Abstract
Despite the various and newly developed chemotherapeutic agents in recent years, cisplatin is still used very frequently as a chemotherapeutic agent, even though cisplatin has toxic effects on many organs. The aim of our study is to show whether ghrelin reduces the liver toxicity of cisplatin in the rat model. Twenty-eight male Sprague Dawley albino mature rats were chosen to be utilized in the study. Group 1 rats (n = 7) were taken as the control group, and no medication was given to them. Group 2 rats (n = 7) received 5 mg/kg/day cisplatin and 1 ml/kg/day of 0.9% NaCl, Group 3 rats (n = 7) received 5 mg/kg/day cisplatin and 10 ng/kg/day ghrelin, Group 4 rats (n = 7) received 5 mg/kg/day cisplatin and 20 ng/kg/day ghrelin for 3 days. Glutathione, malondialdehyde (MDA), superoxide dismutase (SOD), plasma alanine aminotransferase (ALT) levels, and liver biopsy results were measured in rats. It was determined that, especially in the high-dose group, the MDA, plasma ALT, and SOD levels increased less in the ghrelin group as compared to the cisplatin group, and the glutathione level decreased slightly with a low dose of ghrelin, while it increased with a higher dose. In histopathological examination, it was determined that the toxic effect of cisplatin on the liver was reduced with a low dose of ghrelin, and its histopathological appearance was similar to normal liver tissue when given a high dose of ghrelin. These findings show that ghrelin, especially in high doses, can be used to reduce the toxic effect of cisplatin.
Introduction
Cisplatin is a highly effective agent used in chemotherapy, though it is also a cytotoxic drug with varying side effects that can be very serious. Cisplatin can be used in high doses in aggressive cancers; thus, its toxicity increases accordingly. 1 The most critical side effect of chemotherapeutic agents is diffuse cytotoxicity. A wide variety of these side effects especially include nephrotoxicity and hepatotoxicity, which can have a significant effect on vital functions. Thus, they are the most important factors limiting the clinical use of cisplatin.2,3 Medical treatments try to reduce mortality and morbidity, but it still seems to be a significant problem. 3 Despite its powerful antineoplastic effectiveness, hepatotoxicity limits cisplatin’s therapeutic application due to its toxic side effects during therapy such as nephrotoxicity, neurotoxicity, ototoxicity, myelosuppression.4–8 The majority of studies looking at the connection between cisplatin dosage and toxic side effects have found a link between cisplatin total dose and toxic side effects. This is most evident in the liver and hematopoietic system. Hepatotoxicity, on the other hand, has been shown to occur after a single dose or low repetitive doses. 4
In recent years, it has been reported in many studies that increased oxidative stress due to increased free oxygen radicals is a source of nephrotoxicity. 9 According to these studies, cisplatin produces reactive oxygen species such as hydroxyl radicals, hydrogen peroxide, single oxygen, and superoxide ions. Increasing the reactive oxygen species also inhibits the antioxidant enzymes, negatively affecting the natural antioxidant defense, leading to lipid peroxidation in the membranes, and a decrease in the activity of protective enzymes against peroxidation. This increased oxidative stress, which results in DNA damage, is also responsible for the toxicities formed. 10 Cisplatin, during the excretion of toxic metabolites, metabolizes mostly in the liver after the kidneys. This is because hepatocytes contain a high number of mitochondria, and hence some authors have reported that the damage to these organelles is the first step of cisplatin hepatotoxicity. 11 Cisplatin-induced oxidative stress primarily affects the mitochondrion, resulting in the depletion of mitochondrial protein-SH, suppression of calcium uptake, and a decrease in mitochondrial membrane potential and its function. 12 Higher doses of cisplatin, which may be needed for successful tumor suppression during aggressive treatment protocols, may cause hepatotoxicity, which is often seen during low-dose repetitive cisplatin therapy.1,13 Hepatotoxicity is a lesser-known side effect of cisplatin therapy and the underlying cause is unknown. The generation of oxidative stress due to reactive oxygen species (ROS), 14 a decreased antioxidant protection mechanism including antioxidant enzymes, 15 and the non-enzymatic molecule reduced glutathione (GSH) 16 have all been identified as significant alterations in cisplatin toxicity. Furthermore, mitochondrial dysfunction, apoptosis, Ca2+ homeostasis defects,17,18 and the role of pro-inflammatory genes such as COX-2 and inducible nitric oxide synthase (iNOS) may all play crucial roles in the underlying mechanisms of cisplatin-induced hepatotoxicity. 19
Even though mechanism of cisplatin-induced hepatotoxicity is unknown, the two most commonly accepted theories are a change in the antioxidant balance against oxidative stress and apoptosis acceleration.4,6,20 Endogenous enzymatic antioxidants (SOD, CAT, GSH Px, GSR), endogenous non-enzymatic antioxidants (GSH, melatonin, thioredoxin), and exogenous antioxidants (α-carotene, β-carotene, ascorbic acid, α-tocopherol, retinol, lycopene, flavonoids, phenols, phenolic acid) are used by cells subjected to oxidative stress to remove reactive oxygen (superoxide, hydroxyl, hydrogen peroxide) and nitrogen species [nitric oxide (NO), nitrogen dioxide, and peroxynitrite]. Some studies have been performed using a variety of antioxidants, free-radical scavengers, and natural herbal medicinal compounds to reduce Cisplatin’s side effects while maintaining its antineoplastic efficacy. Apocynin, thymoquinone, curcumin, silymarin, resveratrol, gingko, methionine, taurine, and dexpanthenol have been the most highly researched antioxidant compounds in the literature, in addition to the above mentioned antioxidants.
Ghrelin is a 28-amino acid, peptide-mediated hormone, isolated as a specific endogenous ligand for the growth hormone secretagogue receptor (GHS-R) that stimulates the growth hormone release. The primary site of release is the A-like cells in the gastric oxyntic mucosa.11,21 Apart from the stomach, the synthesis of this hormone in the liver, central nervous system, intestines, kidneys, heart, pancreas, lungs, gonads, and other organs such as the placenta shows that it is a peptide that plays a role in regulating many biological activities. 21 Ghrelin has many biological functions in addition to its involvement in homeostasis of energy and feeding, including cardioprotective activity and improvement of cardiac function, 22 neuroprotective activities, 23 a high anti-inflammatory 24 and antioxidant effects on many types of cells and tissues, such as in the liver, heart and lung.25–28 The anti-inflammatory activity of acylated ghrelin is primarily based on its particular effect on T lymphocytes and monocytes, where it suppresses the proinflammatory cytokines expression including IL-1β, IL-6, and TNF-α. 29 Recent studies have also reported that ghrelin has antioxidant and anti-apoptotic properties. 30 In this sense, Kheradmand et al. stated that ghrelin increases the activity of antioxidant enzymes and decreases lipid peroxidation in the ovaries of rats. 31 Obay et al. discovered that ghrelin protects erythrocytes, liver, and brain of rats from pentylenetetrazole-induced oxidative stress by suppressing lipid peroxidation and decreasing antioxidant enzyme activities and glutathione levels. 32 Likewise, Xu et al. found that ghrelin significantly decreased malondialdehyde levels while increasing the activity of antioxidant enzymes including catalase and superoxide dismutase in primary cell cultures of cardiomyocytes. 33 Furthermore, Moreno et al. discovered that ghrelin has a hepatoprotective function, which could help to prevent fibrosis. 34 Nevertheless, it is unknown if ghrelin inhibits lipid peroxidation or improves antioxidant enzyme activity in rats with cisplatin-induced liver injury. Hence, the aim of this study was to examine the potential protective role of ghrelin by measuring the activities of antioxidant enzymes (SOD, GSH), oxidative stress markers (MDA), and liver injury (ALT), as well as detecting hepatic histopathological changes in rat model of cisplatin-induced liver injury.
Materials and methods
Animals and laboratory
All experiments performed in the study were carried out according to the rules in the Guide for the Care and Use of Laboratory Animals, as adopted by the National Institutes of Health (U.S.) and Animal Ethics Committee’s consent (Ethical N:004548/2). Twenty-eight rats, male Sprague–Dawley mature rats, weighing 200–250 g, were used. The rats were housed in quiet rooms with a 12–12 h light-dark cycle (light from 07.00 to 19.00) and a 22–24°C ambient temperature; and they were given standard laboratory food and tap water ad libitum.
Chemicals and drugs
Cisplatin (Kocak Pharma Ilac, Turkey), Ghrelin (Sigma Chemical Co., USA), Ketamine and Xylazine (Alfasan International B.V., Holland), ALT elisa kit (USCN, Life Science Inc.). All other chemicals used for various analyses were purchased either from Sigma Chemical Co. (St Louis, MO, USA).
Experimental protocol
28 male Sprague Dawley albino mature rats were recruited to study. Group 1 rats (n = 7) were taken as the control group and no medication was given. Group 2 rats (n = 7) received 5 mg / kg / day cisplatin (Kocak Pharma Ilac, Turkey) and 1 ml/kg/day 0.9% NaCl, Group 3 rats (n = 7) received 5 mg/kg/day cisplatin and 10 ng/kg/day ghrelin (Sigma-Aldrich), Group 4 rats (n = 7) received 5 mg/kg/day cisplatin and 20 ng/kg/day ghrelin (Sigma-Aldrich) for 3 days.
Then, the animals were euthanized, and their blood samples were collected by cardiac puncture for biochemical analysis. Subsequently, bilateral hepatectomies were performed for histopathological examination.
Histopathological examination of liver
For histological and immunohistochemical studies, all animals were anesthetized by ketamine (40 mg/kg, Alfamine®, Alfasan International B.V., Holland) and xylazine (4 mg/kg, Alfazyne®, Alfasan International B.V., Holland) i.p. and perfused with 200 ml of 4% formaldehyde in 0.1 M phosphate-buffer saline (PBS). Formalin-fixed kidney sections (4 μm) were stained with hematoxylin & eosin. All sections were photographed with an Olympus C-5050 digital camera mounted on an Olympus BX51 microscope.
Measurement of liver lipid peroxidation (MDA)
Lipid peroxidation was determined in tissue samples by measuring malondialdehyde (MDA) levels as thiobarbutyric acid reactive substances (TBARS).
35
Briefly, trichloroacetic acid and TBARS reagent were added to the tissue samples, then mixed and incubated at 100
Measurement of liver protein levels
Total protein concentration in liver samples was determined according to Bradford’s method using bovine serum albumin as standard.
Determination of SOD activity
Total SOD activity was determined according to the method of Sun et al. 36 The principle of the method is the inhibition of nitrobluetetrazolium (NBT) reduction by the xanthine-xanthine oxidase system as a superoxide generator. One unit of SOD was defined as the enzyme amount causing 50% inhibition in the NBT reduction rate. SOD activity was given as units per milligram protein (U/mg protein).
Measurement of plasma ALT levels
Plasma ALT levels were measured using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (USCN, Life Science Inc.)
Measurement of tissue glutathione (GSH) levels
GSH content in tissue samples was measured spectrophotometrically according to Ellman’s method.
37
In this method, thiols interact with 5,5’-dithiobis-(2-nitrobenzoic acid) (DTNB), and form a colored anion with a maximum peak at 412 nm. GSH levels were calculated from the standard calibration curve and expressed as nmol
Statistical analysis
Statistical evaluation was performed by a one-way analysis of variance (ANOVA). The differences among groups regarding GSH, SOD, ALT, and MDA levels were evaluated by using one-way ANOVA. When the P-value from the variance analysis was statistically significant, the post hoc Tukey test was utilized. Results were presented as mean ± SEM. A value of p < 0.05 was considered to be significant.
Results
Effect of ghrelin on histopathological changes
Histopathological analysis showed that cisplatin, when compared to the control group (Figure 1(A)), induced vacuolar changes in pericentral hepatocytes, central venous dilatation and extensive hepatocellular necrosis (Figure 1(B)). Microscopic evaluations showed that the severe hepatic lesions induced by cisplatin were significantly decreased by the treatment with ghrelin (10 ng/kg) (Figure 1(C)). In the cisplatin + 20 ng/kg ghrelin group, nearly normal liver histology was observed (Figure 1(D)).

Liver histopathology H&E (×40). (A) Normal liver (control group). (B) Vacuolar changes of pericentral hepatocytes (arrow), central venous (cv) dilatation (cisplatin and saline group) and hepatocyte necrosis (asterisk). (C) Decreased central venous dilatation and hepatocyte necrosis (cisplatin and 10 ng/kg ghrelin group). (D) Nearly normal liver histology (cisplatin and 20 ng/kg ghrelin group).
Effect of ghrelin on oxidative stress marker MDA levels
The MDA level increased in rats given only cisplatin, as compared to the control group, while the MDA level increased significantly less in the group treated with 10 ng/kg ghrelin and cisplatin compared to the group given only cisplatin, and that when 20 ng/kg ghrelin and cisplatin were given, it was determined that the MDA level increased statistically much lesser manner than the cisplatin given group (p < 0.0001) (Table 1).
Effect of ghrelin on MDA and SOD levels.

** p < 0.0001 (different from control), *p < 0.01 (different from Cisplatin + saline).
##p < 0.000 (different from control), #p < 0.01 (different from Cisplatin + saline).
† p > 0.05 (different from Cisplatin+ ghrelin 10 ng/kg).
Effect of ghrelin on antioxidant enzyme SOD and GSH levels
SOD level decreased in rats given only cisplatin compared to the control group, and SOD level decreased statistically less in the group given 10 ng/kg ghrelin and cisplatin compared to the group given only cisplatin (p < 0.01), and when 20 ng/kg ghrelin and cisplatin were administered, it was determined that the level of the SOD decreased statistically lesser than the group given cisplatin. Moreover, it was observed that the SOD value decreased more in the group given 10 ng/kg ghrelin, but not in a statistically significant manner (p > 0.05) (Table 1).
GSH level decreased in rats given only cisplatin compared to the control group, while it was observed that the GSH level increased statistically in the group given 10 ng/kg ghrelin and cisplatin compared to the control as well as the cisplatin-only group (p < 0.01), and when 20 ng/kg ghrelin and cisplatin were given, the GSH level increased further in a statistically insignificant manner as compared to the control group and cisplatin-only group (p < 0.05) (Table 2).
Effect of ghrelin on ALT and GSH levels.
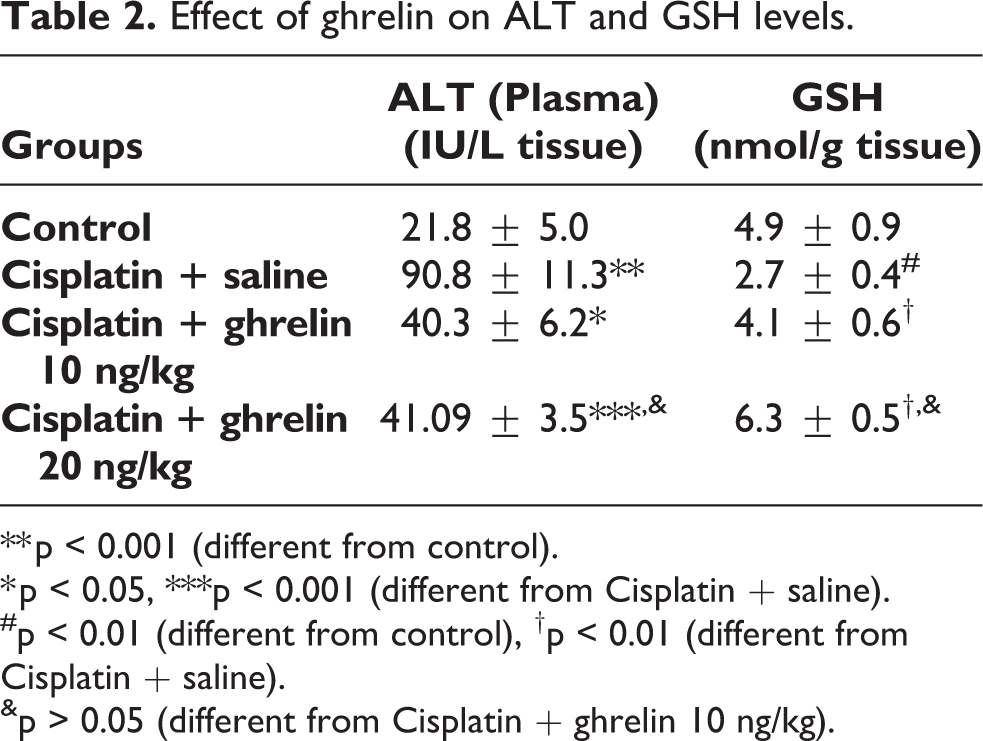
** p < 0.001 (different from control).
* p < 0.05, ***p < 0.001 (different from Cisplatin + saline).
#p < 0.01 (different from control), †p < 0.01 (different from Cisplatin + saline).
&p > 0.05 (different from Cisplatin + ghrelin 10 ng/kg).
Effect of ghrelin on cisplatin-induced liver injury
The effects of treatment with ghrelin on the cisplatin-induced elevation of serum ALT levels were shown in Table 2. The ALT level increased in rats given only cisplatin compared to the control group, while the ALT level significantly decreased in the groups given 10 ng/kg ghrelin + cisplatin and 20 ng/kg ghrelin + cisplatin compared to the group given only cisplatin (p < 0.01). Also, it was determined that ALT level increased in a statistically insignificant manner when 20 ng/kg ghrelin + cisplatin group were compared with 10 ng/kg ghrelin + cisplatin group (p > 0.05) (Table 2).
Discussion
Cisplatin is a widely used chemotherapeutic agent in the treatment of many cancers and has a toxic effect on many organs, especially hepatotoxicity and nephrotoxicity, which can cause vital problems. Cisplatin produces reactive oxygen species such as hydroxyl radicals, hydrogen peroxide, single oxygen, and superoxide ions. Increasing reactive oxygen species also inhibits the antioxidant enzymes, negatively affecting the natural antioxidant defense, leading to lipid peroxidation in the membranes, and a decrease in the activity of protective enzymes against peroxidation. This increased oxidative stress, which results in DNA damage, is responsible for the toxicities formed. Cisplatin, during the excretion of toxic metabolites, metabolizes most in the liver after the kidneys. 11 This is because hepatocytes contain a high number of mitochondria, and some authors have reported that the damage to these organelles was the first step of cisplatin hepatotoxicity. 11 It is well understood that cisplatin is rapidly absorbed by the human liver and that large doses of the drug cause hepatotoxicity.38,39 Cisplatin therapy causes a variety of responses in tumor cells, including DNA damage, membrane peroxidation, mitochondrial dysfunction, and protein synthesis inhibition.40,41 One of the pathogenic causes of cisplatin’s toxic effects in kidney and liver has been found to be the production of free radicals, which leads to oxidative stress. 42 Cisplatin has been shown to induce histopathological and ultrastructural changes in the liver, including inflammatory invasion, hyperplasia, fibrosis of periportal area, severe damage of hepatic cords, and blood sinusoids dilatation. Numerous hepatocytes exhibited karyomegaly and pyknotic nuclei, all of which are indicative of apoptosis.43,44 Numerous mechanisms and agents have been investigated to mitigate cisplatin-induced toxicity, but none have been shown to be effective for therapeutic use.45–47 Therefore, we aimed to show in this study whether ghrelin reduces the liver toxicity of cisplatin in the rat model.
Whereas the pathophysiological cause of cisplatin-induced hepatotoxicity is still unknown, recent research has shown that oxidative stress and apoptosis are responsible for a significant portion of the cisplatin’s toxicologic side effects.4,8 Cisplatin toxicity studies have shown decreased hepatic GSH levels and increased hepatic MDA levels, confirming the theory that cisplatin toxicity is due to increased oxidative stress in hepatocytes. 8 Thus, laboratory trials to date have used a variety of antioxidants, free-radical scavengers, and natural herbal extracts believed to possess defensive properties against cisplatin-associated cytotoxicity. Due to cisplatin’s ability to produce active oxygen species such as superoxide ion and hydroxyl radical and to inhibit active antioxidant enzymes in normal tissue; experimental studies have been conducted to investigate the protective effects of antioxidant activity of well-known substances (Vitamin e, Vitamin c, Ginkgo Biloba, melatonin, etc.) on the prevention of cisplatin hepatotoxicity.9,48,49 In these studies, it has been reported that accumulation in the liver during cisplatin metabolism causes sinusoidal dilatation and congestion, hepatocellular degeneration and toxicity manifested by inflammatory cell infiltration in the liver, and the aforementioned antioxidants have curative effects against this mechanism. In our study, it was found that venous dilatation and hepatic necrosis decreased in the ghrelin-treated groups.
Following cisplatin therapy, increased MDA levels and decreased GSH concentrations in liver tissue indicate that oxidative stress due to free-radical injury is one of the potential pathophysiologic mechanism of cisplatin hepatotoxicity. Cisplatin hepatotoxicity, like nephrotoxicity, has been linked to oxidative damage caused by ROS production. 50 Following cisplatin therapy, lipid peroxidation and GSH depletion were observed in the liver tissue. According to Pratibha et al., cisplatin enhanced lipid peroxidation and resulted in a substantial decrease in GSH levels in rat liver tissue. 1 MDA is the end product of lipid peroxidation and reflects the level of lipid peroxidation in the tissue. The increase in MDA level depends on the severity of tissue damage, lipid peroxidation, and inflammation. Toxic effects in humans and animals include nausea, vomiting, myelosuppression, sensitivity reactions, nephrotoxicity, ototoxicity, neurotoxicity, and bone marrow suppression; but the main dose-limiting side effect is cumulative nephrotoxicity. 51 Despite extensive prophylactic measures, irreversible kidney damage occurs in approximately one-third of the patients who undergo cisplatin treatments. In our study, it was observed that the MDA level was lower in rats given Ghrelin, especially with high-dose ghrelin, as compared to those given only cisplatin. This finding is consistent with previous observations that ghrelin decreased the elevated levels of MDA in preadipocyte cell culture, 52 rat testis and ovaries.31,53 Likewise, Obay et al. showed that pretreatment of rats with various doses of ghrelin inhibited an increase in lipid peroxidation caused by pentylenetetrazole. 32 Similarly, Işeri et al. demonstrated that ghrelin treatment significantly reduced MDA levels in rats suffering from alendronate-induced gastric tissue injury. 54 The decline in MDA level caused by ghrelin treatment demonstrates that ghrelin’s anti-lipid peroxidation function is involved in hepatoprotection.
GSH is a critical component of the non-enzymatic antioxidant mechanism and was previously shown to be involved in the removal of cisplatin. 55 As a result, reduced GSH levels can also play a direct role in lipid peroxidation induced by cisplatin. Cisplatin hepatotoxicity has also been linked to changes in the enzymatic component of antioxidant defense mechanisms in the liver. Cisplatin can inhibit the activities of superoxide dismutase, catalase, and glutathione peroxidase, as well as elevate MDA levels in the rat liver.9,56 It is one of the essential compounds for the regulation of Glutathione (GSH), and it is responsible for a diversity of cell functions. Moreover, it has a direct antioxidant function. Reaction with superoxide radicals, peroxyl radicals, and singlet oxygen is followed by oxidized glutathione formation and other disulfides. Glutathione S-transferase and Glutathione peroxidase are GSH-dependent antioxidant enzymes. 57 Glutathione S-transferase catalyzes the conjugation group of reduced glutathione via sulfhydryl to electrophilic centers on a wide variety of substrates. This activity is beneficial in the detoxification of endogenous compounds such as the metabolism of peroxalate lipids and xenobiotics. 58 GSH is the most important protection from antioxidative tripeptide-reactive metabolites that play an important role in the cell. 59 In addition, it is necessary to detoxify toxic substances, including chemotherapeutic drugs, and regulation of cellular homeostasis. 51 Glutathione contains reduced GSH along with peroxidase. GSH is important for maintaining the integrity of mitochondria and cell membranes. SOD, CAT, and GPx are antioxidant enzymes that act as a defensive mechanism to oxidative tissue damage. 60 O2 is converted to H2O2 by SOD. H2O2 is converted to water by GPx and catalase. Sustaining equilibrium between reactive oxygen species and antioxidant enzymes is critical, and this can serve as a significant mechanism for preventing oxidative stress-induced damage. 61 In our study, it was observed that the GSH level was higher in rats given ghrelin, especially with high-dose ghrelin, compared to those given only cisplatin. Our findings are compatible with previous findings about ghrelin’s antioxidative effects in other tissues.32,33,52,62,63 Our results showed that ghrelin protects the liver from cisplatin-induced damage by increasing SOD and GSH levels. Furthermore, ghrelin’s protection against cisplatin-induced liver damage may also be based on its inducible role on heme oxygenease-1 (HO-1), a redox-sensitive inducible protein that allows good cytoprotection against oxidative stress in a number of experimental models, including liver ischemia/reperfusion subsequent to transplantation either hemorrhage/resuscitation. 64 Evaluations of liver transaminase levels and histopathologic analysis of liver have demonstrated that HO-1 induction protects against CCl4-induced hepatotoxicity. 65 The nuclear factor erythroid 2-related factor 2 (Nrf2) is believed to control HO-1 induction. 66 Nrf2 is crucial for the antioxidant protection mechanism of the cell against oxidative damage.67,68 In our study, the induction in SOD and GSH levels in the groups of ghrelin treatment could also be a result of ghrelin-induced Nrf2 increase.
Transaminases are the most sensitive biomarkers, including cellular damage and toxicity. This is because they are cytoplasmic in location and are released into the circulation after cellular damage. 69 There may be ALT changes in liver disease and myocardial infarction. 70 As previously reported, treatment with cisplatin alone resulted in a substantial increase in serum ALT levels in this study, which are indices of liver cell injury.71–73 Our findings discovered that ghrelin therapy effectively prevented rats from cisplatin-induced hepatic toxicity as demonstrated by lower serum ALT levels. This finding is consistent with the observations that ghrelin reduced the elevated activity of AST and ALT in plasma of acetaminophen-treated rats. 74 Likewise, Moreno et al. found that pretreatment with ghrelin reduced AST activity in CCl4-treated rats. 34 Histological examination validated our results as well. The vacuolar changes in pericentral hepatocytes, central venous dilatation and extensive hepatocellular necrosis were observed in cisplatin-induced liver damage. Ghrelin treatment significantly ameliorated these histopathological modifications.
Plasmatic ghrelin levels are lower in patients with alcoholic hepatitis and chronic hepatitis C than in normal subjects and associate adversely with the seriousness of fibrosis. 34 Furthermore, ghrelin treatment inhibits hepatic damage in rats with chronic hepatic fibrosis induced by bile duct ligation (BDL) through suppressing the BDL-induced raise in TNF-α, IL-1, and IL-6 plasma levels. 62 Thirdly, ghrelin administration reduces the amount of myofibroblast aggregation in the damaged liver and reduces neutrophil infiltration.34,62 As a consequence, ghrelin inhibits collagen-1 and TGF-1 expression in primary hepatic stellate cells (HSC), the major hepatic fibrogenic cells, 34 resulting in less collagen deposition.34,62 Ghrelin has also anti-inflammatory and antifibrotic properties in rats with thioacetamide (TAA)-induced hepatic damage, where it reduces liver inflammation and collagen deposition by inhibiting hepatic cell apoptosis and antioxidative function, which is partly regulated by nitric oxide induction (NO). 75 Eventually, the physiological function of the ghrelin gene in the development of liver fibrosis was examined using ghrelin knock-out mice, which exhibit significantly more extreme CCl4-induced liver damage and fibrosis than wild type mice, implying that endogenous ghrelin is essential for a suitable response to liver injury. 34
In conclusion, this study shows that ghrelin has a significant hepatoprotective function against cisplatin-induced liver damage through decreasing MDA and ALT levels, and enhancing SOD and GSH activities. It has been observed that the administration of ghrelin, especially in high doses, can reduce the hepatoxic effect of cisplatin. Further research would be conducted to determine ghrelin’s possible beneficial role in the prevention or treatment of hepatic diseases.
Footnotes
Author contributions
R.B.: principal author. M.A.E.: writing, data collection and work. E.E.: data collection and work. A.M.: data collection and work. A.E.: data collection and work. Ö.A.: data collection and work. O.E.: data collection and work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
