Abstract
Objective:
To explore the potential function of MDM2-mediated Notch/hes1 signaling pathway in cisplatin-induced renal injury.
Methods:
The acute renal injury models of mice after intraperitoneal injection of cisplatin in vivo, and the apoptotic models of human renal tubular epithelial (HK-2) cells induced by cisplatin in vitro, were conducted respectively. The renal function-related parameters were measured. The renal tissue pathological changes and apoptosis were observed by PAS staining and TUNEL staining, respectively. Cell viability and apoptosis were detected by MTT and flow cytometry. Notch/hes1 pathway-related proteins were tested by Western blotting.
Results:
After mice injected by cisplatin, the levels of Cr, BUN, urine cystatin C, urine NGAL and urine ACR were increased and GFR was decreased with the elevation of renal tubular injury scores, the upregulation of the expressions of MDM2, N1ICD, Hes1 and Cleaved caspase-3, as well as the enhancement of cell apoptosis accompanying decreased ratio of Bcl-2/Bax. However, these cisplatin-induced renal injuries of mice have been improved by MDM2 inhibition. Besides, the declined viability, increased cytotoxicity, and enhanced apoptosis were observed in cisplatin-induced HK-2 cells, with the activated Notch/hes1 pathway. Notably, the phenomenon was alleviated in cisplatin-induced HK-2 cells transfected with MDM2 shRNA, but was severer in those co-treated with AdMDM2. Moreover, Notch1 siRNA can reverse the injury of AdMDM2 on HK-2 cells.
Conclusion:
Inhibiting MDM2 could reduce cell apoptosis through blocking Notch/hes1 signaling pathway, thus alleviating the acute renal injury caused by cisplatin.
Introduction
Cisplatin, a broad-spectrum antineoplastic drug, acts by binding to and inducing cross-linking of deoxyribonucleic acid, ultimately triggering cell apoptosis, which has been widely used in clinical chemotherapy of various solid tumors. 1 But unfortunately, the optimal usefulness and efficacy of cisplatin-based chemoradiotherapy is limited by its dose related toxicity, particularly nephrotoxicity. 2 The manifestations of cisplatin nephrotoxicity are various, which include acute and chronic renal failure. 3,4 While acute renal injury has been reported to be the most serious complication of cisplatin treatment, and its pathogenesis involves oxidative stress, vascular factors, inflammation, and the apoptosis of renal tubular epithelial cells. 5,6 Current evidence has shown that cisplatin-induced apoptosis of the renal tubular epithelial cell is one of the primary elements in cisplatin nephrotoxicity. 7 Therefore, to find out a molecular target concerning for the prevention and treatment of the apoptosis of renal tubular epithelial cells, may have important clinical significance in reducing cisplatin nephrotoxicity. 8
Murine double minute 2 (MDM2), an E3 ubiquitin ligase, are responsible for various biological processes, like cell cycle, differentiation, DNA repair injury and so on, via regulating the stability of numerous substrate molecular proteins. 9,10 The published data have shown that the expression of MDM2 in kidney is mainly located in renal tubular epithelial cells and podocytes. 11 Mulay et al. pointed out that inhibiting MDM2 can restrain the regeneration of renal tubular epithelial cell in a p53-dependent manner in the mice models of acute renal injury after ischemia, which could also reduce the inflammatory response after ischemia through blocking NF-κB pathway. 12 Thomasova et al. have shown that MDM2 can restrain renal tubular epithelial cell death and acute renal injury. 13 Besides, knockout MDM2 has been demonstrated to regulate high glucose-induced podocyte mitotic mutation by restraining Notch1/hes1 pathway. 14 Notch1/hes1 pathway, an important pathway to regulate cell proliferation and differentiation, has obtained great attention in glomerular-related diseases in recent years. 15,16 More importantly, there was evidence stating that cisplatin-induced acute kidney injury would be alleviated through limiting Notch1/hes1 pathway. 17 Therefore, we speculated that MDM2 may mediate cisplatin-induced acute renal injury by regulating Notch1/hes1 pathway.
To address this question, the acute renal injury models of mice after intraperitoneal injection of cisplatin in vivo, and the apoptotic models of human renal tubular epithelial cells induced by cisplatin in vitro, were conducted to explore the potential function of MDM2-mediated Notch/hes1 signaling pathway in cisplatin-induced acute renal injury.
Materials and methods
Ethical statement
Animal experimental designs were approved by the Laboratory Animal Ethics Committee of our institution, and all the researches on experimental animals were conducted in strict compliance with the relevant regulations of the National Institutes of Health (NIH). 18
Animals
The experimental animals consist of 60 C57BL/6J male mice (8–10 weeks old, weighing 150–180 g) with clean grade, which was brought from Shanghai SLAC Laboratory Animal Co., Ltd. These mice were fed in clean grade animal rooms at room temperature (22–25°C) in normal circadian rhythm, with freely access to food and water.
Establishment and grouping of mouse models
The renal injury model was established by intraperitoneal injection of cisplatin in mice, and the experiment was divided into four groups with 15 mice in each group, including: Control group (mice without any treatment), Cisplatin group, MDM2 inhibitor group, and Cisplatin + MDM2 inhibitor group. Mice in the Cisplatin + MDM2 inhibitor group intraperitoneal injected with 20 mg/kg of the MDM2 inhibitor nutlin-3a (Selleck Chemicals, TX, USA) in 50% dimethyl sulfoxide (vehicle) and 15 mg/kg cisplatin l h later, followed by daily nutlin-3a injection for 4 days. For mice in the Cisplatin group, a single intraperitoneal injection of 15 mg/kg Cisplatin (A14207156601, Qilu Pharmaceutical Co., Ltd) was conducted in mice after equivalent vehicle injection with the volume of 10 μl/g. 17 Mice in the MDM2 inhibitor group were only injected nutlin-3a and didn’t inject Cisplatin.
Sample collection
On the fifth day, the 24 h urine was collected, and the blood was obtained from ophthalmic venous plexus of mice, placed at room temperature for 2 h, centrifuged at 4000 rpm for 10 min at 4°C. Then, the serum was stored in refrigerator at −80°C for the determination of biochemical indicators. The kidney was taken out, and some kidney tissues were fixed at room temperature with 4% neutral paraformaldehyde for making pathological sections, and the rest of kidney tissues were separated and stored in a refrigerator at −80°C for Western blotting.
Detection of renal function indexes
The serum Cr and BUN levels were detected by German Beckman automatic biochemical analyzer, and the urine cystatin C and urine NGAL levels were detected by ELISA kit (AAR-AKI-G1, RayBiotech, Norcross, GA, USA). The urine microalbumin levels were measured with kits purchased from Exocell Inc. (Philadelphia, PA, USA), and the glomerular filtration rate (GFR) was detected by using FIT-GFR Inulin kits (FIT-0415; BioPAL, Worcester, MA, USA).
PAS staining
Paraffin slices were dewaxed to water and subsequently treated with a 1% aqueous solution of periodic acid, then treated with Schiff’s reagent for 10 min. According to the previous literature, 19 the renal tubular injury score was as follows: 0 points: normal kidney tissue; 1 points: lesion area <10%; 2 points: lesion area in 10–25%; 3 points: lesion area in 25–50%; 4 points: lesion area in 50–75%; 5 points: lesion area >75%.
TUNEL staining
Kidney tissues were cut into slices. After being deparaffinized using xylene and ethanol dilutions and rehydration, the protease working K solution was added to react at 21–37°C for 20 min. Then, 0.3% methanol solution was added at room temperature for 30 min, and immersed in 0.1% Triton X-100 (dissolved in 1×SSC solution) for 5–10 min. According to the instructions of TUNEL test kit (Roche, Basel, Switzerland), the apoptotic rate (100%) = (TUNEL positive cell/total cells) × 100% was observed under fluorescence microscope by adding (4′,6-diamidino-2-phenylindole) DAPI to stain nucleus and neutral gum seals.
HK2 cell culturing and grouping
The human renal tubular epithelial cell HK2, brought from American Type Culture Collection (Manassas, VA, USA), were cultured in DMEM-F12 containing 5% FBS in an incubator at 37°C with 5% CO2. After stabilization, HK2 cells were digested and centrifuged in 6 or 12-well plates. When cells grew to 80%, they were conducted starvation treatment, which used DMEM-F12 medium containing 0.5% FBS instead. Then, cells were divided into groups as follows: Control group (without any treatment); Cisplatin group (with Cisplatin (10 μM) stimulated for 48 h 20 ); Cisplatin + MDM2 shRNA group (with the transfection of MDM2 shRNA and the treatment of Cisplatin); Cisplatin + empty vector group (with the transfection of control empty vector and the treatment of Cisplatin); Cisplatin + AdMDM2 group (with the transfection of AdMDM2 and the treatment of Cisplatin); and Cisplatin + AdMDM2 + Notch1 siRNA group (with the transfection of AdMDM2 and Notch1 siRNA and the treatment of Cisplatin). The recombinant adenovirus vector carrying human MDM2 sequence (AdMDM2), recombinant lentiviral vector harboring a short hairpin (sh)RNA targeting the gene encoding the MDM2 (MDM2 shRNA) and control empty lentivirus and were constructed by JikaiGene (Shanghai, China). Notch1 specific small interfering RNA (Notch1 siRNA) was purchase from Ribobio, Guangzhou, China to knock down Notch1 expression. HK2 cells were incubated with lentivirus or adenovirus or transfected with siRNA according to the manufacturer’s instructions.
MTT assay
HK2 cells in each group were inoculated into 96-well plates. About 5 × 103 cells were suspended in 200 µl of culture medium and added into 96-well plates. Then, 20 µl of 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (5 mg/ml) was added into each well. The supernatant was discarded after avoiding-light incubated for 4 h at 37°C. Then, 150 µl of dimethyl sulfoxide (DMSO) was added into each well to dissolve intracellular crystals, and the absorbance was measured at 492 nm by microplate reader.
LDH detection
HK2 cells in each group were inoculated into 96-well plate, and the instruction of LDH colorimetric assay kit (Life Technologies) was used. Then the optical density (OD) was determined at 490 nm wavelength by enzyme labeling instrument. Cytotoxicity (%) = (sample absorbance − control absorbance)/(maximum amount of releasable enzyme activity absorbance − control absorbance) × 100%. 21
Flow cytometry
HK2 cells were digested by trypsinase without EDTA and collected in 15 ml centrifugal tube. The cells were centrifuged at 1500 rpm for 5 min at 4°C. Cells were suspended with cold PBS, then suspended with 400 µl 1×Annexin V conjugate solution and blended lightly. Then, 5 µl of Annexin V-FITC staining solution was added to the prepared cell suspension, gently blended, and were incubated at 2–8°C for 15 min without light. Thereafter, 5 µl of PI (propidium iodide) was added into each tube and the cell apoptosis was detected by flow cytometry.
Western blotting
The protein from renal tissue and cells was extracted and its concentration was determined with a BCA method (Wuhan Boster Biological technology Co., Ltd). Protein (50 µg per lane) was electrophoresis separated by 10% polyacrylamide gel (Wuhan Boster Biological technology Co., Ltd), PVDF transfer membrane, 5% BSA blocked for 1 h at room temperature, added anti 1:500 dilution. Then the membranes were incubated with the primary antibodies at 4°C overnight. The primary antibodies were Notch1 intracellular domain (N1ICD) (ab8925, Abcam), Hes1 (ab71559, Abcam), Bcl-2 (ab114016, Abcam), Bax (ab77566, Abcam), Cleaved caspase-3 (ab2302, Abcam) and β-actin (ab8227, Abcam). The corresponding second antibody (1:1000 dilution, Abcam) was incubated at room temperature for 1 h, and the membrane was washed for three times/5 min. β-actin was used as internal reference, and the image was developed by Bio-rad Gel Dol EZ imager (GEL DOC EZ IMAGER, Bio-rad, California, USA). The gray value of the target band was analyzed by Image J software.
Statistical method
SPSS 21.0 statistical software was used to analyze the data. The experiment was repeated at least three times, and the data was presented as the means ± SD. Differences among groups were compared by the One-way ANOVA by a Tukey’s post hoc test. The difference was significant when p < 0.05.
Results
Renal function index of mice in each group
As shown in Figure 1, compared with Control group, the Cr, BUN, urine cystatin C, urine NGAL, and urine ACR levels were increased in the mice from Cisplatin group, with the declined GFR (all p < 0.05), while no obvious difference was found between MDM2 inhibitor group and Control group (p > 0.05). Besides, the levels of Cr, BUN, Urine cystatin C, Urine NGAL, and Urine ACR were lower while GFR was higher in mice from the Cisplatin + MDM2 inhibitor group than those from the Cisplatin group (all p < 0.05).

Comparison of renal function indexes of mice in each group. Comparison of the levels of Cr (a), BUN (b), urine cystatin C (c), urine NGAL (d), urine ACR (e), and GFR (f) in the mice among each group. *p < 0.05: compared with Control group; # p < 0.05: compared with Cisplatin group.
Pathological changes of kidney tissue in mice of each group
The result of PAS staining has been shown as Figure 2. The kidney structure was clear with complete glomerulus and well-formed tubular epithelial cells in the Control group and MDM2 inhibitor group. In Cisplatin group, the renal tubular epithelial cells had unclear cell boundaries, partial necrosis, karyopycnosis or disappearance, and the homogeneous red staining transparent tube seen in the tubular. As for Cisplatin + MDM2 inhibitor group, the phenomenon of necrosis, and karyopycnosis or disappearance, was improved, but it was still seen some transparent tubular types with clear ballon lumen and normal cell morphology. The renal tubular injury score was increased significantly in Cisplatin group when compared to Control group and MDM2 inhibitor group, but was lower in MDM2 inhibitor group than in Cisplatin group (all p < 0.05).

Comparison of PAS staining and renal tubular injury score in mice among each group. *p < 0.05: compared with Control group; # p < 0.05: compared with Cisplatin group.
Cell apoptosis of mice in each group
TUNEL staining (Figure 3) demonstrated the increased apoptosis of kidney tissues in Cisplatin group (p < 0.05), but no significant changes in mice from MDM2 inhibitor group when compared with Control group (p > 0.05). Furthermore, the apoptosis was decreased significantly in Cisplatin + MDM2 inhibitor group by comparison with the Cisplatin group (all p < 0.05).

Comparison of the apoptosis of kidney tissues in mice among each group. *p < 0.05: compared with Control group; # p < 0.05: compared with Cisplatin group.
Expression of Notch1/hes1 signaling pathway in mice of each group
The protein expression of Notch1/hes1 signaling pathway was detected by Western blotting (Figure 4). Consequently, no significant difference was found between Control group and MDM2 inhibitor group in Notch1/hes1 pathway (all p > 0.05), whereas the protein levels of MDM2, N1ICD, Hes1, and Cleaved caspase-3 were elevated with the decreased Bcl-2/Bax ratio in Cisplatin group (all p < 0.05). Compared with Cisplatin group, these protein expressions decreased significantly with the increased ratio of Bcl-2/Bax in the Cisplatin + MDM2 inhibitor group (all p < 0.05).

The protein expression of Notch1/hes1 signaling pathway in the kidney tissue of mice in each group detected by Western blotting. *p < 0.05: compared with Control group; # p < 0.05: compared with Cisplatin group.
Comparison of HK2 cell viability, cytotoxicity and apoptosis in each group
The declined viability, increased cytotoxicity (LDH levels), as well as enhanced apoptotic rate of HK-2 cells were observed in Cisplatin group and Cisplatin + empty vector group, as compared to Control group in Figure 5 (all p < 0.05). When compared with Cisplatin group, the increased viability with the decreased cytotoxicity and apoptosis rate of HK-2 cells were exhibited in the Cisplatin + MDM2 shRNA group; and however, the cells in the Cisplatin + AdMDM2 group presented an opposite result (all p < 0.05). Moreover, cells in the Cisplatin + AdMDM2 + Notch1 siRNA group had higher cell viability, and lower cytotoxicity and apoptosis than those in the Cisplatin + AdMDM2 group (all p < 0.05).

Comparison of HK-2 cell viability, cytotoxicity and apoptosis in each group. (a) The viability of HK-2 cells determined by MTT assay, (b) comparison of HK-2 cell cytotoxicity (LDH release) in each group, and (c and d) the apoptotic rate of HK-2 cells determined by flow cytometry. *p < 0.05: compared with Control group; # p < 0.05: compared with Cisplatin group; & p < 0.05: compared with Cisplatin + MDM2 shRNA group; % p < 0.05: compared with Cisplatin + AdMDM2 group.
Expression of Notch1/hes1 signaling pathway in HK-2 cells
As shown in Figure 6, the protein expression of MDM2, N1ICD, Hes1 and Cleaved caspase-3 in HK-2 cells was detected to be increased with the decreased Bcl-2/Bax ratio in the Cisplatin group and Cisplatin + empty vector group, as compared with Control group (all p < 0.05). Compared with Cisplatin group, the expression of above proteins decreased significantly with the enhanced ratio of Bcl-2/Bax in Cisplatin + MDM2 shRNA group; and however, cells in the Cisplatin + AdMDM2 group presented an opposite expression tendency(all p < 0.05). Additionally, the lower expression of these proteins and higher ratio of Bcl-2/Bax were discovered in HK-2 cells from the Cisplatin + AdMDM2 + Notch1 siRNA group than those from the Cisplatin + AdMDM2 group (all p < 0.05).
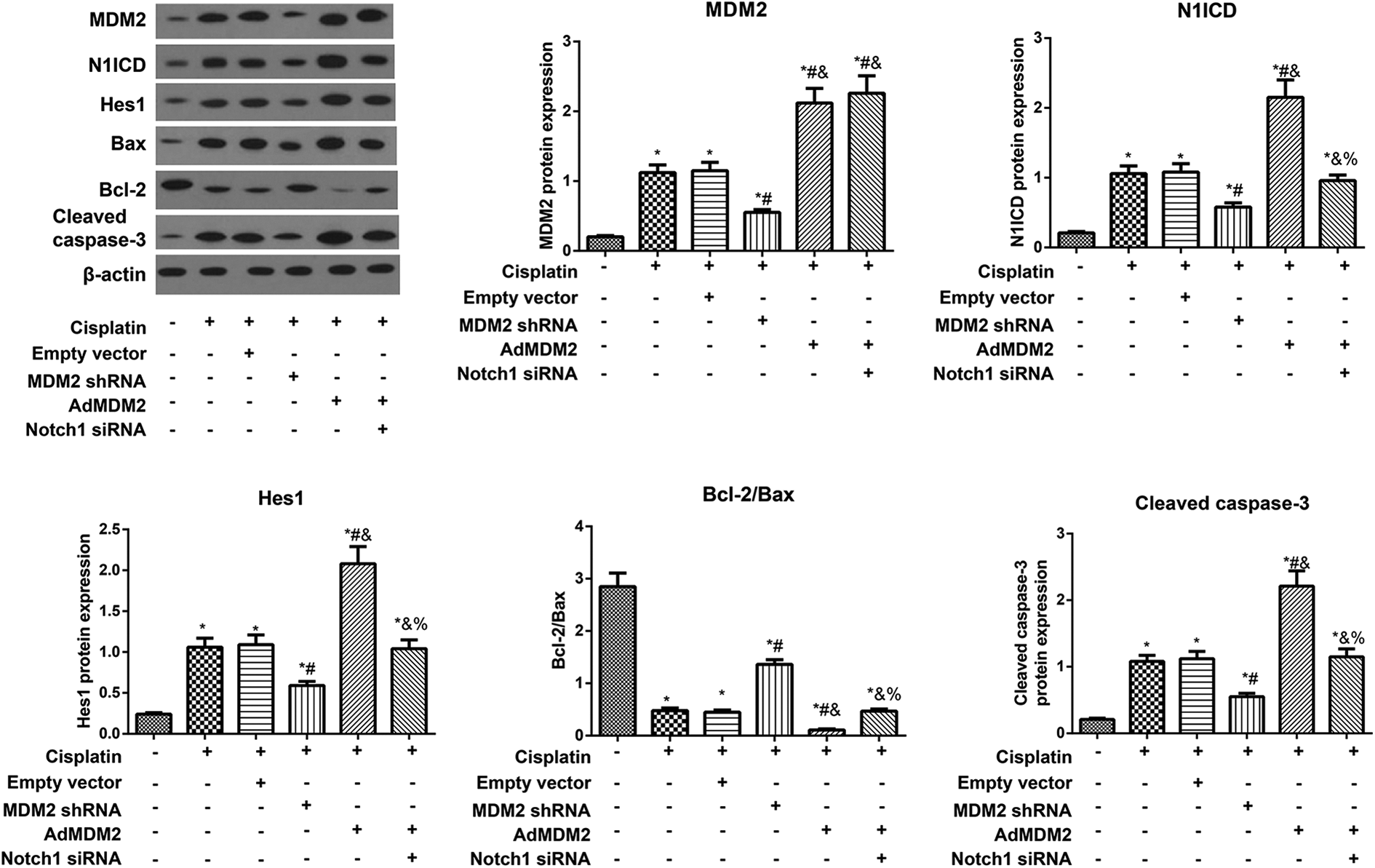
The protein expression of Notch1/hes1 signaling pathway in HK-2 cells among each group. *p < 0.05: compared with Control group; # p < 0.05: compared with Cisplatin group; & p < 0.05: compared with Cisplatin + MDM2 shRNA group; % p < 0.05 compared with Cisplatin + AdMDM2 group.
Discussion
Cumulative evidence indicates that the main manifestation of acute renal injury is the alteration of renal function, especially the glomerular filtration rate, and the major traditional diagnostic markers of renal injury are blood urea nitrogen (BUN) and serum creatinine (Cr). 22 During renal injury, the damaged tubular epithelial cells would produce a large number of NGAL, a classic biomarker for early diagnosis of acute renal injury, which could further induce the apoptosis of infiltrating neutrophils in tubulointerstitium. 23,24 Besides, Cystatin C and ACR are also good sensitive biomarkers reflecting glomerular filtration rate. 25,26 In this study, the acute renal injury mice models were established by intraperitoneal injection of cisplatin and the levels of the above markers were detected. As a result, the levels of Cr, BUN, Urine cystatin C, Urine NGAL and Urine ACR were increased significantly with the decreased GFR in cisplatin-treated mice, which was consistent with previous findings. 19,27 However, all the above-mentioned indexes were improved in those mice after the treatment of MDM2 inhibitor, suggesting that inhibiting MDM2 may improve cisplatin-induced renal function of acute renal injury.
Cisplatin is excreted mainly through the kidney, and the decreased renal function may result in the increased toxicity. 28 Several lines of evidence have confirmed that cisplatin preferentially accumulates in proximal tubular epithelial cells, and then damage these cells. 29 Worth mentioning, the apoptosis of renal tubular epithelial cells plays an important role in the pathogenesis of cisplatin-induced acute renal injury. 30,31 Through pathological observation, the renal tubular epithelial cells of cisplatin-treated mice had unclear cell boundaries, partial necrosis, karyopycnosis or disappearance in vivo, and TUNEL staining showed that the apoptotic rate was increased significantly. While the in-vitro experiments also demonstrated that the activity of cisplatin-induced HK-2 cells was decreased significantly with the increased apoptosis, which was in line with other previous researches. 5,32,33 Nevertheless, the apoptotic rate of renal tubular cells decreased significantly after inhibition of MDM2. For example, MDM2 blockade during postischemic acute kidney injury could alleviate the inflammatory response to restrict the necrosis of renal tubular epithelial cells by blocking the NF-κB pathway, as indicated by Mulay et al. 12 On the other hand, cisplatin has been reported to activate Bax and then increase the permeability of mitochondrial membrane; meanwhile, cytochrome c could release from mitochondria to activate caspase, thus initiating apoptosis mediated by mitochondrial pathway. 34 As analyzed by western blotting in our study, the expression of Cleaved caspase-3 was increased with the decreased ratio of Bcl-2/Bax in both cisplatin-treated mice and cisplatin-induced HK-2 cells, but MDM2 inhibition could improve these expressions. Similarly, Man Jiang and colleagues also found that MDM2 inhibitor (Nutlin-3) can restrain cisplatin-induced oligomerization of Bax and Bak and reduce cisplatin-induced renal cell apoptosis, 35 indicating that suppression of MDM2 may attenuate cisplatin-induced apoptosis of renal tubular epithelial cells by mediating mitochondrial apoptosis.
To further explore the regulation mechanism of MDM2, Notch1/hes1 pathway-related proteins were detected by western blotting, and consequently, cisplatin could increase the expression of N1ICD and Hes1 in renal tubular epithelial cells. Overexpression of MDM2 further promoted whereas inhibition of MDM2 reduced the expression of them. There was evidence that MDM2 can inactivate Numb, a substrate of MDM2 which could suppress Notch1, through ubiquitination degradation, thus activating its downstream Notch1 pathway. 36 It has also been found that MDM2 can activate Notch1 pathway directly through ubiquitination non-degradation pathway without relying on Numb. 37 Moreover, glomerular mesangial cells, the activation of Notch1 signaling was attenuated by MDM2 depletion, and MDM2 interacted with N1ICD was independent of Numb in the study of Lei et al. 38 Therefore, we speculated that MDM2 may activate Notch1 pathway in renal tubular epithelial cells without depending on Numb. Generally, the activation of Notch would give rise to the nuclear localization of the intracellular portion (NICD) of the Notch receptor, and then activates Hes1 gene to regulate cell turnover. 39,40 In agreement with our results, the cisplatin-induced tubular injury would be alleviated via inhibiting Notch2/Hes-1 expression, 17 which would further decrease the expression of Bax but increase the expression of Bcl-2 to reduce cell apoptosis. 41 It is also important to emphasize here that Notch1 siRNA treatment in our research can reverse the effect of MDM2 overexpression on the apoptosis of renal tubular epithelial cells, suggesting that inhibition of MDM2 can regulate downstream apoptosis-related protein expression by regulating Notch1/hes1 pathway, thus delaying apoptosis and protecting against cisplatin-induced acute renal injury.
In summary, our research has got the point that inhibiting MDM2 may improve reduce cisplatin-induced tubular injury and apoptosis via limiting Notch1/hes1 pathway, which provides a new method for the treatment of cisplatin-induced acute renal injury, alleviating its toxic and side effects, and expanding its clinical application.
Footnotes
Acknowledgment
We would like to give our sincere appreciate to all the reviewers for this work.
Author contributions
XL designed the manuscript, LZ conducted the study, G-DHan illustrated the results, PL and YZ wrote and revised the manuscript. XL and LZ contributed equally to this work.
Availability of data and material
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Cangzhou Science and Technology R&D Instruction Project (No. 183302086).
