Abstract
Cisplatin is an effective anticancer drug used against a variety of cancers. The full therapeutic potential of cisplatin is often hampered due to concurrent development of various side effects in the hosts. Rutin, a naturally occurring bioflavonoid shows several pharmacological activities. It has been earlier reported by us that rutin and cisplatin in combination show better antitumor activity against murine ascites Dalton’s lymphoma. As cisplatin is given to cancer-bearing hosts only, the present study was undertaken to explore the histoprotective effect of rutin against some toxicities induced by cisplatin in tumor-bearing mice. Cisplatin treatment caused severe damages in tissue architecture such as degenerated hepatocytes with nuclear condensation and sinusoidal dilatation in the liver, glomerular deterioration, infiltration of cells, and tubular congestion in the kidney, and vacuolization of Sertoli cells or dense granules in the cytoplasm and damaged seminiferous tubules in the testes. In the rutin plus cisplatin combination-treated mice, all the abnormal tissue architectural features were decreased. Further, as compared to cisplatin treatment, combination treatment did not show any significant elevation in the liver functional biomarkers (serum aspartate aminotransferase, alanine aminotransferase and alkaline phosphatase) and renal functional biomarkers (serum urea and creatinine levels). The combination treatment reduced the sperm abnormalities also as compared to the cisplatin alone treatment. The in vitro hemolysis assay of red blood cells and scanning electron microscopy revealed that combination treatment lessened the cisplatin-induced hemolysis and abnormalities in RBCs. Thus, the present findings demonstrate that rutin has histoprotective ability against cisplatin-induced toxicities in tumor-bearing mice.
Introduction
Cancer is a grave human health-related concern responsible for the second leading cause of death worldwide. As per the facts and figures of the American Cancer Society (2018), the global cancer burden indicates that about one in every seven deaths worldwide is caused by cancer. 1 Cisplatin is a cancer chemotherapy drug commonly used against a wide spectrum of cancers, 2 but its use is limited due to the development of various severe toxicities in the hosts such as nephrotoxicity, hepatotoxicity, hematotoxicity. 3 The hepatotoxicity of cisplatin has been reported to arise from the induced generation of reactive oxygen species (ROS) in the epithelial cells of the sinusoids, particularly in the mitochondria. 4 –6 The accumulation of high concentrations of cisplatin in the kidneys causes nephrotoxicity through apoptosis and necrosis, vascular factors, and inflammation of the tubules. 7 –9 For cisplatin-induced nephrotoxicity, numerous mechanisms such as mitochondrial dysfunction, inflammation, DNA damage, oxidative stress and apoptosis have suggested that the free radical scavengers and the antioxidants agent can prevent cisplatin-induced nephrotoxicity. 10
Rutin is a common dietary flavonoid present in common fruits and vegetables. 11 It belongs to a class of flavonoids called flavonols 12 and exhibits a wide range of biological activities such as antitumor, antioxidant, anti-diabetic, nephroprotective, hepatoprotective, hematoprotective effects. 13,14 Several studies have also reported that rutin exhibits hepatoprotective effects against various drug-induced toxicities. 15 –17 The protective effect of rutin on carfilzomib-induced renal injury via the inhibition of oxidative stress and inflammation has been reported. 18 Further, the ameliorative effect of rutin against isoniazid-induced alterations in certain hematological and biochemical parameters of Albino rats has been reported. 19 The protective ability of rutin against cisplatin-induced hematotoxicity in tumor-bearing mice has also been demonstrated. 20
As the anticancer drugs are given to cancer bearing hosts only, it is of prime significance to assess the modulatory effects of rutin on cisplatin-induced toxicological parameters in the same cancer-bearing hosts. Thus, considering the pharmacological importance of rutin, the present study was conducted to assess further the protective effects of rutin against other toxicities parameters induced by cisplatin in tumor-bearing mice.
Materials and methods
Chemicals
Rutin (CAS No. 153-18-4, rutin hydrate) with ≥94% purity was purchased from Sigma (St. Louis, MO, USA). Cisplatin solution (1mg/ml of 0.9%, NaCl) was obtained from Biochem Pharmaceutical Industries, Mumbai, India. Hematoxylin (Delafield’s) and eosin (2% w/v) were purchased from HiMedia Laboratories Pvt. Ltd., Mumbai, India.
Animals and tumor maintenance
Inbred Swiss albino mice colony is being maintained in a standard laboratory condition with food pellets and drinking water ad libitum. Ascites Dalton’s lymphoma (DL) tumor was maintained in vivo in 10–12 weeks old mice of both sexes as per details described earlier. 13 The maintenance, use of the mice and the experimental protocols have been approved by the Institutional Animals Ethics Committee (No. NEC/IEC/2018/005, dated October 01, 2018) of the North-Eastern Hill University, Shillong, India.
Drugs treatment schedule
The dose and the schedule of drug treatment were based on our previous reports.
13,20
Tumor-transplanted animals were divided into four groups keeping 10 mice (5 males and 5 females) per group and treated as follows: Group 1: mice served as tumor-bearing control received drug vehicle (i.e.phosphate-buffered saline, pH 7.4) only. Group 2: mice were treated with rutin (30mg/kg body weight, i.p.) on the 8th and 10th-day post-tumor transplantation Group 3: mice were treated with a single therapeutic dose of cisplatin (8mg/kg body weight, i.p.) on the 10th day post-tumor transplantation. Group 4: mice were treated with rutin on the 8th and 10th day followed by a single dose of cisplatin on the 10th day post-tumor transplantation keeping a gap of 6 h post rutin treatment.
Organosomatic index (OSI)
The weight of mice from different treatment groups was weighed on Docbel Braun common balance (capacity 500 g). Then, mice were sacrificed and dissected out the fresh liver, kidney, and testes and weighed (g) in the Systronic electronic single pan balance (sensitivity 0.001 g). The organosomatic index was calculated as per the method described by Gupta et al. 21 using the following formula:
Histopathological studies
It has been earlier reported that the pathological features in tissues are developed after 5 days of cisplatin treatment. 22 On the 5th day post-treatment, the liver, kidney and testes of mice in different experimental groups were collected and processed following established steps for histopathological studies. The histological sections were thoroughly examined under a light microscope (Leica) to analyze changes in the cellular organization in tissues of the hosts under different experimental groups. For a comparative analysis, the pathological damages in the tissue sections were graded as (+) low/minimal (< 10%), (++) mild/moderate (< 30%), (+++) high (< 50%) and (++++) very high/severe (> 50%).
Liver function test (LFT) and renal function test (RFT)
The blood sample was collected from the mice in the same group used for histological studies by retro-orbital venous sinus bleeding and serum was separated. The changes in the serum levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP) were assessed for liver function and serum urea and creatinine level for renal function using Clinical Chemistry Analyzer (SYNERGY BIO-1904C).
Sperm abnormality analysis
The sperm abnormality analysis under different groups was performed as described by Wyrobek and Bruce. 23 Briefly, the male mice were sacrificed in different groups and the cauda epididymis was removed and then minced into pieces and kept for 20 min undisturbed. The spermatozoa were spread on a clean slide, air-dried, and then stained with 1% aqueous eosin-Y. For the sperm abnormalities analysis in terms of head and tail shapes, 500 sperms from each mouse were examined.
Scanning electron microscopy (SEM) of red blood cells (RBCs)
Blood was collected from the eye orbit of mice using a capillary tube and processed for scanning electron microscopy of RBCs as per details described by Kalita et al. 24
In vitro hemolysis assay
In vitro hemolysis assay was carried out as per the method described by Deepika et al. 25 To test hemolytic activity, 0.2 mL of diluted RBC suspension was added to 0.8 mL of rutin, cisplatin, and rutin plus cisplatin solutions in PBS at different concentrations. To obtain positive and negative controls, RBCs were suspended in deionized water and PBS, respectively. The samples were incubated in a static condition at room temperature for 3 h, followed by centrifugation at 10000×g for 10 min. The absorbance of hemoglobin in the supernatant was measured at 545 nm, with 630 nm as a reference, in a spectrophotometer (Bio-Rad, USA) at 37°C. The assay was repeated thrice and the percentage of hemolysis was calculated using the following equation:
Statistical analysis
The results were expressed as the mean ± standard deviation (SD). Statistical significance was determined by one-way analysis of variance (ANOVA). The difference among multiple groups was analyzed by a post hoc (Tukey test). The statistical significance of difference was taken as P-value ≤ 0.05.
Results
Organsomatic index
Organosomatic indices obtained for liver, kidney, and testes in the control and rutin treated mice were almost comparable and equivalent, whereas cisplatin-treated mice showed a significant increase in OSI for all the organs. The OSI of the organs of the mice treated with rutin plus cisplatin was significantly reduced as compared to that in the cisplatin-treated mice (Table 1).
The organosomatic index (OSI) values from mice under different treatment conditions.

* P ≤ 0.05 as compared to the control group and #P ≤ 0.05 as compared to cisplatin treatment. The number of mice analyzed was 5 (i.e., n = 5).
Histopathology of liver
Histopathology of the liver from control and rutin-treated mice exhibited proper central vein and regular arrangement of hepatocytes (Figure 1a, b). Cisplatin treatment developed congestion in a central vein, hepatocytes damage, and dispersed sinusoidal distortion (Figure 1c). On the other hand, mice treated with the combination of rutin and cisplatin revealed minor alterations/damages in central vein and hepatocytes as compared to cisplatin treatment demonstrating recovery in the impaired hepatocytes (Figure 1d).
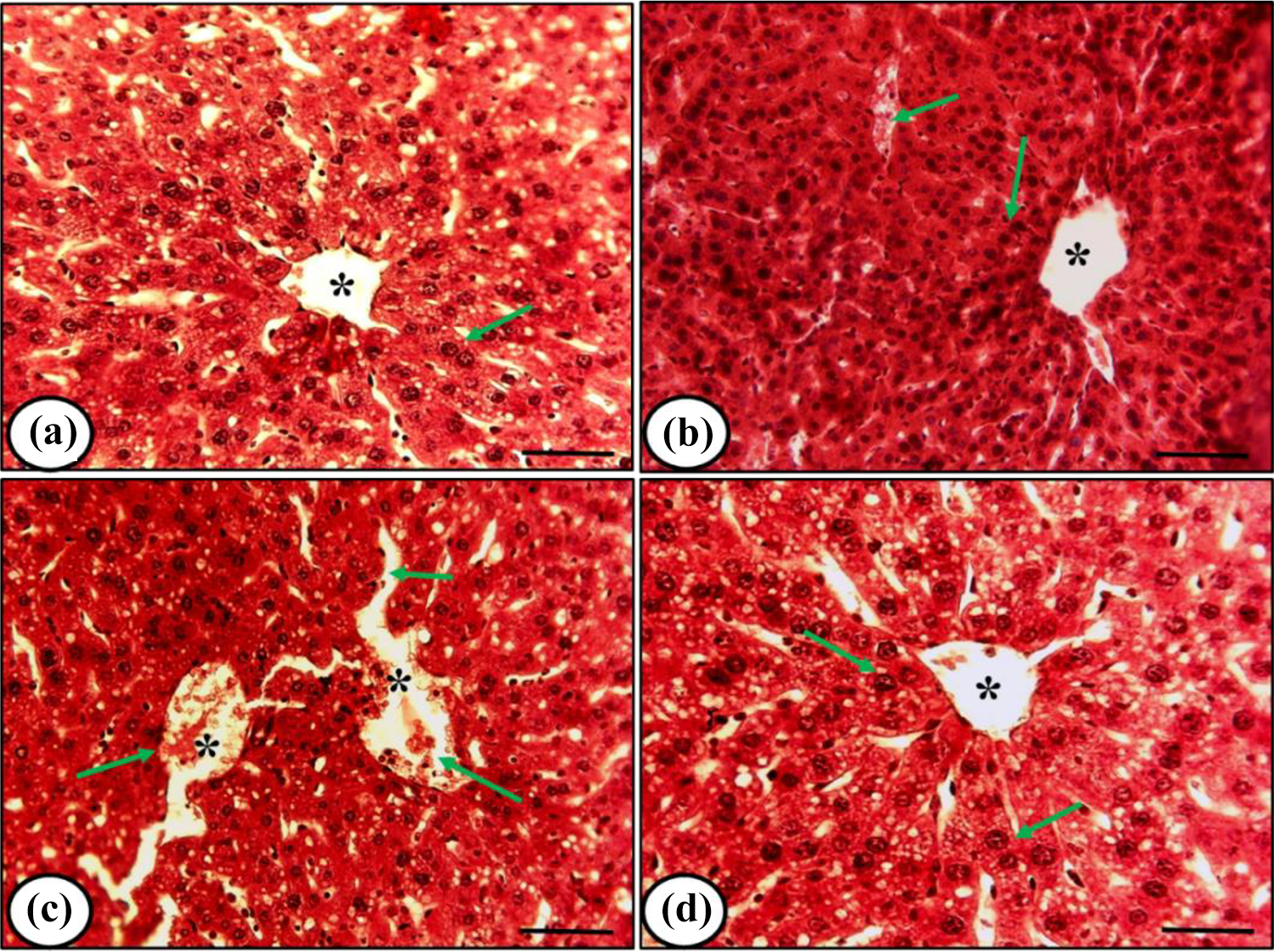
Representative histomicrographs of liver from mice under different treatment conditions. (a), (b), (c) and (d) denote control, rutin alone treatment, cisplatin alone treatment, and rutin plus cisplatin treatment respectively. Control and rutin alone treatment showed normal hepatocytes arrangement (arrow) and proper central vein (asterisk). Cisplatin alone treatment developed damage in hepatocytes (arrow) and congestion in a central vein (asterisk). Rutin plus cisplatin treatment led to a recovery in damaged hepatocytes (arrow). Scale bar= 20μm.
Liver function test (LFT)
The level of liver function biomarkers such as ALT, AST and ALP did not change after the rutin treatment. However, cisplatin treatment caused a significant elevation in these enzymes. The combination treatment of mice showed a significant reduction in these enzymatic activities as compared to cisplatin alone treatment (Figure 2).
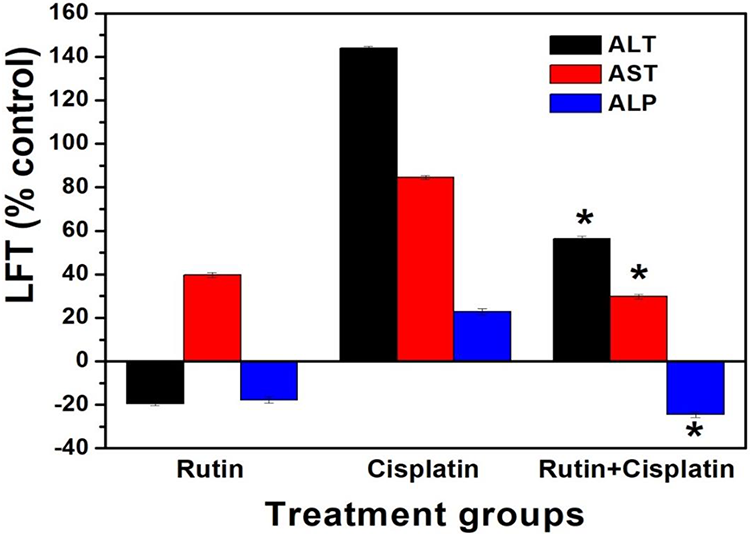
Changes in liver functional biomarkers in mice under different treatment conditions. Results are expressed as mean ± S.D., n = 3. ANOVA, *P ≤ 0.05 as compared to cisplatin alone treatment. ALT= Alanine aminotransferase, AST= Aspartate aminotransferase, and ALP= Alkaline phosphatase.
Histopathology of kidney
Histological features of the kidney from control and rutin treated-mice indicated intact epithelial cells and normal renal glomeruli and tubules (Figure 3a, b). However, cisplatin treatment caused damages in renal cells, infiltration of cells, tubular congestions, and glomerular degeneration indicating severe nephrotoxicity in the mice (Figure 3c). These damages in renal cells and glomerular features were reduced in the kidney of mice treated with rutin plus cisplatin signifying the nephroprotective ability of rutin (Figure 3d).

Representative histomicrographs of the kidney from mice under different treatment conditions. (a), (b), (c) and (d) denote control, rutin alone treatment, cisplatin alone treatment and rutin plus cisplatin treatment respectively. Control and rutin alone treatment showed normal intact glomerular (arrow) and tubular arrangement. Cisplatin treatment resulted in congested vein and degenerated glomeruli (arrow). After rutin plus cisplatin treatment fewer damages in glomerular (arrow) and tubular cells in the kidney were observed. Scale bar= 20μm.
Renal function test (RFT)
Rutin treatment did not show much variation in the serum urea and creatinine level. An increase in the level of serum urea and creatinine were observed after cisplatin treatment. However, the mice treated with the combination of rutin and cisplatin showed a substantial reduction in the levels of serum urea and creatinine levels (Figure 4).

Changes in the level of renal function biomarkers in mice under different treatment conditions. Results are expressed as mean ± S.D., n = 3. ANOVA, *P ≤ 0.05 as compared to cisplatin alone treatment.
Histopathology of testes
Histological features of testes from mice in the control and rutin treated mice showed distinct spermatogenic cells, Leydig cells in the interstitial tissue, and seminiferous tubules (Figure 5a, b). Cisplatin treatment showed damages in seminiferous tubules, degeneration of Leydig cells, and vacuolization in Sertoli cells or dense granules in the cytoplasm (Figure 5c), whereas the mice treated with rutin and cisplatin combination exhibited a reduction in vacuolated tubules, damages in spermatogenic cells and seminiferous tubules (Figure 5d).
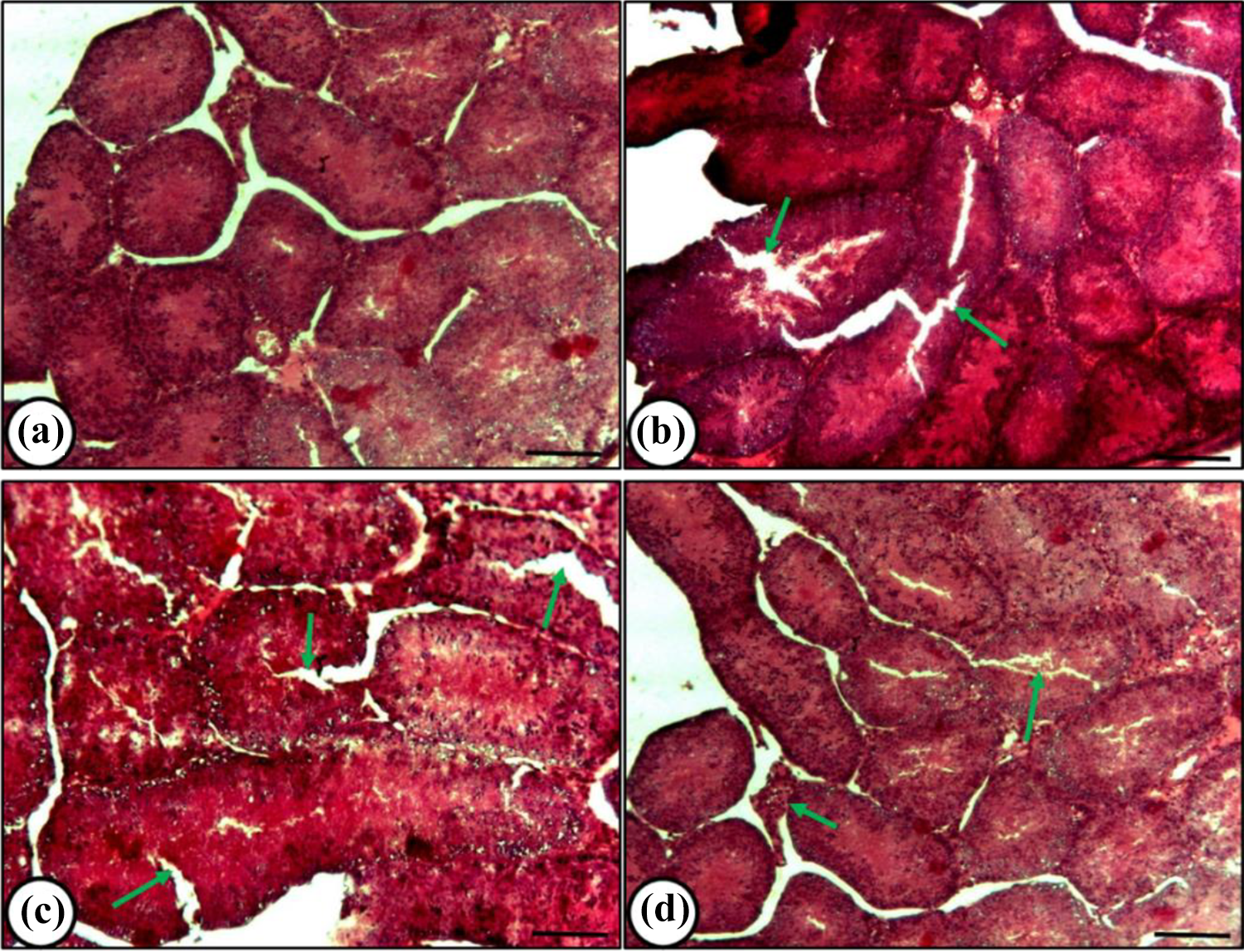
Histological features of testes from mice under different treatment conditions. (a) Control-showing well-defined seminiferous tubules (arrow); (b) Rutin treatment- showing almost normal features (arrow); (c)-Cisplatin alone treatment-showing damaged tubules with vacuolated Sertoli cells (arrow); (d) Rutin plus cisplatin treatment-showing reduced vacuolated tubules (arrow) with less deranged features. Scale bar= 20μm.
Various pathological damages observed in the liver, kidney, and testes under different treatment groups were graded (Table 2).
Grading of pathological damages in the liver, kidney, and testes of tumor-bearing mice under different treatment groups. The number of mice analyzed was 3 (i.e., n = 3).

(−) Nil, (+) Low/Minimal (< 10%), (++) Mild/moderate (< 30%), (+++) high (< 50%), (++++) very high/severe (> 50%).
Sperm abnormality analysis
The sperm abnormality analysis showed that cisplatin treatment developed the highest abnormalities as compared to other groups (Figure 6, Table 3). The swollen head, amorphous head, coiled tail, headless, hookless head, banana-like head, beaked head, bent tail, incorrect head-neck joining and balloon-like head were major abnormalities observed in the sperms (Figure 6). However, the mice treated with the combination of rutin and cisplatin significantly abridged these sperm abnormalities as compared to the cisplatin alone treatment (Table 3).
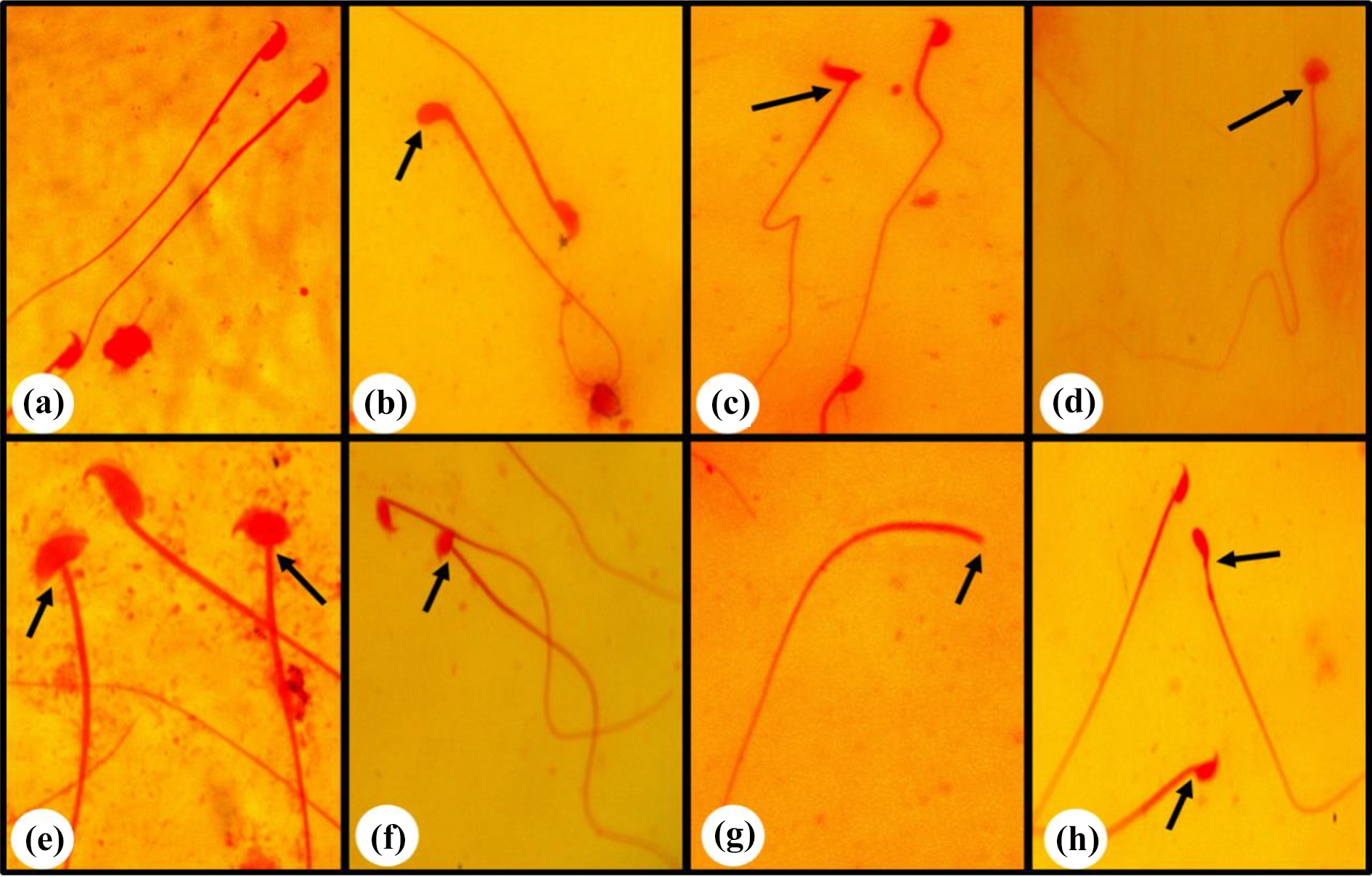
Representative photographs of normal and different abnormal sperms observed. (a) normal, (b) hookless head, (c) incorrect head-neck joining, (d) balloon-like head, (e) swollen head, (f) amorphous head, (g) headless and (h) banana head.
Quantitative analysis of various types of sperm abnormalities developed in mice under different treatment conditions.

* P ≤ 0.05 as compared to cisplatin alone treatment and #P ≤ 0.05 as compared to the control group.
Morphological abnormalities in RBCs
The different types of morphological abnormalities observed in RBCs under different treatment conditions were echinocytes (occurrence of evenly toothed projections), acanthocytes (thorn-like cells), elliptocytes (elliptical-shaped cells), scalloping, and burr type (rough edge cells) (Figure 7). The mean percent normocytes (RBCs with normal shape and size) in the control group were approximately 86%, whereas, in the rutin alone, cisplatin alone and rutin plus cisplatin-treated group, it was about 59, 46 and 64% respectively. As compared to control, the number of abnormalities in RBCs was significantly increased in the cisplatin alone treatment group. There was a significant reduction in the occurrence of abnormal RBCs in the tumor-bearing mice treated with a combination of rutin and cisplatin as compared to the cisplatin alone treatment (Figure 8).
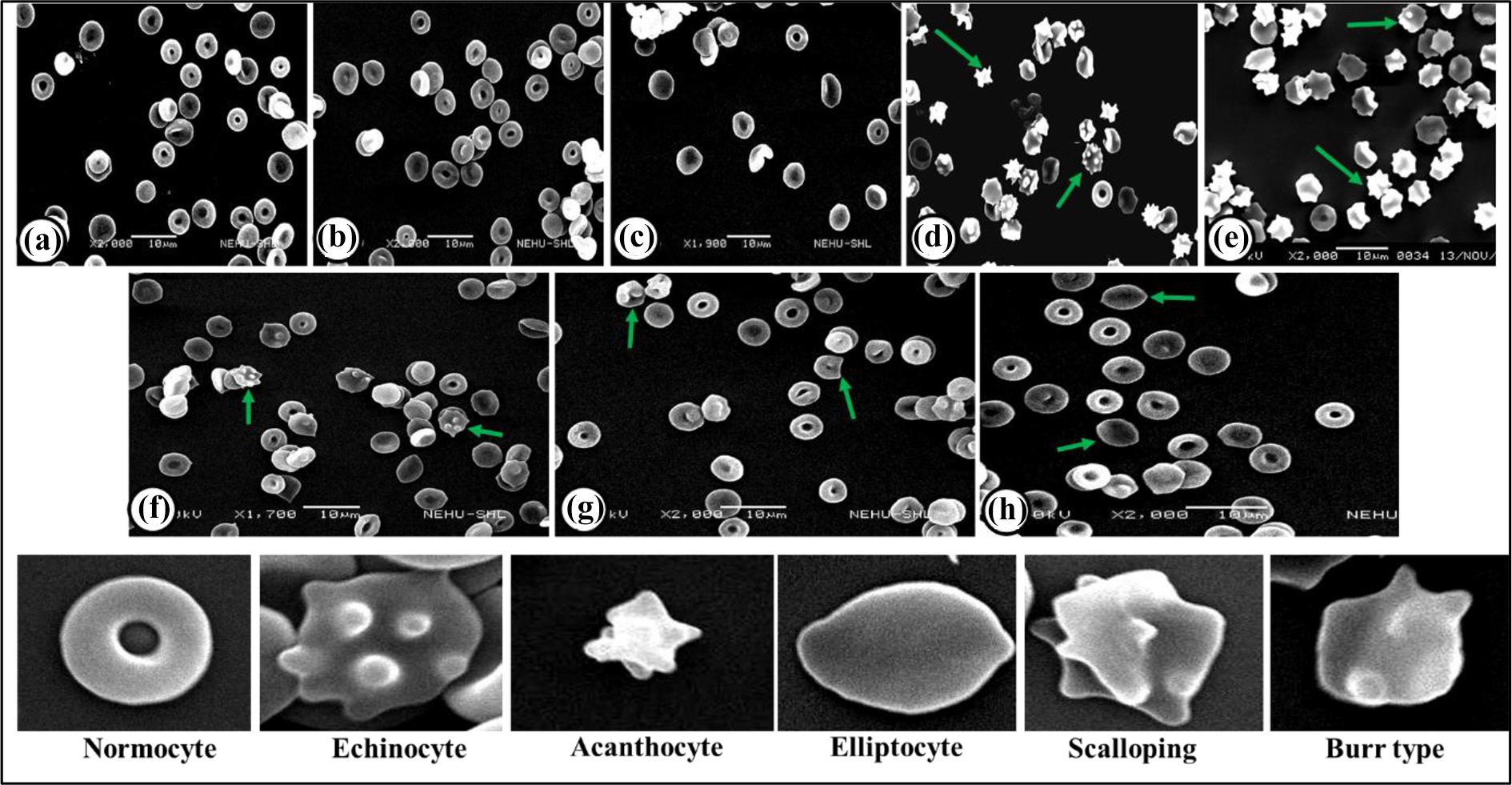
Scanning electron micrographs of RBCs showing various types of abnormalities (arrows) under different treatment conditions. Control (a); Rutin alone treatment for 24 h (b) and 96 h (c); Cisplatin alone treatment for 24 h (d) and 96 h (e); Rutin plus cisplatin treatment for 24 h (f), 48 h (g) and 96 h (h).The lower row shows an enlarged view of normal and different abnormalities in RBCs.

Comparative analysis of total abnormalities developed in RBCs under different treatment conditions. Results are expressed as mean ± S.D., n = 3. ANOVA, *P ≤ 0.05 as compared to the control group; #P ≤ 0.05 as compared to cisplatin treatment.
Hemolytic assay
The hemolytic activity was determined by the release of hemoglobin upon the treatment of RBCs with rutin alone, cisplatin alone, and rutin plus cisplatin combination (Figure 9a). The hemolytic percentage obtained below 5% at the experimental concentration was considered as the safest dose. The hemolysis at all the tested concentrations with rutin alone was found to be <5% except at 150 µg/ml (6.03%). In the cisplatin alone treatment, except at 4µg/ml and 7.5 µg/ml, all the tested concentrations showed a significant increase in the hemolytic percentage i.e. 12.69, 11.30, 15.45 and 18.47% for 15µg/ml, 45µg/ml, 75µg/ml and 150µg/ml, respectively. However, RBCs treated with the combination of rutin and cisplatin showed a significant reduction in the hemolytic activity at all the tested concentrations as compared to the cisplatin alone treatment indicating that rutin in combination with cisplatin reduces the hemolytic effects of cisplatin (Figure 9b).
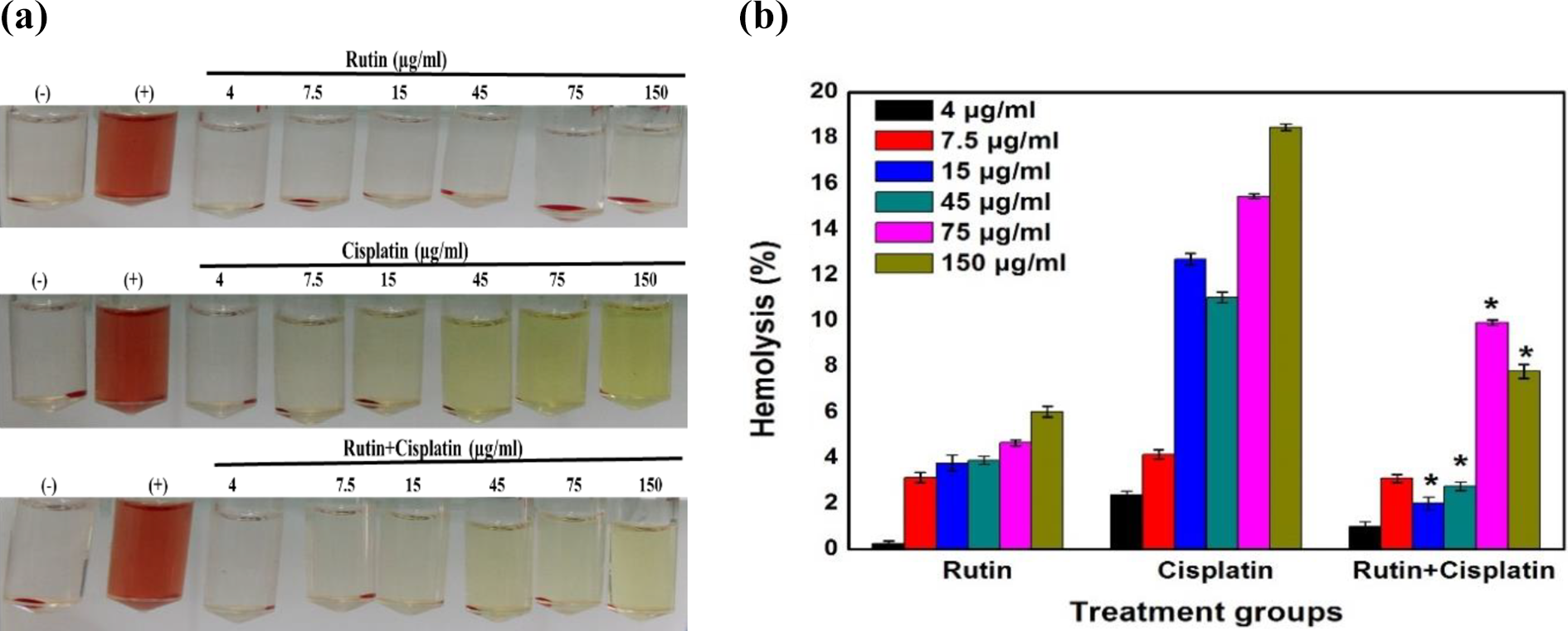
Effect of rutin, cisplatin, and rutin plus cisplatin on RBC hemolysis. (a) RBCs suspensions exposed to rutin, cisplatin, and rutin plus cisplatin at different concentrations (4, 7.5, 15, 45, 90, and 150 μg/ml) for 3 h followed by centrifugation. (b) Comparative percent hemolysis of RBCs incubated with drugs at different concentrations ranging from 4 to 150 μg/mL for 3 h.Results are expressed as mean ± S.D., n = 3. ANOVA, *P ≤ 0.05 as compared to cisplatin alone treatment.
Discussion
Phytochemicals are gaining much attention in the development of risk-free chemo-preventive agents against various diseases including cancer. In an attempt to overcome diverse side effects of anticancer drug cisplatin in the hosts, baicalein, a naturally occurring bioflavonoid and ascorbic acid have been used in combination with cisplatin. 26,27 In the present study, rutin, a kind of flavonoid was used in combination with cisplatin to investigate its likely modulatory effect on the cisplatin-induced toxicities in tumor-bearing mice as the cisplatin is given to cancer-bearing hosts only.
Organosomatic indices may help in signifying the health status of the mice. 21 The findings on the organosomatic index (OSI) of the liver, kidney and testes in cisplatin-treated mice showed a significant increase in the OSI values (Table 1) which may indicate that cisplatin treatment caused pathological damages in these tissues. However, the OSI for these tissues in the combination treatment was significantly reduced as compared to the cisplatin alone treatment showing almost normal values (Table 1) which suggests that rutin in combination with cisplatin has protective/restoring effects. The protective effects of rutin against various drug-induced toxicities in the hosts have been reported. 28 –30 Khan et al. 31 demonstrated that treatment of rats with rutin significantly restores the increased absolute liver and the relative liver weight caused by carbon tetrachloride (CCl4) treatment. Kamel et al. 32 reported that cisplatin treatment caused a significant elevation in the relative kidney weight, whereas rutin in combination with cisplatin ameliorated the cisplatin-induced alterations in the relative kidney weight. The present findings from OSI also revealed that rutin has histoprotective ability against pathological damages induced by cisplatin alone treatment.
The histopathological observations revealed that cisplatin alone treatment caused maximum alterations in the architecture of liver such as congestion in a central vein, hepatocytes damages, and dispersed sinusoidal distortion, whereas in combination treatment of mice with rutin plus cisplatin showed a noteworthy restoration in the architecture of the liver (Figure 1). The various studies on the protective ability of rutin against CCl4-induced liver damage in mice and rats have been reported showing that rutin has hepatoprotective action. 31,33 Reddy et al. 34 reported that rutin can improve hepatic function by regenerating the histoarchitecture and functioning of the liver in acetaminophen-induced liver damage due to the antioxidant and free radical scavenging properties of the rutin. A study on 5-Fluorouracil (5-FU) -induced hepatotoxicity in rats reported that rutin and quercetin treatment lessen the liver damage caused by 5-FU in rats by the antioxidant effects of quercetin and rutin. 30
Liver injury causes membrane damage or necrosis, which allows intracellular enzymes to be detected in serum and enzyme levels such as ALT, AST, and ALP are often used as biomarkers to assess hepatic damage. Higher concentrations of these enzymes in the serum indicate that the hepatic membrane’s functional integrity has been lost. The results on the liver function test revealed that cisplatin alone treatment caused a significant increase in serum ALT, AST, and ALP levels. However, the mice treated with rutin plus cisplatin showed a reduction and restoration in these enzyme levels as compared to cisplatin alone treatment (Figure 2). Rutin is a powerful free radical scavenger and has been reported to possess the ability to protect the liver from the adverse effects. 17,18,35 The pre-treatment with rutin has been reported to significantly reduce the activity of serum ALT, AST, and ALP and improve the histological signs of acute liver damage induced by CCl4 33 and acetaminophen. 34 Abdel-Ghaffar et al. 16 demonstrated that the activities of serum AST, ALT and ALP were remarkably elevated in isoniazid-treated rats and the animal group treated with rutin 1 hour before isoniazid administration caused a significant reduction in the activities of these enzymes. Thus, our results are in quite an agreement with findings from other earlier studies supporting the hepatoprotective ability of rutin when used in combination with the cisplatin.
The histological features of the kidney revealed normal histoarchitectural features with intact epithelial cells and normal renal glomeruli and tubules in control and the rutin alone treated mice. However, cisplatin treatment caused severe alterations in histoarchitecture of the kidney such as infiltration of cells, tubular congestions and glomerular degeneration, damages in renal cells, whereas treatment of mice with rutin plus cisplatin showed improvement in the histopathological alterations and fewer damages in glomerular and renal tubular cells, thus, indicating the nephroprotective effect of rutin (Figure 3d). It has been reported that rutin mitigates cisplatin-induced renal inflammation and apoptosis via reducing caspase-3 activity and activating nuclear factor kappa-B (NF-κB). 36 Other studies also sustain the nephroprotective effects of rutin against various drugs/chemicals-induced toxicities in the hosts. 29,37 –40
Renal dysfunction and nephrotoxicity induced by cisplatin are characterized by a reduction in renal function that leads to an increase in serum creatinine and blood urea levels. 38,39 Our results on the renal function test revealed that cisplatin alone treatment caused a markedly increase in the creatinine and urea levels in serum. However, in the rutin plus cisplatin-treated group, the levels of these renal functional biomarkers were significantly restored as compared to the cisplatin alone treatment indicating the nephroprotective properties of rutin (Figure 4) and supports both histological observations and OSI findings discussed above.
The histological features of testes revealed that cisplatin alone treatment caused severe damages in seminiferous tubules and form vacuolated tubules whereas control and rutin alone treated mice did not show many changes in the architecture of testes. However, the rutin plus cisplatin-treated mice reversed the histopathological damages caused by cisplatin alone treatment and showed restoration in the histoarchitecture of the testes (Figure 5d). It has been demonstrated that oxidative stress caused by free radicals plays an important role in the development of cisplatin-induced testicular damage and the use of rutin in combination with cisplatin reversed the cisplatin-related pathological effects on testicular tissue. 41 Aksu et al. 42 reported that cisplatin treatment induced testicular degeneration and increased oxidative stress and apoptosis in adult male rats whereas rutin plus cisplatin treatment ameliorated the cisplatin-induced reproductive damage in adult male rats by the reduction in necrosis and degeneration of seminiferous tubules. Several other researchers have also reported the protective effects of rutin against different drug-induced testicular toxicities. 28,43 –46 Altogether, the findings on histological studies support the previous reports that rutin plays a crucial histoprotective role when used in combination with cisplatin and has the potential to lessen the pathological damages caused by cisplatin (Table 2).
Longchar and Prasad 22 have demonstrated that cisplatin treatment causes a significant increase in sperm abnormalities in tumor-bearing male mice and acts as potential germ cell mutagen. In the present study also, the results from sperm abnormality analysis showed that cisplatin alone treatment raised the abnormalities in sperm morphology (Figure 6, Table 3). As compared to cisplatin alone treatment, tumor-bearing mice treated with rutin plus cisplatin showed a significant reduction in abnormal sperms indicating the testicular protective effect of rutin (Table 3). These findings are in good agreement with the earlier reports 41 –43
Longchar and Prasad 27 reported that cisplatin alone treatment caused the development of various hematotoxic features and morphological abnormalities in erythrocytes in mice bearing ascites Dalton’s lymphoma. In the present study also, cisplatin alone treatment significantly raised the number of abnormal RBCs (Figure 7). However, combination treatment of mice with rutin plus cisplatin lessens the abnormalities in the RBCs as compared to cisplatin alone treatment indicating the rutin’s hematoprotective ability (Figures 7 and 8). The present findings strengthen our previous findings on the protective ability of rutin against cisplatin-induced hematotoxicity. 20 The findings from the in vitro hemolysis analysis also support the morphological changes in RBCs that cisplatin alone treatment produced significant hemolysis in RBCs compared to the control (Figure 9a). The percent of hemolysis in the combination treatment with rutin plus cisplatin revealed a significant reduction in the hemolysis values as compared to cisplatin alone treatment, thus, indicating anti-hemolytic properties of rutin(Figure 9b). Thus, the assessment of these toxicity parameters further establishes that rutin has the protective ability against cisplatin-induced toxicities in the cancer-bearing hosts.
In conclusion, the overall findings from these toxicity parameters study showed that cisplatin alone treatment causes severe distortions in histoarchitecture of liver, kidney and testes and also induces a high percentage of abnormalities in RBCs and sperms (Figure 10). However, the combination treatment of tumor-bearing mice with rutin plus cisplatin leads to the restoration in histoarchitecture alterations caused by cisplatin. This combination treatment also exhibited minimal abnormalities in RBCs and sperms as compared to cisplatin alone treatment (Figure 10).
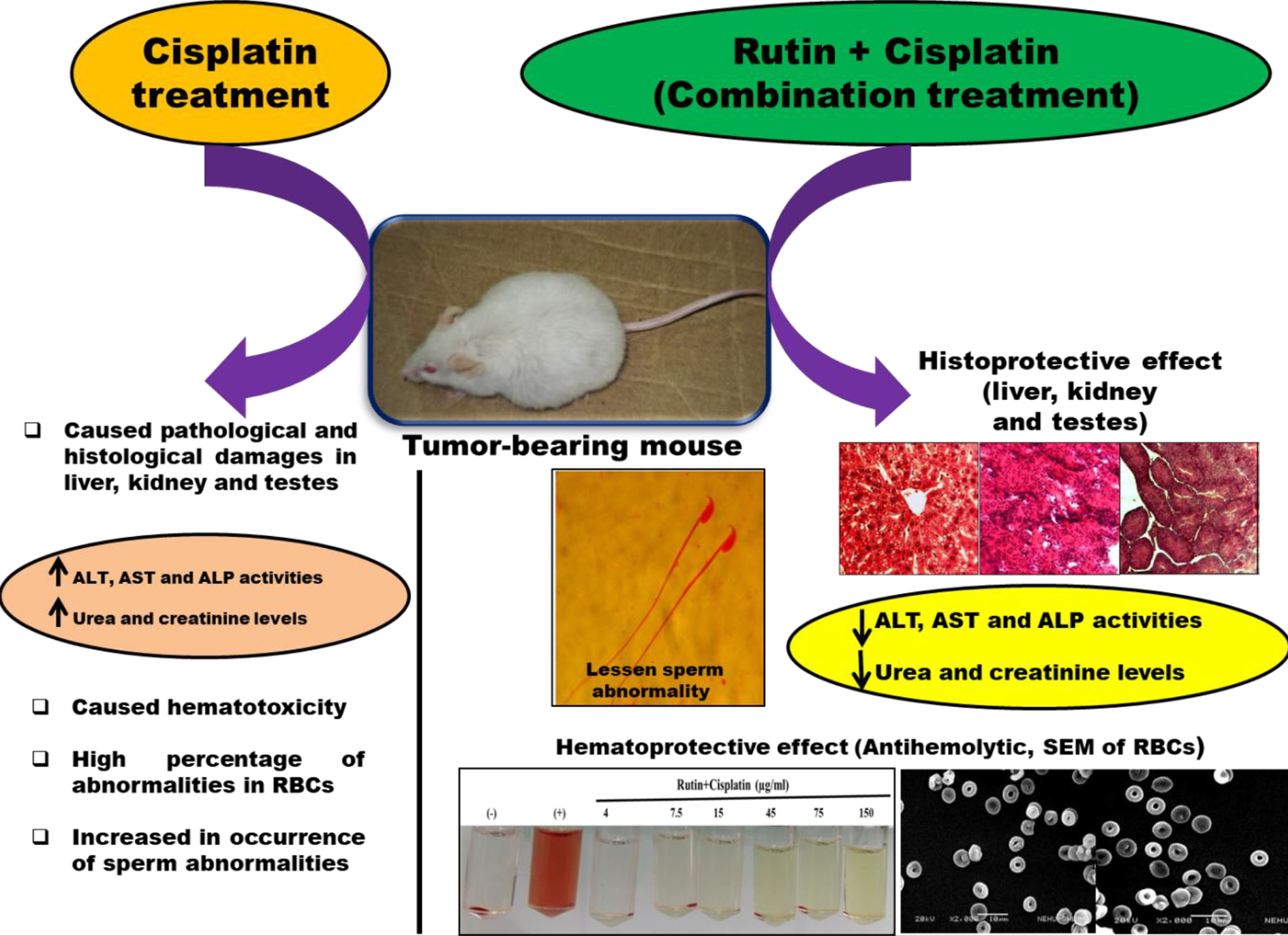
Summary of the various toxicity related changes observed after the cisplatin alone and rutin plus cisplatin combination treatment of mice.
Thus, the rutin plus cisplatin treatment approach could be used in chemotherapy to prevent the toxicities induced by cisplatin. Further, the details of the molecular mechanisms behind the protective ability of rutin in combination with cisplatin in the hosts may need further exploration.
Footnotes
Acknowledgments
We gratefully acknowledge the research facilities provided by the Department of Zoology, North-Eastern Hill University, Shillong in the completion of the present study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial support for this research work was provided by the University Grants Commission, New Delhi in the form of a non-NET fellowship to Rajesh Prasad.


