Abstract
Amyloid beta (Aβ) peptide aggregation and cholinergic neurodegeneration are involved in the development of cognitive impairment. Therefore, in this article, we examined rosuvastatin (RSV), an oral hypolipidemic drug, to determine its potential as a dual inhibitor of acetylcholinesterase (AChE) and Aβ peptide aggregation for the treatment of cognitive impairment. Molecular docking study was done to examine the affinity of RSV with Aβ1–42 and AChE in silico. We also employed neurobehavioral activity tests, biochemical estimation, and histopathology to study the anti-Aβ1–42 aggregation capability of RSV in vivo. Molecular docking study provided evidence that RSV has the best binding conformer at its receptor site or active site of an enzyme. The cognitive impairment in female Wistar rats was induced by high-salt and cholesterol diet (HSCD) ad libitum for 8 weeks. RSV ameliorated serum cholesterol level, AChE activity, and Aβ1–42 peptide aggregations in HSCD induced cognitive impairment. In addition, RSV-treated rats showed greater scores in the open field (locomotor activity) test. Moreover, the histopathological studies in the hippocampus and cortex of rat brain also supported that RSV markedly reduced the cognitive impairment and preserved the normal histoarchitectural pattern of the hippocampus and cortex. Taken together, these data indicate that RSV may act as a dual inhibitor of AChE and Aβ1–42 peptide aggregation, therefore suggesting a therapeutic strategy for cognitive impairment treatment.
Keywords
Introduction
Alzheimer’s disease (AD) is the most common form of dementia, associated with neuropathological and neurobehavioral changes accompanied by memory and cognitive impairment. 1,2 It is characterized by excessive production and accumulation of Aβ peptide, hyperphosphorylated tau protein, and low levels of acetylcholine (ACh). 3 Accumulating evidence indicates that a sequence of events contributes to the development and progression of AD, including oxidative stress, inflammation, and altered cholesterol metabolism. 4,5 Due to sedentary lifestyle and increase in consumption of salt and oil by people, certainly the number of patients and the economic impact of AD will grow extraordinarily in the future without advances in therapy or prevention. 6 Owing to the increasing average lifespan, the prevalence of AD is sharply on the rise, with the World Health Organization predicting above 20 million cases by 2020. 7
The metabolic disorders seem to be closely associated with impairment of cognitive function in humans. Accumulating evidence has demonstrated an association between metabolic disorder and the onset of cognitive impairment. 8,9 In recent years, increased intake of high-salt and cholesterol diet (HSCD) have become a worldwide health problem, predisposing individuals to coronary, cerebrovascular, and peripheral vascular diseases; type-2 diabetes; as well as hyperlipidemic disorders. 10 –12 Hyperlipidemia encourages brain function deficits, cognitive impairments, and behavioral changes. 13,14
Statins (3-hydroxy-3-methylglutaryl coenzyme-A reductase inhibitors), a class of hypolipidemic drugs, have been anticipated as potential agents for the treatment of cognitive impairments. 15,16 Although their main effects are related to the inhibition of cholesterol synthesis, the decrease in the levels of low-density lipoproteins and triglycerides, they also strongly modulate inflammatory cells around atherosclerotic plaques. 17 Apart from their therapeutic use in cardiovascular diseases, statins may also have beneficial effects in the central nervous system. 18,19
In the present study, we examined the effects of rosuvastatin (RSV), the most widely used and arguably the most effective statin. 20,21 We assumed that RSV might inhibit acetylcholinesterase (AChE) activity and Aβ1–42 peptide aggregation simultaneously during cognitive impairment pathogenesis. This effect may be attributed to the inhibitory effects of RSV directly on the Aβ1–42 peptides, which differs from atorvastatin, another statin that reduces Aβ1–42 aggregation through modulating amyloid precursor protein cleavage. 22 –24 We attempted to explain the potential molecular mechanism of the inhibitory features by using molecular docking, assessing neurobehavioral activity along with a series of tests that include total cholesterol (TC) level, AChE level, Aβ1–42 level, histopathology, and ultimately examining the inhibition of AChE activity and Aβ1–42 peptide aggregation in HSCD-induced cognitive impairments in vivo.
Materials and methods
Drugs and chemicals
RSV was obtained as a gift sample from Sun Pharmaceutical Industries Limited (Gurgaon, Haryana, India). Piracetam (PCT) was obtained as a gift sample from Arbro Pharmaceuticals Limited (New Delhi, India).
Animal procurement
The experimental protocol was approved by Institutional Animal Ethics Committee of Jamia Hamdard (Hamdard University), New Delhi, India (registration no. JH/993/CPCSEA) as per the guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals. Female Wistar rats in the range of 150–200 g of body weight were issued from Central Animal House Facility, Jamia Hamdard, New Delhi, and housed in standard polypropylene cages and had access to commercial standard pellet diet (Amrut rat feed; Nav Maharashtra Chakan Oil Mills Ltd, New Delhi, India). The rats were maintained under controlled room temperature (23 ± 2°C) and relative humidity (60 ± 5%) with 12 h light/12 h dark (day/night) cycle in the departmental animal house.
Drug preparation
Suspension of RSV and PCT was prepared by triturating the weighed amount of RSV (5, 10, and 15 mg/kg) and PCT (200 mg/kg) in 0.5% carboxy methyl cellulose suspension (w/v) in normal saline, respectively. 25,26 High-salt saline was prepared freshly by adding 2% w/v sodium chloride in water. Pellets of high-cholesterol diet were prepared freshly by adding 1.25% cholesterol and 10% coconut oil in standard diet pellets and dried at room temperature.
Experimental design
Prior to experimental studies, rats were acclimatized for 7 days and fed with standard diet. After that, animals were fed to HSCD ad libitum for 8 weeks to induce cognitive impairment. 9,27 Rats were treated with RSV orally, PCT intraperitoneally, and a combination of both for 7 weeks as described below.
The animals were randomly divided into nine groups, that is (N = 6 in each group): normal control (NC) group, received normal saline and original pellets diet for 15 weeks; per se RSV (PSR) group, received the normal saline and original pellets diet for 15 weeks + last 7 weeks RSV (10 mg/kg body weight (b.wt)); per se PCT (PSP) group, received the normal saline and original pellets diet for 15 weeks + last 7 weeks PCT (200 mg/kg b.wt); toxic control (TC) group, received HSCD for 15 weeks; R5 group, received HSCD for 15 weeks + last 7 weeks RSV (5 mg/kg b.wt); R10 group, received the HSCD for 15 weeks + last 7 weeks RSV (10 mg/kg b.wt); R15 group, received the HSCD for 15 weeks + last 7 weeks RSV (15 mg/kg b.wt); P200 group, received the HSCD for 15 weeks + last 7 weeks PCT (200 mg/kg b.wt); R10+P200 group, received the HSCD for 15 weeks + last 7 weeks RSV (10 mg/kg b.wt) + PCT (200mg/kg b.wt). At the end of treatment, rats were observed for behavioral parameters followed by blood collection and then rats were immediately killed. Brain were separated and preserved for biochemical and histopathological examination.
Molecular docking analysis
The ligand docking was performed using solution structure of acetyl cholinesterase (AChE) and amyloid beta (Aβ) and was carried out on Maestro 16.0 program (Schrodinger Inc., USA). The three-dimensional structure of human brain AChE and Aβ in apo form were taken from the protein data bank (PDB ID: 1IYT and 3LII) to be used for the present docking study. 28,29 The protein preparation was carried out in three steps, that is, preprocess, review, and modify, followed by refinement using “protein preparation wizard” in Maestro 16.0. In these steps, water molecules were deleted and hydrogen atoms were added. Energy of the structure was minimized using OPLS 2005 force field Maestro 10.5 program (Schrodinger Inc. USA). Similarly, ligands were prepared again using Force Field 2005. Receptor grid generation program was run by clicking any atom of the ligand and the default box was prepared. The ligand was docked into the grid generated from the protein using extra precision. The results were evaluated by docking scores. 30 The compound was docked three times and the minimum of the three scores was used. The docking score, binding free energy, and hydrogen bonds and π–π interaction formed with the surrounding amino acids are used to predict their binding affinities and proper alignment of these compounds at the active site of the AChE and Aβ.
Locomotor activity
The neurobehavioral tests in each group were performed by locomotor activity (LA) in the video path analyzer. The experiment was performed between 8:30 a.m. and 1:30 p.m. in standard laboratory conditions. LA was carried out in the open-field arena consisting of an acrylic box (40.6 × 40.6 × 40.6 cm 3 ), which is fitted with two photo beam frames (16 beams/dimensions; 2.5 cm between beams). The activity meter was joined to a computer interface (TrueScan 2.0 version, Coulbourn Instruments, Allentown, Pennsylvania, USA) that recorded beam breaks (100 ms sampling rate). LA of each animal was counted for 20 min after a period of 30 min for acclimatization. 31 The LA of a rat is counted through interruptions of IR beams caused by movements of rat placed on the grid floor of the activity meter is converted into counts and digitally displayed on the panel. After each animal was tested, the motility cage was thoroughly wiped with 70% isopropyl alcohol to minimize possible interference from animal odors. A total of five LA components were recorded for each groups as move time (s), rest time (s), horizontal activity (cm), mean velocity (cm/s), and total movement (#).
Body weight and brain weight
Body weight gain was calculated as a difference of initial and final weights of animals. Brain weight was measured after killing the animals. 32,33
Determination of TC estimation in serum
Serum TC activity was assayed by enzymatic methods using commercial assay kits according to the manufacturer’s protocol. 34
Determination of brain AChE activity
AChE activity assays were carried out based on the colorimetric method. 35,36 Concisely, 20 mg of rat brain was homogenized with 1.0 ml of potassium phosphate buffer, pH 8.0 in the homogenizer. In all, 0.4 ml aliquot of homogenate was added to a cuvette containing 2.6 ml of phosphate buffer (pH 7.4) followed by the addition of 100 μl of the 5,5-dithiobis-2-nitrobenzoate reagent. The absorbance was measured at 412 nm wavelength using a ultraviolet spectrophotometer. Twenty microliters of the acetylthiocholine iodide was added and change in absorbance per minute was calculated. The enzyme activity was expressed as micromolar of substrate hydrolyzed per minute per milligrams of protein.
Determination of Aβ content in brain and serum
Frozen brains were thawed and minced. Then, the tissues of the brain were weighed and homogenized in 50 mM Tris-HCl buffer, pH 8.0, containing a cocktail of protease inhibitors. The homogenates were centrifuged (100,000 × g, 1 h, 4°C), and the supernatants were stored at −80°C for additional analysis of soluble Aβ. Then, the pellets were sonicated in 5 M guanidine and incubated for 30 min at room temperature then centrifuged (100,000 × g, 1 h, 4°C) and then supernatants were stored for analysis of insoluble Aβ. 28,37 The levels of Aβ1–42 in brain and serum were measured by using the ELISA method. The concentrations of insoluble and soluble Aβ1–42 were estimated by using Aβ ELISA kits (GenxBio Health Sciences Pvt. Ltd. Shakarpur, Delhi-110092, India.), as per manufacturers’ instructions.
Histopathological analysis of brain
For histological analysis, the brain was fixed in 10% neutral buffered formalin and embedded in paraffin. Standard sections of 5 µm thickness were cut and stained with hematoxylin and eosin (H&E). Photomicrographs were taken using the Meiji fluorescent microscope enabled with a lumenera camera. The images were analyzed with infinity analyze 3 (version 6.3.0) software. 38,39
Statistical analysis
Results were expressed as the mean ± standard error of mean (SEM). The statistical significance of difference between groups was determined using one-way analysis of variance followed by Tukey’s test. p Value <0.05 was considered statistically significant. Error bars represent the SEM. All statistical tests were performed using the Prism software package (version 4, GraphPad, San Diego, California, USA).
Results
Molecular docking analysis
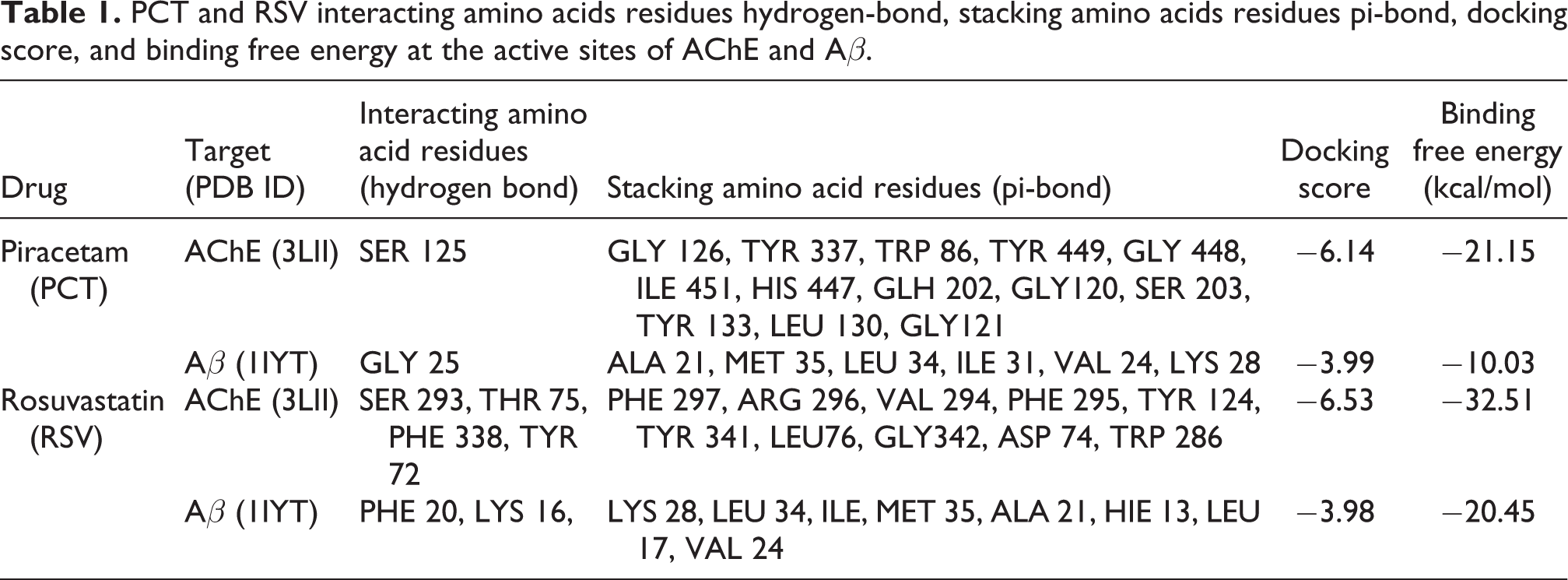
The results were evaluated by interacting amino acid residues’ hydrogen-bond, stacking amino acid residues’ pi-bond, docking score, and binding free energy at the active sites of AChE and Aβ. The docking scores determine the strength of interaction between a ligand and an enzyme (Figures 1 to 4). The lowest docking scores are the outcome of the best binding conformer at its receptor site or the active site of an enzyme. The docked ligand (PCT and RSV) displayed acceptable docking scores and binding free energy for AChE and Aβ. These results suggest that the above-mentioned drug could ameliorate cognitive impairment. These reports strongly suggest that the recognition of key structural features of RSV template will be helpful in designing and synthesizing new analogues with improved AChE inhibitory activity along with decreased Aβ peptide deposition and aggregation. All these results are summarized in Table 1.
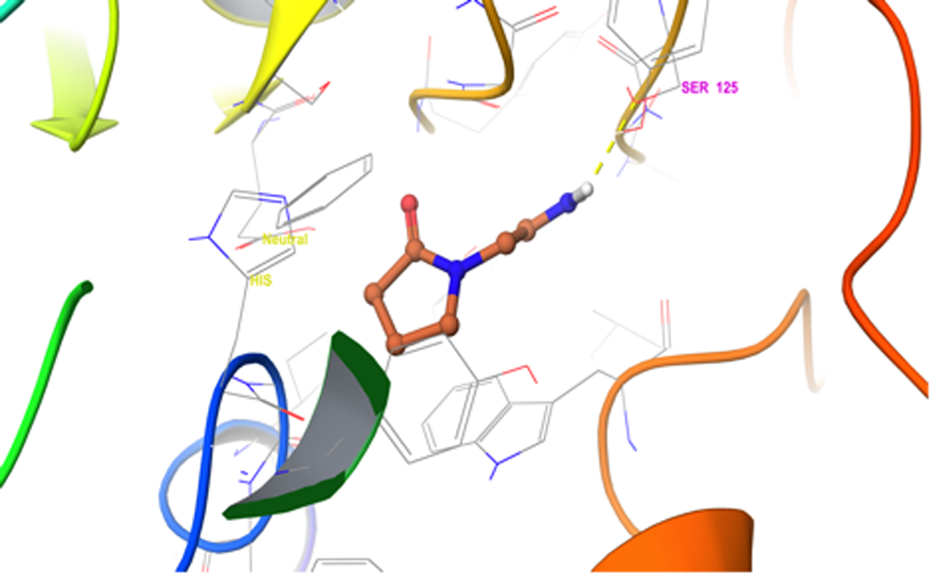
Interaction of PCT docked to 3LII and the ligand (PCT) has been shown in ‘stick’ representation. PCT: piracetam.

Interaction of PCT docked to 1IYT and the ligand (PCT) has been shown in ‘stick’ representation. PCT: piracetam.

Interaction of RSV docked to 3LII and the ligand (RSV) has been shown in ‘stick’ representation. RSV: rosuvastatin.

Interaction of RSV docked to 1IYT and the ligand (RSV) has been shown in ‘stick’ representation. RSV: rosuvastatin.
PCT and RSV interacting amino acids residues hydrogen-bond, stacking amino acids residues pi-bond, docking score, and binding free energy at the active sites of AChE and Aβ.
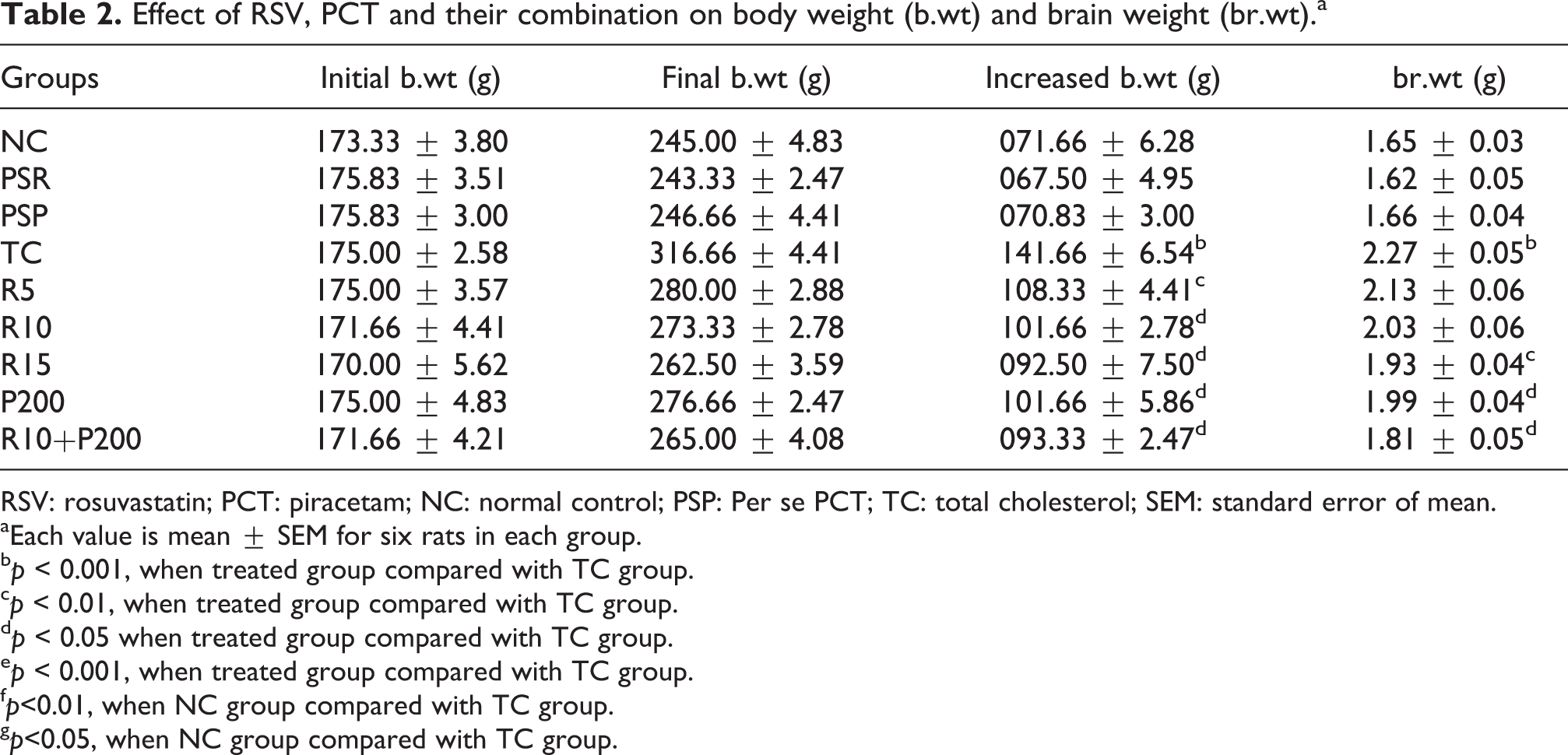
Body weight and brain weight
Body weight and brain weight increased during HSCD in all experimental groups but the maximum increase was observed in the TC group (Table 2).
Effect of RSV, PCT and their combination on body weight (b.wt) and brain weight (br.wt).a
RSV: rosuvastatin; PCT: piracetam; NC: normal control; PSP: Per se PCT; TC: total cholesterol; SEM: standard error of mean.
aEach value is mean ± SEM for six rats in each group.
b p < 0.001, when treated group compared with TC group.
c p < 0.01, when treated group compared with TC group.
d p < 0.05 when treated group compared with TC group.
e p < 0.001, when treated group compared with TC group.
f p<0.01, when NC group compared with TC group.
g p<0.05, when NC group compared with TC group.
Locomotor activity
Locomotor responses, such as move time, rest time, horizontal activity, mean velocity, and total movements, were observed. Mean time, horizontal activity, means velocity, and total movements were significantly decreased in the TC group when compared with the NC group. However, rest time was significantly (### p < 0.001) increased. Locomotor responses of RSV and PCT combination dose group such as mean time, horizontal activity, mean velocity, and total movements exhibit increased (***p < 0.001) as compared to the TC group. Concurrently, rest time was significantly (***p < 0.001) decreased in comparison to the TC group (Table 3).
Effect of RSV, PCT and their combination on locomotor activity.a

RSV: rosuvastatin; PCT: piracetam; NC: normal control; PSP: Per se PCT; TC: total cholesterol; SEM: standard error of mean.
aEach value is mean ± SEM for six rats in each group.
b p < 0.001, when NC group compared with TC group.
c p<0.01, when treated group compared with TC group.
d p < 0.001, when treated group compared with TC group.
e p<0.01, when NC group compared with TC group.
f p<0.05, when NC group compared with TC group.
g p<0.05, when treated group compared with TC group.
Effect on serum TC level
A significant increase (### p < 0.001) in the TC level in the TC group was observed in comparison to the NC group. This effect was significantly reversed by dose-dependent RSV administration in the fashion of R5 (*p < 0.05), R10 (**p < 0.01), and R15 (***p < 0.001) in comparison to the TC group. Combination of R10+P200 also showed a significant (***p < 0.001) and marked reduction in the TC level (Figure 5).
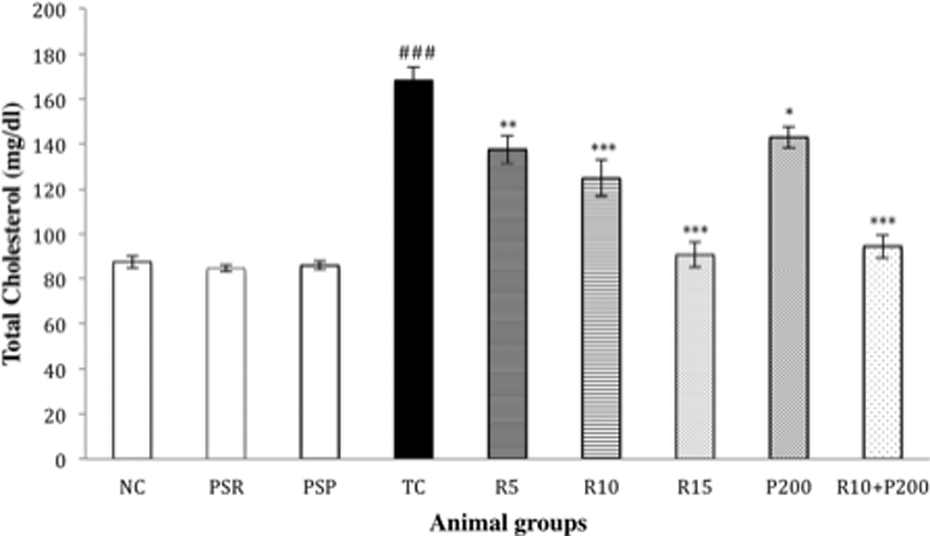
Effect of RSV, PCT, and their combination on serum TC level. Data are presented as mean ± SEM for six rats in each group. ###p < 0.001 versus TC group; ***p < 0.001; **p < 0.01; *p < 0.05 versus TC group. RSV: rosuvastatin; PCT: piracetam; TC: toxic control; SEM: standard error of mean.
Effect on AChE activity
Effects of RSV on the activity of neurotransmitter AChE in the brain of different experimental groups are summarized in Figure 6. A significantly (### p < 0.001) increased AChE activity in the TC group was observed in comparison to the NC group. A non-significant (p > 0.05) amelioration in AChE activity in the brains of R5 group rats was observed in comparison to the TC group. While RSV administration significantly (R10, *p < 0.05 and R15, ***p < 0.001) reversed the decreased AChE activity in comparison to TC group dose dependently. The combination of both R10+P200 also showed a significant (***p < 0.001) as well as more marked reduction in the AChE activity.
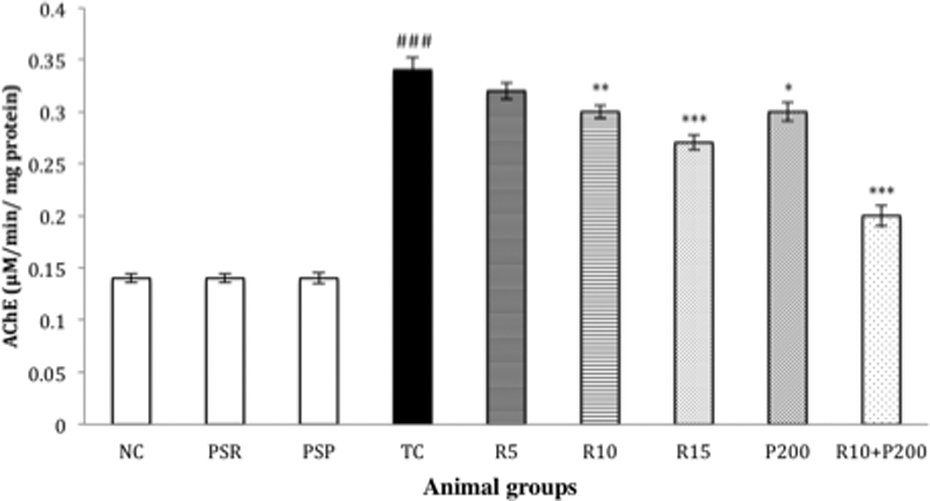
Effect of RSV, PCT, and their combination on AChE level in the brain. Data are presented as mean ± SEM for six rats in each group. ###p < 0.001 versus TC group. ***p < 0.001; **p < 0.01; *p < 0.05 versus TC group. RSV: rosuvastatin; PCT: piracetam; AChE: acetylcholinesterase; SEM: standard error of mean; TC: toxic control.
Effect on Aβ1–42 content in brain and serum level
Figure 7 illustrates the different levels of serum Aβ1–42 along with soluble and insoluble Aβ in brain. A significantly (### p < 0.001) increased level of serum Aβ1–42 along with soluble and insoluble Aβ1–42 in the brain of the TC group was observed as compared to the NC group. This effect was significantly reversed by RSV (R5, R10, and R15) administration dose dependently (**p < 0.01, ***p<0.001, ***p<0.001). The combination of R10+P200 also showed a significant reduction in all the above different Aβ1–42 levels (***p<0.001).
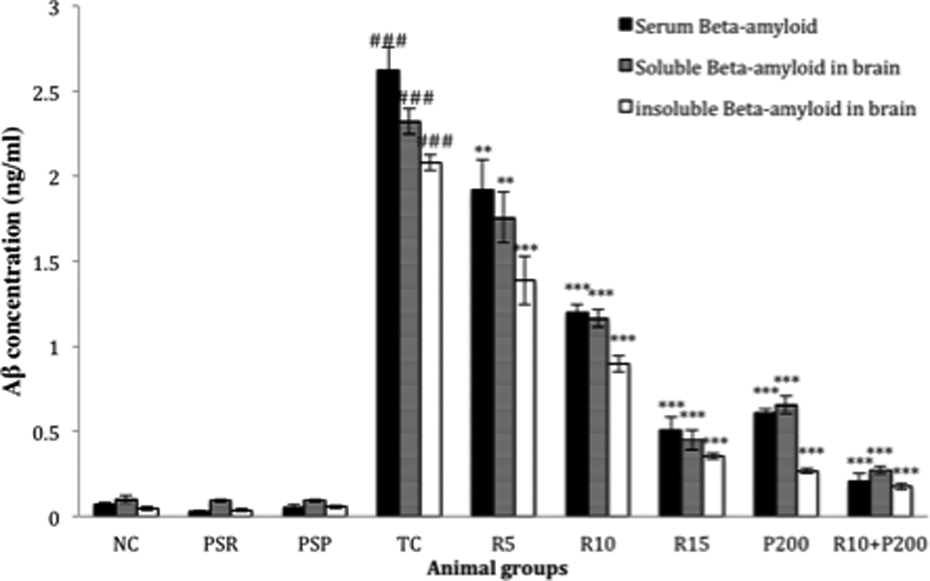
Effect of RSV, PCT, and their combination on the level of insoluble and soluble Aβ1–42 in the brain and serum. Data are presented as mean ± SEM for six rats in each group. ###p < 0.001 versus TC group. ***p < 0.001; **p < 0.01; *p < 0.05 versus TC group. SEM: standard error of mean; TC: toxic control; RSV: rosuvastatin; PCT: piracetam.
Effect on brain histopathology
As illustrated in Figure 8, an irregular shape of neurons and the nuclear membrane were seen in hippocampus of the TC group. These neuronal damages were reversed after RSV and PCT administration to different groups. There was a closely arranged neurons along with intact neuronal shape of nuclear membrane was observed clearly in the NC, RSV, and PCT treated group. The combination of both RSV and PCT more markedly preserved the neuronal architecture. The same pattern of morphological improvement in the treated group was observed in cortex also (Figure 9).
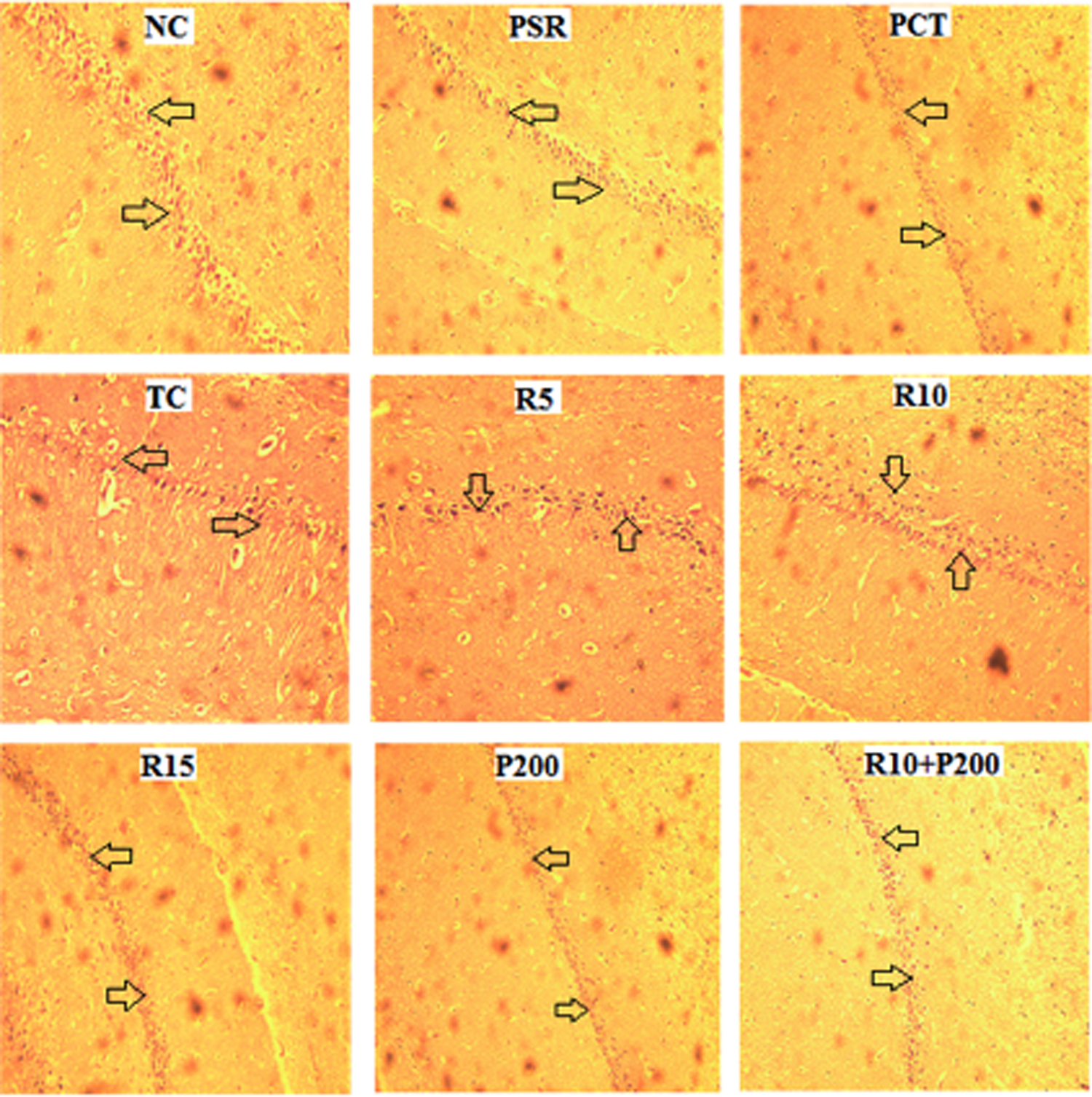
Effect of RSV, PCT, and their combination on neuron density in hippocampus of rat brain. The photomicrographs of coronal section of hippocampus stained with hematoxylin and eosin (×100). RSV: rosuvastatin; PCT: piracetam.

Effect of RSV, PCT, and their combination on neuron density in cortex of rat brain. The photomicrographs of coronal sections cortex stained with hematoxylin and eosin (×100). RSV: rosuvastatin; PCT: piracetam.
Discussion
In the present study, we used molecular docking to predict the affinity of RSV on Aβ1–42 and AChE. We also performed in vivo experiments to further prove that RSV could up-regulate cholinergic system and decrease Aβ load. Many progresses were made recently on the “one molecule-one target” compounds. 40,41 However, a new therapeutic concept “one molecule-multiple targets” is considered to be a better paradigm for treating cognitive impairment (Figure 10). AChE and Aβ peptide aggregation seems to be a critical node within the complex pathological network of cognitive impairment. 42,43 Cholinergic degeneration can accelerate Aβ burden in vivo. 44 Therefore, discovering some dual inhibitors of both Aβ aggregation and AChE activity will be necessary for potential therapeutic approaches that can slow or mitigate the progression of cognitive impairment.

Therapeutic approach of RSV “one molecule-multiple targets” for better paradigm in treating cognitive impairment. RSV: rosuvastatin.
Our molecular docking study provides evidence that RSV binds the active sites of AChE and Aβ1–42. RSV has a similar glide score in comparison to PCT for Aβ1–42 and AChE. These reports strongly suggest that the recognition of key structural features of RSV template will be helpful in designing and synthesizing new analogues with improved AChE inhibitory activity along with decreased Aβ1–42 peptide deposition and aggregation. The molecular docking study was performed to gain insights into the interaction of RSV with the residues of the active sites of AChE and Aβ1–42 as well as to investigate the underlying mechanism of action. It has been proposed that an ideal AChE inhibitor should bind to the catalytic and peripheral sites simultaneously, which could disrupt the interactions between the enzyme and the Aβ1–42 peptide, and hence, slow down the progression of the disease. 45 Further insights into the nature and number of bonds formed revealed that hydrogen bonds, polar bonds, and hydrophobic interactions play an important role in the “RSV–AChE” interaction. The binding energy determines the strength of interaction between a ligand and an enzyme. The lowest binding energy is the outcome of the best binding conformer at its receptor site or active site of an enzyme. 46 The docked ligand (RSV) displayed acceptable binding energy values for AChE. These results suggest that the above-mentioned drug could serve as a dual inhibitor for hyperlipidemic and cognitive impairment.
Body weight was gradually increased in all experimental groups, but TC group rats gained more marked increase in body weight. 47,48 Concurrent to previous studies, RSV and PCT either alone or in combination ameliorated the change in BW. Moreover, studies also suggest that the decrease in weight may reduce the progression of memory impairment. 49,50
Consistent to previous reports, we observed that administration of RSV decreased the serum cholesterol concentration in TC rats, suggesting that RSV could improve hypercholesterolemia associated Aβ1–42 deposition. On the other hand, a high-cholesterol diet may lead to Aβ1–42 peptide aggregation. 51 Moreover, it has been suggested that cholesterol fractions could be involved in both AD and vascular dementia. 52,53 It has been suggested that the useful effects of statins on neurodegeneration may not be only due to their cholesterol lowering effects, but also due to their cholesterol-independent or pleiotropic effects, such as improving blood flow, reducing coagulation, modulating the immune system, and reducing oxidative damage. 54,55
All animal groups showed a significant difference in the total amount of photocell beam crossing during LA testing, ruled out the possibility that higher body weight in TC group rats that could be an attribute for the poor performance on memory task. Consistently, impairment of motor performance test in TC group rats was found and the decreased locomotor activities such as move time, horizontal activity, mean velocity, and total movement and increase in rest time. 56 RSV and PCT either alone or in combination improved these alterations in motor performances. These observations indicate that the improvement in motor performance might be due to the reduction in AChE and Aβ1––42 peptide aggregation. 56,57
It is well accepted that the cholinergic system, which is important for learning and memory, is closely related to age-related diseases such as AD and particularly important in the pathogenesis of AD. 58 ACh is a neurotransmitter necessary for memory tasks and retrieval. ACh synthesis depends on the availability of acetyl Co-A, provided by the breakdown of glucose and insulin, which regulates the activity of choline acetyl transferase. On the other hand, activity of AChE, a hydrolyzing enzyme for ACh, has been considered as one of the markers of cholinergic function. AChE regulates the cholinergic neurons and neuromuscular transmission. Increased activity of AChE leads to rapid break down of ACh, which correlates cholinergic system abnormalities with intellectual impairment. We observed marked increased activity of AChE in TC rats in consistent with the previous report. 59 Furthermore, RSV treatment markedly attenuated AChE activity in the brain that ameliorated the HSCD diet-induced cognitive impairment. Collectively, data from behavioral test and cholinergic estimation showed the beneficial effects of RSV in enhancing the memory and cognition in HSCD-fed rats.
In the amyloid cascade evidence, the accumulated and oligomerized Aβ1–42 causes progressive and neuritic injury, thereby inducing oxidative injury, which may be responsible for the synaptic dysfunction and memory impairment in the AD patients. 60 Therefore, measuring the quantity of Aβ1–42 in the serum and brains in our HSCD-fed rats could indicate the therapeutic effects of RSV. On the basis of the results, we found a decrease in the levels of the soluble Aβ1–42, as well as insoluble Aβ1–42 in serum and brain, featuring a remarkable reduction in surface area of senile plaque deposits after treatment with RSV. This might correspond to our assumptions in the in silico study by binding to the Aβ1–42, RSV halted the aggregation of Aβ1–42, thereby reducing the formation of senile plaques.
Our results demonstrated that the brain weight of the TC group was significantly higher than that of NC groups. This finding has been supported by the previous studies. 61,62 The increase in brain weight is indicative of the brain pathological condition, which was confirmed by the histopathological analysis of hippocampus and cortex in our study. 63 RSV and PCT either alone or in combination maintained the neuronal architecture in HSCD-fed rats. This further strengthens the earlier suggested protective effects of RSV and PCT in HSCD-fed rats.
Conclusion
Taken together, we can conclude that HSCD was significantly associated with cognitive impairment in rats. Treatment with RSV ameliorated cognitive impairment by improved LA, reducing cholesterol deposition, AChE activity, and Aβ1–42 peptide aggregation which in turn supported by in silico study. Our findings encourage using of RSV, as a preventive approach for cognitive impairment in humans.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work was supported by the grant funded by the Department of Science and Technology (IF130014), Government of India, New Delhi.
