Abstract
Arsenic, a major environmental toxicant and pollutant, is a global public health concern. Among its many adverse effects, arsenic is immunotoxic, but its effects on human neutrophil functions are not yet well-defined. In this study, we aimed to evaluate the in vitro effects of acute low-dose NaAsO2 exposure on human polymorphonuclear neutrophils (PMNs) for 12 h on the following innate defense mechanisms: formation of neutrophil extracellular traps (NETs), production of reactive oxygen species (ROS), and phagocytosis. Phorbol myristate acetate (PMA) was added to induce NETs formation, which was quantified by measuring cell-free extracellular DNA (cf-DNA), myeloperoxidase-conjugated (MPO)-DNA and neutrophil elastase-conjugated (NE)-DNA, and confirmed by immunofluorescence labeling and imaging. Extracellular bactericidal activity by NETs was evaluated by co-culturing Escherichia coli and PMNs in the presence of a phagocytic inhibitor. Levels of NETs in the culture medium after PMA stimulation was significantly lower in PMNs pre-exposed to arsenic than those not exposed to arsenic. Immunofluorescence staining and extracellular bactericidal activity by NETs revealed similar results. Phagocytosis and ROS production by PMNs were also significantly reduced by arsenic pre-exposure. Together, our findings provide new insights in arsenic immunotoxicity and suggest how it increases susceptibility to infectious diseases in humans.
Keywords
Introduction
Arsenic, classified by the International Agency for Research on Cancer as a Group 1 carcinogen, is a major environmental toxicant and pollutant, and a global health concern. 1 Its main sources are natural and anthropogenic. 2 Arsenic compounds are frequently found in drinking water, foodstuffs, air, and soil, 3,4 and elevated concentrations of arsenic in groundwater pose a huge threat to public health in some regions, such as Bangladesh, Taiwan, and Argentina. 5 –8 Humans and food-producing dairy animals inadvertently consume arsenic through contaminated drinking water and food. According to the WHO guidelines, the safe limit of arsenic in drinking water is 10 µg/l and the maximum permissible limit is 50 µg/l. 9 The Agency for Toxic Substances and Disease Registry ranked arsenic at the top of their Substance Priority List in 2019. 10
Arsenic intensifies non-infectious pathologies, including cancer, skin lesions, vascular diseases, diabetes mellitus, and neuropathy, 11,12 but also damages hematological and immunological systems. 13 –16 Recent human and animal studies revealed that arsenic exposure causes neutropenia 17 and damages lymphocyte and macrophage function, 18 –20 increasing susceptibility to recurrent opportunistic infections like tuberculosis 21 as well as Influenza A 22 and parasitic infections, thus acting as an immunosuppressive agent. 12,16,17,22 –24
Brinkmann et al. 25 first described a neutrophil-derived antimicrobial defense mechanism of innate immunity, termed neutrophil extracellular traps (NETs). Stimulation of NETs causes the extrusion of a meshwork of chromatin fibers expressing antimicrobial proteins like myeloperoxidase (MPO), neutrophil elastase (NE), and cathepsin G. NETs are essential in innate immunity and captures, immobilizes, and kills intruding gram-negative and gram-positive bacteria, fungi, and parasites, as well as invading microorganisms extracellularly. 25,26
In a recent in vitro study, arsenic was shown to have potent granulotoxic effects in mammals and cause a marked decrease in phagocytosis by human and bovine neutrophils. 14 But how arsenic exposure influences NET formation is yet to be investigated. In the present study, we explored the possibility that NET formation by neutrophils would be altered under arsenic exposure. We further sought to examine phagocytosis, bactericidal activity, and generation of reactive oxygen species (ROS) to assess neutrophil function in the presence of arsenic.
Materials and methods
Isolation and primary culture of human neutrophils
All experiments were approved by the Institutional Review Board of Aichi Medical University (17-H341). Written informed consent was obtained from each subject. Peripheral blood was collected by venipuncture from 10 healthy Japanese adults, 5 men and 5 women, with a median age of 30 years (range, 18–54 years), by using a lithium heparin blood collection tube (BD Vacutainer; Becton, Dickinson and Company, Franklin Lakes, NJ, USA). Exclusion criteria were a history of malignant, degenerative, or infectious diseases; diabetes mellitus; and the use of corticosteroids or other immunosuppressive agents at the time of the study. All participants completed a questionnaire that included items on exposure, such as sources of drinking water and periods of living and working abroad. Polymorphonuclear neutrophils (PMNs) were isolated by discontinuous density gradient centrifugation on 1-Step Polymorphs (Accurate Chemical and Scientific Corporation, Westbury, NY, USA). To prevent neutrophil activation during the separation procedure, resuspension of pellets was performed slowly, and the vortex setting was kept in the low- to mid-range so that cells were not activated. Freshly isolated PMNs were resuspended using phenol red-free RPMI-1640 medium containing 2 mM L-glutamine (Wako Pure Chemicals, Osaka, Japan) supplemented with 6% heat-inactivated fetal bovine serum. This method yields samples of >95% viability assessed by trypan blue dye exclusion. Cell density was determined by manual counting, i.e. a researcher counted cells in the microscope field with a hemocytometer.
Arsenic treatment
Arsenic in the form of NaAsO2 (Sigma-Aldrich, St. Louis, MO, USA) was diluted with PBS to a final concentration of 20 ng/ml. 14 This concentration reflects the level at which arsenic is toxic in humans (the blood concentration of arsenic in residents with arsenic intoxication in Bangladesh was reported as 14.3 ng/ml, 27 and the mean [SD] arsenic concentration in whole blood collected from residents of the Lanyang Basin, on the northeast coast of Taiwan, where well water has a high arsenic content, was 9.6 [9.96] ng/ml 28 ).
In our study, for all experiments, freshly isolated PMNs were washed twice with 0.45% NaCl solution, resuspended in phenol red-free RPMI-1640 medium, seeded at 1 × 106 cells/ml in 35 mm culture dishes, and incubated with or without arsenic for 12 h at 37°C in a humidified atmosphere containing 5% CO2. In general, humans are unintentionally exposed to arsenic via drinking water and foods that contain high levels of the element. Li et al. 29 showed that after administration of a single gavage to animals, blood levels of arsenic increase over the first few hours and then decrease again over the next few hours, indicating that blood components, including neutrophils, would be exposed to high levels of arsenic for approximately 12 h after a single gavage. On the basis of this animal experiment, we chose an incubation time of 12 h. However, further experiments with a wider range of incubation times would be valuable to improve our understanding of the chronic effects of arsenic.
PMN viability assay
Calcein-AM (Dojindo, Kumamoto, Japan) was used as a fluorescent probe for labeling and monitoring cellular functions of live PMNs. After 12 h incubation with or without arsenic, the contents of culture dishes were collected and centrifuged at 250 × g for 5 min. The cells were resuspended in culture dishes with fresh RPMI-1640 (phenol red-free) and 10 µM calcein-AM was added. After 30 min of incubation, samples were examined by fluorescent calcein retention in the cell with a fluorometric reader (SpectraMax M spectrophotometer, Molecular Devices, San Jose, CA, USA) at 480-nm excitation and 520-nm emission.
cf-DNA, MPO-DNA, and NE-DNA
Release of NET components, including cell-free extracellular DNA (cf-DNA), myeloperoxidase-conjugated DNA (MPO-DNA), and neutrophil elastase-conjugated DNA (NE-DNA), was assayed as described previously. 30 After 12 h of incubation with or without arsenic, the cultured PMNs were stimulated with 25 nM phorbol-12-myristate-13-acetate (PMA) for 4 h at 37°C. Then the PMNs were digested with 500 mU/ml micrococcal nuclease (Worthington Biochemical Corp, Lakewood, NJ, USA) for 30 min. Nuclease activity was subsequently stopped by the addition of 5 mM EDTA, and the supernatant was collected and stored at 4°C until use. cf-DNA from the culture supernatant was quantified using the Quant-iT PicoGreen dsDNA fluorescence assay (Life Technologies, Carlsbad, CA, USA) at 485-nm excitation and 538-nm emission. Fluorescence intensity, which reflects the amount of DNA in the sample, was measured with a QUBIT® 2.0 Fluorometer (Life Technologies). The medium levels of MPO- and NE-DNA were measured by ELISA. 31 In brief, quantitative detection of MPO- and NE-DNA were performed using a “sandwich” ELISA with monoclonal antibodies against MPO (catalog #07-496, Merck Millipore Corp., Burlington, MA, USA) and NE (catalog #MABF759, Merck Millipore Corp.), and a peroxidase-conjugated monoclonal anti-DNA antibody (Cell Death Detection ELISA #1154467500: bottle 2; Roche Diagnostics, Indianapolis, IN, USA). The wells of microtiter strips were coated with monoclonal antibodies specific for MPO and NE to capture MPO- and NE-DNA derived from NETs. A peroxidase substrate (2,2’-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) was added, which reacted with the bound peroxidase to yield a soluble green product detected at 405 nm. Absorbance readings were proportional to the amount of bound horseradish peroxidase-labeled anti-DNA monoclonal antibody. Results are expressed in arbitrary units.
Detection of NETs by immunolabeling
PMNs incubated with or without arsenic for 12 h were seeded onto 0.001% poly-D-lysine coated glass coverslips, incubated for 1 h to allow adherence, and stimulated with 25 nM PMA for 4 h at 37°C. Then, after fixing with 4% paraformaldehyde, the cells were permeabilized with 0.5% Triton X-100 (Wako Chemicals, Tokyo, Japan) and blocked with 1% skim milk. Cells were subsequently incubated for 1 h with diluted primary monoclonal anti-MPO (0.1 µg) and anti-NE (0.7 µg) antibodies, before being incubated overnight with a secondary antibody (Alexa Fluor-647-conjugated antibody) (all antibodies from Abcam, Cambridge, UK). After incubation, DNA was stained with 1 μg/ml Hoechst 33342 for 5 min. Specimens were mounted in Mowiol (Sigma-Aldrich) and viewed under a fluorescence microscope (BZ-9000, Keyence, Osaka, Japan). The excitation wavelengths for NE, MPO, and Hoechst 33342 were 650 nm, 495 nm, and 528 nm, respectively; and the emission wavelengths were 665 nm, 515 nm, and 617 nm, respectively. Image analysis was performed using Lucia imaging software (Laboratory Imaging s.r.o., Praha, Czech Republic). NETs were counted from six different fields of vision in each slide and expressed as the percentage of NET-forming cells per total number of cells in the field. A NET was defined as a discrete area of bright blue fluorescence larger than a neutrophil. 32
Reactive oxygen species measurement
ROS production in PMA-activated PMNs was measured using cell-permeant 2’,7’-dichlorodihydrofluorescein diacetate (DCFH-DA; Sigma-Aldrich) according to the manufacturer’s instructions. Briefly, after loading 10 µM of DCFH-DA, PMNs pretreated with or without arsenic were stimulated with PMA for 1 h, and ROS production was assayed by oxidation of nonfluorescent DCFH to a highly green-fluorescent dichlorofluorescein with a SpectraMax M5 spectrophotometer at 485-nm excitation and 525-nm emission.
Extracellular bactericidal activity
Freshly isolated PMNs were incubated for 12 h with or without arsenic, and NETs were induced by PMA. E. coli (K12 strain) was added to each plate in the presence of 10 µg/ml cytochalasin D (Wako Chemicals), which is a phagocytosis inhibitor. This concentration was chosen according to a previous bactericidal activity assay. 33 The samples were then centrifuged at 700 × g for 10 min. After co-culture for 30 min at 37°C, each well was scraped off, and a 10 µl aliquot from each well was seeded onto an agar plate and cultured overnight at 37°C. The number of colonies that grew was counted, and bactericidal activity was expressed as a percentage, with 100% representing the number of colonies obtained when bacteria were incubated without neutrophils. 34
Phagocytic assay
PMN phagocytic capacity was examined by their ingestion of FITC-labeled opsonized Escherichia coli using the Phagotest kit (ORPEGEN Pharma, Heidelberg, Germany) according to the manufacturer’s instructions. In brief, after incubating with or without arsenic for 12 h, E. coli bacteria were added and incubated at 37°C. Phagocytosis was stopped by placing the samples on ice and adding quenching solution. Propidium iodide was added to stain DNA, and flow cytometric analysis was performed using the BD LSRFortessa X-20 cell analyzer (BD Biosciences, Heidelberg, Germany).
Statistical analysis
All results are presented as the mean ± SEM. Statistical analysis was performed in SigmaPlot 14 (Systat Software Inc., Richmond, CA, USA). We compared differences in continuous variables with the nonparametric Mann–Whitney test. Repeated measures ANOVA was used to compare NET and ROS production in different groups. p < 0.05 was considered statistically significant.
Results
Arsenic inhibited PMA-induced cf-DNA, MPO-DNA, and NE-DNA release from PMNs, but not PMN viability
PMA significantly increased cf-DNA, MPO-DNA, and NE-DNA levels compared with NaAsO2(−)/PMA(−), whereas arsenic pretreatment for 12 h significantly inhibited PMA-induced cf-DNA, MPO-DNA, and NE-DNA release (Figure 1). Arsenic exposure per se did not affect cf-DNA, MPO-DNA, or NE-DNA levels compared with NaAsO2(−)/PMA(−), and arsenic exposure for up to 12 h did not affect PMN viability. Fluorescence intensity of calcein after incubation with and without arsenic was 110 ± 5.3 and 108 ± 9.8, respectively (not significantly different).
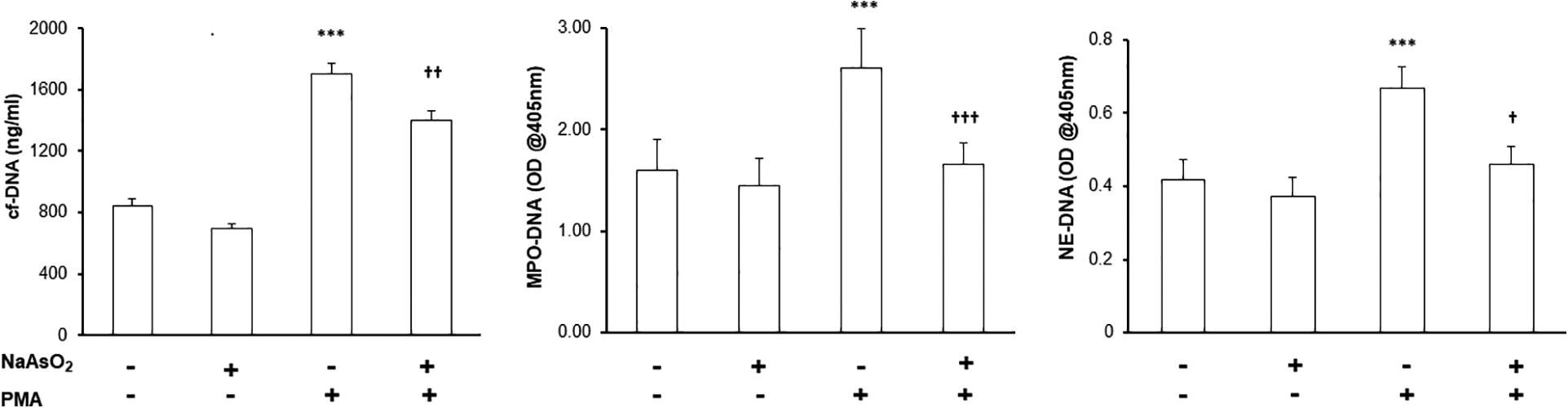
cf-DNA, MPO-DNA, and NE-DNA levels after exposure of PMNs to arsenic. Data are presented as the mean ± SEM (n = 10). ***p < 0.001 vs. NaAsO2(−)/PMA(−); †p < 0.05, ††p < 0.01 and †††p < 0.001 vs. NaAsO2(−)/PMA(+).
Arsenic inhibited PMA-induced NET formation
PMNs pretreated without arsenic released MPO, NE, and DNA extracellularly after stimulation with PMA, whereas PMNs pre-exposed to arsenic showed little response to PMA (Figure 2).

Detection of NETs by immunolabeling. (A) Direct immunofluorescence staining of DNA (blue), myeloperoxidase (green), elastase (red), and merge (yellow) in PMNs without (b) or with (c) arsenic pretreatment. Scale bar, 10 μm. (B) Quantification of NETs after PMA stimulation of isolated PMNs pretreated without or with arsenic. Arsenic reduced NET formation by approximately 45%. Data are presented as mean ± SEM (n = 10). **p < 0.01 vs. NaAsO2(−)/PMA(−); ††p < 0.01 vs. NaAsO2(−)/PMA(+).
Bactericidal activity assay under a phagocytosis inhibitor
The concentration of cytochalasin D used in this assay (10 μg/ml) was sufficient to inhibit phagocytosis, but it did not affect the formation of NETs (data not shown). Co-incubation of E. coli and PMA-activated PMNs exposed to arsenic led to extracellular bactericidal activity of only 40.7 ± 3.7%, whereas 53.5 ± 4.8% of bacteria were killed extracellularly by PMA-induced PMNs not exposed to arsenic (p < 0.01).
Arsenic inhibited ROS production and phagocytic capacity
ROS production was significantly increased by PMA and significantly inhibited by arsenic (Figure 3). Furthermore, 12 h exposure to arsenic significantly reduced the phagocytic capacity of PMNs compared with that of PMNs not exposed to arsenic (Figure 4).

ROS production in PMNs after arsenic exposure. ROS production after PMA stimulation of isolated PMNs pretreated without or with arsenic. Data are presented as the mean ± SEM (n = 10). ***p < 0.001 vs. NaAsO2(−)/PMA(−); †††p < 0.001 vs. NaAsO2(−)/PMA(+).

Phagocytic capacity in PMNs after arsenic exposure. Phagocytic capacity of isolated PMNs pretreated without or with arsenic. Representative histogram is shown in Figure 4A. PMNs were gated in the software program by using forward and side scatter. The percentage of phagocytosing cells was determined by counting the number of events above this marker position (M2/M1+M2). Figure 4B summarizes the phagocytic capacity of PMNs after arsenic exposure. Data are presented as the mean, and error bars indicate the SEM (n = 5). **p < 0.01 vs. NaAsO2(−).
Discussion
This is the first study to demonstrate how acute low-dose arsenic exposure in vitro impairs human PMN function, affecting NET formation, phagocytosis, and ROS production. PMA-stimulated release of MPO-DNA, NE-DNA, and cf-DNA into the cell culture medium was significantly lower in arsenic-pre-exposed PMNs than in PMNs not exposed to arsenic, indicating that NET formation was impaired. NET-related bactericidal activity, which was evaluated in the presence of a phagocytosis inhibitor, was also significantly reduced in arsenic-pretreated PMNs. Immunofluorescence staining of released DNA, NE, and MPO confirmed these results. Phagocytosis and PMA-stimulated ROS production, which are other important functions of PMNs, were also impaired by arsenic.
NET lattices are formed by activated PMNs and can trap both gram-positive and gram-negative bacteria. 25,26 Capture and containment of pathogens by intracellular factors released into the extracellular fluid by innate immune effector cells is an evolutionarily conserved host defense mechanism. 35 NETs are formed in response to a variety of pro-inflammatory stimuli, such as lipopolysaccharides, interleukin-8, and tumor necrosis factor, as well as by various microorganisms and pathogens, including experimental models of shigellosis, 25 necrotizing fasciitis, 32 and pneumococcal pneumonia. 36 Therefore, NETs would be expected to form part of the host defense system during inflammation. Extrapolating our findings to the clinical situation suggests that the decreased formation of NETs by PMNs, and the impaired extracellular bactericidal activity by this mechanism, may impair the elimination of microbes in patients with sepsis, rendering them highly susceptible to the spread of infection.
Only one study to date, conducted by Wei et al. 37 has investigated the influence of arsenic on NET formation by PMNs, which showed that exposing PMNs to arsenic for 90 min induced NET formation as assessed by DNA release into the medium and by immunofluorescence imaging. Our results, however, show that arsenic per se cannot induce NET formation by PMNs, although it does decrease PMA-induced NET formation. This discrepancy could be explained by different exposure times, arsenic concentration, or the experimental method used to detect NET formation. For example, in our study, the dose used was 100 times lower than that in the Wei et al. report. We assessed NETs by measuring NE-DNA and MPO-DNA in addition to cf-DNA level. cf-DNA in the medium could come from several sources in addition to release from NETs, including cellular necrosis and apoptosis. 35,38,39 Several previous studies have shown that in vitro arsenic exposure to PMNs induces apoptosis and necrosis due either to increased ROS production or to activation of caspases. 40,41 MPO-DNA and NE-DNA are more specific for NETs than cf-DNA 42,43 because these web-like structures released by activated PMNs are composed of DNA associated with neutrophil granule proteins such as NE, cathepsin G, and MPO. 25
Arsenic treatment has been shown to increase 44 –46 or decrease 14,47 –49 ROS production, depending on the experimental conditions. In our experiment, we incubated neutrophils with a low level of sodium arsenic for 12 h. The discrepancies in findings on ROS production after arsenic exposure could be accounted for by differences in the arsenic compound used, 44 length of exposure, 50,51 cell type, 14,50,51 and concentration of arsenic. 50
The arsenic-induced impairments we observed in PMN functions, including phagocytosis and ROS production, are consistent with previous data. Taheri et al. showed that exposing PMNs to arsenic in vitro significantly decreased both phagocytosis and PMA-induced hydrogen peroxide production. They concluded that arsenic possesses granulotoxic properties and may impair innate immunity. In zebrafish, PMA-induced ROS production assessed by oxidation of dihydrodichlorofluorescein diacetate to dichlorofluorescein was also decreased upon arsenic exposure. 49,52
Humans and mice exposed to arsenic show elevated incidence and severity of infections, respiratory problems, and fungal and parasitic infections. 19,21,53 In mice, chronic arsenic exposure resulted in a severely compromised response to influenza A infection accompanied by decreased cytokine production and increased mortality. 22 In Bangladesh, prenatal arsenic exposure from drinking water was found to increase the risk of lower respiratory tract infection during infancy. 54 Other studies have shown that ex vivo peripheral blood mononuclear cell proliferation and IL-2 secretion were reduced in arsenic-exposed individuals. 40 The impairments in PMN function observed in the present study would explain, at least in part, the enhanced risk of immune dysregulation and infection in arsenic-exposed individuals.
This study had some limitations. The influence of short-term arsenic exposure is not identical to that of chronic exposure. To clarify the pathogenesis of chronic arsenic poisoning, which is clinically of great significance, another experimental approach is necessary. A further limitation is that the bactericidal activity observed in this study does not apply to gram-positive bacteria, because we investigated bactericidal activity in gram-negative bacteria. Moreover, we studied the influence of only one dose of arsenic exposure. Further investigations with multiple doses would be desirable to clarify the influence of arsenic on PMN function and innate immunity.
Conclusions
In summary, this study shows that acute low-dose arsenic exposure in vitro impairs the function of human PMNs, including their formation of NETs, phagocytosis, and production of ROS. Our findings provide new insight that arsenic induces immunotoxicity and increases susceptibility to infectious diseases in humans.
Footnotes
Abbreviations
Acknowledgments
The authors thank Dr Aminul M Huq for his valuable comments and advice.
Author contributions
Study conception and design: MMI and NT. Data collection: MMI. Analysis and interpretation: MMI. Writing the manuscript: MMI and NT. Critical revision: NT and MMI.
Availability of data and material
The data generated during and/or analyzed during this study are not publicly available due to issues of confidentiality but are available from the authors upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Written informed consent was obtained from each healthy volunteer included in the study, and the study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki as reflected a priori by the individual institutions’ Medical Ethics Committees.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by a JSPS KAKENHI Grant-in-Aid for Young Scientists B (17K11598) and Grant-in-Aid for Scientific Research C (18K08927).
