Abstract
Spinal cord injury (SCI) is one of the most common destructive injuries, which may lead to permanent neurological dysfunction. Currently, transplantation of bone marrow mesenchymal stem cells (BMSCs) in experimental models of SCI shows promise as effective therapies. BMSCs secrete various factors that can regulate the microenvironment, which is called paracrine effect. Among these paracrine substances, exosomes are considered to be the most valuable therapeutic factors. Our study found that BMSCs-derived exosomes therapy attenuated cell apoptosis and inflammation response in the injured spinal cord tissues. In in vitro studies, BMSCs-derived exosomes significantly inhibited lipopolysaccharide (LPS)-induced PC12 cell apoptosis, reduced the secretion of pro-inflammatory factors including tumor necrosis factor (TNF)-α and IL (interleukin)-1β and promoted the secretion of anti-inflammatory factors including IL-10 and IL-4. Moreover, we found that LPS-induced protein expression of toll-like receptor 4 (TLR4), myeloid differentiation factor 88 (MyD88) and nuclear transcription factor-κB (NF-κB) was significantly downregulated after treatment with BMSCs-derived exosomes. In in vivo studies, we found that hindlimb motor function was significantly improved in SCI rats with systemic administration of BMSCs-derived exosomes. We also observed that the expression of pro-apoptotic proteins and pro-inflammatory factors was significantly decreased, while the expression of anti-apoptotic proteins and anti-inflammatory factors were upregulated in SCI rats after exosome treatment. In conclusion, BMSCs-derived exosomes can inhibit apoptosis and inflammation response induced by injury and promote motor function recovery by inhibiting the TLR4/MyD88/NF-κB signaling pathway, which suggests that BMSCs-derived exosomes are expected to become a new therapeutic strategy for SCI.
Keywords
Introduction
Spinal cord injury (SCI) is usually a damage to the spinal cord or cauda equina caused by devastating trauma, often causing temporary or permanent neurological dysfunction, such as paralysis and neuralgia.1,2 In recent years, the incidence of SCI in China has gradually increased, but clinically there is a lack of effective treatment for patients with SCI. For a long time, nerve fibers in the injured spinal cord have been considered non-regenerative, but recent studies have shown that nerve fibers in the injured spinal cord can regenerate as long as the adverse microenvironment of the central nervous system for axon growth is overcome.3,4
Bone marrow mesenchymal stem cells (BMSCs) have multidirectional differentiation characteristics and potential neuroprotective capacity. Their transplantation therapy has been applied in a variety of animal models and clinical studies.5–7 Recent studies have shown that BMSCs have a therapeutic effect not mainly relying on the characteristics of stem cells, but rather relying on their paracrine and endocrine effects at the transplantation sites, affecting the microenvironment of the body, exerting immunoregulatory functions, and promoting the nutritional regeneration of damaged tissues.8–10 Among them, exosomes dominate the paracrine role of BMSCs. As an important mediator of intercellular material transfer and information exchange, exosomes are extracellular vesicles with a diameter of 30–100 nm actively secreted by cells, which carry a variety of proteins, lipids, mRNAs, microRNAs and other molecules. In addition, exosomes can selectively sort intracellular substances into vesicles and discharge them into the extracellular space through endosomal pathways and membrane fusion, affecting and altering the biological behavior of other cells.11–13
At present, the therapeutic role of exosomes derived from mesenchymal stem cells (MSCs) in a variety of diseases has been reported. In the field of renal injury repairment, MSCs-derived exosomes can play renal protective and regenerative roles by regulating proliferation and promoting growth factor expression. 14 Exosomes from adipose-derived mesenchymal stem cells can activate the Wnt/β-catenin pathway to regulate myocardial autophagy levels and reduce myocardial ischemic injury. 15 As the main mediator of intercellular communication, exosomes play an important role in the occurrence and development of nervous system diseases, as well as in the repair of nerve injury. Studies have revealed that exosomes play therapeutic roles in ischemic stroke and other neurological diseases, such as promoting regeneration of injured areas and reducing neuronal necrosis.16–18 Other studies have found that after SCI, MSCs-derived exosomes can significantly attenuate apoptotic levels and inflammatory response, promote vascular regeneration of injured spinal cord, inhibit lesion size and promote functional recovery of injured spinal cord. 19
Based on the current research status at home and abroad, we treated LPS-induced PC12 cells and rat SCI models with BMSCs-derived exosomes, and explored the main mechanisms regulating apoptosis and inflammation response, aiming to provide a new reference for reducing SCI and promoting nerve repairment.
Materials and methods
Animals
Adult male Sprague-Dawley rats (200–250 g) were provided by Henan Laboratory Animal Center (Henan, China). All animals were housed in individual cages in light cycle-controlled environment with free access to food and water. Eighteen SD rats were divided into sham group, control group and exosome group, with 6 rats in each group. In sham group, the T10 lamina was removed and sutured layer by layer without spinal cord impact. Both the control group and the exosome group were subjected to SCI modeling. 20 After modeling, 200 µL of exosomes were injected into the tail vein daily for 7 consecutive days, and an equal volume of PBS treated was used as a control. BBB scores of hindlimb motor function were performed in each group at days 1, 3, 7, 14, 21 and 28 after operation. All animal care and experimental procedures were approved by the Ethics Committee of Second Affiliated Hospital of Xi’an Jiaotong University (XJ2020106).
Cell culture
The BMSCs were purchased from the American Type Culture Collection (ATCC; https://www.atcc.org/) and stored in Dulbecco’s modified Eagle’s medium (DMEM, Sigma-Aldrich Chemical Company, St Louis, MO) containing 15% FBS (Sigma-Aldrich Chemical Company) and penicillin-streptomycin solution (100×; Thermo Fisher Scientific Inc, Waltham, MA). When cells were 85% confluent, they were trypsinized and subcultured at a ratio of 1:2.
Exosome isolation and identification
When BMSCs were subcultured to passage 3-passage 5, extracellular vesicles were collected. The specific procedures were as follows: the medium was collected and centrifuged at 300 × g for 10 min, followed by centrifugation at 2000 × g for 10 min at 4°C. After centrifugation, a 0.22-µm sterile filter (Steritop™ Millipore, Burlington, MA) was used to remove the whole cells and cellular debris followed by obtaining the cell supernatant. After 24 h of incubation, the conditioned medium (CM) was collected and exosomes were isolated by using exosome extraction kit (Wako pure Chemicals Industry, 293-77601) according to the manufacturer’s instructions. The expression levels of exosome-specific biomarkers CD9, CD63, CD81, TSG101 and Alix were detected by Western blotting, and protein concentration was determined by using a bicinchoninic acid protein assay (BCA; Thermo Fisher Scientific, Waltham, MA), and then stored in PBS at −80 °C for further experiments.
Flow cytometry
The cells were collected and washed twice with PBS. The cell suspension (100 μL) with a cell density of 1 × 106 cells/mL was transferred into the culture tube and then incubated with 5 µL Annexin V-FITC and 5 µL propidium iodide at room temperature for 20 min in the dark. Finally, 400 µL of binding buffer was added, and apoptotic cells were determined by flow cytometry (BD Biosciences, USA, 556570).
Western blotting
Total protein was extracted from PC12 cells or spinal cord tissues by using Pro-prep TM protein Extraction Solution (Daejeon, Korea). The protein content of each sample was determined using the BCA Protein Assay Kits (Thermo Scientific). Then, equal amounts of proteins (15 μg/lane) were separated on a 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidenedifluoride (PVDF) membranes (Bio-Rad, Hercules, CA, USA). The membranes were blocked in 5% (w/v) nonfat dry milk in TBST (Tris-buffered saline-0.1%Tween) at 25°C for 3 h and then incubated with the following primary antibodies: rabbit polyclonal anti-β-actin antibody (1:1000, Abcam, ab8227), rabbit monoclonal anti-CD9 antibody (1:2000, Abcam, ab92726), rabbit monoclonal anti-CD63 antibody (1:1000, Abcam, ab217345), mouse monoclonal anti-CD81 antibody (1:1500, Abcam, ab79559), rabbit monoclonal anti-TSG101 antibody (1:3000, Abcam, ab125011), rabbit monoclonal anti-Alix antibody (1:2500, Abcam, ab186492), rabbit polyclonal anti-cleaved caspase3 antibody (1:1100, Abcam, ab2302), rabbit monoclonal anti-Bax antibody (1:2000, Abcam, ab32503), rabbit polyclonal anti-TLR4 antibody (1:500, Abcam, ab13556), rabbit monoclonal anti-MyD88 antibody (1:1000, Abcam, ab199247), rabbit monoclonal anti-TRAF6 antibody (1:800, Abcam, ab227560). Then, the membranes were incubated with horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (1:3000, Abcam, ab6721) or goat anti-mouse IgG (1:3000, Abcam, ab6728) for 1 h at room temperature. The bands were visualized by using an ECL Plus Chemiluminescence Reagent Kit (Pierce, Rockford, IL, USA) and were photographed by a chemiluminescence imaging system. Image J software was used to quantify the band densities.
ELISA
The secretion of pro-inflammatory factors including TNF-α (SEKR-0009-96T), IL-1β (SEKR-0002-96T), MCP-1 (SEKR-0024-96T) and MIP-1 (SEKR-0025-96T) in PC12 cell supernatant was detected by ELISA kits (Solarbio, China) following the manufacturer’s instructions. Similarly, the levels of anti-inflammatory factors including IL-10 (SEKR-0006-96T) and IL-4 (SEKR-0004-96T) were detected by ELISA kits (Solarbio, China).
RT-qPCR
Total RNA was isolated from PC12 cells or spinal cord tissues by using the TRIzol (Invitrogen, Carlsbad, CA, USA). Then the first strand of cDNA was synthesized using MMLV Reverse Transcriptase kit (TaKaRa, Da Lian, China). Real-time PCR was conducted by using SYBR Premix Ex TaqTM Kit (Applied Biosystems, Foster City, CA, USA). The reaction was run in ABI7500 Real-time PCR system (Applied Biosystems, Carlsbad, CA). GAPDH was used as an endogenous control. Briefly, 2 µL of cDNA was added to 10 µL of the 2×SYBR green PCR master mix with 0.4 µL of Taq polymerase enzyme (RiboBio, China), 0.8 µL of each primer and 6 µL ddH2O to a final volume of 20 µL. The RT-qPCR cycling conditions consisted of: 95°C for 3 min; then 35 cycle amplification for 20 s at 95°C, 30 s at 55°C, 15 s at 72°C; followed by 1 min at 72°C. The primers used in this study were synthesized by Sangon Biotech (Shanghai, China). The relative expression levels were normalized by using the 2−ΔΔCt method. The following primers were used: CD9 (forward 5’ TTG GAC TAT GGC TCC GAT TC 3’, reverse 5’ GGC GAA TAT CAC CAA GAG GA 3’); CD63 (forward 5’ ATG GCA CTC GGG TGT GGA TAC 3’, reverse 5’ TAC AGT ATA ATT CGT TTT GT 3’); CD81 (forward 5’ TGT ATG TGT CGT GGG TC 3’, reverse 5’ T GTC GTG ATC GCG 3’); TSG101 (forward 5’ GGC TGT CCT TAC CCA CCT G 3’, reverse 5’ CCA TTT CCT CCT TCA TCC GC 3’); Alix (forward 5’ TGC GGA TTA CTT TGG CGA TG 3’, reverse 5’ TGC GAT CTC TTC CCC AAA CT 3’); GAPDH (forward 5’ TGA CCA CAG TCC ATG CCA TCA 3’, reverse 5’ GCC TGC TTC ACC ACC TTC TTG 3’).
Statistical analysis
All statistical analyses were performed by using the SPSS software (ver.22.0; SPSS, Chicago, IL). All data were shown as mean ± SEM. Comparisons between two groups were made by Student’s t-test. Data between multiple groups were analyzed with one-way ANOVA followed by post hoc analysis with LSD test. P < 0.05 was considered statistical significance.
Results
Isolation and identification of BMSCs-derived exosomes
The cell culture supernatant was collected, and the extracellular microvesicles of BMSCs were separated and purified by differential centrifugation. After repeated centrifugation, the sediment at the bottom of the centrifuge tube was taken for identification. The results of qPCR and Western blotting confirmed the presence of exosomes, and we observed that surface markers of exosomes, including CD9 (195% increase compared to supernatant group, p < 0.01) (Figure 1A & 1F), CD63 (320% increase compared to supernatant group, p < 0.01) (Figure 1B & 1F), CD81 (169% increase compared to supernatant group, p < 0.01) (Figure 1C & 1F), TSG101 (311% increase compared to supernatant group, p < 0.01) (Figure 1D & 1F) and Alix (221% increase compared to supernatant group, p < 0.01) (Figure 1E & 1F), were significantly enriched in the extracts compared with the normal cultured BMSCs or the medium supernatant.

Isolation and identification of BMSCs-derived exosomes. The supernatant of cell culture was collected and the exosomes derived from rat bone marrow mesenchymal stem cells (BMSCs-Exo) were extracted by differential centrifugation. Relative expression of exosome-specific genes such as CD9, CD63, CD81, TSG101 and Alix was detected by qPCR (1A–1E). Western botting was used to detect the relative expression of exosome marker proteins (1F). β-actin was used as an internal reference. N = 6, ** P < 0.01.
BMSCs-derived exosomes inhibit LPS-induced apoptosis in PC12 cells
Conditioned medium or BMSCs-derived exosomes were incubated with LPS-induced PC12 cells, respectively. Flow cytometry results suggested that LPS strongly induced apoptosis in PC12 cells (126% increase compared to control group, p < 0.01), and when LPS treated cells were incubated with conditioned medium, we observed reduced apoptosis (20% decrease compared to LPS group, p < 0.01), whereas LPS treated cells incubated with BMSCs derived exosomes exhibited less apoptosis (32% decrease compared to LPS + medium group, p < 0.01) (Figure 2A). Similarly, Western blotting results showed that BMSCs-derived exosomes significantly downregulated the expression of LPS-induced pro-apoptotic proteins including cleaved caspase3 (51% decrease compared to LPS + medium group, p < 0.01) (Figure 2B & 2C) and Bax (44% decrease compared to LPS + medium group, p < 0.01) (Figure 2B & 2D), but the expression of anti-apoptotic protein Bcl-2 was significantly upregulated (39% increase compared to LPS + medium group, p < 0.01) (Figure 2B & 2E).

BMSCs-derived exosomes inhibit LPS-induced apoptosis in PC12 cells. PC12 cells were stimulated with 50 ng/ml LPS (cell density was 1 × 106) for 6 h. Apoptosis of LPS-induced PC12 cells were detected by flow cytometry (2A). The protein expression of Cleaved caspase3 (2B & 2C), Bax (2B & 2D) and Bcl-2 (2B & 2E) in LPS-induced PC12 cells was analyzed by Western botting. β-actin was used as an invariant internal control for calculating protein fold-changes. N = 6, ** P < 0.01.
BMSCs-derived exosomes inhibit LPS-induced inflammatory factor secretion in PC12 cells
ELISA was used to detect the regulatory effect of BMSCs-derived exosomes on the secretion levels of inflammatory factors. As expected, LPS significantly induced the secretion of pro-inflammatory factors including TNF-α (706% increase compared to control group, p < 0.01) (Figure 3A), IL-1β (339% increase compared to control group, p < 0.01) (Figure 3B), MCP-1 (293% increase compared to control group, p < 0.01) (Figure 3C) and MIP-1 (313% increase compared to control group, p < 0.01) (Figure 3D), while BMSCs-derived exosomes significantly reversed the LPS-induced secretion of pro-inflammatory factors. Furthermore, ELISA results showed that BMSCs-derived exosomes significantly upregulated the secretion levels of anti-inflammatory factors including IL-10 (54% increase compared to LPS + medium group, p < 0.01) (Figure 3E) and IL-4 (76% increase compared to LPS + medium group, p < 0.01) (Figure 3F).

BMSCs-derived exosomes inhibit LPS-induced inflammatory factor secretion in PC12 cells. PC12 cells were stimulated with 50 ng/ml LPS (cell density was 1 × 106) for 6 h, and the cell culture supernatant was collected. The secretion levels of TNF-α (3A), IL-1β (3B), MCP-1 (3C), MIP-1 (3D), IL-10 (3E) and IL-4 (3F) were analyzed by ELISA kits, respectively. N = 6, ** P < 0.01.
BMSCs-derived exosomes inhibit LPS-induced activation of the TLR4/MyD88/NF-κB signaling pathway
In this study, Western blotting results showed that the protein expression of Toll-like receptor 4 (TLR4) (332% increase compared to control group, p < 0.01), myeloid differentiation factor (MyD88) (389% increase compared to control group, p < 0.01) and tumor necrosis factor receptor-associated factors 6 (TRAF6) (256% increase compared to control group, p < 0.01) was upregulated in LPS-induced PC12 cells (Figure 4A). In turn, LPS activated the inhibitor of NF-κB kinase complex IκKβ (IκB), promoted the phosphorylation of IκB (168% increase compared to control group, p < 0.01), and aggravated the inflammatory response (Figure 4B & 4C). However, BMSCs-derived exosomes significantly inhibited LPS-induced activation of TLR4/MyD88/NF-κB signaling pathway, suggesting that BMSCs-derived exosomes play a protective role in inflammatory injury.
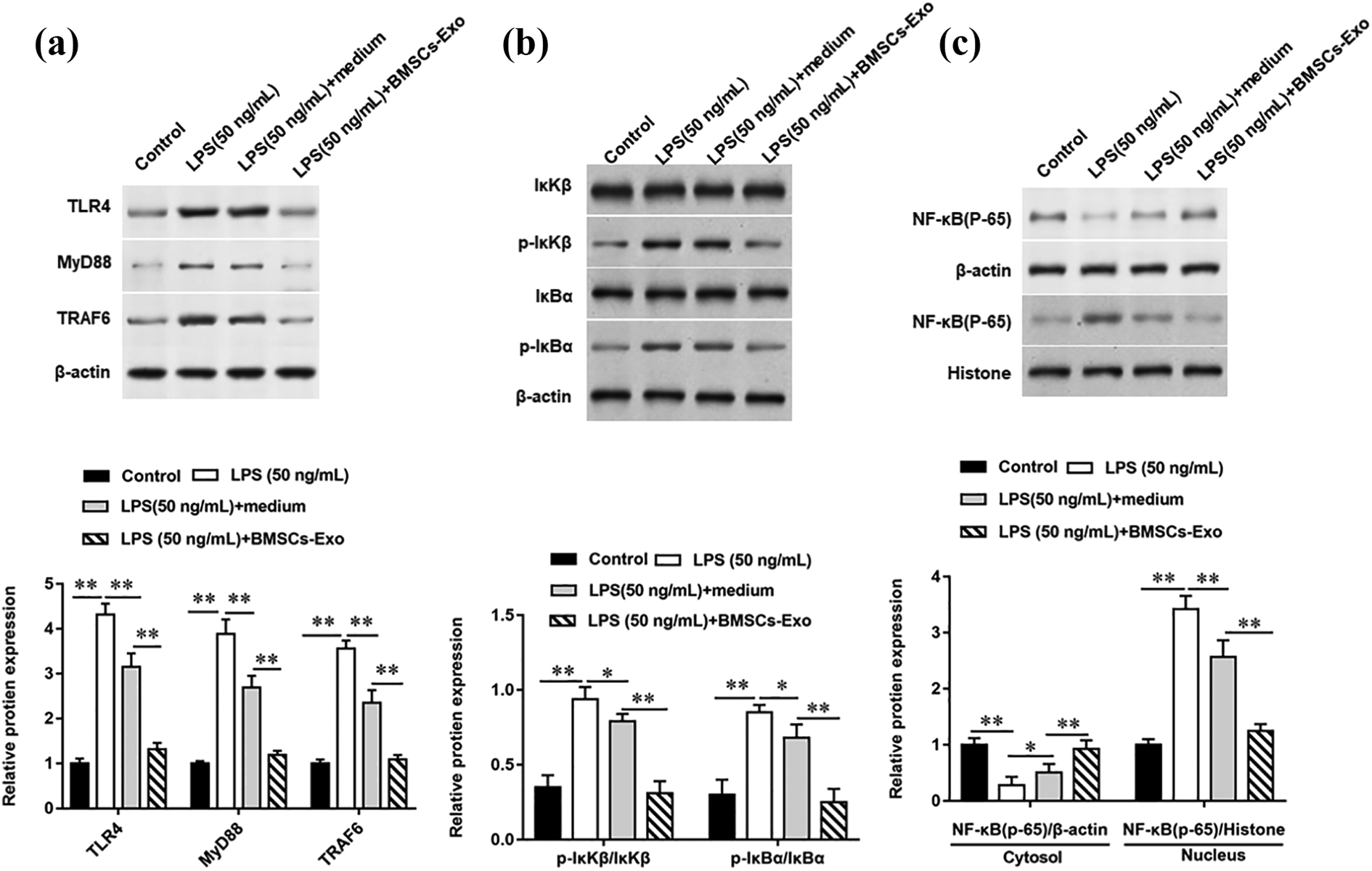
BMSCs-derived exosomes inhibit LPS-induced activation of TLR4/MyD88/NF-κB signaling pathway. PC12 cells were stimulated with 50 ng/ml LPS (cell density was 1 × 106) for 6 h. Relative expression of inflammatory signaling pathway-related proteins including TLR4, MyD88, TRAF6 and NF-κB was determined by Western botting. β-actin was used as the loading control. N = 6, ** P < 0.01.
BMSCs-derived exosomes inhibit cell apoptosis in rats with SCI
The BBB scores of the model animals were scored on the days 1, 3, 7, 14, 21 and 28 after modeling. As shown in Figure 5A, BMSCs-derived exosomes significantly increased BBB scores in rats with SCI, suggesting that they have positive significance for SCI repair. In addition, Western blotting results suggested that BMSCs-derived exosomes significantly downregulated the expression of pro-apoptotic proteins including cleaved caspase 3 (71% decrease compared to SCI group, p < 0.01) (Figure 5B) and Bax (62% decrease compared to SCI group, p < 0.01) (Figure 5C) in injured spinal cord of SCI rats compared with SCI model group, but the expression of anti-apoptotic protein Bcl-2 was significantly upregulated (718% increase compared to SCI group, p < 0.01) (Figure 5D). In addition, we observed that TLR4, MyD88 and TRAF6 proteins were upregulated and NF-κB phosphorylation levels were increased in the injured spinal cord tissues of SCI rats compared with the sham group, while BMSCs derived exosomes inhibited the expression of TLR4 pathway related proteins and decreased NF-κB phosphorylation in the injured spinal cord tissues of SCI rats (Figure 5D).

BMSCs-derived exosomes inhibit cell apoptosis in rats with SCI. Eighteen SD rats were divided into three groups, six in each group, named sham group, SCI group and exosomes group. BBB scores of hindlimb motor functions were performed in each group on days 1, 3, 7, 14, 21 and 28 after operation (5A). Total protein was extracted from injured spinal cord tissues of rats after full grinding, and Western blotting was used to detect the expression of apoptosis-related proteins such as cleaved caspase 3 (5B), Bax (5C) and Bcl-2 (5D). Relative expression of inflammatory signaling pathway-related proteins including TLR4, MyD88, TRAF6 and NF-κB was determined by Western botting (5E). β-actin was used as the loading control. N = 6, ** P < 0.01.
BMSCs-derived exosomes inhibit the inflammatory factors secretion in rats with SCI
The SCI tissues of rats were taken on the day 7 after modeling, and the regulatory effect of BMSCs-derived exosomes on the secretion of inflammatory factors in SCI rats was detected by ELISA. As expected, BMSCs-derived exosomes significantly reversed the secretion of pro-inflammatory factors including TNF-α (74% decrease compared to SCI group, p < 0.01) (Figure 6A), IL-1β (75% decrease compared to SCI group, p < 0.01) (Figure 6B), MCP-1 (72% decrease compared to SCI group, p < 0.01) (Figure 6C), and MIP-1 (56% decrease compared to SCI group, p < 0.01) (Figure 6D). Furthermore, ELISA results showed that BMSCs-derived exosomes significantly upregulated the secretion levels of anti-inflammatory factors including IL-10 (132% decrease compared to SCI group, p < 0.01) (Figure 6E) and IL-4 (107% decrease compared to SCI group, p < 0.01) (Figure 6F).

BMSCs-derived exosomes inhibit the inflammatory factor secretion in rats with SCI. Eighteen SD rats were divided into three groups, six in each group, named sham group, SCI group and exosomes group. The secretion levels of TNF-α (6A), IL-1β (6B), MCP-1 (6C), MIP-1 (6D), IL-10 (6E) and IL-4 (6F) were analyzed by ELISA kits, respectively. N = 6, ** P < 0.01.
Discussion
SCI can lead to pathological pain, limb paralysis and other diseases, even death. 21 However, persistent neuroinflammation, abnormal microenvironment, and both hinder neuronal functional recoveries. Extracellular vesicles are important components of paracrine factors released by MSCs, which can specifically fuse with target cells and release the contents into target cells, thus playing a specific pathophysiological role.22,23 In this study, BMSCs-derived exosomes could significantly reduce LPS-induced PC12 cell apoptosis and inhibit the secretion of proinflammatory factors. Furthermore, systemic administration of BMSCs-derived exosomes promoted the recovery of motor function in SCI rats.
The important role of inflammation in SCI and injury repair cannot be ignored. In the subacute phase of SCI, local inflammation initiates multiple responses that hinder the repair process of SCI.24,25 Effective avoidance and reduction of inflammation is one of the effective means to eliminate the obstacles of SCI repair and promote the repair and functional regeneration of central nervous system injury. 26 In this study, ELISA results showed that TNF-α, IL-1β, MCP-1 and MIP-1, the four proinflammatory factors, were expressed at a lower level in BMSCs-derived exosomes repair models than in injury models, while the secreted level of anti-inflammatory factors including IL-10 and IL-4 was higher, suggesting that BMSCs-exosomes could inhibit the inflammatory response during SCI. This is similar to the results of previous studies. Studies have reported that exosomes derived from hUCMSCs can downregulate the secretion of inflammatory cytokines, such as TNF-α, MIP-1α, IL-6 and IFN-γ, and improve the functional recovery of SCI mice. 27 In addition, it was found that after systemic administration of MSCs-derived exosomes in SCI rats, the expression levels of pro-apoptotic protein (Bax) and pro-inflammatory cytokines (TNF-α, IL-1β) were significantly decreased, while the expression of anti-apoptotic protein (Bcl-2) and anti-inflammatory cytokine (IL-10) was significantly upregulated. 28 Other studies have reported that BMSCs-derived exosomes can attenuate neuronal apoptosis, inhibit glial scar formation, reduce lesion size, inhibit inflammation, promote axon regeneration, and ultimately improve functional and behavioral recovery after traumatic SCI. 29 Furthermore, a recent study has showed that exosomes derived from MSCs rich in miR-126 can promote angiogenesis and neurogenesis, inhibit apoptosis, and promote functional recovery after SCI. 30
TLR4/MyD88/NF-κB signaling pathway is an important pathway to mediate apoptosis and inflammatory activation. 31 Our study found that BMSCs-derived exosomes alleviated the inflammatory response of SCI of SCI rats by inhibiting the expression of downstream genes of TLR4, including MyD88 and TRAF6, and then inhibiting the release of NF-κB from the cytoplasm into the nucleus. Our experimental results were supported by previous studies. Studies have found that Dl-3-n-butylphthalide can improve the motor recovery of SD rats after SCI, significantly reduce neuronal apoptosis 7 days after operation, inhibit the activation of microglia, and reduce the release of inflammatory mediators after injury. 32 Other studies reported that ROS production and pro-inflammatory cytokines were increased in HO-induced NSPCs, while thymosin β4 reversed these effects and decreased TLR4 and MyD88 expression. Furthermore, HO-exposed NSPCs treated with the TLR4/MyD88 pathway inhibitors showed the same reversal effect as thymosin β4 treatment. 33 Sun et al. have found that the inhibition of TLR4/NF-κB signaling can significantly inhibit the inflammatory responses of spinal astrocytes induced by hypoxia-glucose/reoxygenation and reduce edema. 34 A clinical study of 105 SCI patients and 40 healthy subjects showed that the expression levels of TLR4, MyD88 and NF-κB in PBMCs of SCI patients were significantly higher than those of healthy subjects. 35
This study confirmed the protective effect of BMSCs-derived exosomes on neurological and motor function recovery after SCI through in vivo and in vitro experiments, which further suggested that BMSCs-derived exosomes may become a new therapeutic strategy for SCI.
Footnotes
Abbreviations
SCI: spinal cord injury; BMSCs: bone marrow mesenchymal stem cells; MSCs: mesenchymal stem cells; LPS: lipopolysaccharide; DMEM: Dulbecco’s modified Eagle’s medium; SDS-PAGE: sodium dodecyl sulfate polyacrylamide gel electrophoresis; PVDF: polyvinylidene difluoride; HRP: horseradish peroxidase; IL-1β: interleukin-1β; IL-10: interleukin-10; IL-4: interleukin-4; TNF-α: tumor necrosis factor-α; MCP-1: monocyte chemotactic protein-1; MIP-1: macrophage inflammatory protein-1; TLR4: toll-like receptor 4; MyD88: myeloid differentiation factor 88; TRAF6: tumor necrosis factor receptor-associated factors 6
Author contributions
Liying Fan: Conceptualization, Supervision; Jun Dong: Writing-original draft, Methodology; Xijing He: Resources, Data curation; Chun Zhang: Formal analysis, Software; Ting Zhang: Writing-review & editing
Data availability statement
The datasets used during the present study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures involving animal experiments in this study were in accordance with the ethical standards of the institutional and/or National Research Committee. All animal care and experimental procedures were approved by the Ethics Committee of Second Affiliated Hospital of Xi’an Jiaotong University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No: 81771349) and the general project grant of Shaanxi key R&D program (No: 2018SF-178).
