Abstract
In this study, both diabetes and Dunning prostate cancer were induced for the first time in Copenhagen rats in vivo. Thus, the effects of metformin against heart tissue damage of these rats were investigated by biochemical methods. Dunning prostate cancer was induced in Copenhagen rats using high metastatic MAT-LyLu cells. The rats were divided as follows: Control group: only injected with 0.9% NaCl for 14 days; Diabetic group: only injected single dose of streptozotocin (STZ) (65 mg/kg); Cancer group: subcutaneously (s.c) inoculated with 2 x 104 MAT-LyLu cells only; Diabetic + cancer (DC) group: inoculated with 2 x 104 MAT-LyLu cells and STZ injection, Cancer + metformin (CM) group: injected with metformin for 14 days after Mat-LyLu cells application; Diabetic + cancer + metformin (DCM) group: metformin administered for 14 days together with STZ and Mat-LyLu cells. At the end of the experimental period, heart tissues were taken. Reduced glutathione and total antioxidant status levels in heart tissues were decreased, whereas lipid peroxidation, advanced oxidized protein products, nitric oxide, homocysteine, and reactive oxygen species levels, total oxidant status and catalase, superoxide dismutase, glutathione peroxidase, glutathione reductase, glutathione-S-transferase, alanine aminotransferase, aspartate aminotransferase, lactate dehydrogenase and xanthine oxidase activities increased in the diabetic, cancer and DC groups. Treatment with metformin reversed these effects. In conclusion, the present study shows that metformin has a protective effect against heart tissue damage in STZ-induced diabetic rats with Dunning prostate cancer.
Introduction
It is expected that 44140 new cancer cases will be diagnosed in 2020 when compared with new cases diagnosed in 2019. Thus, prostate cancer (PCa) is estimated to be on the first rank among the newly diagnosed cancer cases in 2019. On the other hand, prostate cancer is expected to be the second most common cause of cancer related deaths after lung/bronchial cancer. 1,2
PCa is a disease of the prostate gland, which is located just below the bladder and surrounds the urethra. 3 It has been indicated that acinar adenocarcinoma developing in the glandular cells of the prostate is the main type of PCa that accounts for about 99% of cases. Besides, there are much less common types of PCa such as ductal adenocarcinoma, urothelial, squamous cell, neuroendocrine (carcinoid) tumors and sarcomas. 4
In general, diabetes is a multifactorial disease that is characterized by insulin deficiency or resistance, which lead to hyperglycaemia. The persistence of hyperglycaemia causes a further increase in oxidative stress status due to increased free radical formation. Increased cellular oxidative stress elevates the risk of cancer by damaging important biomolecules such as DNA. 5 Both cancer and diabetes are prevalent diseases whose incidence is increasing globally. Diabetes is associated with high risk factors that progress into many types of cancer, especially hepatocellular carcinoma, 6,7 breast, 8 lung, colorectal, gastric and PCa. 9,10 Diabetes also complicates the prognosis of cardiovascular heart disease and increases the mortality rate associated with coronary heart disease. 11 Both type 1 diabetic (T1DM) and type 2 diabetic (T2DM) patients are at a high risk of developing heart diseases. 12 Moreover, individuals with T2DM have a markedly higher risk of cardiovascular morbidity and mortality as compared to healthy subjects. 13 The risk of developing heart failure in healthy individuals has been reported to be less than two-times that of diabetic patients. 14 Therefore, increased prevalence of diabetes contributes significantly to the etiology of cardiovascular dysfunction, with heart failure increasingly observed in diabetic individuals. 15 Recent studies suggest that chronic hyperglycaemia is associated with ROS-induced oxidative stress, which in turn plays a crucial role in the development and progression of impaired cardiac function. 16 –18 In a population-based study, cancer patients were found to have a higher risk of dying from cardiovascular disease and the risk is negatively related to the diagnosis at an early stage. 19
Metformin (a biguanide drug) is a commonly used as an oral antihyperglycaemic agent that is not only well-tolerated, but easily accessible and is the first treatment option for T2DM. 20 Moreover, metformin has been used as a beneficial drug in the treatment of T2DM in Europe for over 60 years, and in USA for over 25 years. The main reason for the continues use of this drug in T2DM is that it has not enhanced insulin-sensitivity, but also glucose-lowering effect in hepatic cells. 21,22 Beyond its antidiabetic role, metformin treatment is associated with decreased cancer incidence and as well decreased cancer-related mortality. 23,24
In the past decade, clinical studies have shown that diabetic subjects may have a decreased risk of PCa. 25 On the other hand, recent data suggested that the decrease in risk of PCa is associated with the use of antidiabetic drugs like metformin. 26 The main purpose of this study was to determine the possible effect of metformin on heart damage in STZ-induced diabetic rats with Dunning PCa.
Materials and methods
Animals
In this study, Copenhagen breed adult rats obtained from “TÜBİTAK MAM Genetic Engineering and Biotechnology Institute” were used. All experimental protocols were approved by the Animal Care and Use Committee of the Istanbul University, Istanbul, Turkey (2014/28-the ethics committee decision number). Rats weighing 150–220 g were housed individually in a light and temperature-controlled room on a 12 h/12 h light–dark cycle and fed a standard pellet lab chow.
Cell culture
Dunning model strongly metastatic Mat-LyLu cells were grown in RPMI culture medium (RPMI-1640, Gibco; Life Technologies, USA), supplemented with 1% fetal bovine serum (FBS) (Gibco), 2 mM L-glutamine (Gibco; Life Technologies, USA) and 250nM dexamethasone (Sigma; Sigma-Aldrich, USA). 27,28 Cells were maintained in a 37°C/5% CO2 incubator.
Pharmacological agents
Streptozotocin (STZ) (65mg/kg) (Sigma, SB.S0130-1G) was diluted in 1 mL of +4°C physiological saline (PS) and injected intraperitoneally (i.p.) into rats in order to induce diabetes. Fasting blood glucose of rats left unfed was examined at the 72nd hour, those with fasting glucose of 200 mg/dL were considered diabetic. Metformin (250 mg/kg) (Sigma, D150959-5G) was dissolved in 0.2 mL PS at +4°C and given to the rats systemically via gavage.
Experimental design
Experimental rats were randomly split into six groups as follows; I) Control group: only injected with 0.9% PS for 14 days, II) Diabetic group: only injected single dose of streptozotocin (STZ) (65mg/kg), 29 III) Cancer group: subcutaneously (s.c) inoculated with 2 x 104 MAT-LyLu cells only, IV) Diabetic + cancer (DC) group: inoculated with 2 x 104 MAT-LyLu cells and STZ injection, V) Cancer + metformin (CM) group: injected with metformin 30 for 14 days after Mat-LyLu cells application VI) Diabetic + cancer + metformin (DCM) group: metformin administered for 14 days together with STZ and Mat-LyLu cells. The 2 x 104 MAT-LyLu cells were inoculated into the upper-right extremities of the rats in order to create PCa after the diabetic assay. The experiment was terminated on the 14th day to procure early PCa. 31 At the end of the study, all animals were dissected under ketamine hydrochloride (Ketalar®, Eczacıbaşı) and xylazine HCl (Alfazyne®, Holland) anesthesia.
Biochemical analyses
Heart tissue samples were taken and immediately washed in ice-cold with PS solution and homogenized in PS with a glass homogenizer to make up to 10% homogenate (w/v). These homogenates were centrifuged at 10000×g at +4°C for 10 minutes. After centrifugation, clear supernatants were collected and the pellets were discarded. The clear supernatants were used for biochemical analyses. Reduced glutathione (GSH), lipid peroxidation (LPO), advanced oxidized protein products (AOPP), and nitric oxide (NO) levels were determined according to by the methods of Beutler 32 using Ellman’s reagent, Ledwozyw et al., 33 Witko-Sarsat et al., 34 and Miranda et al., 35 respectively. Homocysteine (HCys) level was assayed using ELISA kit according to instructor’s manual. Reactive oxygen species (ROS) levels, total oxidant status (TOS), and total antioxidant status (TAS) in the heart tissues were analyzed according to Zhang et al., 36 Erel, 37,38 respectively. Catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione reductase (GR) and glutathione-S-transferase (GST) activities were assessed by the methods described by Aebi, 39 Mylorie et al., 40 Paglia and Valentine 41 and modified by Wendel, 42 Beutler, 43 and Habig and Jacoby, 44 respectively. Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were assayed according to Reitman and Frankel. 45 Lactate dehydrogenase (LDH) and xanthine oxidase (XO) activities were estimated according to Bais and Philcox 46 and Corte and Stirpe, 47 respectively. The protein content in the supernatants was estimated according to Lowry et al. 48 method using Bovine Serum Albumin (BSA) as standard.
Statistics
Statistical analysis was performed using GraphPad Prism 6.0 (GraphPad Software, San Diego, California, USA). All data were expressed as means ± standard deviation (SD). The results were evaluated using an unpaired t-test and analysis of variance (ANOVA) followed by Tukey’s multiple comparison tests. A p value of less than 0.05 represented significance.
Results
Reduced glutathione (GSH) and lipid peroxidation (LPO) levels are shown in Figure 1. The levels of GSH in diabetic and DC group were slightly elevated, but significantly lower in cancer group than the control group (p < 0.0001). On the other hand, metformin treatment to DC group resulted in a slighter declined, but caused a remarkable increase in heart GSH levels of the cancer group (p < 0.0001) (Figure 1). LPO levels in diabetic group was insignificantly elevated, whereas in the cancer and DC group, it was remarkably raised compared to the control group (p < 0.01 and p < 0.0001). Supplementation of metformin to both cancer and DC groups resulted in a significant decrease in the heart LPO levels (p < 0.05 and p < 0.0001) (Figure 1).

Effect of metformin on heart tissue glutathione and lipid peroxidation levels in diabetic, cancer and DC groups of rats: (a) GSH, (b) LPO. All the values are expressed as mean ± SD. DC: Diabetic+cancer; CM: Cancer+metformin; DCM: Diabetic+cancer+metformin; GSH: Reduced glutathione; LPO: Lipid peroxidation; MDA: Malondialdehyde.
Advanced oxidized protein products (AOPP), nitric oxide (NO), and homocysteine (HCys) levels are presented in Figure 2. AOPP levels in diabetic, cancer and DC groups were significantly increased when compared to control groups (p < 0.001, p < 0.0001 and p < 0.01). There was a significant decrease in the level of AOPP in cancer group (p < 0.0001), but an insignificant decrease in DC group when metformin was administered to cancer and DC groups (Figure 2). NO and HCys levels in the heart homogenates of diabetic, cancer and DC groups were notably increased as compared with control group (p < 0.0001, p < 0.001 and p < 0.05). The administration of metformin to cancer and DC groups gave rise to a notable decrease in NO and HCys levels respectively (p < 0.001 and p < 0.01) (Figure 2).
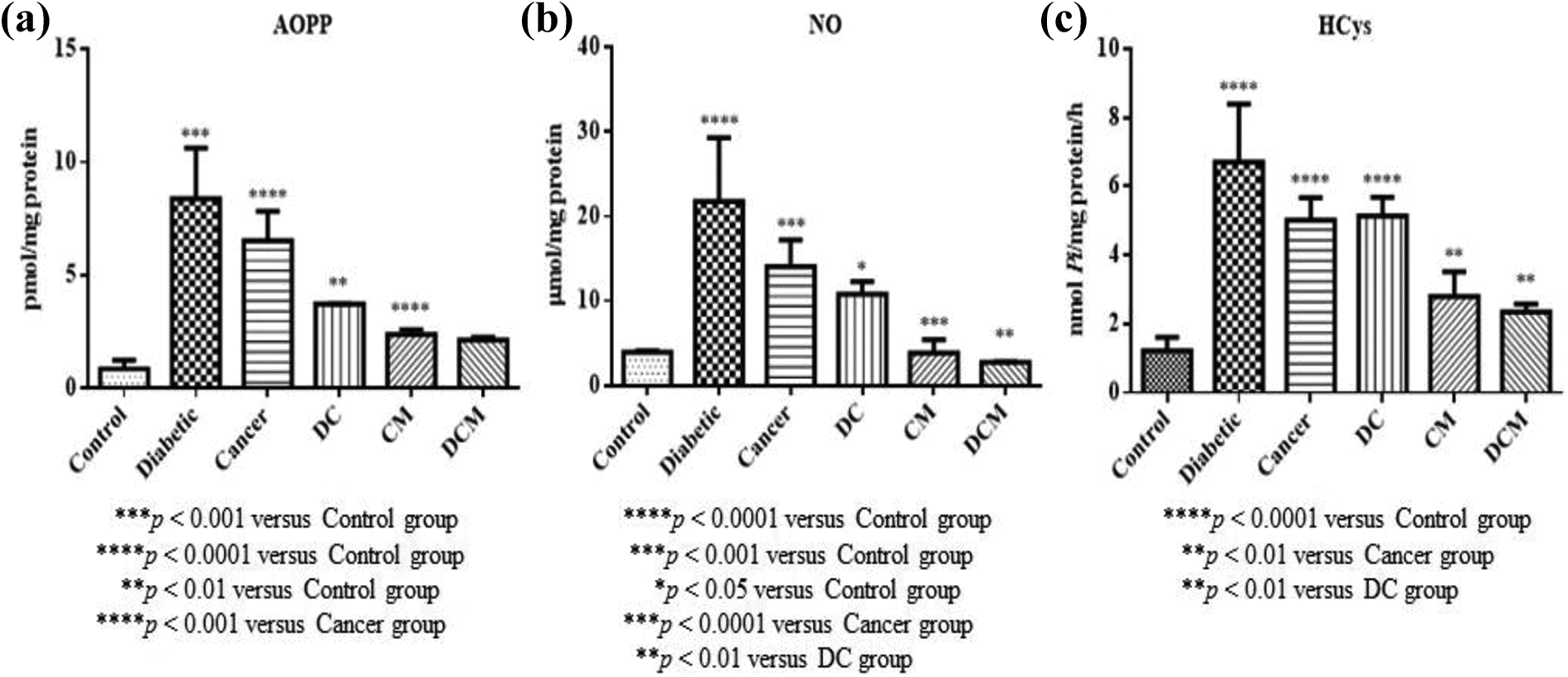
Effects metformin on AOPP, NO, and HCys levels in diabetic, cancer and DC groups of rats: (a) AOPP, (b) NO, (C) HCys. All the values are expressed as mean ± SD. DC: Diabetic+cancer; DCM: Diabetic+cancer+metformin; AOPP: Advanced oxidized protein products; NO: Nitric oxide; HCys: Homocysteine.
Reactive oxygen species (ROS), total oxidant status (TOS) and total antioxidant status (TAS) levels are shown in Figure 3. A remarkable elevation of ROS activity and TOS levels were observed in diabetic (p < 0.001 and p < 0.01), cancer (p < 0.0001 and p < 0.001) and DC groups (p < 0.0001) respectively when compared to control rats (Figure 3). On the contrary, TAS level remarkably decreased in diabetic (p < 0.0001), cancer (p < 0.001) and DC group (p < 0.01) when compared to control rats. Supplementation of metformin in both cancer and DC groups notably reversed these effects (Figure 3).
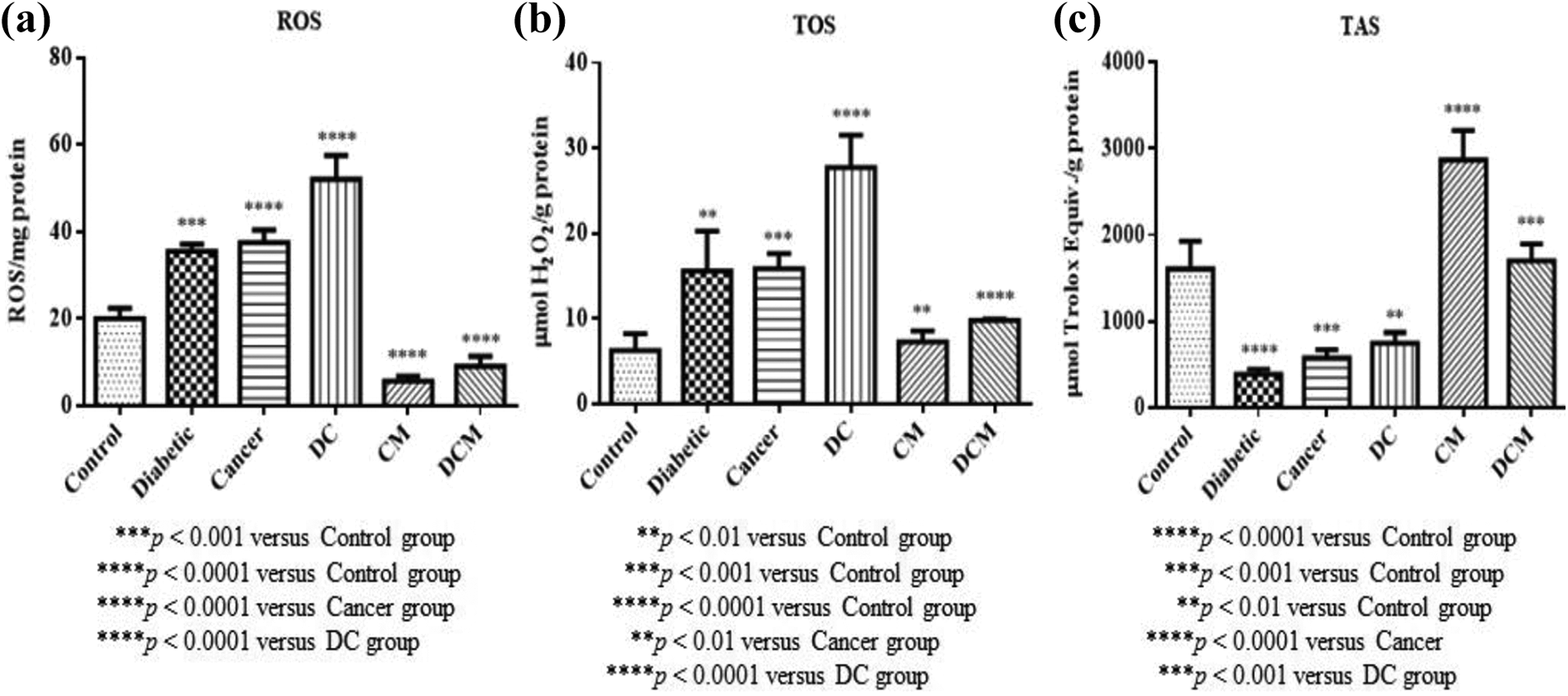
Effects of metformin on ROS, TOS, and TAS levels in diabetic, cancer and DC groups of rats: (a) ROS, (b) TOS, (c) TAS. All the values are expressed as mean ± SD. DC: Diabetic+cancer; CM: Cancer+metformin; DCM: Diabetic+cancer+metformin; ROS: Reactive oxygen species; TOS: Total oxidant status; TAS: Total antioxidant status.
Catalase (CAT) and superoxide dismutase (SOD) activities of all groups are shown in Figure 4. A statistically significant elevation of both CAT and SOD activities were observed in diabetic (p < 0.0001 and p < 0.01), cancer (p < 0.001 and p < 0.05) and DC (p < 0.0001 and p < 0.001) groups as compare to control group. The administration of metformin to the cancer and DC groups noticeably dropped CAT and SOD activities (p < 0.01 and p < 0.001) (Figure 4).

Effects of metformin on heart tissue CAT and SOD activities in diabetic, cancer and DC groups of rats: (a) CAT, (b) SOD. All the values are expressed as mean ± SD. DC: Diabetic+cancer; CM: Cancer+metformin; DCM: Diabetic+cancer+metformin; CAT: Catalase; SOD: Superoxide dismutase.
Glutathione peroxidase (GPx), glutathione reductase (GR) and glutathione-S-transferase (GST) activities of heart tissues of all groups were presented in Figure 5. In the heart of diabetic, cancer and DC rats, GPx activity was significantly increased when compared to the control group (p < 0.05 and p < 0.0001). After supplementation with metformin to both cancer and DC group, a significant decrease in GPx activity (p < 0.0001) (Figure 5) was noticed. We observed a notable increase in GR and GST activities all experimental groups when compared to control rats (p < 0.01, p < 0.0001 and p < 0.05). Metformin treatment resulted to a remarkable decrease in GR (p < 0.0001 and p < 0.001) and GST activities in both cancer and DC groups (p < 0.0001) (Figure 5).
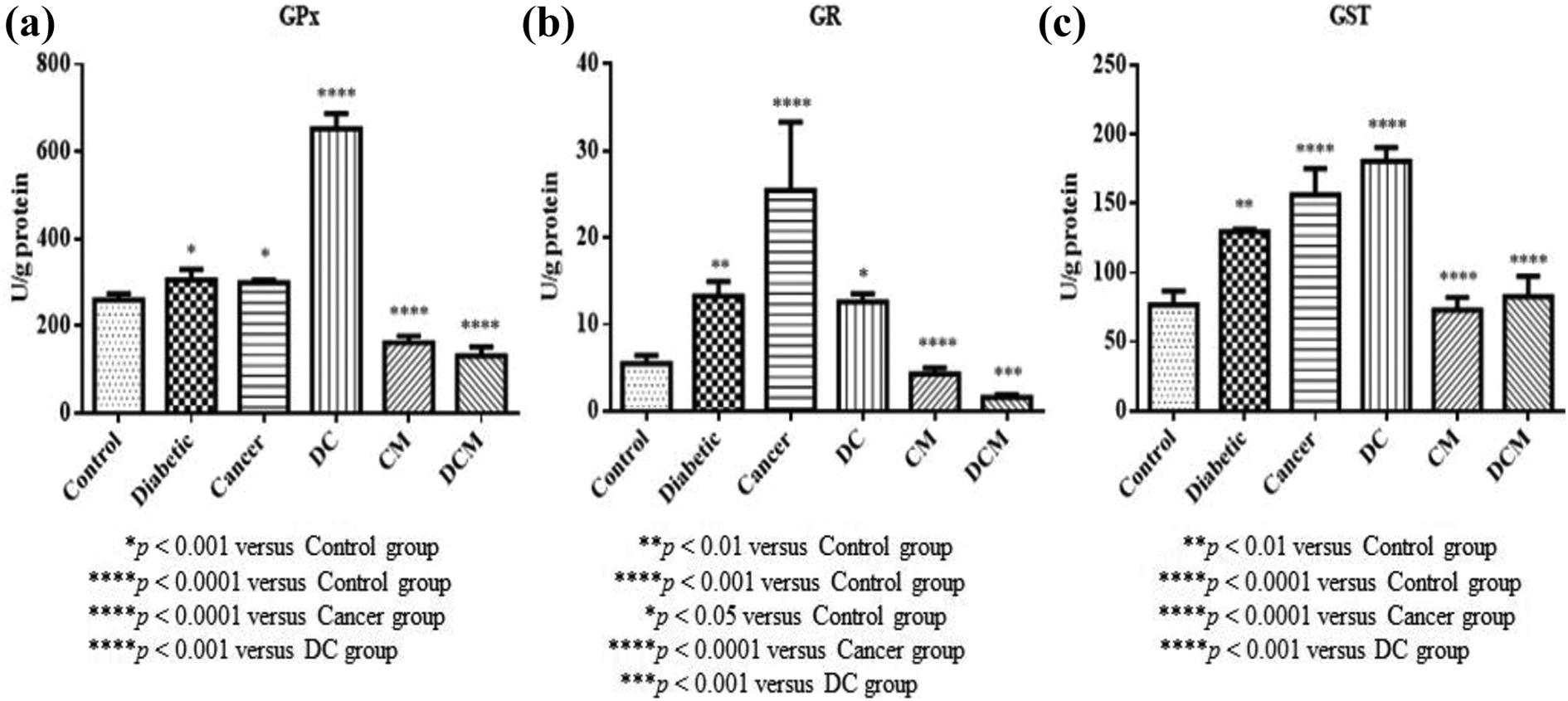
Effects of metformin on GPx, GR and GST activities in diabetic, cancer and DC groups of rats: (a) GPx, (b) GR, (c) GST. All the values are expressed as mean ± SD. DC: Diabetic+cancer; CM: Cancer+metformin; DCM: Diabetic+cancer+metformin; GPx: Glutathione peroxidase; GR: Glutathione reductase; GST: Glutathione-S-transferase.
Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities are presented in Figure 6. It was observed that ALT and AST activities were significantly elevated in diabetic, cancer and DC group as compared to control rats (p < 0.0001 and p < 0.05). Metformin administration to cancer and DC group significantly reverted the activities of both ALT and AST (p < 0.001, p < 0.0001 and p < 0.01) (Figure 6).

Effects of metformin on ALT and AST activities in diabetic, cancer and DC groups of rats: (a) ALT, (b) AST. All the values are expressed as mean ± SD. DC: Diabetic+cancer; CM: Cancer+metformin; DCM: Diabetic+cancer+metformin; ALT: Alanin aminotransferase; AST: Aspartate aminotransferase.
Lactate dehydrogenase (LDH) and xanthine oxidase (XO) activities of all experimental groups are presented in Figure 7. In diabetic, cancer and DC groups, we observed a notable increase in LDH (p < 0.001 and p < 0.0001) and XO activities (p < 0.0001, p < 0.001, and p < 0.01) when compared to control rats. The administration of metformin to cancer group caused a significant decrease in the LDH (p < 0.0001) and XO activities of cancer and DC groups (p < 0.001 and p < 0.05) (Figure 7).
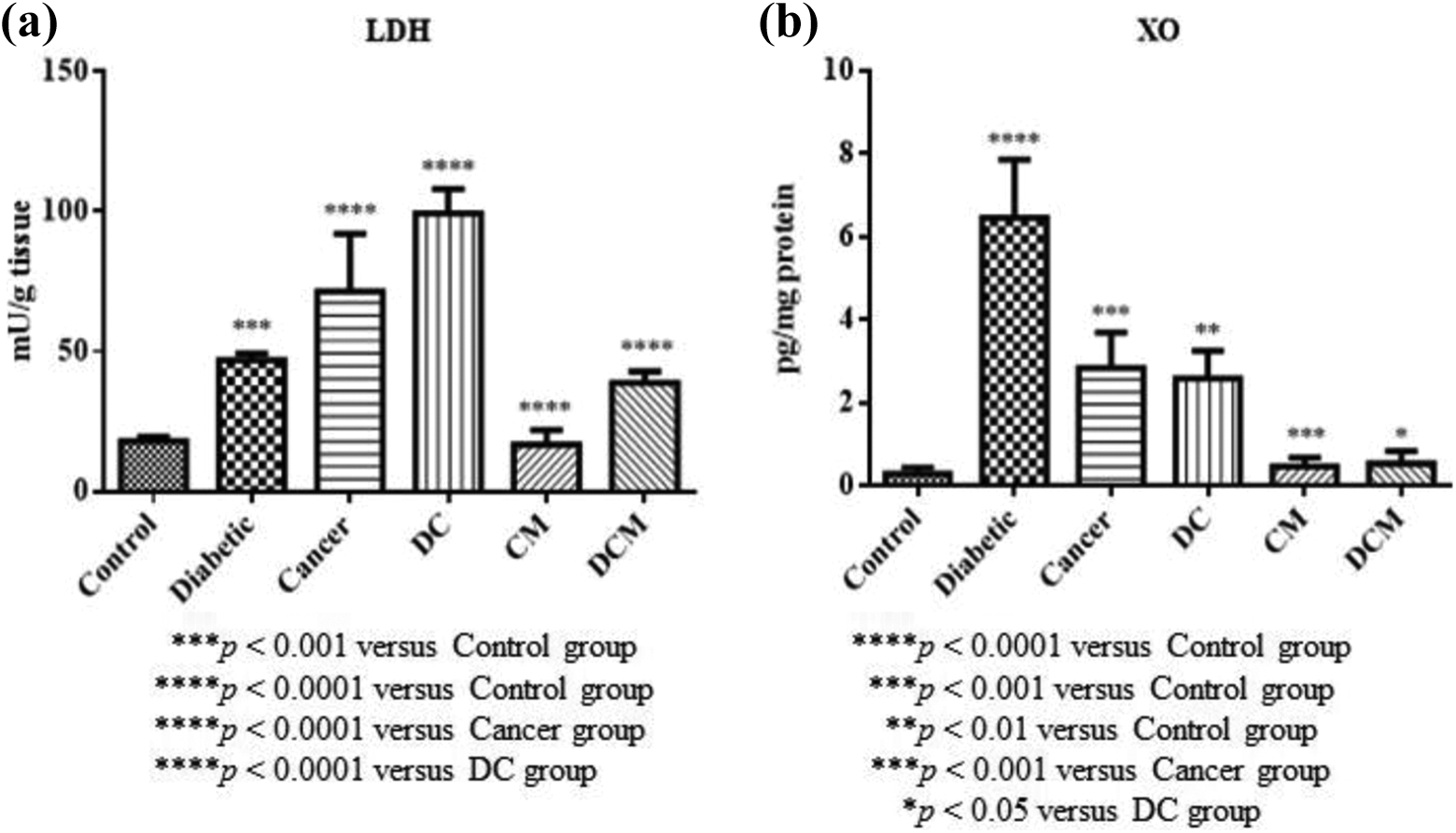
Effects of metformin on LDH and XO activities in diabetic, cancer and DC groups of rats: (a) LDH, (b) XO. All the values are expressed as mean ± SD. DC: Diabetic+cancer; CM: Cancer+metformin; DCM: Diabetic+cancer+metformin; LDH: Lactate dehydrogenase; XO: Xanthine oxidase.
Discussion
Diabetes and cancer continue to be among the most important health problems in terms of both morbidity and mortality, and are becoming increasingly critical day by day. However, diabetes alone and/or its related factors are a possible micro-environment that can trigger formation or progression of cancer. 49 It is stated that diabetic individuals have higher risk of death due to cancer as compared to patients without diabetes. 50 Scientists and research groups all over the world are making great efforts to find the treatment of both diabetes and various types of cancers. For this purpose, they are constantly working on new drugs or molecules that may be useful in medication for use in treatment. Upon literature search, we found little or no information on the possible beneficial effects of metformin on heart tissues of STZ-induced diabetic rats with Dunning prostate cancer model.
Under normal physiological conditions, there is a balance between reactive oxygen species (ROS) that are constantly being formed in cells and antioxidants that neutralize them. Disruption of this balance in favors of ROS, i.e. oxidative stress set in which is characterized by the accumulation of cellular oxy-radicals or compromised endogenous defense systems. 51
Reduced GSH is a major intracellular component of antioxidant defense, which neutralizes ROS as well as regulates cellular survival by offering redox homeostasis in a variety of tumors including lung, breast, colon and PCa. 52,53 On the other hand, quantification of malondialdehyde (MDA) levels as secondary metabolite of LPO in either serum or tissue homogenates are useful biomarker for assessing oxidative damage. 54 In the present study, we observed that GSH levels apparently declined in the cancer group only, but moderately increased in both diabetic and DC groups, whereas LPO levels were notably elevated when compared to control group. Depletion of GSH and elevation of LPO levels in experimental groups may be as a result of increased ROS formation. Hajra et al. 55 stated that doxorubicin-induced cardiotoxicity caused a remarkable decline of GSH levels, whereas LPO levels significantly increased. These outcomes were similar to the LPO levels of cardiac tissues of diabetic rats reported by Naderi et al., 56 and Aju et al., 57 and of breast cancer subjects reported by Kangari et al. 54 In our study, it was observed that the levels of both GSH and LPO were almost normalized to levels similar to that of the control group after metformin treatment. The attenuating properties of metformin may be due to its antitumor, antioxidant, and antihyperglycaemic effects. 58,59
AOPPs are oxidized plasma proteins of albumin and its aggregates, which are important biomarkers for determining oxidative stress. It is observed that increased AOPP levels may be an independent risk factor of atherosclerotic plaques in coronary artery disease. 60 It is also suggested that AOPP’s accumulation may be linked to vascular lesions in diabetes, chronic renal insufficiency and atherosclerosis. 61 NO, a ubiquitous short-lived signaling molecule in nature and a free radical as well as, has a vasodilating effect under physiological circumstances in the vascular endothelium. 62 In addition, NO plays a dual role in cancer development depending on its concentration or where it is formed. It may also have a tumouricidal or tumour-inhibiting effect. 63,64 HCys is a non-proteinogen α-amino acid that is produced from methionine, an elevation of its levels is clinically associated with primary risk factor for coronary and cardiovascular mortality. 65 In the current study, we found that the levels of AOPP, NO and HCys in cancer, diabetic and DC group rats were significantly higher than control rats. The alteration of AOPP, NO and HCys levels may be due to altered oxidant/antioxidant balance which favored oxidants or the progression of the inflammatory process. Similar results were reported by Škvařilová et al. 66 for AOPP, by Stadler et al. 67 for NO and by Beyramzadeh et al. 68 for HCys, respectively. We observed that all these parameters were nearly restored by metformin administration to the cancer, diabetic and DC rats. Metformin may be responsible for decreasing protein oxidation, as well as HCys levels in the heart. Our findings are in accordance with that of Meaney et al. 69 and Kelleni et al. 70
To assess the redox status, we focused on ROS activity, TOS and TAC levels. These parameters are frequently studied because they are useful biomarkers for evaluating the biochemical and pathophysiological conditions underlying various diseases associated with oxidative stress. 71,72 In the present study, we found that ROS and TOS levels in diabetic, cancer and DC groups were higher than values found in control group, whereas TAS level in these group was lower than in control rats. Our findings are consistent with the Bozkurt et al. 71 and Zińczuk et al. 73 Metformin treatment to cancer and DC groups resulted in restoration of oxidant/antioxidant status in heart tissues of rats, which may be related to the anticancer and antidiabetic effects of metformin.
The antioxidant enzyme system is a complex antioxidant mechanism ubiquitously found in cells against harmful effects of free radicals on tissues. SOD, CAT, GPx, GR, and GST are among the basic and first line of defense against deleterious effects of free radicals in the cell. 74 SOD catalyzes the dismutation of superoxide radical to H2O2. Produced by dismutation of superoxide radicals, H2O2 is converted into H2O by CAT and/or GPx. During the reaction catalyzed by GPx, GSH is oxidized to it disulphide i.e. GSSG. On the other hand, GSSG is reduced by the catalytic action of the GR enzyme. 75 In our study, SOD, CAT, GPx, GR and GST activities in diabetic, cancer and DC groups were found to elevate as compared to control rats. These results suggest the increased activity of these first line antioxidant enzymes may be due to adaptive response against overproduction of ROS in heart tissues of both diabetes and PCa models. In a by Stefek et al., 76 both SOD and CAT activities were increased in myocardium of diabetic rats. Kumar et al. 77 conducted a study on effects of multiple antioxidants on cardiac complications and they found that CAT activities were higher in the test than in control rats. Similar findings were reported by Tchamgoue et al. 78 On the other hand, Kotrikadze et al. 79 and Taheri et al. 80 reported that GPx and GR activities were sharply increased in prostate tumor and T2DM patients, respectively. GPx activities were found to rise in prostate tissues of diabetic rats. 81 Moreover, it was found that both GPx and GR activities significantly increased in gastrocnemius muscle of diabetic rats. 82 It was shown that GST activities in myocardium tissues were notably increase in diabetic rats. 83 In the present study, metformin administration reversed the alterations of these defensive enzymes. The reason for the superior effect of metformin may be a compensatory response to oxidative stress and possibly the increased expression of antioxidant enzymes in tissues of rats. 84
ALT and AST have been used as diagnostic biomarkers for assessing both liver and heart tissue necrosis. 85,86 In the current study, both ALT and AST activities significantly increased in all experimental groups as compared to control group. Our findings were in accordance with reports of Bayramoglu et al. 87 and Hajra et al. 55 Metformin administration restored the activities of these enzymes in heart tissues of experimental rats. This indicates that metformin reduces oxidative stress, leading to reduced formation of ROS. The findings of the present study are in accordance with results of previous studies. 78,88
LDH, a soluble cytoplasmic enzyme, is ubiquitously found in almost all cells. It is released into extracellular space when the integrity of plasma membrane is compromised for any reason. 89 Its increased activity is an important biomarker of tissue damage especially the cardiac tissue. 90 XO is a well-known enzyme that is responsible for generation of free radicals when it catalyzes the conversion of hypoxanthine and xanthine to uric acid. 91 Enhanced XO activity in vascular endothelium is association with elevated serum uric acid levels which result in oxidative damage. Moreover, the activity of vascular endothelium-bounded XO is reported to increase more than two-fold in both heart failure and coronary arteries disease when compared to healthy subjects. 92 In the current study, we found that LDH and XO activities in cancer, diabetic and DC groups of rats significantly increased as compared to control rats. In a study by Farshid et al. 93 and Aliciguzel et al., 94 increase in LDH and XO activities in heart tissues in diabetic rats were reported. Both LDH and XO activities notably diminished when metformin was administered to diabetic, cancer and DC groups. In our study, MDA levels increased in all damaged heart tissues. In addition, the increased level of MDA may be associated with the activity of XO which is an important free radical source. Therefore, metformin may have reduced MDA level and thus XO activity with its antioxidant effect. Our results were consistent with the findings of Kelleni et al. 70 and Cosić et al. 95
Conclusions
In the present finding, the administration of metformin in both STZ-induced diabetes and Dunning PCa model resulted in positive changes on total oxidant-antioxidant status. Metformin treatment also exhibited beneficial effect by altering the activities of first line antioxidant defense enzyme in heart tissues of rats. In conclusion, it can be suggested that metformin may recuperate heart damage by stabilizing oxidative stress parameters as well as cardiac enzyme activities in STZ-induced diabetes and Dunning PCa model via its antioxidant, antitumor and anti-inflammatory effects.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
