Abstract
Background:
Acute kidney injury (AKI) is the main clinical concern resulted from ischemia-reperfusion injury (IRI). Ample clinical data indicates that AKI is associated with distant organ dysfunctions and poor patients’ outcomes. Oxidative stress and inflammation have a critical role in the pathogenesis of organ injuries following IRI. The objectives of this study were to determine the impact of Gamma Oryzanol (GO), extracted from rice bran oil, on distant organs in rats after IRI.
Methods:
Twelve out of 24 Wistar rats were treated by one dosage of GO (100mg/kg) 1 h before I/R induction through both oral gavage and intraperitoneal injection. Then, the AKI model rats were induced by IRI. Oxidative stress and antioxidant protein levels were assessed in the brain, heart, and liver tissues in the experimental groups. Furthermore, the effects of GO on IRI-induced liver dysfunction, apoptosis, and inflammation were measured by Western blot.
Results:
GO pretreatment could significantly restore the levels and activity of antioxidant proteins in the brain, heart, and liver tissues (P < 0.05). Moreover, GO pretreatment could decrease the inflammatory cytokine (IL-1, IL-6, and TNF-α) in the liver (P < 0.01). By reducing Bax/Bcl-2 ratio and down-regulating caspase-3, GO could significantly diminish apoptosis in the liver tissue after the kidney I/R (P < 0.01). Additionally, GO could significantly diminish the deterioration of liver function in the kidney I/R model.
Conclusion:
GO protects distant organs against renal IRI-induced oxidative stress. Furthermore, it ameliorates liver function and remarkably exerts anti-oxidative, anti-inflammatory, and anti-apoptotic roles in the liver as an important detoxifying organ.
Keywords
Introduction
Acute kidney injury (AKI) is a growing global concern among severe hospitalized patients. AKI can result from ischemia-reperfusion injury (IRI), 1 triggered during surgery. 2,3 An overload in fluid volume, uremia, and disruption in the electrolyte and acid-base balances are well-known AKI outcomes that are obviously manifested by a higher risk of mortality. 4 However, this mortality rate cannot be only described by loss of renal functions or by AKI treatment complications. Instead, AKI triggers organ-specific and systemic immunologic, humoral, and hemodynamic imbalances explaining the patients’ mortality from AKI. AKI-induced multi-organ dysfunctions play major roles in outcomes of patients with AKI. 5,6 An increase in organ cell death and endothelial injury, production of reactive oxygen species (ROS), dysregulation of the immune system, elevated leukocyte infiltration, and inflammation in distant organs that are mediated by organ-organ communication 7 are common complications in IRI-induced distant organ dysfunctions. 8 –10
AKI induces oxidative stress, increases lipid peroxidation, and decreases the activities of enzymatic and non-enzymatic antioxidants levels in the brain, heart, and liver. 11 AKI-induced cardiac injury is mostly mediated by activation of the sympathetic nervous system (SNS) or unstable renin–angiotensin–aldosterone system (RAAS). 12 Cardiomyopathy and cell apoptosis are elevated in the heart tissue 24 h after IRI 13 and defective cardiac mitochondria may be important in heart injury of AKI patients. 14 Loss of memory and neural cell apoptosis along with blood-brain barrier (BBB) disruption were found during bilateral renal I/R as a result of kidney-brain crosstalk. 15 RAAS is activated by reno-cerebral reflex after I/R-induced AKI 16 and RAAS inhibitors, sympatholytic drugs, or renal nerve blockage may initiate reno-cerebral protection in AKI patients. 16 The pathophysiology of renohepatic crosstalk during AKI is related to alterations in renal and liver drug metabolism that are mediated by leukocytes activation, inflammation, uremic toxins, and other neuro-humoral factors. 7
Gamma Oryzanol (GO) is an essential bioactive component derived from rice bran oil containing a mixture of lipids; sterols and ferulic acid. In lesser amounts, GO can be found in wheat bran and some vegetables and fruits. Its powder is inodorous, white or yellowish-white, crystalline, and oil soluble with high purity (≥99%). The anti-inflammatory and antioxidant effects of GO are reported in different studies. 17 –21
Understanding the complex connections between renal and distant organs can result in the development of novel diagnostics and therapeutic strategies to recover outcomes in cases with AKI. In the present study, the effects of GO on I/R-induced oxidative stress in distant organs including brain, heart, and liver of rats were evaluated. Furthermore, considering the importance of the liver in drug detoxification and its role in the administration of antioxidants, the impact of GO on inflammatory and apoptosis pathways was assessed in liver tissues of the AKI-rat model at the protein level using western blot assay. Moreover, oral gavage and intraperitoneal injection (IP) routes of administration were applied to compare the effects of GO in rats.
Materials and methods
Reagents
Rats were bought from Pasteur Institute (Tehran, Iran). Gamma-Oryzanol was purchased from Syna Vison Co., (Iran). Primary and secondary antibodies for western-blotting were obtained from Santa Cruz Biotechnology, Inc. (Texas, USA). The other reagents that used in this study were obtained from standard commercial suppliers.
Animals
In this study, 24 adult male Wistar rats (230 ± 20 gr) were used. After 10 days adaptation to the new condition, the experiment was begun. Rats were divided randomly into 4 similar groups, including: (1) sham group, (2) I/R group, (3) I/R+ GO treatment group (100mg/kg body weight) by gavage, (4) I/R+ GO treatment group (100 mg/kg body) 22 by intraperitoneal injection (IP). The GO treated rats (3 and 4 groups) were given a single dose of GO only 1 hour before ischemia induction. Maintenance and feeding of all groups were similar, free access to food and water was provided for all rats and all animals were kept at standard condition (21 ± 2°C) under light/dark cycle (12 hours).
Induction of ischemia-reperfusion
Before I/R induction, the animals were anesthetized by xylazine and ketamine (10 and 90 mg/kg, IP). In the Sham group, just renal artery vascular manipulation (without clamp) was performed and in other groups, the abdominal wall was cut and renal vascular were obstructed for 45 min by non-traumatic vascular clamps. Ischemia was induced successfully when red-colored kidneys became pale. Then, clamp and occlusion were removed and the abdominal cavity was stitched and animals were returned into their own cages. After 6 h reperfusion, blood samples were collected directly from the left ventricle of the present rats for some biochemical factors. Then, rats were euthanized and their main organs such as the brain, heart, and liver in all groups were taken for further analyses.
Biochemical analysis
To investigate the effect of GO on antioxidant capacities of the studied organs, the enzymatic and non-enzymatic antioxidant protein levels were measured. The total glutathione (GSH) levels in the isolated homogenates tissues were assessed by a commercial assay kit (Cat Na. CS0260, Sigma Aldrich, Darmstadt, Germany). The activity of glutathione peroxidase (GPx), catalase (CAT), superoxide dismutase (SOD), total antioxidant capacity (TAC) level along with malondialdehyde (MDA) levels were assessed in heart, brain and liver tissues using special kits (Zellbio, Biocore, Germany) according to their protocols. Furthermore, levels of liver enzymes including AST (aspartate transaminase) and ALT (alanine aminotransferase), and total serum protein levels were evaluated in the serum samples of the studied rats with a photometric method using related kits (Pars Azmoon, Iran).
Western blot analysis
To examine the impact of GO on inflammatory and apoptosis pathways in the liver tissues, protein levels of TNF-α, IL-1, and IL-6 and Bax, Bcl2, and caspase-3 were evaluated in liver tissue homogenates using western-blotting. Total cellular proteins were extracted from the liver tissues using lysis buffer. The concentration of total protein in the supernatant was measured by Bradford protein assay. Then, 12 µg/ml of the protein samples were separated on SDS-PAGE (10%) gels and transferred to PVDF membranes (polyvinylidene difluoride). After blocking, primary antibodies including β-actin (cat. no. sc-47778), IL-1β (cat. no. sc-32294), IL-6 (cat. no. sc-28343), TNF-α (cat. no. sc-130349), Bax (cat. no. sc-7480), Bcl2 (cat. no. sc-492), and caspase-3 (cat. no. sc-7272) were incubated with the membranes at 4°C overnight. Following general washing, the bindings of primary antibodies were detected with anti-rabbit IgG-HRP (cat. no. sc-2357) as a secondary antibody at 1:1,000 dilution. Finally, the bands were visualized, photographed, and quantified.
Statistical analysis
All values were presented as the mean ± SD. Comparisons between groups were analyzed by one-way ANOVA followed by Tukey’s post-hoc multiple comparison test. The IBM SPSS 16.0 Software (SPSS, Inc.) used for statistical analysis. P < 0.05 was set as a significant level.
Results
To reveal the impact of GO on kidney IRI-induced distant organ injuries, rats were subjected to IRI with or without pretreatment with 1 dosage of GO 1 h before IRI induction. GO pretreatments were performed by IP and gavage.
GO suppressed oxidative stress in the brain, heart, and liver after kidney IRI
To study the antioxidant activity of GO in different organs in the experimental groups, the levels of GSH and TAC, and enzymatic activities of GPx, CAT, and SOD were measured. Kidney I/R significantly decreased the levels of these factors in the brain and heart tissues compared to the Sham group (P < 0.001), Tables 1 and 2. However, GO pretreatment could significantly elevate their protein levels especially when applied by IP in the brain (P < 0.001) and liver (P ≤ 0.45) (Tables 1 and 2).
Protective effects of GO on brain after kidney IRI.

Values are expressed as the mean ± SD. I/R, ischemia-reperfusion; GO, Gamma Oryzanol; IP, treated by intraperitoneal; SOD, superoxide dismutase; GPx, glutathione peroxidase; MDA, malondialdehyde; TAC, Total antioxidant capacity; GSH, total glutathione.
a I/R group vs. Sham.
b treatment groups vs. I/R group.
Cardioprotective effects of GO after kidney IRI.

Values are expressed as the mean ± SD. I/R, ischemia-reperfusion; GO, Gamma Oryzanol; IP, treated by intraperitoneal injection; SOD, superoxide dismutase; GPx, glutathione peroxidase; MDA, malondialdehyde; TAC, Total antioxidant capacity; GSH, total glutathione.
a I/R group vs. Sham.
b Treatment groups vs. I/R group.
Compared with the Sham-surgery group, significant reduction was observed in the TAC (79.57 ± 10.28 vs. 51.39 ± 4.91, P < 0.001) and GSH (29.28 ± 2.07 vs. 19.27 ± 4.46, P = 0.001) levels along with GPX (100.08 ± 8.75 vs. 75.98 ± 8.75, P = 0.008), CAT (178.65 ± 11.72 vs. 118.84 ± 5.93, P < 0.001) and SOD (14.58 ± 1.80 vs. 11.79 ± 0.93, P = 0.043) activities in the IR groups in liver tissue. However, GO treatment could significantly increase their levels in the treated groups compared to the IR group (P < 0.05), consequently, suppress the IRI-induced oxidative stress in the liver (Table 3). GO could increase the total antioxidant capacity (51.39 ± 4. 9 vs. 62.38 ± 8.63, P = 0.170) and GPX (75.98 ± 8.75 vs. 88.47 ± 12.47, P = 0.198) of the liver, but they were not statistically significant, Table 3. Both gavage and IP GO-pretreatment could improve the antioxidant activity of the studied tissues, the IP method was much more significant.
Hepatoprotective effects of GO after kidney IRI.

Values are expressed as the mean ± SD. GO, Gamma Oryzanol; IP, treated by intraperitoneal injection; SOD, superoxide dismutase; GPx, glutathione peroxidase; MDA, malondialdehyde; TAC, Total antioxidant capacity; GSH, total glutathione.
a I/R group vs. Sham.
b Treatment groups vs. I/R group.
GO pretreatment reduced lipid peroxidation in the main organs after kidney IRI
The impact of GO on kidney IRI-produced lipid peroxidation (MDA) levels were evaluated in the studied organs and groups (Tables 1, 2, and 3). In the brain (343.71 ± 6.45, P < 0.001), heart (942.32 ± 50.09, P < 0.001), and liver (447.35 ± 55.65, P < 0.001) tissues of the IR group elevated levels of MDA were observed compared to the Sham group. Administration of GO to IRI-induced rats resulted in a significant reduction of MDA in the brain (284.70 ± 14.83, P = 0.01), heart (353.07 ± 24.76, P < 0.001), and liver (236.19 ± 24.20, P = 0.001) compared to the IRI group. The treatment by IP was more significant except for the MDA level in the brain (Tables 1, 2, and 3).
GO diminished inflammation in the liver after kidney IRI
As presented in Figure 1A–D, an increase was detected in the protein expression levels of inflammatory cytokines, including TNF-α (P = 0.001), IL-6 (P = 0.037), and IL-1 (P = 0.012) in I/R group compared to the Sham group. GO treatment could significantly reduce the levels of these cytokines when compared with the IR group. These reductions in inflammatory cytokine were much more significant in IP (P < 0.001) than gavage (P < 0.01) group (Supplementary Table S1).

Effect of GO on IRI-induced inflammation in the liver. (A) Represents Western blots. The bar graphs reveal the intensity of inflammatory cytokines including (B) TNF-α, (C) IL-1, and (D) IL-6 in the studied group. β-actin was used as a loading control. Values are expressed as the mean ± SD. */#P < 0.05, **/##P < 0.01, and ***/###P < 0.001. * I/R group compared with Sham; # treatment groups compared with I/R group. I/R, ischemia-reperfusion; GO, Gamma Oryzanol; IP, treated by intraperitoneal injection; ns, not significant.
GO reduced hepatic cells apoptosis after kidney IRI
Compared with the Sham group, kidney I/R resulted in a significant elevation in Bax and a decrease in Bcl protein levels in the I/R group, where GO could remarkably reverse these alterations (Supplementary Table S1, Figure 2A and B). Quantification of the intensity of immunoreactive bands indicated the significance of these changes in the treated groups in comparison to the I/R group, P < 0.001 (Figure 2B). Likewise, compared with the I/R group, treatment with GO significantly reduced the caspase-3 expression at the protein level, P < 0.05 (Figure 2C and D).
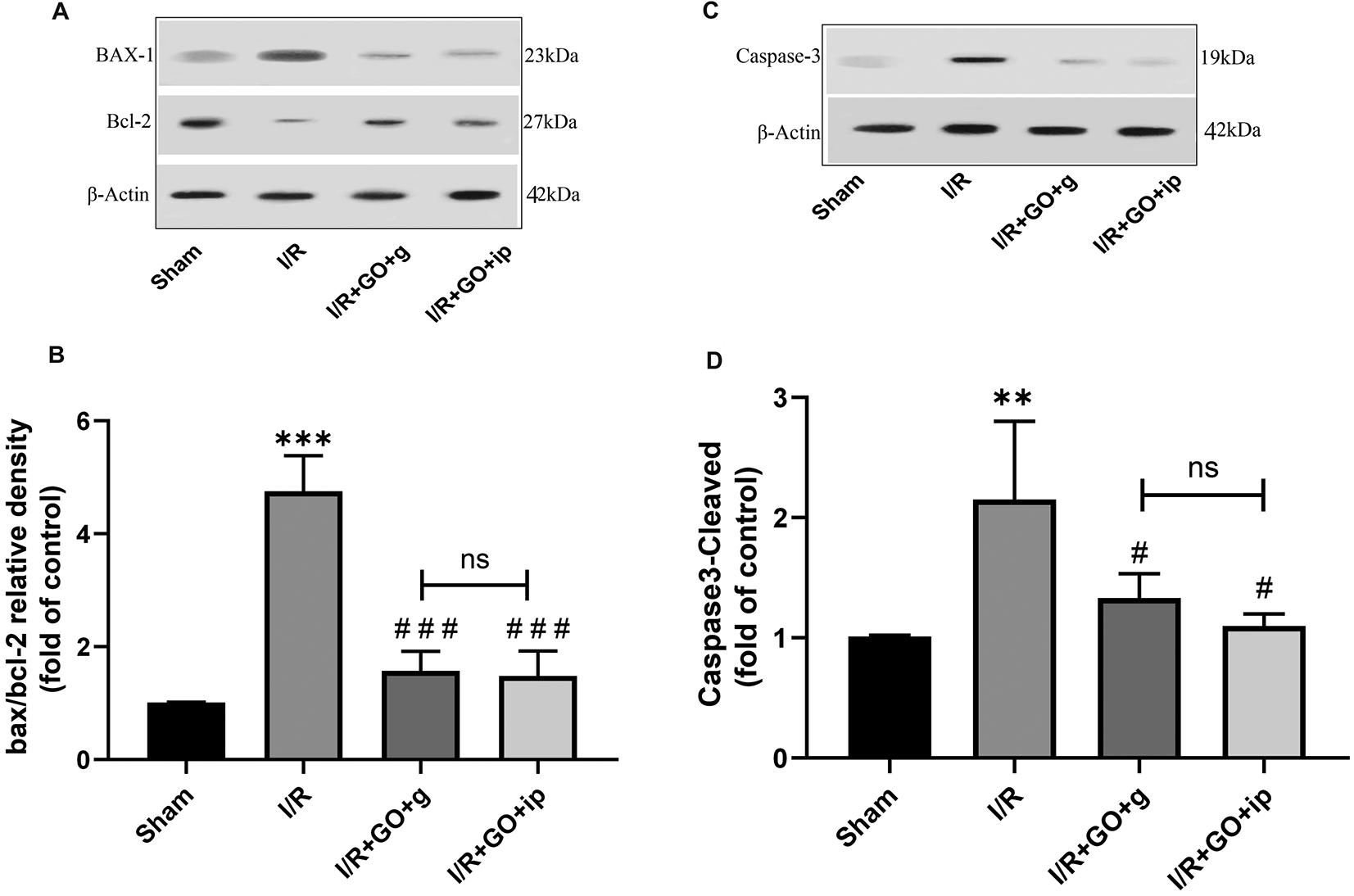
Effect of GO on IRI-induced inflammation in the liver. (A, C) Represents Western blots. The bar graphs reveal the intensity of apoptotic factors; (B) Bax/Bcl ratio (C) caspase 3 in the studied group. β-actin was used as a loading control. Values are expressed as the mean ± SD. */#P < 0.05, **/##P < 0.01, and ***/###P < 0.001. * I/R group compared with Sham; # treatment groups compared with I/R group. I/R, ischemia-reperfusion; GO, Gamma Oryzanol; IP, treated by intraperitoneal injection; ns, not significant.
GO improves the IRI-induced liver dysfunction
The levels of hepatic functional parameters including AST (aspartate transaminase) and ALT (alanine aminotransferase) were also evaluated in the serum samples of the rats. Elevated levels of AST and ALT were significantly observed in the I/R group compared to the sham group. GO treatment could significantly diminish the levels of these enzymes in the treated groups (Supplementary Table S1, Figure 3A and B). Additionally, GO treatment could improve the decreased concentrations of serum total proteins after I/R; however, it was not statistically significant (Figure 3C).

Effect of GO on liver function after kidney IRI. The levels of hepatic functional parameters (A) AST, (B) ALT, and (C) liver total proteins in the experimental groups. Values are expressed as the mean ± SD. */#P < 0.05, **/##P < 0.01, and ***/###P < 0.001. * I/R group compared with Sham; # treatment groups compared with I/R group. I/R, ischemia-reperfusion; GO, Gamma Oryzanol; IP, treated by intraperitoneal injection; AST, aspartate transaminase; ALT, alanine aminotransferase; ns, not significant.
Discussion
In the clinical setting, IRI is a common source of AKI that can be resulted in irreversible renal injury, distant organ dysfunctions, and high mortality. Since oxidative stress and inflammation in the kidney and other distant organs are the hallmarks of IRI-induced injury, interventions that target these events would be beneficial in managing the harmful effects of IRI. 1,21,23 In this study, GO pretreatment could significantly improve the antioxidant capacities of brain, heart, and liver against kidney IRI-induced oxidative stress. The hepatoprotective effects of GO were mediated by its antioxidative, antiapoptotic, and anti-inflammatory effects. Moreover, GO could significantly diminish the deterioration of liver function in a I/R model.
Until now, researchers find multiple compounds that are effective in reducing complications of distant organs in AKI patients. As an antioxidant, GO has been applied to ameliorate oxidative stress in lung and liver. 21,24 It also exerts anxiolytic effects under high-fat diet and chronic stress circumstances. 17,18 The anti-inflammatory effect of Oryzanol is reported in glaucoma. 20 GO could suppress ROS accumulation and ROS-activated mitochondrial apoptotic pathway in a hepatic cell line. 19 However, the impact of GO on renal IRI-induced distant organ dysfunction has not been reported to our knowledge. In this study, the results pointed out the potential of GO to ameliorate extrarenal organ dysfunctions after IRI in vivo.
It is reported that after IRI, MDA level, as an index of lipid peroxidation, is increased and GSH (a central free radical scavenger), SOD, CAT and GPX levels are decreased in brain hippocampus tissue of rats. 25 Zhang et al. discovered that Bcl-2 family proteins (including decreased Bcl-2 expression/increased Bax expression) are dysregulated and the NF-κB pathway is activated in the brain tissue after renal IRI. 26 In the present study, GO treatment of rats 1 h before I/R could increase the antioxidant capacity of the brain, indicated by increased levels of GSH, TAC, SOD, CAT and GPX and decreased level of MDA compared to I/R group in the brain tissue. These results indicated a protective impact of GO on the brain against kidney IRI.
Amini et al. reported that Naringin and Trimetazidine improved the activity of antioxidant enzymes and reduced myocardial injury after kidney IRI. 23 Additionally, Mdivi-1 as a Drp1 (Dynamin-related protein 1) inhibitor, protected the heart against renal I/R-induced cardiac injury in rats. 14 Likewise, in this study, GO exerted cardioprotective effects by increasing the total antioxidant capacity of the heart, diminishing lipid peroxidation indicated by lower MDA level, and increasing the antioxidant enzymes at protein levels.
In the rat IRI model, not only apoptotic hepatocytes are seen after I/R injury, but also an increase in liver MDA, ALP (alkaline phosphatase), AST, and ALT levels and also a decrease in the activity of SOD are observed. 27,28 IRI also increases IL-1 and TNF-α levels in liver 29 and plasma 30 leading to reduce antioxidant ability in liver tissue. IR increases lipid peroxidation and plasma ALT level after liver injury in AKI patients. 31 Likewise, in the present study, kidney IRI could trigger hepatic oxidative stress, as evidenced by an increase in liver MDA, decrease levels of total GSH, SOD, and CAT, and diminished liver TAC. Elshazly and her colleagues discovered that Pioglitazone as a PPARγ agonist, reduces inflammatory and apoptotic responses in the liver after renal IRI. 32 Vitamin E reduced plasma levels of ALT and AST levels in patients with AKI-induced liver injury. 33 Another antioxidant, Thymoquinone (THQ) (the main component of Nigella sativa), was approved for improvement of renal and hepatic functions. 34 Similarly, in the present study, one dosage of GO before IRI could enhance liver antioxidant defense ability. Moreover, it decreased liver lipid peroxidation that identified by the lower level of MDA when compared to the I/R group. GO pretreatment could also decrease the levels of inflammatory cytokine including IL-1, IL-6, and TNF-α in comparison to the I/R group. An increased Bax/Bcl-2 ratio in the liver tissue could up-regulate caspase-3 and elevate apoptosis in the I/R group. Treatment of rats with GO protected the liver against kidney IRI-induced apoptosis confirmed by decreased levels of proapoptotic Bax, increased levels of antiapoptotic Bcl, a decline in Bax: Bcl ratio, and decreased levels of caspase-3 at protein levels in liver tissue. As well, GO could significantly diminish the deterioration of liver function in the kidney I/R model, reflected by the decreases in levels of AST and ALT after IRI. Alterations in protein synthesis is reported in hepatic dysfunction in AKI patients, GO could increase the diminished concentration of total liver proteins after IRI. These results confirm the hepatoprotective effects of GO after kidney IRI. Both injection and oral administration of GO were effective in the protection of distant organs against IRI-induced oxidative stress; indicating the bioavailability of GO in rats by both administration routes. However, the results of IP administration were more significant.
There are some limitations to this study. We did not study the toxicity of GO in the IRI model in rats. The receptor(s) and molecular mechanism(s) for GO action are still unclear. We found that at the early stage, GO pretreatment can protect against renal IRI-induced multiple organ dysfunction, however, it is important to reveal whether it can induce protective effects on different distant organs at the chronic stage.
Conclusion
The results of this study indicated a significant protective effect of GO on IRI-induced oxidative stress in brain, heart, and liver; GO improved the antioxidant defense potential of these organs after kidney IRI. It also ameliorated liver function, remarkably lessened hepatocytes apoptosis, and attenuated inflammation in the liver as an important detoxifying organ. Based on the current findings that links kidney damage with distant organs injury, GO pretreatment would be a successful therapeutic strategy in reducing the IRI-induced organ dysfunctions after surgery and organ transplantation.
Supplemental material
Supplemental Material, Supplementary_Table_S1 - Protective effects of Gamma Oryzanol on distant organs after kidney ischemia-reperfusion in rats: A focus on liver protection
Supplemental Material, Supplementary_Table_S1 for Protective effects of Gamma Oryzanol on distant organs after kidney ischemia-reperfusion in rats: A focus on liver protection by Yasin Bagheri, Shadi Aghajani, Mahla Hosseinzadeh, Farid Hoshmandan, Abdollah Abdollahpour and Sepideh Zununi Vahed in Human & Experimental Toxicology
Footnotes
Ethical approval
Animal experimental procedures and protocols were performed according to the standards of the National Institutes of Health for Laboratory Animals Care and approved by the Animal Care Committee at Tabriz University of Medical Sciences, Tabriz, Iran (IR.TBZMED.VCR.REC.1399.123).
Acknowledgment
The authors wish to thank the personnel of Faculty of Veterinary Medicine of Islamic Azad University of Tabriz and Kidney Research Center of Tabriz University of Medical Sciences for guidance and help.
Author contributions
YB, SA, MH, AA, and FH contributed to experimental work and data analysis. SZV did drafting and revising the article. All authors gave final approval of the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study financially supported by Kidney Research Center of Tabriz University of Medical Sciences, Tabriz, Iran.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
