Abstract
Introduction:
Carbon monoxide (CO) poisoning is a crucial cause of delayed neuropsychiatric syndrome (DNS). However, most biomarkers are not satisfactory for the prediction of DNS caused by CO poisoning. Thus, we evaluated the adequacy of the serum glucose/potassium (GLU/K) ratio, which may be an easy, quick, and readily available parameter that can be used in the emergency department for predicting DNS.
Methods:
We evaluated 281 patients who were admitted to our emergency department between January 2012 and December 2018. The patients were divided into two groups: DNS (+) and DNS (−). The GLU/K was compared for the groups.
Results:
Glucose, blood urea nitrogen, carboxyhemoglobin, and GLU/K ratios of patients in the DNS (+) group were statistically significantly higher than those patients in DNS (−) group (140 ± 34 vs. 110 ± 24, p < 0.001; 17.58 ± 6.14 vs. 14.27 ± 5.08, p = 0.003; 29 ± 5.1 vs. 18.9 ± 7.6, p < 0.001; and 38.35 ± 10.11 vs. 28.65 ± 6.53, p < 0.001, respectively). The area under the curve for GLU/K to predict DNS was measured as 0.791, and 35.9 as a cut-off value had 63.6% sensitivity and 89.6% specificity.
Conclusions:
DNS development in CO poisoning is a serious and feared complication. We suggest that the GLU/K ratio has a high potential as a rapid, easy preliminary marker for the exclusion of patients who will not subsequently develop DNS.
Introduction
Background
Carbon monoxide (CO) is a colorless, odorless, tasteless, nonirritating gas in low concentrations. Due to these features, CO intoxication is also called a silent killer. Intoxication is seen with the incomplete burning of carbon-containing compounds, with CO reaching high concentrations in the air and with inhalation. 1,2 After inhalation, it is rapidly absorbed from pulmonary capillaries and binds to hemoglobin with 240 times greater affinity than oxygen, causing a left-sided oxyhemoglobin curve and decreased tissue oxygenation. 3
CO poisoning is also important in terms of leaving neurologic and cardiac sequelae in the long term. It can cause ataxia, dementia, lack of concentration, and behavioral disorders. 2 It is believed that CO-induced brain damage is generally related to hypoxia. Tissue hypoxia causes oxidative stress and neuronal death. As a result, loss of consciousness and delayed neuropsychiatric syndrome (DNS) are thought to develop, but the pathophysiology of late neurologic sequelae is not fully understood. 4,5
The incidence of DNS after CO poisoning is thought to be between 3% and 40% because of the lack of established diagnostic criteria. 6 The pathophysiology of DNS has not been fully revealed. Brain lipid peroxidation due to CO intoxication causes intramyelinic edema or inflammation and antigenic alterations of myelin basic protein, resulting in white matter damage. 7 –9
Predictors associated with the severity of brain injury and neurologic sequelae in previous studies among patients with CO poisoning included old age, prolonged CO exposure time, unconsciousness or prolonged coma, acidosis, affected cardiovascular system, high blood glucose, aspartate aminotransferase (AST), leukocyte, and brain biomarkers such as S100B and copeptin levels. However, S100B and copeptin are not available in many hospital laboratories, and other clinical conditions such as fundamental diseases, duration of unconsciousness, and intoxication time are not satisfactory for the prediction of DNS. 10 –12
When evaluating patients who present with CO intoxication in emergency departments, acute complications are considered, but late complications may be ignored. Therefore, the prediction of late neuropsychiatric complications should be considered as a preventative measure.
Accordingly, we evaluated the adequacy of the serum glucose/potassium ratio (GLU/K), which may be an easy, quick, and readily available parameter that can be used in the clinic for predicting DNS due to CO poisoning.
Patients and methods
Study design and setting
From our hospital database, 414 patients aged 18 years or older with CO intoxication between January 2012 and December 2018 were included in the present study. Patients with blood carboxyhemoglobin (CO-Hb) saturation <10% (n = 90) at the admission time, patients with missing data (n = 25), death due to CO poisoning in the emergency department (n = 5), patients with a history diabetes mellitus or admission serum glucose level >200 mg/dL (n = 13) were excluded from the study. Patients were also evaluated for neuropsychiatric diseases in their background before CO intoxication and a disease/or drug use that could induce hyperkalemia or hypokalemia for exclusion from the study, but no such patients were found. As a result, 281 patients who met our inclusion criteria were included in the study.
The included patients were evaluated as to whether they were admitted to a neurology or psychiatry clinic within 1 year for DNS complications after CO intoxication. The diagnosis for DNS was made according to the International Classification of Diseases (ICD-10) code from the hospital records. The main presentations of DNS are psychosis (e.g. dementia, catatonia, hallucination), psychoneurosis (e.g. depression, anxiety, insomnia), strial syndrome (e.g. parkinsonism, chorea, ballismus, dystonia), motor deficit (e.g. hemiplegia, apraxia), sensory deficit (e.g. hemianopsia, agnosia, cortical blindness), speech deficit (motor or sensory aphasia, agraphia), seizure disorder, spinal cord deficit, and peripheral nerve deficit (e.g. polyneuropathy, mononeuropathy, facial palsy). 4,10,13
On hospital admission, clinical data were recorded such as Glasgow Coma Score (GCS), blood pressure, and pulse rate, venous and arterial blood samples were taken, and laboratory parameters were recorded: complete blood count (hemoglobin, red blood cell (RBC), platelet count, white blood cell (WBC)), biochemistry (alanine aminotransferase (ALT), AST, gamma-glutamyl transferase (GGT), blood urea nitrogen (BUN), serum creatinine, sodium, potassium, glucose, GLU/K), and arterial blood gas (partial pressure of carbon dioxide (PCO2), partial pressure of oxygen (PO2), CO-Hb) levels.
Hyperbaric oxygen is an approved therapy after exposure to CO for loss of consciousness, syncope, seizure, coma, blood CO-Hb levels >25%, focal neurologic deficits, and evidence of acute myocardial ischemia. 2 However, hyperbaric oxygen therapy (HBOT) could not be administered to patients due to the absence of a hyperbaric treatment center in our hospital and nearby centers, or for reasons such as patients who refuse to be referred to receive HBOT treatment at another center. Thus, all patients received normobaric oxygen therapy (oxygen concentration 100% and 10/L per minute) using a non-rebreather mask.
Statistical analyses
All statistical analyses were performed using the IBM SPSS Statistics for Windows version 23.0 software (IBM, Armonk, New York, USA). Continuous data are reported as mean ± standard deviation (SD). The comparison of quantitative variables between the two groups was performed using the Mann–Whitney U test. Correlations between categorical variables were evaluated using the χ 2 test. Logistic regression analysis was used to determine GLU/K and glucose affecting DNS. Receiver operating characteristics (ROC) analysis was performed for the GLU/K ratio and glucose for predicting DNS. The area under the ROC curve (AUC), cut-off values, sensitivity, specificity, positive predictive value, and negative predictive values were calculated to evaluate the performance of the GLU/K ratio and glucose. The level of statistical significance was set at p < 0.05.
Results
Twenty-two (7%) of the 281 patients developed DNS. The mean age in the DNS (+) patients was 36 ± 17 (mean ± SD) years and in the DNS (−) group it was 39 ± 13 (mean ± SD) years. The distribution of the DNS (+) and DNS (−) patients according to their gender were as follows: DNS (+) %59.1% (13) female, %41.9 (9) male patients, DNS (−) 61.4% (159) female, %38.6 (100) male. There was no statistically significant difference between the groups in terms of age and sex (p value: 0.162 and 0.832, respectively).
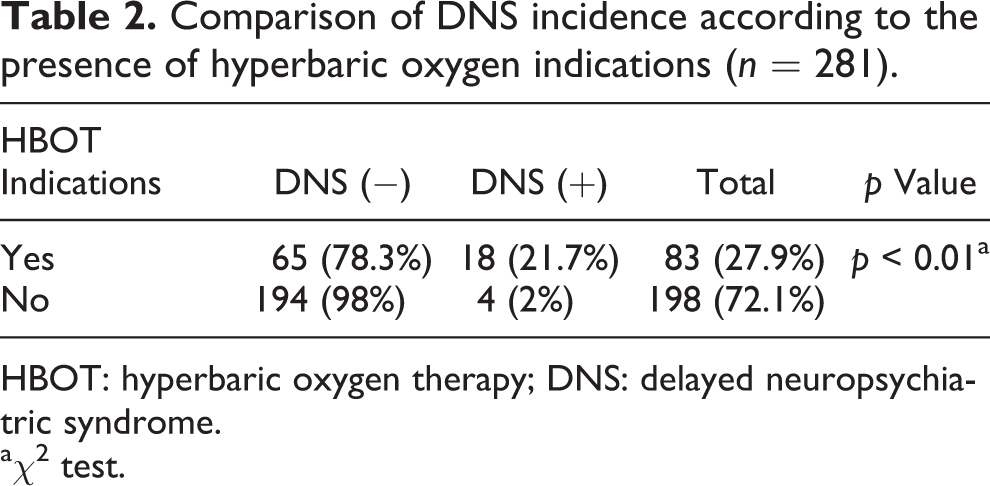
Table 1 summarizes the laboratory admission results of the patients in both groups. The glucose, BUN, CO-Hb, and GLU/K ratios of the patients in the DNS (+) group were statistically significantly higher than in patients in the DNS (−) group (140 ± 34 vs. 110 ± 24, p < 0.001; 17.58 ± 6.14 vs. 14.27 ± 5.08, p = 0.003; 29 ± 5.1 vs. 18.9 ± 7.6, p < 0.001; and 38.35 ± 10.11 vs. 28.65 ± 6.53, p < 0.001, respectively). The GCS scores of the patients in the DNS (+) group were statistically significantly lower than in patients in the DNS (−) group (13.95 ± 1.6 vs. 14.6 ± 0.3, p < 0.05). There were no statistically significant differences between the other laboratory parameters of the patients. The incidence of DNS was significantly higher in patients with hyperbaric indications (Table 2).
Comparison of clinical variables and laboratory parameters according to DNS status.a

GCS: Glasgow Coma Scale; RBC: red blood cell; WBC: white blood cell; BUN: blood urea nitrogen; ALT: alanine aminotransferase; AST: aspartate aminotransferase; GGT: gamma-glutamyl transferase; pH: power of hydrogen; PCO2: partial pressure of carbon dioxide; PO2: partial pressure of oxygen; CO-Hb: carboxyhemoglobin; GLU/K: glucose/potassium; DNS: delayed neuropsychiatric syndrome.
a Normally distributed data expressed as mean ± standard deviation, non-normally distributed data expressed as median ± interquartile range. Student t-test was used for normally distributed data; Mann–Whitney U test was used for non-normally distributed data.
b Normally distributed.
c Statistically significant.
Comparison of DNS incidence according to the presence of hyperbaric oxygen indications (n = 281).
HBOT: hyperbaric oxygen therapy; DNS: delayed neuropsychiatric syndrome.
a χ 2 test.
The odds ratios between GLU/K ratio, serum glucose levels, and DNS rates were 1.135 (95% confidence interval (CI): 1.081–1.192; p < 0.001) and 1.030 (95% CI: 1.017–1.043; p < 0.001), respectively.
Table 3 presented the ROC curve analysis of GLU/K and glucose in discriminating DNS (+) patients among all the studied patients and in HBO indication group. For all studied patients, the AUC for GLU/K was measured as 0.791, and 35.9 as a cut-off value had 63.6% sensitivity and 89.6% specificity. For glucose, the AUC was measured as 0.787, and 128.5 mg/dL as a cut-off value had 63.6% sensitivity and 86.9% specificity (Figure 1). The AUC for GLU/K was measured as 0.704, and 36.8 as a cut-off value had 54.6% sensitivity and 90.7% specificity among the HBOT indications group. For glucose, the AUC was measured as 0.699, and the cut-off value of 133.5 mg/dL had 59.1% sensitivity and 88.4% specificity among the HBOT indications group.
ROC curve analysis of GLU/K ratio and glucose in discriminating DNS (+) patients among all studied patients and in the HBO indications group.

SEN: sensitivity; SPE: specificity; PPV: positive predictive value; NPV: negative predictive value; ROC: receiver operating characteristics; GLU/K: glucose/potassium; HBO: hyperbaric oxygen; AUC: area under the curve.
a DNS (+) patients among all studied patients.
b DNS (+) patients among HBO indications.

ROC analyses of GLU/K ratio and glucose. ROC: receiver operating characteristics; GLU/K: glucose/potassium.
Discussion
One of the serious complications of CO poisoning is DNS. In this study, we analyzed the potential of the GLU/K ratio as a predictor of DNS. We found that the GLU/K ratio was significantly higher in patients developing DNS compared with patients without DNS, and we also showed that an increase in the GLU/K ratio could be a risk factor for developing DNS. In addition, we found a statistically higher incidence of DNS among patients with HBOT indications.
The glucose modifiers that cause hyperglycemic reactions are mainly catecholamines, glucagon, and corticosteroids. 14 After stress and injuries, catecholamines in particular increase serum glucose levels. Catecholamines increase glucose levels directly or by increasing glucagon secretion and inhibiting insulin secretion indirectly. 15 Potassium is stored in the cells of the human body. It is actively transported by the sodium–potassium adenosine triphosphatase pump (Na+/K+-ATPase) from plasma to cells. In this arrangement, catecholamines, insulin, and β2 adrenergic hormones lower serum potassium levels. 16
There are many studies investigating blood glucose levels and clinical disease progression. Thirty-day mortality rates were higher in patients with ST-elevation myocardial infarction whose admission blood glucose levels were >140 mg/dL. 17,18 Chao et al. evaluated the high glycemic variability in critically ill patients and determined increased 30-day mortality among these patients. 19
The increase of serum glucose levels and CO intoxication was evaluated in the 1980s. Sokal and Kralkowska evaluated the relationship between blood glucose, pyruvate, and lactate and the severity of CO poisoning. The authors found no correlation between the blood glucose levels and the severity of intoxication, and blood lactate was the only marker accepted for severity. In another study by Sokal, blood glucose, pyruvate, and lactate levels in patients with CO poisoning were compared with a control group. They determined that pyruvate and lactate levels increased with exposure time and clinical severity, whereas blood glucose levels were more increased among patients with short exposure time and decreased among patients with long exposure. These two studies show that blood glucose levels alone cannot be used to assess CO intoxication severity. 20,21
In a new study by Barghash et al., blood glucose levels were also evaluated among three patient and control groups. The patient groups were classified according to their poisoning severity scores. They determined that blood glucose was especially high among patients with severe poisoning (131.8 mg/dL). These results indicated that blood glucose could determine the outcome of CO intoxication. 22 In our study, the glucose levels (140 ± 34 mg/dL) of patients who developed DNS were similar to those reported in these studies and were statistically significantly higher in the DNS-developing group than in the non-DNS-developing group.
Ogura et al. found that patients who were admitted with poor clinical status had significantly higher levels of catecholamines than those with better clinical status in patients with subarachnoid hemorrhage (SAH). They suggested that sympathetic activation levels in the acute phase of SAH reflected the severity of the disease. 23 Matano et al. reported that there was a significant correlation between cerebral vasospasm and ischemic complications occurring after SAH, and they also showed that the GLU/K ratio and serum GLU/K ratio were high in patients with poor prognosis. 24 Fujiki et al. reported that a high serum GLU/K ratio in patients with aneurysmal SAH would be useful in predicting poor prognosis. 25 Park et al. suggested that catecholamine crises in globus pallidus and deep white matter were key pathophysiologic factors in CO intoxication. 26 In another study, norepinephrine and dopamine were found to be high in pericardial and cerebrospinal fluids in patients who died of CO intoxication. 27 Oh and Choi reported that due to the increase in sympathetic activity in CO intoxication, catecholamine levels might increase in synapses, nerve terminals, and the limbic system, leading to DNS. 13 According to these studies, catecholaminergic crises are likely to occur in CO intoxication resulting in elevation of blood glucose level and decrease of potassium level. The abovementioned studies revealed the relationship between rises in the GLU/K ratio and poor prognosis. Likewise, our study revealed a high serum GLU/K ratio at admission in patients who developed DNS. The GLU/K ratio cut-off value was able to predict 63.6% of patients who had DNS and correctly identified 89.6% of patients who developed DNS. In addition, the high GLU/K ratio was a risk factor for DNS development.
In the literature, studies have been conducted on many markers that may be DNS predictors of CO intoxication. The cut-off value of 0.165 μg/L for S100B had a sensitivity of 90% and a specificity of 87% for predicting DNS. 28 In another study, the anion gap, lactate, osmolality, S100B, and interleukin (IL)-6 were examined, and the sensitivity and specificity of these parameters were found as 90%, 84.8% (anion gap), 70%, 97.8% (lactate), 60%, 88% (osmolality), 90%, 84.8% (S100B), and 100%, 80% (IL-6), respectively. The cut-off value was updated as 0.103 μg/L for S100B. 29 In a study of plasma copeptin levels, sensitivity was found as 77.8%, specificity 82.1%, cut-off 40.6 pmol/L, and the AUC was 849. 12 In our study, the GLU/K ratio had lower sensitivity, but a better specificity than S100B and copeptin. S100B and copeptin are costly biomarkers, frequently unavailable, and not routinely analyzed in all hospitals. According to these results, the GLU/K ratio can be a useful, simple preliminary marker that can be used for the exclusion of patients who will not develop DNS.
HBOT is still a controversial issue in the prevention of DNS. Thom et al. concluded that successful HBOT might require the administration of therapy within 6 h of CO poisoning, which was associated with a significant reduction in the incidence of DNS. 30 Weaver et al. stated that HBOT treatment was effective against the development of DNS. 31 In a review, it was stated that hyperbaric therapy should be preferred to prevent neurologic sequelae. 32 In contrast, the effect of HBO treatment on the incidence of negative neurologic results in CO intoxication was found to be uncertain in another study. 33 In our study, the presence of DNS in patients with indications for HBO treatment was significantly higher and also the GLU/K ratio (AUC: 0.709) and blood glucose levels (AUC: 0.699) were statistically useful for the prediction of DNS among patients who needed HBOT.
Strengths and limitations
We could not analyze the levels of glucagon, corticosteroid, and catecholamine hormones at the time of presentation. Also, the causes for hyperkalemia or hypokalemia were only determined through the evaluation of the patient’s background history from the charts. The patient and control groups were not designed according to their smoking habits.
Conclusions
DNS development in CO poisoning is a serious and feared complication. Physicians should evaluate parameters that could be used for prognosis. We suggest that the GLU/K ratio has a high potential as a rapid, easy preliminary marker for the exclusion of patients who will not subsequently develop DNS.
Footnotes
Author contributions
The conception and design of the study; the acquisition, analysis, and interpretation of data for the work; drafting the work and revising it critically for important intellectual content; and final approval of the manuscript were done by ED. The drafting of the study and its critical revision for important intellectual content was performed by İK. Analysis, and interpretation of data for the study were made by YKT, EsinD, and İÇ.
Data availability statement
The data were retrieved from the Clinical Data Analysis and Reporting System, the Clinical Management System, and the Electronic Patient Record System of the Hospital Authority, Sivas.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by the institutional ethics committee of the Sivas Cumhuriyet University (2020-01/28).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
This study was authorized by the medical authorities of our hospital and patients were informed and signed their consent to their data collection.
