Abstract
Chrysene, one of the basic polycyclic aromatic hydrocarbons (PAHs), has been reported to make damages to human health and living environment. Chronic obstructive pulmonary disease (COPD) is a progressive disorder with high morbidity and mortality. To investigate the role of chrysene in the development of COPD, male C57BL/6 mice were exposed to the cigarette smoke (CS) followed with the administration of chrysene. Morphological analyses indicated that chrysene caused earlier and severer pathological changes in CS-exposed mice. Besides, CS-exposed mice with chrysene treatment showed obvious collagen deposition, elevated α-smooth muscle actin (α-SMA) expression and reduced E-cadherin abundance at earlier stage, which suggested the acceleration and aggravation of pulmonary fibrosis. Moreover, quantification of leukocytes and pro-inflammatory cytokines in bronchoalveolar lavage fluid (BALF) and lung tissues implied that chrysene significantly exacerbated the proceeding of inflammation in CS-exposed mice. Furthermore, significantly increased apoptotic rates, augmented expressions of apoptotic related proteins and highly expressed TRPV1 were determined in CS-exposed mice with chrysene treatment, which indicated the association between COPD pathogenesis and TRPV1 channel. In summary, our findings elucidate that chrysene accelerates the development of COPD in a murine model with new molecular mechanisms.
Introduction
Chronic obstructive pulmonary disease (COPD) is a progressive disease with high global morbidity and mortality. 1 It is predicted that COPD will become the third leading cause of worldwide deaths by 2020. 2,3 Despite that great efforts have been made, the effective therapeutic strategies of COPD remain indistinct, which accounts for the increasingly elevated amount of COPD patients. 4 Various pathogenic factors contributed to the development of COPD has been clarified including the cigarette smoke, biological dusts, mineral dusts and gases/fumes, while cigarette smoking is well recognized as the major cause of COPD progression. 5,6 The severity of COPD varies a lot between the smokers and nonsmokers. In brief, it has been documented that cigarette smokers take over the 80% of the total COPD cases in the world. 7 COPD is pathologically characterized by the airflow limitation, strong inflammatory responses, and protease imbalance. 8 More specifically, toxic chemicals contained in cigarette smoke can induce the abnormal inflammatory response in airway and promote the recruitment of neutrophils and inflammatory cells, 9 which consequently facilitates the proceeding of COPD. In addition, previous study also suggests that the abnormal apoptosis has a critical effect on the destruction of pulmonary tissue in COPD, 3 and cigarette smoke could catalyze the cell apoptosis in human pulmonary microvascular endothelial cells. 3 Besides, the alterations of apoptosis-related genes have also been determined in COPD, 10 further validating the implication of apoptosis to the pathogenesis of COPD.
Chrysene is a member of the polycyclic aromatic hydrocarbon (PAH) family which is a large family consisting of various toxic compounds that come from natural and anthropogenic sources. 11 –13 PAHs always exist in the byproducts of incomplete burning of fossil fuels, oil, wood and tobacco, leading to huge damages to human health and living environment. 14 Commonly, PAHs with low molecular weight get easy access to degrade in environment. Additionally, PAHs are structurally featured with two or more fused benzene rings, and it is indicated that the more benzene rings they obtain, the more difficult for them to degrade in enviroment. 12,15 In accordance with previous study, chrysene is a basic PAH structurally characterized with the higher molecular weight and four fused benzene rings, 12 which makes it hard for biodegradation. Furthermore, it should be noted that chrysene is also a type of PAH with multiple biological properties, and it has shown the potential to induce DNA damage and apoptosis via reactive oxygen species. 13 Additionally, chrysene is capable of triggering the perturbation of DNA repair machinery. 16 Moreover, hydroxylated chrysene metabolites are reported to show developmental toxicity in Zebrafish Embryos, 17 and the connection between chrysene and cancer has also been revealed by several previous studies. 18 Nevertheless, there is little evidence to illustrate the association between chrysene and COPD. Consequently, our study focuses on that whether chrysene aggravates the proceeding of cigarette smoke induced COPD and emphasizes the increasing severity of air pollution.
Methods
Reagents and antibodies
Chrysene (C107019, purity ≥ 98%) was purchased from Aladdin reagents (Shanghai, China). Bcl-2 antibody (AF6139), Bax antibody (AF0120), cleaved caspase-3 antibody (AF7022), collagen I antibody (AF0134) and collagen III antibody (AF0136) were obtained from Affinity (Shanghai, China). TRPV1 antibody (A8564) was purchased from ABclonal (Wuhan, China). β-actin antibody (sc-47778) was purchased from Santa Cruz (CA, USA). Goat anti rabbit IgG (A0208), goat anti mouse IgG (A0216) were obtained from Beyotime Biotechnology (Haimen, China).
Ethics statement
The Animal Ethics Committee of Shengjing Hospital of China Medical University approved all experimental protocols with the official approval number (No.2016PS179K). All performances were conducted in accordance with the guideline and regulations for the care and use of laboratory animals. All efforts have been made to minimize the suffering of experimental animals in our study.
Animals and treatment
Male C57BL/6 mice (20–22 g, 7–8 weeks old) were obtained from the Liaoning Changsheng biotechnology (Liaoning, China). All the animals were kept at the temperature of 25 ± 1°C and the humidity of 44–55% with 12/12 light/dark cycle. Animals were allowed to get the water and food freely during the whole experiment. Afterward, mice were randomly divided into four groups: control group, cigarette smoke (CS) exposure group, chrysene (Chry) group and CS exposure with chrysene group (CS + Chry). The control animals were exposed to the normal room air during the proceeding of modeling. For CS-induced COPD mice, they were exposed to cigarette smoke twice a day with 10 cigarettes each time. The CS exposure took 1 hour each time with interval of 5 minutes, and it was conducted 5 days a week. For chrysene group, mice were administrated with chrysene (10 μg/per mouse) by intratracheal instillation each day for continues 5 days in a week. For the CS + Chry group, mice received the intratracheal instillation of chrysene (10 μg/per mouse) after the second CS exposure each day with the 5-day conduction each week. BALF and lung tissues were collected at the time points of 1st week, 2nd week, 4th week, 8th week and 16th week.
Bronchoalveolar lavage fluid (BALF) collection
The repeated lavage was performed with 0.5 mL pre-cooled normal saline for three times and the fluid was collected for further analysis.
Hematoxylin and eosin (HE) staining
The lung tissues were fixed in 4% paraformaldehyde followed with further dehydrating and embedding in paraffin. Samples were cut into sections with 4-µm width and subsequently subjected to the rehydration and hematoxylin staining (H8070, Solarbio, Beijing, China). Thereafter, sections were differentiated by 1% acid alcohol for 3 seconds and rinsed with running water for 20 minutes. After being further soaked in distilled water for 2 minutes, sections were assigned to the staining of eosin (A600190, Sangon Biotech, Shanghai, China) for histological examination. Ultimately, the representative images of HE staining were captured using the microscope (Olympus BX53, Tokyo, Japan) after the rehydration, transparency and mounting. The morphological analyses on lung injury and diffuse alveolar damage (DAD) were conducted as previously published 19,20 and presented as scores in our study.
Masson’s trichrome staining
Sections were incubated with Masson’s trichrome stains to visualize the collagen deposition. 21 The relative quantity of Masson’s trichrome was demonstrated by the percentage of positive staining within the total tissue area.
Immunofluorescence
Immunofluorescence assay was conducted to detect the expressions of E-cadherin and α-SMA. Lung tissue sections were blocked by receiving the incubation of normal goat serum (SL038, Solarbio) for 15 min at room temperature after antigen retrieval. Subsequently, they were incubated with the primary antibodies against α-SMA (1:200, Ab5694, abcam) and E-cadherin (1:100, #14472, CST) at 4°C overnight. On the following day, slides were washed and incubated with the secondary antibodies Cy3-conjugated anti-rabbit IgG (A0516, 1:300, Beyotime), FITC-conjugated anti-mouse IgG (A5608, 1:300, Beyotime) for 90 min and DAPI (C1002, Beyotime) for 1 min at room temperature in the dark. Images were captured using the fluorescence microscope (Olympus BX53, Tokyo, Japan) after washing and sealing. Besides, the fluorescence intensity of fluorescent images was quantified by the image J software.
Enzyme-linked immunosorbent assay (ELISA)
The production of TNF-α, IL-6, IL-1β, IL-10, IL-17A and IL-17F were detected by ELISA kits according to the manufacture’s instruction. Briefly, mouse TNF-α (EK0527), IL-6 (EK0411), IL-1β (EK0394), IL-10 (EK0417) and IL-17A (EK0431) were measured using the ELISA kits from Boster Biological Technology. Mouse IL-17F (SEB955Mu) was assessed by the ELISA kit from USCN Life Science (Wuhan, China).
Real-time quantitative PCR
Total RNA from the lung tissues was extracted using the TRIpure reagent (RP1001, BioTeke). The quantification of total RNA was performed using the ultraviolet spectrophotometer (NANO 2000, Thermo, USA). cDNA was next synthesized using the reverse transcriptase (PR6502, BioTeke). Real-time PCR was performed according to the protocol using the SYBR Green (S9430, sigma) on ExicyclerTM96 machine (Bioneer Co, Daejeon, Korea) for fluorescence quantification. Primer sequences were listed in Table 1. The data was analyzed and calculated using 2−△△CT with β-actin as internal reference.
Real-time PCR primer information and product length

TUNEL assay
TUNEL assay was performed according to the instructions of an apoptosis detection kit (WLA030a, wanleibio) after the sections being prepared. The representative images were obtained using a microscope (Olympus BX53, Tokyo, Japan), and the apoptotic rate was calculated as the amount of apoptotic cells divided by the total.
Immunohistochemical staining
Immunohistochemical staining was performed to detect the expression of TRPV1. In general, sections were prepared as described above, and incubation of the normal goat serum (SL038, Solarbio) was further performed for 15 min at room temperature after the antigen retrieval and 3% H2O2 treatment. Subsequently, sections were covered with the primary rabbit anti-TRPV1 (1:200, bs-23926R, Bioss) antibody at 4°C overnight. The incubation of the HRP-conjugated IgG (#31460, 1:500, thermoFisher) was next performed at 37°C for 1 h followed by the DAB staining. The images were captured using a microscope (Olympus BX53, Tokyo, Japan) after washing and sealing.
Western-blot analysis
Total proteins from the lung tissues were quantified according to the BCA Protein Quantification Kit (P0011, Beyotime). Total protein was separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to a polyvinylidene fluoride membrane (PVDF). The membrane was next blocked with 5% (M/V) dissolved skimmed milk powder (Q/NYLB 0039S, Yili, China) for 1 hour. The incubation of primary antibody against Bcl-2 (1:1000, AF6139, Affinity), Bax (1:1000, AF0120, Affinity), cleaved caspase-3 (1:1000, AF7022, Affinity), collagen I (1:1000, AF0134, Affinity), collagen III (1:1000, AF0136, Affinity) and TRPV1 (1:1000, A8564, ABclonal) was then performed at 4°C overnight with appropriate dilution ratio, reference was β-actin (1:1000, sc-47778, Santa cruz). After rinsing with TBST, the membrane was incubated in the IgG-HRP secondary antibody (1: 5000, A0208, A0216, beyotime, China) for 45 minutes at 37°C. ECL solution (P0018, beyotime, China) was used for chemiluminescence detection. The Gel-Pro-Analyzer (Media Cybernetics, Bethesda, MD, USA) was used to analyze the optical density of the bands.
Statistical analysis
All data were expressed as mean ± SD. Statistical analyses were performed by using GraphPad Prism 8.0. Comparisons between groups were made by two-way ANOVA followed by Tukey’s tests. P < 0.05 was considered statistically significant.
Results
Chrysene aggravated the pathological changes in lungs of cigarette smoke induced COPD mice
As shown in Figure 1A, results of H&E staining displayed typical pathological changes of COPD in lungs of CS exposed mice since the 8th week. Importantly, these obvious pathological changes had been observed since the 2nd week in CS + Chry group, which indicated the acceleration of COPD progression with chrysene treatment. Besides, the lung injury (LI) score was indicated in Figure 1B based on the severity of alveolar congestion, hemorrhage, infiltration and aggregation of neutrophils together with alveolar wall thickening. 20 The analysis of diffuse alveolar damage (DAD) score was presented in Table 2 according to the degree of edema, inflammation and hemorrhage. Accordingly, remarkably increased score of lung injury and DAD had also been found in mice after the co-treatment of CS and chrysene when compared to CS exposure since the 4th (DAD) or 8th (LI) week within one–week group comparison.

Chrysene aggravates the pathological changes in lungs of cigarette smoke induced COPD mice. (A) The representative pathological images of lung tissues were assessed by H&E staining along with the analysis of lung injury score (B) and alveolar damage score (Table 2), scale bar: 200 μm. The pathological changes were marked as follows: inflammatory cell infiltration together with small bronchi wall thickness (black arrow), increased neutrophil aggregates (red arrow) and augmented secretion into the bronchial lumen (green arrow). All data were presented as mean ± SD of six separate experiments. Versus control group: #P < 0.05, ##P < 0.01, ###P < 0.001, ####P < 0.0001; Versus CS group, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
The effect of chrysene on diffuse alveolar damage (DAD) score.

Note: All values are expressed mean ± SD (n = 6). Compared with control group: #P < 0.05, ##P < 0.01, ###P < 0.001, ####P < 0.0001; Compared with CS group, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Chrysene worsened the fibrosis in lungs of cigarette smoke induced COPD mice
The result of Masson’s trichrome staining in Figure 2A–B showed that the mice developed an obvious pulmonary fibrosis after 2-week CS exposure with significantly increased airway thickening and blue staining. However, these kinds of alterations had been observed in CS + Chry group since the 1st week. Besides, COPD mice with chrysene treatment displayed an earlier and severer collagen deposition with significantly increased expressions of collagen I, collagen III, α-SMA and decreased E-cadherin according to the western-blot and immunofluorescence analyses (Figure 2D–F). Moreover, chrysene also prominently exacerbated these alterations in CS-exposed mice from the 2nd week within group comparison, which further validated that chrysene could worsen the fibrosis in lungs of cigarette smoke induced COPD mice.

Chrysene worsens the fibrosis in lungs of cigarette smoke induced COPD mice. The lung fibrosis was detected by Masson’s trichrome staining (A) with the quantification of positive areas (B) scale bar: 100 µm or 200 μm. The protein levels of collagen I and III were measured by western-blot analysis (C) with the quantitative analysis. The expressions of E-cadherin and α-SMA were determined by immunofluorescence assay (D) with the quantification of fluorescence intensity (E–F), scale bar: 50 μm. All data were presented as mean ± SD of six separate experiments. Versus control group: #P < 0.05, ##P < 0.01, ###P < 0.001, ####P < 0.0001; Versus CS group, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Chrysene exacerbated the inflammation in BALF of cigarette smoke induced COPD mice
The result in Figure 3A indicated that the amount of total leukocytes in BALF was significantly increased in CS group after 2-week CS exposure, while the combination of CS and chrysene expedited the procedure evidenced by the earlier observation of significant increased amount of leukocytes at the 1st week. Within group comparison, remarkable augment in inflammatory cells was found in CS exposed mice after the treatment of chrysene from the 2nd week. Moreover, the productions of pro-inflammatory cytokines in BALF including the TNF-α, IL-6, IL-1β, IL-10, IL-17A and IL-17F were detected by ELISA. Results in Figure 3B–G indicated that significantly increased contents of TNF-α, IL-6, IL-1β, IL-17A and IL-17F along with decreased IL-10 had been found in CS exposed mice since 8th week after CS exposure, while these changes in pro-inflammatory cytokines were found much earlier in CS + Chry group. Within group comparison, significantly elevated expressions of TNF-α, IL-6, IL-1β, IL-17A and IL-17F accompanied with reduced IL-10 were also observed in CS + Chry group comparing to the CS group from the 8th week.

Chrysene exacerbates the inflammation in BALF of cigarette smoke induced COPD mice. (A) The amounts of leukocytes in BALF were analyzed with the assistance of microscope. The productions of TNF-α (B), IL-6 (C), IL-1β (D), IL-10 (E), IL-17A (F) and IL-17F (G) in BALF were detected by ELISA. All data were presented as mean ± SD of six separate experiments. Versus control group: #P < 0.05, ##P < 0.01, ###P < 0.001, ####P < 0.0001; Versus CS group, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Chrysene enhanced the inflammation in lung tissue of cigarette smoke induced COPD mice
The expressions of pro-inflammatory cytokines in lung tissues were assessed by ELISA and real-time quantitative PCR. Results in Figure 4A–L had shown a much earlier discovery of significantly elevated expressions of TNF-α, IL-6, IL-1β, IL-17A, IL-17F and reduced IL-10 in CS + Chry group since the 2nd week, while remarkable changes in these pro-inflammatory cytokines in CS exposed group were just found after 8-week CS exposure. Furthermore, chrysene significantly up-regulated the expressions of TNF-α, IL-6, IL-1β, IL-17A and IL-17F while down-regulated the content of IL-10 in CS exposed mice from the 8th week within group comparison.

Chrysene enhances the inflammation in lung tissues of cigarette smoke induced COPD mice. The productions of TNF-α (A), IL-6 (B), IL-1β (C), IL-10 (D), IL-17A (E) and IL-17F (F) in lung tissue were detected by ELISA. The relative mRNA levels of TNF-α (G), IL-6 (H), IL-1β (I), IL-10 (J), IL-17A (K) and IL-17F (L) in lung tissue were assessed by real-time quantitative PCR. All data were presented as mean ± SD of six separate experiments. Versus control group: #P < 0.05, ##P < 0.01, ###P < 0.001, ####P < 0.0001; Versus CS group, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Chrysene deteriorated the apoptosis in lungs of cigarette smoke induced COPD mice
TUNEL assay was performed for the detection of apoptosis in lung tissues, and results in Figure 5A–B indicated that the percent of apoptotic cells was significantly raised in CS exposed mice when compared to control each week. Besides, it was also found that the treatment of chrysene dramatically increased the apoptotic rate in CS exposed mice each week. The expression levels of Bcl-2, Bax and cleaved caspase-3 were respectively determined by western-blot. As shown in Figure 5C, significant augment of Bax and cleaved caspase-3 along with the reduction of Bcl-2 were observed in CS exposed mice after the 4/8-week exposure of CS, while distinct changes in these biomarker proteins were found much earlier in the CS + Chry group than in CS exposed group. Within group comparison, combination of CS and chrysene prominently promoted the cell apoptosis from the 4th week or 8th week when compared to the CS treatment.
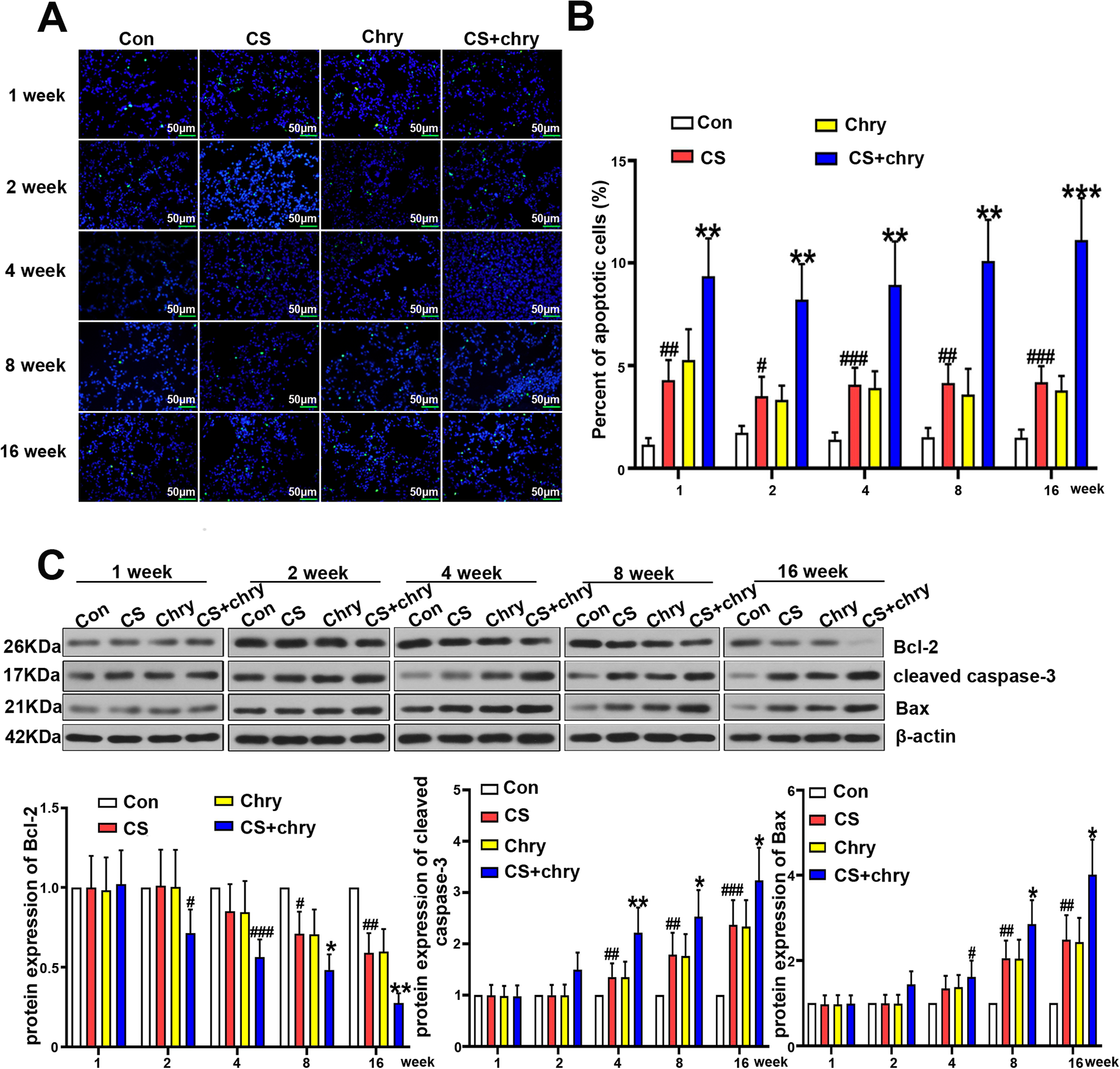
Chrysene deteriorates the apoptosis in lungs of cigarette smoke induced COPD mice. Detection of apoptosis in lung tissue was performed with the TUNEL assay (A) along with the quantification of positive cells (B), scale bar: 50 μm. (C) The protein levels of Bcl-2, Bax and cleaved caspase-3 were detected by western-blot. All data were presented as mean ± SD of six separate experiments. Versus control group: #P < 0.05, ##P < 0.01, ###P < 0.001, ####P < 0.0001; Versus CS group, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Chrysene elevated TRPV1 expression in lungs of cigarette smoke induced COPD mice
TRPV1 has been reported to be partly associated with inflammatory response and apoptosis. 22 Consequently, the expression level of TRPV1 was respectively determined by immunohistochemical staining and real-time quantitative PCR. As indicated in Figure 6A–C, TRPV1 had been highly expressed in the lung tissue of CS exposed mice since the 8th week, and chrysene expedited the proceeding with much earlier observation of significant increased TRPV1 at the 2nd week. Additionally, the expression of TRPV1 was significantly elevated after the treatment of chrysene from the 8th week in CS exposed group within group comparison. Additionally, the protein level of TRPV1 in different time points was also respectively detected in our study. Evidently elevated expression of TRPV1 was found in CS exposed mice after the 4-week CS exposure, while the highly expressed TRPV1 had been discovered in CS + Chry group since the 2nd week. Furthermore, the protein level of TRPV1 was remarkably increased in CS group after the administration of chrysene at 16th week within group comparison.

Chrysene up-regulates the TRPV1 expression in lungs of cigarette smoke induced COPD mice. The expression of TRPV1 in lung tissue was determined by IHC (A) with the analysis of mean density (B). Scale bar: 50 μm. (C) The relative mRNA level of TRPV1 was assessed by real-time quantitative PCR. (D) The protein level of TRPV1 was determined by western-blot. All data were presented as mean ± SD of six separate experiments. Versus control group: #P < 0.05, ##P < 0.01, ###P < 0.001, ####P < 0.0001; Versus CS group, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Discussion
Chronic obstructive pulmonary disease (COPD) is a common respiratory disorder with altered functions and structures. 23 In particular, cigarette smoke is well accepted as the major cause of COPD, and high prevalence of COPD has been found in the general smoking population. 24 To investigate the role of chrysene in the development of COPD, male C57BL/6 mice were exposed to the cigarette smoke followed with the administration of chrysene by tracheal instillation in our study. Subsequently, typical pathological changes including the damaged alveolar walls and pulmonary bullae were observed after the exposure to both CS and Chry at an earlier stage of experiment, and all these abnormalities in structure have been documented to impair the gas exchange and eventually lead to the dysfunction of lung. 25 Moreover, the collagen depositions, elevated expression of α-SMA and reduced E-cadherin were found in CS + Chry exposed mice at the first 2 week according to our findings. Briefly, it has been reported that myofibroblast accumulation, which is characterized with increased α-SMA and collagen deposition, is a key feature for pathological airway remodeling of COPD. 26 Additionally, E-cadherin is implicated with the regulation of epithelial-mesenchymal transition (EMT), the source of myofibroblasts. 27 The results indicated above validate that chrysene accelerated the generation of pathological alterations and pulmonary fibrosis in mice exposed to CS.
Though exact pathogenesis of COPD still remains indistinct, the inflammatory reactions are believed to make huge contributions to the proceeding of COPD. 28 Especially, cigarette smoking has been reported to be able to induce the chronic inflammatory process with ultimate altered pulmonary functions and reduced immune defense capacities. 29,30 Moreover, our findings demonstrated that CS exposure also induced the augment of total leukocytes and pro-inflammatory cytokines like IL-17A along with the reduced anti-inflammatory factor IL-10 in BALF and lung tissues, further validating the connection between COPD and inflammatory response. Accordingly, bronchoalveolar lavage is a well-accepted technique for collecting the fluid of respiratory tract, while it always acts as an important method to assess the airway inflammation in the development of COPD. 31,32 Importantly, the alterations of pulmonary components in BALF, such as the inflammatory cytokines, have been reported to strongly correlate with diagnose and therapeutic strategies of lung inflammation. 31,33 For example, cytokine IL-17A is an elementary member of IL-17 family which consists of six molecules ranging from IL-17A to IL-17F. 34 According to the clinical data, IL-17A serves as a vital effective cytokine in driving inflammatory changes within affected tissues, 35 and the excessive and uncontrolled secretion of these pro-inflammatory cytokines will eventually result in the organ failure. However, IL-10 could exert anti-inflammatory effects on inflammatory disorder via restraining the synthesis of pro-inflammatory cytokines like IL-2, IL-3 and TNF-α to maintain homeostasis. 36
In accordance with the previous reports, apoptosis has been recognized as a key mechanism of the COPD pathogenesis. 37 Especially, tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), a cytokine with both inflammatory and apoptotic features has been documented to promote the development of experimental COPD. Additionally, the reactive oxygen species (ROS), which plays an important role in cell apoptosis, has been the major cause of many pathological conditions related with cigarette smoking. 38,39 Furthermore, it is documented that the destruction of alveolar epithelial cells and endothelial cells through apoptosis could result in the impairment of lung tissue and emphysema. 40 Consequently, cell apoptosis was determined in our study with the observation of significantly increased apoptotic rates and elevated expressions of pro-apoptotic proteins, including Bax and cleaved caspase-3 in CS exposed mice after chrysene treatment. Moreover, the anti-apoptotic protein Bcl-2 was detected with significantly reduced expression. 41 The Bcl-2 protein always functions through the association with pro-apoptotic proteins like Bax and Bak, and the apoptosis event gets initiated when the balance between major apoptosis-related mediators is broken. 41 –43
Considering the observation described above, we could expound that chrysene accelerates the development of COPD by promoting the inflammation and apoptosis in CS exposed mice. However, the target of chrysene in COPD progression has not been found yet. Interestingly, our further study indicated an over-expression of TRPV1 in the earlier stage of COPD development after the co-treatment of CS and chrysene. Meanwhile, higher expressed TRPV1 was also found in CS exposed mice after the treatment of chrysene within group comparison, which suggests that TRPV1 might be the effective molecule in chrysene-mediated COPD progression. As previously reported, TRPV1 is one of the vanilloid transient receptor potential (TRPV) channel family with major expression in sensory neurons. 44,45 It is widely acknowledged that TRPV1 always functions as a pain receptor, while recent study demonstrates the implication of TRPV1 in inflammatory reactions. In details, TRPV1 is documented to up-regulate the expression of pro-inflammatory cytokines in febrile seizure mice, 46 stimulate the JAK2-STAT3 pathway and promote the release of IL-1β in astrocyte. 47 Meanwhile, the TRPV1 channel could also be activated by the pro-inflammatory cytokines. 22 In addition, TRPV1 channel is expounded to facilitate the aggravation of cell apoptosis, which can be induced by air pollutants, one source of chrysene production. 45 Moreover, it is well established that Ca2+ serves as a major messenger in multiple fundamental physiological processes including cell apoptosis, 48 and TRPV1 channel has been documented to strongly associate with calcium signaling, which implies that chrysene may mediate the apoptosis through the regulation of intracellular Ca2+ influx. 22
Conclusions
In summary, our study demonstrates that chrysene accelerates the development of COPD by promoting the inflammation and apoptosis. In addition, highly expressed TRPV1 was found in chrysene-mediated COPD development, suggesting that chrysene might facilitate the inflammation and apoptosis in COPD mice by regulating TRPV1.
Footnotes
Data availability statement
All the data and materials were included in this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
