Abstract
Purpose
In order to seek effective drugs for treating cisplatin-induced acute renal injury and explore the corresponding potential mechanism.
Methods
Mouse kidney injury model was established by intraperitoneal injection of 20 mg/kg cisplatin. The temporal expression of TRPM2 and the regulation of Ginkgolide A on its expression were analyzed by western blot. In order to perform the mechanical analysis, we used TRPM2-KO knockout mice. In this study, we evaluated the repair effect of GA on acute kidney injury through renal function factors, inflammatory factors and calcium and potassium content. Pathological injury and cell apoptosis were detected by H&E and TUNEL, respectively.
Result
Ginkgolide A inhibited inflammatory reaction and excessive oxidative stress, reduced renal function parameters, and improved pathological injury. Meanwhile, we also found that the repair effect of Ginkgolide A on renal injury is related to TRPM2, and Ginkgolide A downregulated TRPM2 expression and inactivated TWEAK/Fn14 pathway in cisplatin-induced renal injury model. We also found that inhibition of TWEAK/Fn14 pathway was more effective in TRPM2-KO mice than TRPM2-WT mice.
Conclusion
Ginkgolide A was the effective therapeutic drug for cisplatin-induced renal injury through acting on TRPM2, and TWEAK/Fn14 pathway was the downstream pathway of Ginkgolide A in acute renal injury, and Ginkgolide A inhibited TWEAK/Fn14 pathway in cisplatin-induced renal injury model.
Introduction
Cisplatin (cis-diaminodichlorplatin (II), CP) is a highly effective chemotherapy drug for various tumor types.1–3 However, the long-term clinical application of CP results in the serious side effects (nephrotoxicity, ototoxicity, neurotoxicity, and emetogenicity. Among these side effects, nephrotoxicity is a main sequela. 4 Approximately one third of patients treated with cisplatin have renal dysfunction. CP causes renal function damage and acute renal failure through a variety of mechanisms, including the production of oxygen/nitrogen substances, DNA damage, renal tubulointerstitial inflammation and apoptosis cell death.5–12
Many studies have confirmed a number of compounds as the potential renal protective agents against CP, including nitric oxide synthesis regulators, natural antioxidants, cell protectors, osmotic diuretics and anti-apoptotic agents. 13 Ginkgolide A is a highly active platelet activating factor antagonist cage molecule, isolated from Ginkgo biloba leaves. 14 Ginkgolide A possesses many biological activities, including anti-inflammatory, anti-cancer, anti-anxiety, anti-atherosclerosis and anti-atherosclerosis, nerve and liver protection. In addition, the evidence has revealed the protection of GA in renal damage, which could improve sepsis-induced renal damage by attenuating renal inflammation and apoptosis via upregulating miR-25 with Nox4 as the target. 14 Researchers have conducted experiments on GA to determine its activity and recorded some encouraging basic findings. However, the protective effect of GA on renal injury is still unclear, and the molecular mechanism is less studied. Transient receptor potential melastatin 2 (TRPM2) is a member of the second TRPM channel subfamily, which can enhance intracellular Ca2+, ROS production and oxidative stress, all of which are related to physiological and pathological processes.15–17 TRPM2 has been found to play a harmful role in ischemia-reperfusion damage of various tissues including kidney, brain, and pancreas.18,19 Based on the existing findings, TRPM2 is a promising therapeutic target for kidney disease.
Tumor necrosis factor-like weak apoptosis-inducing factor (TWEAK) is a type 2 transmembrane protein, which mainly acts as a soluble cytokine with a variety of biological functions, such as pro-inflammatory activity, anti-apoptosis and increasing cell survival and proliferation.3,20,21 TWEAK acts on the activation of fibroblast growth factor-degradable 14 (Fn14) receptor.22–24 Fn14 is expressed by a variety of cell types, including epithelial cells, mesenchymal cells, and endothelial cells. In healthy tissues, the expression of Fn14 is low. However, the level of cellular Fn14 increased induced by stress or injury. Fn14 was found to be expressed in renal tubular cells, mesangial cells, and podocytes. 25 Some studies have also found that renal infiltrating cells such as macrophages also express Fn14. 26
In our study, we mainly focused on the effects of Ginkgolide A on cisplatin-induced renal injury, and the its protective mechanism was also explored. In TRPM2 knockout mice, the effect of this drug on acute kidney injury (AKI) is different from that of TRPM2 wild-type mice, reflecting the role of TRPM2 in it. In order to verify the relationship between TRPM2 and TWEAK/FN14 pathway, we studied the expression difference of TWEAK/FN14 system in TRPM2 knockout mice and wild-type mice, and whether the effect of this pathway on acute renal injury mice is related to TRPM2. At the end of the study, we analyzed whether the drug can protect the cisplatin-induced renal injury by regulating the TWEAK/FN14 system.
Materials and methods
Animals
TRPM2 [-/-:3-979] mice (TRPM2-KO) were provided by Cyagen (Number: SCXK(Su)2018-0003). Adult wild type (WT) male C57BL/6J mice (Number: SCXK (Lu)20190003) were also used in our study as the control. The age and weight of all rats in this study were 8–10 weeks and 18–22 g, respectively. All mice were maintained in a specific pathogen free level animal room at the constant temperature (24 ± 2°C) and relative humidity of 40%–60%. All protocols and procedures in this study were approved by the institutional Animal Care and Use Committee of the Yantai Affiliated Hospital of Binzhou Medical University and followed the Guide for the Care and Use of Laboratory Animals by the United States (US) National Institutes of Health (NIH, 2011).
Induction of AKI
The mice were given intraperitoneally 20 mg/kg cisplatin in normal saline.27–29 Then, the mice were injected with Ginkgolide A solution in DMSO twice. Ginkgolide A (powder, HY-B0355, MedChemExpress, purity ≥ 98.0%) was dissolved in 10% DMSO solution (DMSO: PEG3000: Tween-80: saline = 1:4:0.5:4.5). The control group was injected with DMSO solution alone at the same time points.
Experimental design
The protective effect of Ginkgolide A on acute renal injury is related to TRPM2
In this part, we firstly analyzed the regulation of Ginkgolide A in TRPM2 in renal tissues of TRPM2-WT mice in concentration-dependent manner. Different concentrations (20 mg/kg and 40 mg/kg) of Ginkgolide A were injected into mice. Then TRPM2-KO and TRPM2-WT mice were randomly divided into three groups: normal group, cisplatin induction group, Ginkgolide A group (GA-40), respectively. TRPM2-KO and TRPM2-WT mice were treated with 40 mg/kg GA for three consecutive days after cisplatin injection. 10 mice per group. DMSO was dissolved in DMSO.
The protective effect of TWEAK/Fn14 pathway on acute renal injury in mice is related to TRPM2
In this part, we also used TRPM2-KO and TRPM2-WT mice as the study subject. We firstly analyzed the effect of TRPM2 knockout on TWEAK/Fn14 pathway, and the expression of TWEAK/Fn14 pathway in renal tissues of AKI mice. Next, we also injected lentivirus-transfected TWEAK shRNA or Fn14 shRNA (virus concentration 1011GC/animal) into tail vein of TRPM2-KO and TRPM2-WT mice after cisplatin injection. Both TRPM2-KO and TRPM2-WT mice were randomly divided into four groups: normal group, cisplatin induction group, cisplatin induction + TWEAK shRNA group, cisplatin induction + Fn14 shRNA group. 10 mice in each group.
The protective mechanism of Ginkgolide A against cisplatin-induced acute kidney injury is related to TWEAK/Fn14
In this part, we only used TRPM2-WT mice to perform the mechanical analysis of Ginkgolide A in cisplatin-induced acute renal injury. Mice were randomly divided into five groups: cisplatin induction + GA-40 group, cisplatin induction + GA-40 + TWEAK group, cisplatin induction + GA-40 + TWEAK shRNA group, cisplatin induction + GA-40 group + Fn14 shRNA, cisplatin induction + GA-40 + Fn14. Lentivirus-transfected plasmids (virus concentration 1011GC/animal) were injected into tail vein of TRPM2-WT mice after cisplatin injection. 10 mice in each group. All lentiviral particles were purchased from Merck. TWEAK complementary DNA (cDNA), Fn14 cDNA, TWEAK shRNA, and Fn14 shRNA were inserted into the shuttle vectors pLKO.1-CMV-tGFP vectors. The target sequence of Fn14 shRNA is GCTGGTTTCTAGTTTCCTGGT. The target sequence of TWEAK shRNA is GCGCAGGCAGATGGAGGTTAC.
Sample collection
24 h after the last injection of the drug, the mice were sacrificed via cervical dislocation under 3% pentobarbital sodium (50 mg/kg) anesthesia. Serum and kidney samples were harvested. The serum was stored at −80°C for renal function analysis. The kidney tissues were fixed in 4% paraformaldehyde for histology analysis.
Renal function evaluation
Blood samples were taken 48 h after cisplatin injection to determine the levels of blood urea nitrogen (BUN) and serum creatinine (SCr) using an automated analyzer (Beckman Coulter, USA). The level of serum cystatin C (CysC) was detected by the ELISA kit of mouse CysC (ab201280, Abcam).
Oxidative stress and inflammation response analysis
Renal tissues were collected and homogenized for oxidative stress and inflammation response measurement. Oxidative stress markers SOD, MDA, GSH and MPO, as well as inflammation cytokines (IL-1β, IL-6 and TNF-α) were examined by ELISA methods according to the corresponding kits. All kits were purchased from Nanjing Jiancheng Bioengineering Institute. IL-1β (H002-1-2), IL-6 (H007-1-1), TNF-α (H052-1-2), SOD (A001-3-2), MDA (A003-1-2), GSH (A006-1-1) and MPO (A044-1-1).
Calcium and potassium content in kidney
Kidneys were collected 48 h after cisplatin induction and immediately frozen in liquid nitrogen. The appropriate amount of renal cortex was weighed, and nitric acid solution was added at the vol/wt ratio of 10:1, and then the cortex was digest at 80°C for 1h, and swirling frequently. Continuously, the digest was centrifuged at 10,000g for 5 min, the supernatant was collected and diluted for calcium and potassium detection.
TUNEL staining
To determine the apoptosis of renal cell, paraffin sections (3μm) of kidney tissue were stained by a terminal deoxynucleotidyl transferase (TdT) dUTP nick-end labeling (TUNEL) kit (Beyotime). TUNEL staining was strictly performed according to the manufacturer’s instructions. The TUNEL-positive cells were observed by a light microscope, and the percentage of TUNEL-positive cells was analyzed by Image J pro-plus.
H&E staining
After fixation, paraffin-embedded kidney slices (3μm) were sectioned and stained with hematoxylin and eosin. Renal tissue destruction was also evaluated from these aspects (hemorrhage, inflammatory cell infiltration, tubular cell necrosis and apoptosis, cellular edema, and tubular dilatation) according to the percentage of the kidney involvement, 30 the quantitative grading of the 10 areas corresponding to the kidney tissue is as follows: 1 (<10%), 2 (10%–25%), 3 (26%–50%), 4 (51%–75%), and 5 (>75%), this assessment was performed in a blinded manner by two researchers. 31
Western blot
In short, the kidney tissues were ultrasonically homogenized in RIPA lysis buffer, and centrifuged at 4°C for 12,000g for 10 min to obtain the supernatant. After electrophoretic separation, proteins were transferred onto polyvinylidene fluoride (PVDF) membrane, and the membranes were sealed with 5% skimmed milk powder for 1 h, and incubated with antibody against TRPM2 (1:1200, PB0601, Boster), TWEAK (1:1500, A02009-3, Boster), Fn14 (1:1000, BM4635, Boster), p-NF-κB p65 (1:800, bs-0982R, Bioss), NF-κB p65 (1:1000, bs-23,216R, Bioss), p-IKKβ (1:800, ab194519, Abcam), IKKβ (1:2000, ab124957, Abcam), GAPDH (1:2500, ab9485, Abcam). the membranes were incubated overnight at 4°C with secondary antibodies for 90 min at room temperature, and signals generated by enhanced chemiluminescence (Millipore) were recorded. Analysis of protein bands was carried out by Image-Pro Plus. The total protein level was normalized to the GAPDH protein level.
Statistical analysis
All data were analyzed using SPSS software, and statistical difference in multiple groups was analyzed using one-way analysis of variance (ANOVA) followed by least significant difference (LSD) test. Differences at p < .05 were considered significant. Data were represented as mean ± SD
Results
Ginkgolide A administration down-regulates TRPM2 protein in cisplatin-induced renal injury mice
In this study, we first evaluated the expression of TRPM2 protein in renal tissues after cisplatin induction for different time (6 h, 12 h, 24 h). The result (Figure 1(a)) indicated that a slight expression of TRPM2 in renal tissues of TRPM2 wild type mice, and after cisplatin induction, the expression of TRPM2 increased slightly, but there was no significant difference compared with normal group. After 12 h of cisplatin induction, the expression of TRPM2 protein in renal tissues increased significantly. After 24 h, the expression of TRPM2 was further up-regulated. We also analyzed the expression of TRPM2 protein in cisplatin-induced acute kidney injury in rats after Ginkgolide A treatment (Figure 1(b)). We found that compared with cisplatin-stimulated group, 40 mg/kgGA significantly inhibited the expression of TRPM2 protein in renal injury, while 20 mg/kg GA had no significant regulatory effect on TRPM2. Regulation of Ginkgolide A on TRPM2 in kidney tissue of TRPM2 wild-type mice. (a) The expression of TRPM2 protein in renal tissues after cisplatin induction analyzed by western blot. (b) The expression of TRPM2 protein in kidney tissues of TRPM2 wild-type mice after Ginkgolide A treatment detected by western blot. *p < .05, **p < .01.
Ginkgolide A alleviates the oxidative stress and inflammation response, and improves renal function injury through TRPM2
CP treatment leads to renal function damage including oxidative stress, renal tubulointerstitial inflammation and apoptosis cell death, and so on. As shown in Figure 2(a) and 2(b), after treatment with cisplatin, the levels of oxidative stress and inflammation in kidney tissue increased, mainly manifested by increased MPO and MDA content, decreased SOD content, and elevated levels of inflammatory factors TNF-α, IL-6 and IL-1β. After GA administration, the levels of the above factors changed. Meanwhile, renal function was also explored, the levels of BUN, Scr and CysC were also detected (Figure 2(c)). The three indexes of cisplatin-induced group increased, but after GA administration, these three indexes decreased. Interestingly, we also found the difference between TPRPM2-WT mice and TRPM2-KO mice. In TRPM2 wild-type and knock-out mice without cisplatin, there was no statistical difference in the above oxidative stress indicators, inflammatory factor indicators and renal function parameters. In the cisplatin model group, MPO, MDA, inflammatory factors and renal function parameters in TRPM2-KO mice were significantly lower, and SOD value was significantly increased than those in TRPM2-WT mice. These results indicated that TRPM2 knockout could inhibit inflammation levels, and oxidative stress reaction, and reduce the contents of BUN, Scr and CysC, and improve renal injury induced by cisplatin. After the same dose (40 mg/kg) of GA was given to TRPM2 wild-type mice and knockout mice, the expression of factors in TRPM2 knockout mice was significantly improved. It suggested that the role of GA in promoting the repair of renal function injury is related to TRPM2. Ginkgolide A reduces oxidative stress and inflammation response, improves the renal function through TRPM2. (a) Study design overview. (b) Oxidative stress in TRPM2-KO and TRPM2-WT mice suffered from cisplatin induction was detected by ELISA. The levels of MPO, SOD, MDA and GSH in renal tissues were measured using microplate reader. (c) Inflammation response was also detected by microplate reader. The inflammation factors IL-1β, IL-6 and TNF-α in TRPM2-KO and TRPM2-WT mice with acute renal injury were analyzed by ELISA method. (d) Renal function indexes SCr, BUN and CysC in TRPM2-KO and TRPM2-WT mice with acute renal injury were explored by automated analyzer. *p < .05, **p < .01.
Due to the permeability of TRPM2 to calcium and potassium, we analyzed the content of calcium and potassium in renal tissues of AKI mice (Figure 3). The kidney tissues were harvested 48 h after cisplatin induction. After cisplatin, the content of potassium was reduced, and the content of calcium increased, while GA alters the levels of calcium and potassium in renal tissues. Similarly, we also found the levels of calcium and potassium in TRPM2-KO mice were changed significantly than TRPM2-WT mice. Elemental composition of kidney cortex. The kidneys were harvested 48 h after cisplatin induction. The levels of potassium and calcium in renal cortex of TRPM2-KO and TRPM2-WT mice were determined by atomic absorption spectrometry. *p < .05, **p < .01.
Pathologic changes and apoptosis in the mice renal tissues
Kidneys were collected for H&E staining and Tunel staining. As shown in Figure 4(a) and 4(c), in the normal group, the glomerulus and renal tubules were intact without abnormal pathological changes. In cisplatin group, renal interstitial hemorrhage, inflammatory cell infiltration, as well as cell edema and necrosis in different degrees were observed. GA treatment significantly improved the above pathological changes. Meanwhile, we also found the preferable improvement of pathological injury in TRPM2-KO mice than TRPM2-WT mice after cisplatin. The effect of GA on renal pathological damage in TRPM2 knockout mice is better than that in TRPM2-WT mice. Tunel assay was also performed (Figure 4(b) and 4(d)), and the result indicated that there were almost no apoptotic cells in the kidney tissue of normal TRPM2-WT and TRPM2-KO mice, however, in cisplatin group, the percentage of apoptotic cells was significantly elevated. GA showed inhibitory effect on cell apoptosis in injured kidney tissues. Meanwhile, we also observed the reduction of apoptotic cells in renal tissues of TRPM2-KO mice than TRPM2-WT mice after cisplatin. Also, the inhibitory effect of GA on apoptotic cells of renal tissues of TRPM2-KO mice was stronger than TRPM2-WT mice. These results demonstrated that TRPM2 knockout is beneficial for renal injury repair, and GA showed the protective effect on cisplatin-induced renal injury, and this effect is potentially related to TRPM2. Pathologic changes and apoptotic cells in renal tissues of TRPM2-KO and TRPM2-WT mice after Ginkgolide A treatment. (a) H&E staining, the scale bar is 200 μM; (b) tunel staining, the scale bars are 200 μM and 50 μM. (c) Quantitative grading of renal injury; (d) the percentage of tunel-positive cells in renal tissues of TRPM2-KO and TRPM2-WT mice calculated by image pro-plus. *p < .05, **p < .01.
Effect of TWEAK/Fn14 pathway on acute renal injury in mice and its relationship with TRPM2
Many evidences have demonstrated the role of TWEAK/Fn14 in acute renal injury. In our study, we analyzed the relationship between TWEAK/Fn14 axis and TRPM2. We used western blot to analyze the difference expression of TRPM2 in kidney tissues of TRPM2 wild-type mice and knockout mice after cisplatin induction (Figure 5(a)). Cisplatin treatment led to the activation of TWEAK/Fn14, and result in the increased expression of TWEAK, Fn14, phospho-NF-κB p65 and phospho-IKKβ protein in cisplatin group in either TRPM2-WT mice or TRPM3-KO mice. This result indicated that cisplatin induction activated TWEAK/Fn14 pathway. In TRPM2 wild-type and knockout mice, we found that the activation of TWEK/Fn14 pathway in the kidney tissue of knockout mice was inhibited, and the expression of these proteins was significantly lower than that of TRPM2 wild-type mice. This result displayed that TRPM2 knockout could inhibit the TWEK/Fn14 pathway. Next, we continued to study whether the role of TWEAK/Fn14 pathway in cisplatin-induced renal injury is associated with TRPM2 (Figure 5(b)). To verify this purpose, TWEAK shRNA and Fn14 shRNA lentivirus plasmids were injected into TRPM2-WT and TRPM2-KO mice. After injection of plasmids, the expression of TWEAK and Fn14 was also determined. We found that TWEAK shRNA or Fn14 shRNA could decrease the expression of TWEAK and Fn14 proteins in renal tissues in both TRPM2-WT and TRPM2-KO mice, especially in TRPM2-KO mice. The expression of TWEAK/Fn14 pathway in cisplatin-induced TRPM2-WT or TRPM2-KO mice. (a) Expression of TWEAK/Fn14 pathway in TRPM2 wild-type and knockout mice with acute renal injury was analyzed by western blot. (b) The changes of TWEAK/Fn14 pathway in renal tissue after injection of TWEAK shRNA or Fn14 shRNA into TRPM2 knockout and wild-type mice. *p < .05, **p < .01.
Inhibition of TWEAK/Fn14 pathway provided the protective role in renal damage induced by cisplatin through TRPM2
As shown in Figure 6(a), oxidative stress cytokines MPO, SOD and MDA were measured after TWEAK shRNA or Fn14 shRNA. TWEAK shRNA or Fn14 shRNA injection could reduce the levels of MDA and MPO, and elevate the level of SOD in kidneys. In TRPM2 knockout mice, the improvement effect of TWEAK shRNA and Fn14 shRNA on these factors is more significant. Renal function markers BUN, Scr and CysC were also detected (Figure 6(b)). The significant inhibition of TWEAK shRNA and Fn14 shRNA in three indicators BUN, Scr and CysC was found. Compared with TRPM2 wild-type mice, the contents of BUN, Scr and CysC in TRPM2-KO mice decreased more significantly after lentivirus plasmid injection. Similarly, the detection results of calcium and potassium in kidney tissue showed that the expression of TWEAK and Fn14 was inhibited, and the concentration of potassium increased, while the concentration of calcium decreased (Figure 6(c)). After TWEAK shRNA and Fn14 shRNA, the contents of potassium and calcium in renal tissues of TRPM2-KO mice changed significantly. Protective effect of TWEAK/Fn14 pathway on renal injury in TRPM2-KO mice. The alteration of oxidative stress level (a), the improvement of renal function (b), as well as the levels of potassium and calcium (c) in renal tissues after treatment of TWEAK shRNA or Fn14 shRNA. (b) were also evaluated according to the instruction of manufacturer. *p < .05, **p < .01.
Further, H&E staining was also performed to observe the improvement of pathological renal injury after TWEAK shRNA and Fn14 shRNA (Figure 7(a)). TWEAK shRNA and Fn14 shRNA ameliorated the abnormal pathological changes of renal tissues, including renal interstitial hemorrhage, inflammatory cell infiltration, as well as cell edema and necrosis, especially in TRPM2-KO mice. Tunel staining (Figure 7(b)) also showed that TWEAK shRNA and Fn14 shRNA reduced the apoptotic cells in renal tissues after cisplatin induction. These results suggested that inhibition of TWEAK/Fn14 pathway provided the protective role in renal damage induced by cisplatin, and this effect is partially related to TRPM2. This result is consistent with the previous study. The pathological improvement and anti-apoptotic effect of inhibiting TWEAK/Fn14 pathway on cisplatin-induced renal injury is related to TRPM2. (a) H&E stain of renal tissues from TRPM2-WT and TRPM2-KO mice. The score of renal injury was graded according to the scoring rules. Scale bar is 200 μM. (b) The area of Tunel-positive cells in renal tissues of TRPM2-KO and TRPM2-WT mice was assayed and analyzed after lentivirus plasmids of TWEAK shRNA and Fn14 shRNA injection. The scale bars are 200 μM and 50 μM. *p < .05, **p < .01.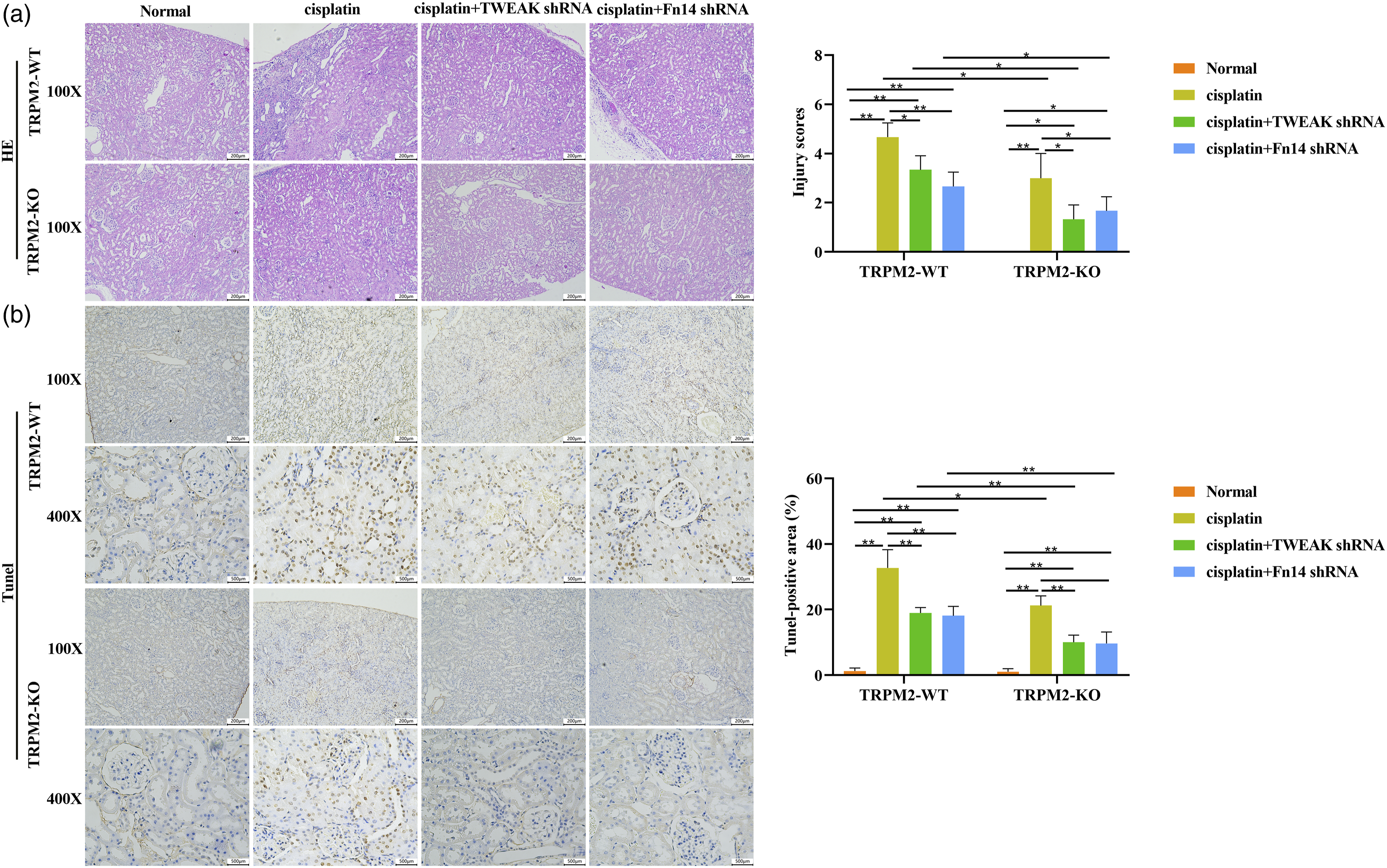
Ginkgolide A protects mice against cisplatin-induced renal damage through TWEAK/Fn14
We demonstrated whether the downstream mechanism of Ginkgolide A is potentially related to TWEAK/Fn14 pathway. In order to confirm this, we injected lentivirus-transfected plasmids into tail vein of TRPM2-WT mice after cisplatin injection. Next, the renal function parameters BUN, Scr and CysC were measured (Figure 8(a)). In previous studies, we have confirmed the role of GA and TWEAK/Fn14 pathway in cisplatin-induced renal injury. We found that after co-injection of GA and silencing plasmids of TWAEK shRNA or Fn14 shRNA, the cytokines (BUN, Scr and CysC) were in a lower level than GA group. However, co-administration of GA and TWEAK or Fn14 overexpressed plasmids could increase the levels of BUN, Scr and CysC in renal tissues. The pathological staining also showed a similar result, and GA + TWEAK and GA + Fn14 administration could aggravate the renal damage, while GA + TWEAK shRNA and GA + Fn14 shRNA could improve the renal injury (Figure 8(b)). Finally, we depicted the regulation of GA in TWEAK/Fn14 pathway in renal injury by western blot. As shown in Figure 8(c), after cisplatin induction, TWEAK and its receptor Fn14 were upregulated, while GA treatment could reduce the levels of these proteins. GA could inactivate TWEAK/Fn14 pathway in renal injury mice. In Figure 8(d), we observed the higher expression of TWEAK and Fn14 in GA + TWEAK group and GA + Fn14 group, and the lower expression of those in GA + TWEAK shRNA group and GA + Fn14 shRNA group. Ginkgolide A provides the protective effect of cisplatin-induced renal injury through TWEAK/Fn14 pathway. (a) Renal function parameters analysis; (b) H&E stain for pathological observation of renal tissues. The scale bar is 200 μM. (c) and (d) TWEAK and Fn14 proteins in TWEAK/Fn14 pathway were analyzed by western blot. *p < .05, **p < .01.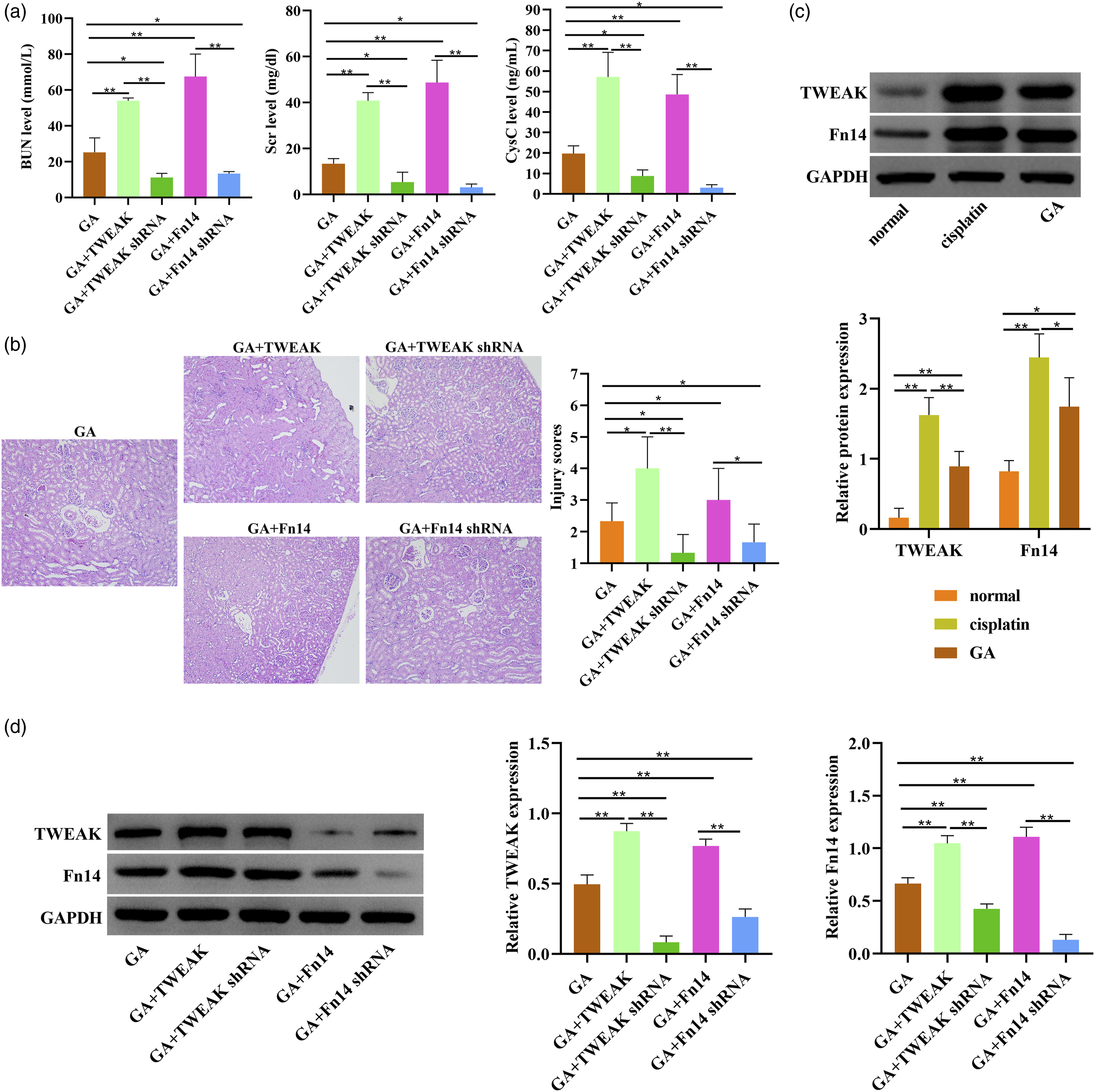
Discussion
The kidney is a dynamic organ, which is one of the main homeostasis systems in the body; they are vulnerable to different kinds of chemicals and drugs.32–34 Platinum complexes (such as cisplatin) are highly nephrotoxic and mainly concentrated in the kidney. 35 It is evident from the current study that cisplatin administration leads to renal injury (acute tubule cell necrosis, inflammation infiltration and glomerular congestion and atrophy). Besides, the biochemical parameters of renal function (blood urea nitrogen, serum creatinine and serum cystatin C) of mice in the cisplatin treatment group were significantly increased, reflecting the early damage of the filter barrier (such as porous capillary endothelium, glomerular basement membrane and visceral epithelial podocytes). Our results are consistent with the reported studies in mouse models.35,36 Ginkgolide A significantly inhibited the increase of biochemical parameters of renal function, which may be due to the improvement of glomerular filtration injury caused by cisplatin.
In addition, cisplatin also leads to renal oxidative damage and irreversible cell damage and necrosis and inflammation response. Excessive production of ROS by cisplatin will lead to the imbalance of antioxidants and lead to developed lipid peroxidation and antioxidant depletion. In our study, we also monitor renal redox parameters: SOD, MDA and MPO. These results agree with previous reports37,38 that cisplatin results in the elevated SOD, MDA and MPO and increased inflammation factors (IL-6, IL-1β and TNF-α) in renal tissues.
Ginkgolide A possesses many biological activities, including anti-inflammatory, anti-cancer, anti-anxiety, anti-atherosclerosis and anti-atherosclerosis, nerve, and liver protection. Only few studies have reported the protective role of Ginkgolide A in renal injury. A study reported the protective role of Ginkgolide A in hemorrhagic shock-induced renal injury in rats. 39 Additional evidence has revealed the protection of Ginkgolide A in sepsis-induced renal damage via upregulating miR-25 with Nox4 as the target. 14 We also demonstrated the role of this drug in cisplatin-induced renal injury. The result confirmed the improvement of Ginkgolide A in cisplatin-induced renal injury.
In this study, TRPM2 is also a research focus. We indicated whether TRPM2 can be used as a downstream molecule of GA, and the value of TRPM2 in TWEAK/Fn14 on renal injury. TRPM2 plays a minor role in acute kidney injury, 40 and displays the necessary roles in the sensitiveness to oxidative stress. 16 Besides, the recent study showed that inhibiting TRPM2 can effectively prevent ischemic acute renal injury by reducing oxidative stress, inflammation, and cell apoptosis. 41 So, whether TRPM2 acts as the downstream molecule of Ginkgolide A in renal tissues is still unknown. TRPM2-KO mice and TRPM2-wild type mice are applied in this study to verify the molecular mechanism of Ginkgolide A in acute renal injury. We found that the effect of GA on the repair of renal injury in TRPM2-KO mice is stronger than that in TRPM2-WT mice, which manifested by low levels of inflammatory factors, reduced oxidative stress, and enhanced renal function.
TWEAK/Fn14 pathway plays a central role in mediating renal damage.42,43 TWEAK plays the effect on intrinsic cells of kidney and inflammatory cells with potential pathophysiological correlation. 44 The role of TWEAK in tubular cells has been studied. In renal tubular cells, TWEAK induces the expression of inflammatory cytokines, down-regulates the expression of Klotho, and promotes cell apoptosis in the presence of sensitizers. A similar effect was also observed on glomerular mesangial cells. TWEAK is expressed in many tissues of the whole body. The high levels of TWEAK were found in heart, brain, pancreas, lung, intestine, ovary, vascular system and skeletal muscle, and low levels were found in liver and kidney. 22 These cells expressing Fn14 are sensitive to TWEAK. In addition, both TWEAK and Fn14 are highly expressed in renal injury, and targeting this system is beneficial to the study of different renal injury models. More importantly, lentivirus-mediated TWEAK and Fn14 knockdown protected against renal injury, inflammation and oxidative stress while restoring renal function in established cisplatin-induced acute renal injury models. We also confirmed whether the effect of TWEAK/Fn14 pathway on cisplatin-induced renal injury is related to TRPM2 through using TRPM2 knockout mice. The results showed that inhibition of TWEAK/Fn14 pathway was more effective in TRPM2-KO mice. Meanwhile, TWEAK/Fn14 pathway is a downstream pathway of Ginkgolide A in acute renal injury, which has been fully confirmed in our study.
In summary, we have demonstrated the following points in this study: (1) Ginkgolide A has a good effect on improving cisplatin-induced acute renal injury, inhibiting inflammatory reaction and excessive oxidative stress, improving renal function, and cisplatin-induced renal injury; (2) The repair effect of Ginkgolide A on renal injury is related to TRPM2, and Ginkgolide A could downregulate TRPM2 expression in renal tissues; (3) The effect of TWEAK/Fn14 pathway on cisplatin-induced renal injury is related to TRPM2. Inhibition of TWEAK/Fn14 pathway was more effective in TRPM2-KO mice; (4) TWEAK/Fn14 pathway is a downstream pathway of Ginkgolide A in acute renal injury, and Ginkgolide A could inhibit TWEAK/Fn14 pathway in cisplatin-induced renal injury model.
Ethical Statement
Ethical Approval
All protocols and procedures in this study were approved by the institutional Animal Care and Use Committee of the Yantai Affiliated Hospital of Binzhou Medical University and followed the Guide for the Care and Use of Laboratory Animals by the United States (US) National Institutes of Health (NIH, 2011).
Data availability statement
The data that support the findings of this study are not publicly available due to confidentiality but are available from the corresponding author.
Footnotes
Author contributions
All authors contributed to conceptualization. Haiyan He: material preparation, data curation and formal analysis, investigation, and methodology. Jun Ge: material preparation, data curation and formal analysis. Shaona Yi: material preparation, investigation, and methodology. Yuhong Wang: project administration and resources. Ye Liu: project administration and resources and software. Ying Liu: project administration. The writing of original draft was written by Xiaoming Liu. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Each author should have participated sufficiently in the work to take public responsibility for appropriate portions of the content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
