Abstract
Allergic rhinitis (AR) is a common allergic inflammatory and chronic reactive disease caused by allergen-induced immunoglobulin E (IgE). Tanshinone IIA (Tan IIA) is one of the active ingredients in Salvia miltiorrhiza Bunge (Danshen) and plays a vital role in inhibiting inflammation. Thus, we hypothesized that Tan IIA has anti-allergic effects and studied the function of Tan IIA in mast cells and an AR animal model. We induced RBL-2H3 cell sensitization with monoclonal anti-2,4,6-dinitrophenyl-immunoglobulin (Ig) E/human serum albumin (DNP-IgE/HSA) and constructed an ovalbumin (OVA)-induced AR model in mice. The role of Tan IIA in AR progression was studied using the MTT assay, ELISA, western blot, toluidine blue staining, HE staining, and Alcian blue and safranin O (A&S) staining. Tan IIA treatment significantly increased IgE/HSA-induced cell viability. However, Tan IIA treatment markedly downregulated the expression levels of β-hexosaminidase, histamine, tumor necrosis factor (TNF-α), interleukin 1β (IL-1β), IL-4, and IL-5 in IgE/HSA-induced cells. Furthermore, Tan IIA improved typical symptoms in the OVA-induced AR model mice by inhibiting the phospholipase Cγ1 (PLCγ1)/protein kinase C (PKC)/IP3R pathway. Additionally, Tan IIA effectively improved the degranulation of RBL-2H3 cells and OVA-induced AR in mice. Together, these results suggest that Tan IIA may be a potential drug for the treatment of AR in the future.
Introduction
Allergic rhinitis (AR) is a chronic inflammatory disease with a high incidence and is characterized by abnormal body reactions in the nose due to increased sensitivity to allergens.1-3 The main symptoms of AR in patients are nasal hypersecretion, nasal itching, sneezing, and swelling of the nasal mucosa.4-6 At present, the recognized pathogenesis of AR is that the allergen-induced immunoglobulin (Ig) E antibody binds to the high-affinity IgE receptor (FcεRI) on the surface of mast cells to activate these cells, inducing intracellular signal cascades, such as the activation of PLCγ1 and PKC, and elevating the expression level of Ca2+, which further causes mast cells to degranulate.2, 7 It can simultaneously secrete a variety of inflammatory cytokines that play important roles in AR, including TNF-α, IL-4, IL-1β, and other factors such as histamine, and β-hexosaminidase.8-11 AR has been reported to affect 10%–20% of the population and interferes with the quality of work, study, and sleep of patients. 12 Therefore, it is necessary to study the pathogenic mechanism of AR and identify potential candidates for treating the disease.
Salvia miltiorrhiza Bunge (Danshen) is a famous traditional Chinese herb that is usually used in China and other Asian countries for the treatment of cardiovascular diseases, asthma, and other diseases.13, 14 Tanshinone IIA (Tan IIA) is an active fat-soluble component of the herb and has a wide range of biological activities, including improving microcirculation, scavenging oxygen free radicals, inhibiting platelet aggregation, and anti-thrombosis activity.15-17 Moreover, studies have shown that Tan IIA plays an indispensable role in anti-inflammatory and antioxidant activity.18, 19 Thus, we hypothesized that Tan IIA may also be an active ingredient with potential anti-allergic rhinitis effect. Therefore, we constructed a model of AR in vivo and in vitro to study the function of Tan IIA in AR and its mechanism of action.
Materials and Methods
Cell culture and IgE-mediated mast-cell activation
The RBL-2H3 cell line was purchased from the Type Cell Culture Collection of the Chinese Academy of Sciences (Shanghai, China). Cells were grown in culture dishes filled with minimum essential medium (MEM, Hyclone Laboratories) plus 10% FBS (Gibco), which consisted of 100 μg/mL streptomycin/penicillin. They were placed in an incubator with an environment of 1.5 mg/mL sodium bicarbonate, 110 μg/mL sodium pyruvate, and 5% CO2 at 37°C. After 48-well plate inoculation for 12 h, cells were treated with 100 ng/mL DNP-IgE (Sigma-Aldrich) for 12 h, resulting in cell sensitization. The cells were pretreated with Tan IIA at a concentration of 5 μM (Tan IIA-L) or 10 μM (Tan IIA-H) (purity: 99% by HPLC, Sigma-Aldrich) for 30 min. 20 Tan IIA was dissolved in methanol to obtain a stock concentration of 2.94 mg/mL tanshinone IIA. The cells treated above were stimulated with 250 ng/mL DNP-HSA (Biosearch, Petaluma) for 12 h. There were five groups including the control group, Tan IIA-H group, IgE/HSA group, IgE/HSA + Tan IIA-L group, and IgE/HSA + Tan IIA-H group.
Cell viability
The five groups were treated as described above, and cell viability was detected by the MTT assay. Cell suspensions were evenly cultured in a 96-well plate at a concentration of 5000 cells in 100 μL per well at 37°C for 24 h. The cells were then treated and cultured for 48 h. MTT solution was added to the cells for 4 h. After full response to the cells, cell viability was detected using a microplate reader (Thermo Fisher Scientific) at an absorbance intensity of 450 nm, and the results were analyzed.
Assessment of histamine and β-hexosaminidase
Cells were seeded in 24-well plates and treated as described above. Supernatants were obtained and centrifuged for 10 min at 4°C; thereafter, 50 μL supernatant was collected into a new 1.5-mL tube and thoroughly mixed with 50 μL of substrate (1 m
Inflammatory factor determination
Cells were seeded in six-well plates, cultured in a 37°C incubator overnight until they reached a density of 80%, and then treated as described above. The supernatants were collected to detect the expression levels of IL-4, IL-5, IL-1β, and TNF-α using ELISA kits (Elabscience Biotechnology Co., Ltd).
Toluidine blue staining
Cells that were treated as described above were rinsed with PBS for 2 min, fixed with 4% paraformaldehyde at room temperature for 30 min, washed with PBS, impregnated with 1% toluidine blue for 2 h, re-dyed with hematoxylin, and washed with ddH2O to remove the excess dye. Cells were then gradually dehydrated in different concentrations of alcohol (70%–95%) and transparent with xylene. 22 After sealing with neutral resin, the images were observed and analyzed using a Leica inverted microscope (Leica Microsystems, Germany).
Western blotting
Protein samples were obtained from the cells and nasal mucosal tissues. The protein lysates were fully lysed for 30 min in ice and centrifuged at 12,000 × g for 5 min at 4°C. After discarding the precipitate, the supernatant was collected, diluted with 5 × loading buffer at a ratio of 1:4, and heated for 5 min at 100°C for full protein denaturation. For western blotting, the samples were electrophoretically separated by SDS-PAGE and electrotransferred to polyvinylidene difluoride (PVDF) membranes. After blocking in the western blocking fluid for 2 h on a shaker, the residual liquid were washed, then the membranes were incubated overnight at 4°C with primary antibody, which was pre-diluted with western primary antibody dilution buffer at a dilution of 1:1000. After washing the membranes three times in 1× TBST, the corresponding secondary antibody conjugated with goat anti-rabbit or goat anti-mouse HRP was incubated for 2 h. After washing the membranes three times, the proteins were detected using the BeyoECL Plus kit. The primary antibodies were procured from Cell Signaling Technology, including anti-PLCγ1, anti-phosphorylated-PLCγ1 (p-PLCγ1), anti-IP3R, anti-phosphorylated-IP3R (p-IP3R), anti-phosphorylated PKC (p-PKC), anti-PKC, and β-actin.
Animals and OVA-induced AR in mice
Female BALB/c mice (25 ± 2 g) were procured from the Shanghai Slake Laboratory Animal Co., Ltd and were subjected to a stable environment with a relative humidity of 50%–60% and a temperature of 23 ± 2°C. In addition, mice were housed under normal laboratory conditions with a 12 h light/dark cycle. In total, 24 mice were used, and six mice were placed in each group. All mice in this study were handled in strict compliance with the National Institute of Health Guide for the Care and Use of Laboratory Animals, and the experimental protocol was approved by the Institutional Animal Care and Use Committee of The Second Affiliated Hospital of Zhejiang University of Chinese Medicine. Mice were injected intraperitoneally with 2 mg of aluminum hydroxide containing OVA (50 μg) every 2 days for 14 days. Then, sensitized mice were obtained by injecting 10 μL of 10% OVA into the bilateral nasal cavities for 10 consecutive days. 23 The Tan IIA-treatment group received an intraperitoneal injection of 5 or 10 mg/kg Tan IIA from days 15–24 prior to intranasal OVA treatment. 20 The control group received only saline injections. The numbers of nasal rubs and sneezes were measured 10 min after intranasal OVA stimulation. At 24 h after the last intranasal stimulation, we obtained serum samples from the mice, and the expression levels of OVA-specific IgE, histamine, IL-4, IL-5, IL-1β, and TNF-α were evaluated by ELISA.
Eosinophil (EOS) and mast cell staining
Nasal mucosa tissues were fixed with 10% formalin and processed into paraffin blocks. These blocks were then cut into tissue sections of 4-μm thickness and stained with hematoxylin and eosin (H&E) and Alcian blue and safranin O (A&S) for the detection of EOS and mast cells. The numbers of EOS and mast cells on the same side were counted. At least five random fields per slide were analyzed in a blinded manner. 23
Statistical analyses
Data were analyzed using GraphPad Prism 7 and expressed as mean ± standard error (SEM). Statistical significance was determined by one-way analysis of variance (ANOVA), followed by the Bonferroni post-hoc test. Statistical significance was set at p < 0.05.
Results
Effect of Tan IIA on mast cell degranulation, which mediated AR
To evaluate the effect of Tan IIA on AR in vitro, we constructed a cell model to induce mast cell degranulation with IgE/HSA and treated cells with low and high concentrations of Tan IIA. The MTT assay was performed to detect cell viability in the five groups. Tan IIA had no toxic effect on RBL-2H3 cells, and cell viability was decreased in the IgE/HSA group compared with that in the control group. However, pretreatment with low or high concentrations of Tan IIA in the IgE/HSA group effectively improved cell viability, compared with that in the IgE/HSA group alone (Figure 1(a)). Moreover, the release of β-hexosaminidase and histamine was measured. The results indicated that the expression levels of β-hexosaminidase and histamine both increased in IgE/HSA-treated cells, compared with that in the control group, in contrast to the IgE/HSA-Tan IIA group (Figures 1(b) and (c)). Activation of mast cells is known to lead to the further secretion of proinflammatory factors. ELISA was used to evaluate the expression of IL-4, IL-5, IL-1β, and TNF-α. Tan IIA markedly inhibited the expression of these proinflammatory cytokines compared to that in the control group (Figures 1(d) to (g)). For a more intuitive observation of Tan IIA-treated model cell degranulation, RBL-2H3 cells were treated as described above and stained with toluidine blue. Normal RBL-2H3 cells were elongated, with purple particles inside the cells. After induction by IgE/HSA, cells appeared in a state of degranulation with irregular cell morphology, and the purple particles were released outside the cell. Tan IIA treatment of the IgE/HSA-sensitized cells ameliorated the morphological changes and decreased the number of degranulated cells (Figure 1(h)). Effect of Tan IIA on mast cell degranulation, which mediated allergic rhinitis. (a) The viability of RBL-2H3 cells was detected using the MTT assay in five groups, namely, the control, Tan IIA-H, IgE/HSA, IgE/HSA+Tan IIA-L, and IgE/HSA+Tan IIA-H groups. (b) β-hexosaminidase activity was measured with the optical density at 405 nm in RBL-2H3 cells. The levels of (c) histamine, (d) IL-4, (e) IL-5, (f) TNF-α, and (g) IL-1β were detected by ELISA. (h) Degranulation of RBL-2H3 cells was evaluated by H&E staining. All data are expressed as the mean ± SE *p < 0.05, **p < 0.01, compared with the control group; &p < 0.05, &&p < 0.01 compared with the IgE/HSA group.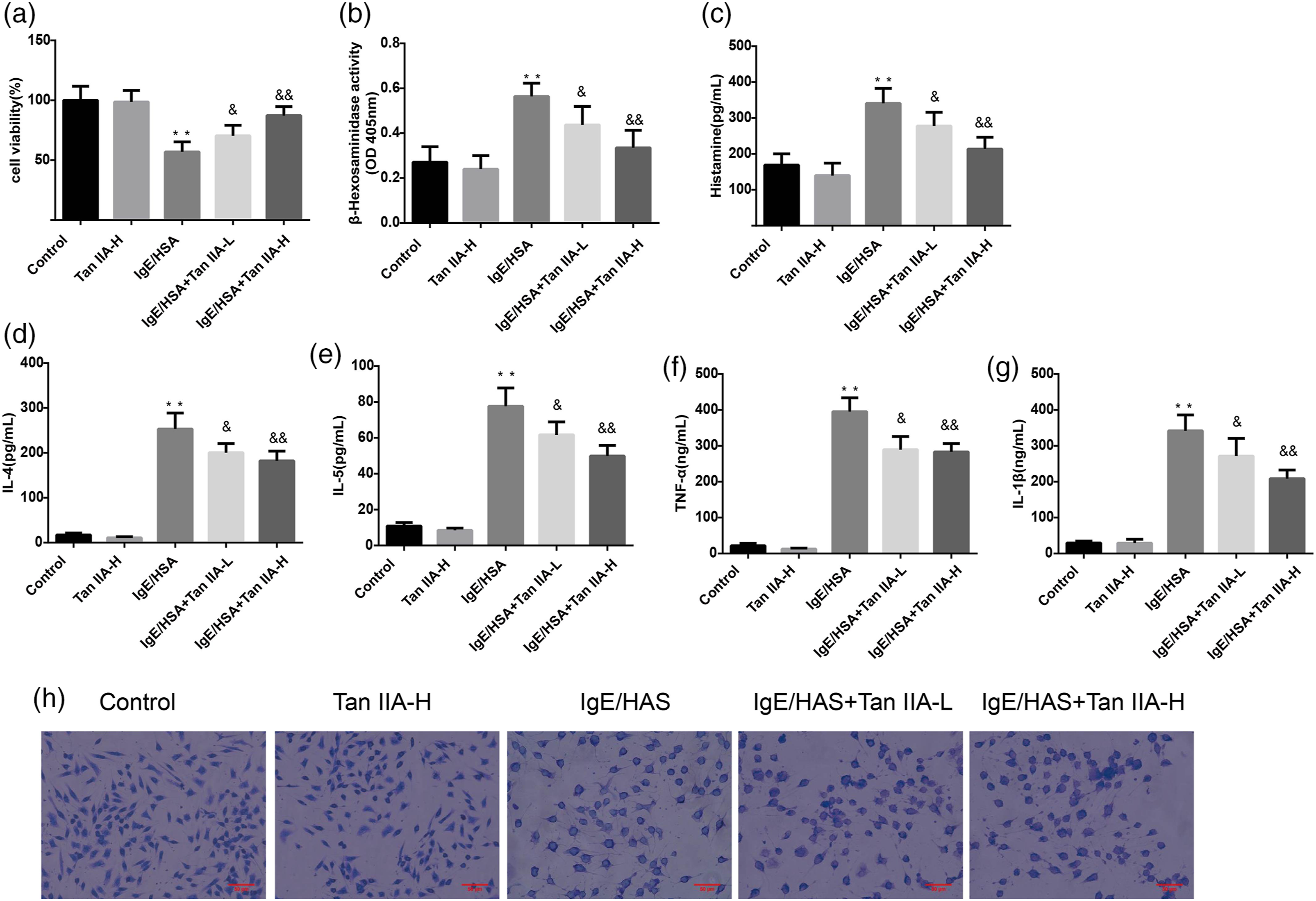
Tan IIA inhibited the activation of the PLCγ1/PKC/IP3R pathway in RBL-2H3 cells
Some studies have reported that the pathological mechanism of AR is closely related to the PLCγ1/PKC/IP3R pathway.24, 25 The allergen first activates mast cells, leading to the phosphorylation of PLCγ1, PKC, and IP3R and the release of Ca2+, which further induces cell degranulation and secretion of proinflammatory cytokines. Thus, the protein expression levels of phosphorylated PLCγ1, PKC, and IP3R were examined by western blotting. Tan IIA could effectively inhibit the IgE/HAS-induced activation of PLC γ1/PKC/IP3R and reduced the release of Ca2+ in RBL-2H3 cells, compared with that in the control group (Figures 2(a) to (g)). Tan IIA inhibited the activation of the PLCγ1/PKC/IP3R pathway in RBL-2H3 cells. (a) Phosphorylation levels of PLCγ1, PKC, and IP3R were measured by western blotting. The statistics for p-PLCγ1/β-actin (b), PLCγ1/β-actin (c), p-PKC/β-actin (d), PKC/β-actin (e), p-IP3R/β-actin (f), and IP3R/β-actin (g) are as shown above. *p < 0.05, **p < 0.01, compared with the control group; &p < 0.05, &&p < 0.01 compared with the IgE/HSA group.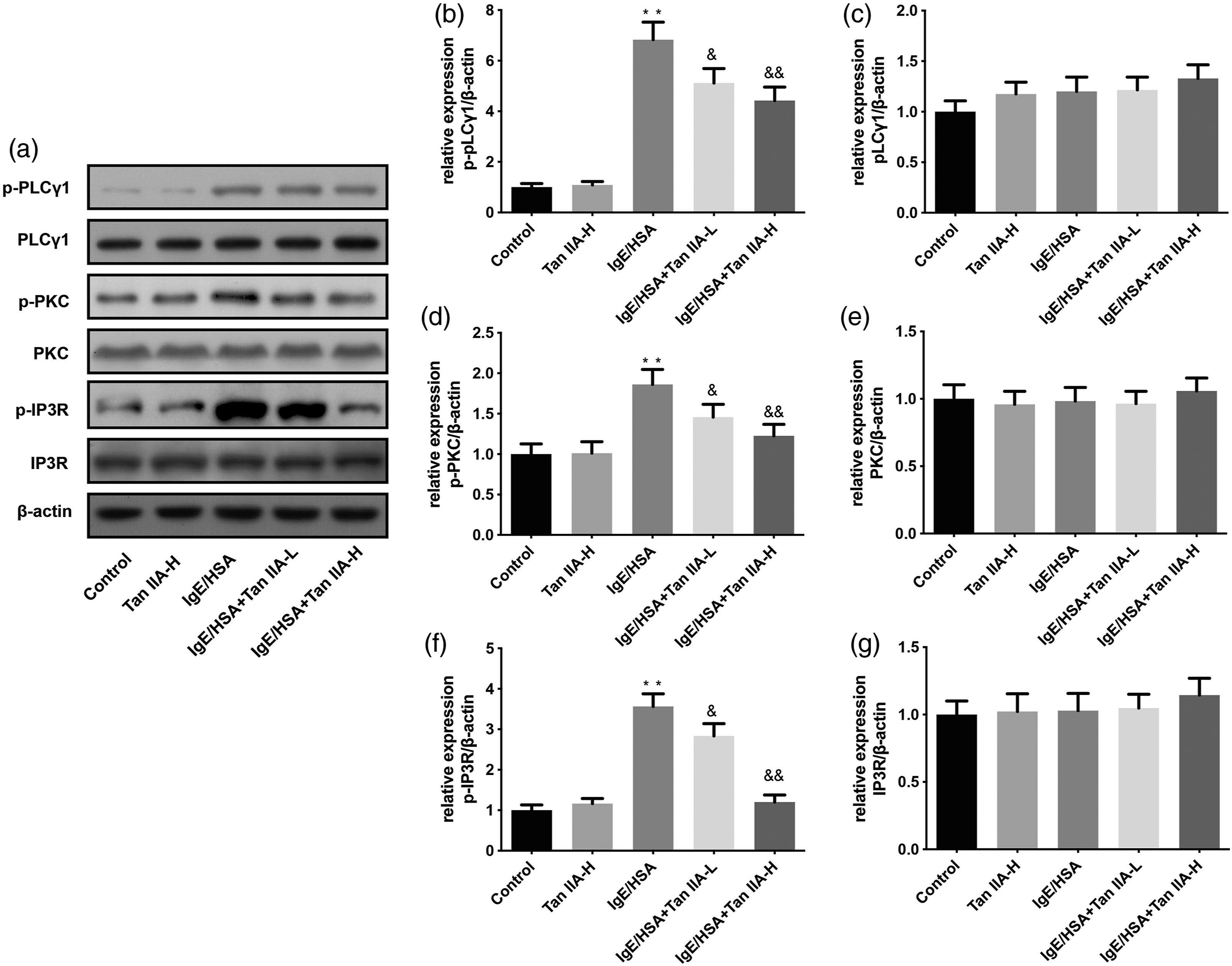
Effect of Tan IIA in OVA-induced AR model mice
To further study the influence of Tan IIA on AR in vivo, we constructed an OVA-induced AR mouse model and recorded the number of nasal rubs every 10 min for 9 days. Compared with normal mice, mice treated with OVA demonstrated a significantly higher degree of friction. However, Tan IIA treatment reduced this effect and reduced the number of nasal rubs in the AR model mice (Figure 3(c)). In addition, histopathological observation of mouse nasal mucosa tissues was performed using the H&E and A&S assays. Our results showed that compared with control mice, mice in the AR group had tissues that showed edema, dilation of small blood vessels, hyperplasia of glands, and a large amount of EOS infiltration. In the AR+Tan IIA-L group, Tan IIA-L partially reduced the nasal damage caused by AR and reduced congestion, edema, and EOS infiltration. In the AR+Tan IIA-H group, these phenomena were almost undetectable (Figures 3(a) and (b)). EOS and mast cell numbers were calculated according to the staining results. EOS and mast cell numbers in the nasal cavity tissues of mice treated with OVA were increased compared to those in normal mice, while Tan IIA treatment markedly reduced the number of these cells (Figures 3(d) and (e)). Consistently, the nasal symptoms score, including itching and rubbing score, among these groups was also displayed in the Figures 3(f) and (g). Effect of Tan IIA on OVA-induced AR model mice. (a) Hematoxylin and eosin staining was used to detect the expression levels of eosinophils. (b) A&S staining was used to detect mast cells. (c) The number of nasal rubs was counted for 10 min after OVA intranasal stimulation in the control, AR, AR+Tan IIA-L, and AR+Tan IIA-L groups. (d) Mast cells and (e) eosinophils were counted in two individuals. (f) Total times of sneezing and (g) rubbing were counted in two individuals. Scale bar = 50 μm. *p < 0.05, **p < 0.01, compared with the control group; &p < 0.05, &&p < 0.01 compared with the AR group.
Tan IIA decreased the expression of inflammatory cytokines in AR model mice
Inflammatory cytokines play indispensable roles in AR pathogenesis and aggravate AR symptoms.
26
To study the effect of Tan IIA on inflammation in AR, we investigated the expression of inflammatory factors in differently treated AR model mice using ELISA. Consistent with the expression levels of proinflammatory factors in vitro, the expression of IgE, IL-4, IL-5, IL-1β, and TNF-α in the serum of OVA-treated mice was higher than that in control mice, and Tan IIA treatment obviously decreased the serum levels of these factors (Figures 4(a) to (e)), similar to the expression of histamine (Figure 4(f)). Tan IIA decreased the expression of inflammatory cytokines in AR model mice. Mice were treated with OVA; at 24 h after the last intranasal stimulation, serum samples of the mice were obtained. (a) IgE, (b) IL-4, (c) IL-5, (d) TNF-α, (e) IL-1β, and (f) histamine levels were evaluated by ELISA. *p < 0.05, **p < 0.01, compared with the control group; &p < 0.05, &&p < 0.01 compared with the AR group.
Tan IIA suppressed the activation of the PLCγ1/PKC/IP3R pathway in OVA-induced AR model mice
To detect the activation of the PLCγ1/PKC/IP3R pathway simultaneously in vivo, we obtained samples of nasal mucosa tissues from the four treatment groups. Western blot assays were performed to detect the phosphorylation levels of PLCγ1, PKC, and IP3R. In OVA-treated mice, the PLCγ1/PKC/IP3R pathway was significantly activated compared to that in the control mice. However, Tan IIA markedly suppressed the activation of PLCγ1, PKC, and IP3R in OVA+Tan IIA mice (Figures 5(a) to (g)). Tan IIA suppressed the activation of the PLCγ1/PKC/IP3R pathway in OVA-induced AR model mice. (a) The relative expression levels of PLCγ1, PKC, and IP3R were determined with western blotting. The statistics for p-PLCγ1/β-actin (b), PLCγ1/β-actin (c), p-PKC/β-actin (d), PKC/β-actin (e), p-IP3R/β-actin (f), and IP3R/β-actin (g) are as shown above. *p < 0.05, **p < 0.01, compared with the control group; &p < 0.05, &&p < 0.01 compared with the AR group.
Discussion
In this study, we investigated the effect of Tan IIA on AR development. Our results illustrated that Tan IIA reduced the degranulation of mast cells and improved OVA-induced AR by inhibiting the PLCγ1/PKC/IP3R pathway. Thus, the findings of this study may provide a theoretical basis for the treatment of AR using traditional Chinese medicine.
AR is a global health problem that can occur in all age groups. 27 AR refers to a non-infectious inflammatory disease of the nasal mucosa, in which when a specific individual is exposed to an allergen, the release of IgE-mediated medium (mainly histamine) is induced and a variety of immunocompetent cells and cytokines are stimulated.28-30 For the clinical treatment of AR, chemical mediator receptor antagonists, mast cell stabilizers, Th2 cytokine inhibitors, steroids, and other drugs are commonly used.31, 32 However, there have been relatively few studies on the treatment of AR using traditional Chinese medicine. Therefore, in our study, we explore the utility of Tan IIA, which has anti-inflammatory, blood circulation, and blood stasis effects, in the treatment of AR.
Tan IIA was extracted from a traditional Chinese medicine, Salvia miltiorrhiza Bunge (Danshen). Chromatographic studies have reported that the obtained content of Tan IIA was higher in the ethanol extract than in the water extract of Danshen.33, 34 Moreover, some studies have demonstrated that daily treatment with the ethanol or water extract of Danshen effectively decreased inflammatory cell infiltration, reduced the number of Th1/Th2 factors, and reduced hyperplasia of goblet cells in an OVA-induced AR mouse model. 20 Madi-Ryuk and its active ingredient tannic acid have also been reported to alleviate AR clinical symptoms by regulating inflammatory mediators in OVA-induced AR model mice. 23 Allergic mediators, pro-inflammatory factors, and chemicals released by mast cells are known to play important roles in the occurrence of AR 35 ; therefore, mast cells are a potential target for the treatment of AR. Recently, the inhibitory effects of Tan IIA on mast cell degranulation as well as its anti-allergic activity in an OVA-induced mouse asthma model were reported, 20 which is similar to our hypothesis about the effect of tanshinone IIA on cell degranulation and OVA-induced AR. However, the mechanism underlying the effects of Tan IIA in an OVA-induced mouse model needs to be further explored.
Therefore, we established models of AR induced by OVA in vivo and by DNP-IgE/HSA-induced RBL-2H3 sensitized cell in vitro, and we demonstrated that Tan IIA significantly reduced cell degranulation and decreased the expression of β-hexosaminidase, histamine, and proinflammatory cytokines. Tan IIA alleviated the symptoms of AR by inhibiting the biological behavior of mast cells. At present, mast cells are the key effector and immunoregulatory cells that act in the pathophysiological process of AR and in pathological changes in the nasal mucosa. 36 A previous study showed that hydroxysafflor yellow A suppressed antigen-induced Ca2+ mobilization and mast cell degranulation by regulating the activation of the PKC/PLCγ/IP3R pathway. 24 ES-62 effectively repressed the mast cell response and blocked the release of proinflammatory cytokines by targeting different PKC isoforms. 37 Our study indicated that Tan IIA acts on mast cells by inhibiting the phosphorylation of PLCγ1, PKC, and IP3R and by downregulating the expression of the downstream inflammatory factors IL-4, IL-5, IL-1β, and TNF-α.
Although these findings provide important insight, there were also limitations to this study. First, the effect of Tan IIA on Ca2+ mobilization in AR requires further research. Second, the effect of the other active ingredients of Danshen on AR should be investigated simultaneously. Third, the mechanism underlying the interaction of Tan IIA with mast cells remains unclear.
In summary, Tan IIA could relieve mast cell-mediated AR by regulating the PLCγ1/PKC/IP3R pathway. The findings of this study provide a theoretical basis for the use of traditional Chinese medicine in the treatment of AR.
Footnotes
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Natural Science Foundation Project of the Zhejiang Province (grant number: LY19H280009; “Study on the Mechanism of the Effective Components of Salvia Miltiorrhiza in Regulating Mast Cell PLCγ/PKC/Ca2+ Pathway Against Allergic Rhinitis”) and the Traditional Chinese Medicine Science and Technology Program of Zhejiang Province [grant number: C2019ZB079].
Ethical approval
All animal experiments were approved by the Animal Investigation Ethics Committee of The Seventh Affiliated Hospital of Sun Yat-Sen University (Shenzhen, China).
