of cTn T and cTn I was measured using a commercially available ELISA kit.21,22
Biochemical estimations
The level of lipid peroxidative markers such as TBARS and lipid hydroperoxides (LOOH) were estimated by the methods of Niehaus and Samuelson, 23 and Jiang et al., 24 respectively. Haemoglobin was estimated by the method of Drabkin and Austin, 25 and protein level was determined by Lowry et al. method. 26 Activities of antioxidant enzymes such as reduced glutathione (GSH), catalase (CAT), glutathione peroxidase (GPx), and superoxide dismutase (SOD) were estimated by the methods of Ellman, 27 Sinha, 28 Rotruck et al., 29 and Kakkar et al., 30 respectively, in circulation and heart tissue.
Determination of lipid regulatory mRNA expression by PCR array
The RNA was extracted from liver tissue using the RNeasy Mini kit (Qiagen, USA). The Nanodrop spectrophotometer was used to test the purity of the isolated RNA. The cDNA was reverse transcribed using a First Strand cDNA Synthesis Kit and used for PCR amplification by SYBR green using the Qiagen kit. A qPCR array analyzed the relative expression pattern of lipid regulatory genes sterol-regulatory-element-binding protein-1c (SREBP-1c), fatty acid synthase (FAS) and acetyl-CoA carboxylase (ACC), acetyl-CoA synthetase (ACS), liver X receptor–alpha (LXR-α), and down-regulated expression of acetyl-CoA oxidase (ACO), peroxisome proliferator-activated receptor-alpha (PPAR-α) and carnitine palmitoyl transferase-1 (CPT-1). The primer sequences were listed in Table 1. Relative gene expression (RQ) was calculated using 2−ΔΔCt and plotted as cluster grams.
The primer sequences for PCR array.

Statistical analysis
All the values were expressed as mean ± standard deviation of six rats from each group and statistically evaluated by one-way analysis of variance (ANOVA) followed by Duncan’s Multiple Range Test (DMRT). A value of p < 0.05 was considered statistically significant.
Results
Effect of zingerone on HCD-induced an atherosclerotic lesion in the aorta (H&E staining)
Figure 1 depicts the effect of zingerone on HCD-induced an atherosclerotic lesion in the aorta of the experimental rats (Figure 1(A) and (B)). Control and zingerone alone treated rats showed normal architecture of aorta. HCD-induced rats showed (Figure 1(C)) wide atherosclerotic plaques containing inflammatory cells, cholesterol deposition and thickening of the aorta. Comparing with HCD group, zingerone intervention (Figure 1(D)) significantly attenuated pathological changes (substantially reduced the atherosclerotic plaques containing inflammatory cells, cholesterol deposition and thickening of the aorta).

Effect of zingeone on HCD-induced an atherosclerotic lesion in the aorta (H&E staining). (A) Control and zingerone alone treated rats (B) showed normal architecture of aorta. HCD-induced rats showed (C) wide atherosclerotic plaques containing inflammatory cells, cholesterol deposition and thickening of the aorta. HCD group + zingerone (D) treated rats showed significantly attenuated pathological changes (substantially reduced the atherosclerotic plaques containing inflammatory cells, cholesterol deposition and thickening of the aorta).
Effect of zingerone on HCD-induced lipid profile and lipoproteins in plasma and heart tissue
We analyzed Lipid profile and lipoprotein status in the plasma of experimental rats. We observed that the HCD-fed rats increased TC, TG, FFA, PL, LDL-C, VLDL-C, and apolipoprotein B and decreased HDL-C level in the blood plasma. Conversely, the zingerone administration prevented HCD-induced circulating lipids and lipoproteins levels and significantly brought them back to a near normal level (Figures 2 and 3).
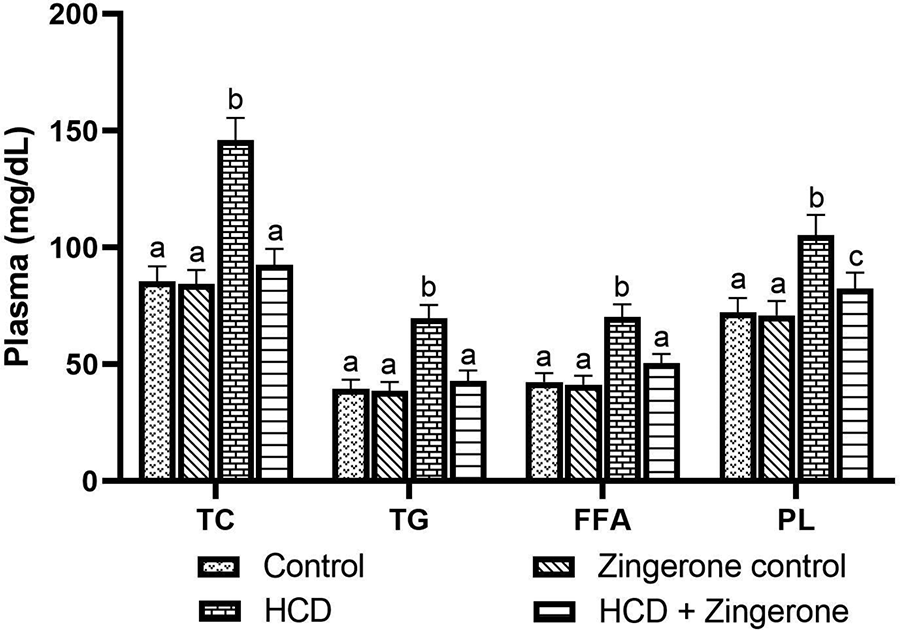
Effect of zingerone on the levels of TC, TG, FFA, and PL in the plasma of control and HCD-induced rats. Values not sharing a common marking superscript (a, b, c) are different significantly at p-value ≤ 0.05 (DMRT). b (HCD—group III) was compared with a (control—group I). c (HCD+ zingerone—group IV) was compared with b (HCD—group III).

Effect of zingerone on the levels of lipoproteins and Apolipo protein-B in the plasma of control and HCD-induced rats. A. HDL-C level, B. VLDL-C level, C. LDL-C level and D. APO-B level. Values are given as means ± S.D (n=6). Values not sharing a common marking superscript (a, b, c) are different significantly at p-value ≤ 0.05 (DMRT). b (HCD-group III) was compared with a (control-group I). c (HCD+ zingerone – group IV) was compared with b (HCD-group III).
Lipid levels in HCD-induced rats in heart tissue were analyzed (Figure 4). HCD-induced rats significantly elevated TC, TG, FFA, and PL levels when compared to control rats. Conversely, zingerone administration prevented TC, TG, FFA, and PL levels and brought them back to near normal in HCD-fed rats. The zingerone alone treatment does not alter the lipids parameters in the plasma and heart tissue.

Effect of zingerone on the levels of TC, TG, FFA, and PL in the heart tissue of control and HCD-induced rats. Values are given as means ± S.D (n = 6). Values not sharing a common marking superscript (a, b, c) are different significantly at p-value ≤ 0.05 (DMRT). b (HCD—group III) was compared with a (control—group I). a (HCD+ zingerone—group IV) was compared with b (HCD—group III).
Effect of zingerone on HCD-induced cardiac marker enzymes
Figure 5(A) and (B) show the impact of zingerone treatment on the activities of CK, CK-MB, cTn T, and cTn I. HCD-induced rats significantly (P < 0.05) increased the activity of cardiac marker enzymes. A substantial decrease in the activity of CK, CK-MB cTn T, and cTn I was observed in zingerone-administered HCD-induced rats when compared to control rats. The zingerone alone treatment does not alter cardiac markers activities than the control rats.

Effect of zingerone on HCD-induced serum cardiac marker enzymes. (A) Effect of zingerone on CK and CK-MB in the serum of control and ISO-induced rats. (B) Effect of zingerone on cTn T and cTn I in the serum of control and ISO-induced rats. Values are given as means ± S.D (n = 6). Values not sharing a common marking superscript (a, b, c) are different significantly at p-value ≤ 0.05 (DMRT). b (HCD—group III) was compared with a (control—group I). c (HCD+ zingerone—group IV) was compared with b (HCD—group III).
Effects of zingerone on HCD-induced lipid peroxidation in plasma and heart tissue
HCD rats showed increased lipid peroxidation products such as TBARS and LOOH levels in plasma and heart tissue. However, zingerone administration significantly reduced TBARS and LOOH in plasma and cardiac tissue compared to the HCD-induced group. The zingerone alone treatment does not alter these lipid peroxidative markers level than the control group (Figure 6(A) to (D)).

Effect of zingerone on the levels of TBARS and LOOH in the plasma and heart tissue of control and HCD-induced rats. Values are expressed as means ± S.D for six rats in each group. Values not sharing a common superscript (a, b, c) differ significantly at p ≤0.05 (DMRT). b (HCD—group III) was compared with a (control—group I). a (HCD+ zingerone—group IV) was compared with b (HCD—group III).
Effects of zingerone on HCD-induced antioxidant status
The antioxidants status in the circulation and heart tissue of experimental rats (Table 2) were analyzed. Animals fed with HCD had considerably reduced enzymatic antioxidants such as SOD, CAT, and GPx. Decreased levels of the GSH level was observed when compared to the sham control group. Whereas zingerone supplementation increased antioxidant status in HCD-induced rats. The zingerone treatment alone does not alter the circulation and heart tissue antioxidants status compared to the sham control group.
Effect of zingerone on Hb, Protein, SOD, CAT, GPx in erythrocytes and GSH in plasma and heart tissue of HCD-fed rat.

Values are expressed as means ± S.D for six rats in each group. Values not sharing a common marking superscript (a, b, c) are different significantly at p-value ≤ 0.05 (DMRT). U* = Enzyme required to scavenge the chromogen formed by 50% in one min. U** = µmole of hydrogen peroxide decayed per min. U*** = µmole of GSH required/min.
b (HCD—group III) was compared with a (control—group I). c (HCD+ zingerone—group IV) was compared with b (HCD—group III)
Effect of zingerone on HCD-induced body weight
Figure 7 shows the effect of zingerone on HCD-induced body weight in male Wistar rats. HCD consumption in rats resulted in a significant and progressive increase in body weight compared to the control group rats. Oral administration of zingerone revealed a substantial reduction in body weight when compared to HCD-induced rats.

Effect of zingerone on body weight in HCD-fed rats. Values are means ± S.D. for six rats. In each group, means with a different superscript letter (a, b, c) differ significantly at p <0.05 (DMRT). b (HCD—group III) was compared with a (control—group I). c (HCD+ zingerone—group IV) was compared with b (HCD—group III).
Effect of zingerone on blood pressure measurements in HCD-induced rats
In this study, we found that HCD-induced caused a significant (P < 0.05) increases in systolic and diastolic blood pressure compared to control rats. Simultaneously, treatment with zingerone reduced the systolic and diastolic blood in HCD-induced rats (Figure 8(A) and (B)).

Effect of zingerone on HCD-induced blood pressure measurement. (A) Effect of zingerone on HCD-induced systolic pressure measurement. (B) Effect of zingerone on HCD-induced diastolic pressure measurement. The data not sharing a common superscript (a, b, c) are different significantly at p-value ≤ 0.05 (DMRT). b (HCD—group III) was compared with a (control—group I). c (HCD+ zingerone—group IV) was compared with b (HCD—group III).
Effect of zingerone on HCD-induced lipid regulatory gene expression by PCR array
We analyzed the transcriptional profile of target genes in the liver tissue of experimental animals. The relative mRNA expression pattern (RQ) of lipid regulatory genes like sterol-regulatory-element-binding protein-1c (SREBP-1c), fatty acid synthase (FAS) and acetyl-CoA carboxylase (ACC), acetyl-CoA synthetase (ACS), liver X receptor –alpha (LXR-α) were up-regulated and down-regulated expression of acetyl-CoA oxidase (ACO), peroxisome proliferator-activated receptor-alpha (PPAR-α) and carnitine palmitoyl transferase-1 (CPT-1) were observed in HCD-fed rats. Conversely, the zingerone administration modulated lipid-regulated genes in HCD-fed rats (Figure 9(A) and (B)). The zingerone treatment alone does not alter the transcriptome pattern of lipid regulatory genes compared to sham control.

Effect of zingerone on HCD-induced lipid regulatory gene expression by the qRT-PCR array. (A) Hierarchical cluster gram analysis of PCR array results for lipid regulatory gene expression in zingerone and HCD-induced rats. Bright red indicates the highest normalized signal values, green-blue represents the lowest signal values and black represents median signal values. The total mRNA was isolated from liver tissues and was detected using a custom PCR array following the manufacturer’s instructions. The clustergram results of three independent experiments were analyzed using online tool (B). Relative gene expression levels lipid regulatory gene expression in HCD and HCD + zingerone-treated rats. The fold changes of gene expression were calculated as per 2−ΔΔCt. **(HCD+ zingerone—group IV) was compared with *(HCD—group III).
Discussion
Hypercholesterolemia usually develops severe atherosclerosis involved in the pathogenesis of coronary heart disease and myocardial infarction. The high fat-rich diet contributes to the development of hyperlipidemia, atherosclerosis, and impaired lipid metabolism. 31 In the current study, we found that zingerone anti-atherosclerosis affects atherosclerotic lesion in aorta, lipid profile, lipoprotein status, cardiac markers, lipid and antioxidant status and lipid regulatory gene expression against HCD-fed rats.
Considering plaque formation is a crucial characteristic of the progression of atherosclerotic disease. 32 In the present study, we mainly observed the pathological change in the aorta and evaluated the effect of zingerone on the atherosclerotic lesion in HCD-induced rats. Histopathological examinations revealed that wide atherosclerotic plaques containing inflammatory cells, cholesterol deposition and thickening of the aorta. Whereas, zingerone treatment significantly attenuated pathological changes (substantially reduced the atherosclerotic plaques containing inflammatory cells, cholesterol deposition and thickening of the aorta), indicating zingerone have a certain therapeutic effect on AS.
In this research, significant increases in TC, TG, FFA, PL, LDL-C, VLDL-C, and apolipoprotein B (Apo B) levels, along with a substantial decrease in HDL-C in HCD-induced rats. In general, TG-rich lipoproteins are predictors for cholesterol-enriched particles depositing cholesterol in the arterial wall.33,34 Besides, excessive LDL-C can cause the development of “arterial atherosclerotic lesions”. 35 Apo B is associated with LDL, which plays a vital role in peripheral tissues and liver ingestion cholesterol-rich LDL particles, and the reduced concentration of HDL-C hinders cholesterol elimination from the arterial wall. 36 In the present study, our results demonstrated that zingerone supplementation significantly reduced hyperlipidemia, suggesting that zingerone can be beneficial for the lipid and lipoprotein profile attenuates the initiation and progression of atherosclerosis. 37 We proved that the preventive effect of zingerone extracts against altered lipid metabolism in high cholesterol-fed rabbits.
The present observations reflect a substantial increase in the cardiac markers CK, CK-MB, cTnT and cTnI, in the serum of HCD rats suggesting that the morphological and functional changes in the cardiac muscle and showing disturbance of the integrity of the cell membrane. 38 Cardiac markers enter into the bloodstream from cardiomyocytes due to the disruption and uncontrolled permeability of cell membranes, 39 demonstrating increased enzyme activity in HCD group was reported. 40 Oral administration of zingerone significantly reduced the CK CK-MB, cTnT, and cTnI in HCD-fed rats. The protective effect of zingerone decreases the degree of cardiac abnormalities in HCD-induced rats compared to control rats and could be related to observed changes. Hemalatha et al. reported that zingerone on altered cardiac markers, lipid peroxidation markers and antioxidant status in the circulation of isoproterenol-induced in experimental rats. 41
In atherosclerosis, lipid peroxidation is an essential pathogenic occurrence, and the accumulation in the tissues causes cellular dysfunction and oxidative stress-related diseases. Elevated levels of lipid peroxidation products such as TBARS and LOOH and diminished levels/activities of GSH, SOD, CAT, and GPx are due to oxidative damage in HCD-induced atherosclerosis rats. Its antioxidant effect results from the decreased levels of lipid peroxidation products and improved antioxidant status were observed in zingerone-administered rats. These data suggest a strong free radical scavenging mechanism for zingerone. 41 This anti-atherosclerotic property of zingerone may be due to its structure of methoxyl and phenolic hydroxyl groups. Typically, the methoxyl group releases a proton from the hydroxyl group. The zingerone has a long-chain ethyl methyl ketone group that increases the coefficient of bulk, stabilization, and partition. This property might help enter the cells with zingerone to make it accessible for free radicals to be quenched. Zingerone antioxidant potential may be attributed to hydroxyl groups present on the aromatic ring and double bonds in zingerone between C-3 and C-4. Several studies also reported that zingerone does not induce ROS formation as it is known to be non-toxic.42,43
In the present study, HCD-fed rats showed a considerable body weight gain compared to control rats. In the previous studies, the HCD-diet group revealed increased body weight compared to the other dietary groups. 44 Previous studies have revealed that the zingerone dissolves body fat in HFD-fed animals. Ginger and their associated compound like zingerone supplementation prevented triglyceride deposition and inhibited preadipocyte differentiation into adipocytes. 45 Further, in our results, HCD-induced atherosclerosis in rats caused a substantial rise in systolic and diastolic blood pressure compared to normally fed control rats. El-Bassossy et al. suggested that cardiac repolarization and AV conduction linked to diabetes are enhanced by oral zingerone administration. 46 Likewise, in this study, oral zingerone dosage substantially decreases the body weight, systolic and diastolic blood pressure in rats fed with HCD.
SREBP-1c is a transcription factor that mediates cholesterol biosynthesis and the regulation of lipogenesis genes, including ACC and FAS.47,48 SREBP-1c will bind to its target genes promoter regions promote the lipogenic enzyme transcript. 49 LXRs, which are upstream transcription variables that increase the activity of SREBP-1c to facilitate the synthesis of fatty acids. 50 LXR-α is a nuclear transcriptional factor that activates through its target genes by binding to DNA sequences and controls cholesterol metabolism. 51 HCD-fed rats showed increased expression in the liver of LXR-α, SREBP1c, ACC, and FAS. Excess intake of cholesterol has been shown to cause increased lipogenic enzymes (ACC and FAS) via SREBP-1c induction, which leads to hepatic lipogenesis. 52 Zingerone administration shows a reduced hepatic expression of LXR-α and SREBP1c lipogenic transcription factors in HCD-fed rats. Besides, the hepatic expression of ACC and FAS were down-regulated by zingerone administration along with HCD. Apart from the redox properties, zingerone exhibits known pharmacological and biological activities, including anti-inflammatory, anti-cancer, and antimicrobial activities. The suppression of SREBP-1c, LXR-α, and its target lipogenic enzymes FAS and ACC may have contributed to this antioxidant property.
PPARα belongs to the superfamily of the nuclear receptor. Once PPARα is triggered, it would connect the PPAR-RXR heterodimer to the downstream DNA-binding site, regulating fatty acid oxidation enzymes like ACOX1, CPT-I, CPT-II, etc. 53 PPAR-α is highly expressed in the tissue with elevated fatty acids oxidation rates, like brown fat, heart and liver. 54 The PPAR-α agonist stimulates the β-oxidation of fatty acid and decreases lipid accumulation on the liver. 55 Hepatic CPT-1 is the β oxidation regulator in hepatocytes that determines the critical rate. The increase in CPT-1 activity to significantly reduce hepatic triglycerides is necessary to increase fatty liver. 56 The first step of the β-oxidation cycle in which a fatty acyl-CoA is oxidized into 2-trans-enoyl-CoA 57 has catalyzed acyl-CoA oxidase (ACO). The levels of mRNAs for ACO and CPT-1 have also been reported to be up-regulated by a PPAR-α.58,59 In the HFD rats, which was in good agreement with previous reports, 60 our findings showed that hepatic CPT-1, ACO, and PPAR-α mRNA expression levels were significantly lower. Zingerone administration increased the expression of hepatic mRNA of these enzymes significantly. Zingerone effects on the liver include an increase in PPAR activities and oxidation of the fatty acids that reduce lipogenesis and prevent liver damage by inhibiting free radical production. Moreover, the expression of lipid regulatory enzymes modulated in link with our previous findings by Kumar et al. 61 These data suggest that the administration of zingerone prevented HCD-mediated hepatic damage.
Conclusion
Our study demonstrates that zingerone reduces atherosclerosis caused by HCD. The anti-atherosclerotic effect of zingerone may reduce the atherosclerotic lesion in aorta, lipid profile, cardiac markers, improve the antioxidant status and regulation of lipid gene expression in HCD-fed rats. Further studies are warranted to clarify the potential beneficial effects and specific molecular mechanisms of zingerone on HCD. Therefore, it is suggested that zingerone use may be helpful as an adjuvant in preventing and managing diet-induced atherosclerosis and its related complications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
