Abstract
Objectives:
Uremic pruritus is common in patients with chronic kidney disease (CKD). The retention of uremic solutes is thought to be associated with uremic pruritus. Meanwhile, activation of protease-activated receptor-2 (PAR-2) has been suggested to play an important role in pruritus. The present study was performed to investigate the effects of uremic solutes on the expression of PAR-2 in the skin.
Methods:
Indoxyl sulfate (IS), p-cresol (PC), and uremic sera from CKD patients were used to stimulate PAR-2 expression in normal human epidermal keratinocytes (NHEKs). Also, NHEKs were additionally pretreated with soybean trypsin inhibitor to evaluate its inhibitory effect on PAR-2 expression. Patterns of cutaneous PAR-2 expression were investigated in skin samples from five CKD patients and CKD mice.
Results:
In NHEKs, IS, PC, and sera from CKD patients significantly induced PAR-2 mRNA and protein expression. Soybean trypsin inhibitor significantly decreased PAR-2 mRNA and protein expression in NHEKs treated with IS, PC, and CKD sera. NHEKs treated with IS and PC exhibited significant increases in protease activity. Skin from both CKD patients and mice exhibited marked upregulation of PAR-2 expression compared to control skin.
Conclusions:
Results from the present study suggest that uremic solutes either directly or indirectly affect PAR-2 expression in the skin of CKD subjects, potentially playing an important role in the pathogenesis of uremic pruritus.
Keywords
Introduction
Uremic pruritus is common in patients with chronic kidney disease (CKD). Previous studies indicated that 60–80% of dialysis patients have uremic pruritus. 1 –3 Although the mechanisms underlying the appearance of uremic pruritus in CKD are not fully understood, the retention of uremic solutes is thought to play a role. 4 –6 Uremic solutes are categorized into three major physiochemical types: free water-soluble, low-molecular-weight solutes; protein-bound solutes; and middle molecules. 6 Among these, protein-bound solutes are not effectively eliminated by dialysis, and are of biochemical significance in CKD. 4,6,7 Two protein-bound solutes in particular, indoxyl sulfate (IS) and p-cresol (PC), are considered toxic as they lead to vascular and renal disease progression in CKD patients. 4 –6 Nevertheless, the effects of IS and PC on the skin of CKD patients have yet to be investigated.
Protease-activated receptors (PARs) comprise a subfamily of G protein-coupled receptors that are activated by proteolytic cleavage of their extracellular domains. 8 Specific proteases for activating PARs include thrombin for PAR-1, -3, and -4, and trypsin for PAR-2. 8 In the skin, PAR-2 is expressed abundantly in various types of cells and acts as a sensor for endogenous and exogenous proteases. 9,10 Marked increases in PAR-2 expression have been reported in primary afferent nerve fibers throughout the skin of atopic dermatitis patients, and these increases are significantly related to histamine-independent itching sensations. 11 In addition, our group reported that cutaneous PAR-2 expression and serine protease activity are greater in pruritic CKD patients as determined by confocal laser microscopy and in situ zymography. 12
This study was performed to investigate the effects of protein-bound uremic solutes on the expression of PAR-2 in human skin and a CKD mouse model. To do so, IS, PC, and sera from CKD patients were used to stimulate PAR-2 expression in human primary keratinocytes. Then, PAR-2 mRNA and protein expression was measured using real-time polymerase chain reaction (RT-PCR) and Western blotting. Finally, immunohistochemical staining of human and mouse skin samples was performed to determine the patterns of PAR-2 expression in CKD.
Materials and methods
Cell culture
Normal human epidermal keratinocytes (NHEKs; Lonza, Basel, Switzerland) were incubated with keratinocyte growth medium (Lonza) and serially subcultured for 5–10 passages. IS and PC were purchased from Sigma-Aldrich (St. Louis, Missouri, USA), and serum samples were obtained from CKD patients and healthy controls. IS and PC were diluted in phosphate-buffered saline and methanol, respectively, to concentrations of 10, 50, 100, and 250 μg/mL. NHEKs were stimulated with different concentrations of IS and PC and with 4% pooled serum samples. NHEKs cultured with 4% bovine serum albumin were used as controls. In further experiments, NHEKs were additionally pretreated with soybean trypsin inhibitor (Sigma-Aldrich) at a dilution ratio of 1:10 in keratinocyte growth medium for each experimental treatment (differing concentrations of IS and PC) to evaluate its inhibitory effect on PAR-2 expression. All experiments were performed in triplicate.
RT-PCR and Western blotting
Total RNA was isolated from NHEKs after 12, 24, 48, and 72 h of incubation with IS, PC, and serum samples using TRIzol reagent (Invitrogen Life Technologies, Carlsbad, California, USA), and cDNA was synthesized using a Takara RNA PCR kit (V2.1; Takara Bio Inc., Shiga, Japan). An ABI 7500 sequence detection system (Applied Biosystems, Foster City, California, USA) was used for quantitative RT-PCR (qRT-PCR). β-Actin expression was used as an internal standard for qRT-PCR. The following primers were used: PAR-2 5′-TCATGATGTTTTGCCTGAGC-3′ (forward) and 5′-CACATAGGCAGAGGCTGTGA-3′ (reverse), and β-actin 5′-ATAGCACAGCCTGGATAGCAACGTAC-3′ (forward) and 5′-CACCTTCTACAATGAGCTGCGTGTG-3′ (reverse). Experiments were repeated at least three times.
For Western blotting, total protein from the NHEKs in each treatment was extracted, and prepared samples were immunoblotted with rabbit polyclonal anti-PAR-2 antibody (ab180953; Abcam, Cambridge, Massachusetts, USA) and mouse monoclonal anti-β-actin antibody (Sigma-Aldrich). Signals were detected using enhanced chemiluminescence, and immunoreactive bands were quantitatively analyzed using ImageJ software (V1.48; National Institutes of Health, Bethesda, Maryland, USA).
Protease activity assay
The EnZCheck Protease Assay (E6638) kit (Invitrogen, Carlsbad, California, USA) was used to measure protease activity in keratinocytes and culture media according to the manufacturer’s instructions.
All samples were incubated with BODIPY fluorescent casein substrate at room temperature for 1 h, and the resultant fluorescence was read using a luminescence spectrometer (LS50B; PerkinElmer Inc., Waltham, Massachusetts, USA) with an excitation wavelength of 485 nm and an emission wavelength of 538 nm.
Patients
Five CKD patients with uremic pruritus (four men and one woman; mean age, 53 years; range, 41–74 years) and five healthy controls (four men and one woman; mean age, 58 years; range, 45–70 years) provided informed consent for participating in this study. CKD status was defined as estimated glomerular filtration rate <15 mL/min/1.73 m2, as calculated using the Modification of Diet in Renal Disease equation. All five CKD patients were undergoing hemodialysis three times per week. None of the participants had a prior history of any dermatological disease other than pruritus within the previous year. Serum samples were obtained from CKD patients prior to hemodialysis and the healthy controls and stored at −70°C. In addition, normal-looking skin samples were taken from the abdomens of the CKD patients and healthy controls. The study was approved by the Institutional Review Board of Severance Hospital, Yonsei University College of Medicine, Seoul, Korea.
Animal model
To induce CKD in a mouse model, 6-week-old male FVB mice (n = 5; KOATECH, Pyeongtaek, Korea) were used, and a two-step procedure was performed as described previously. 13 Briefly, we performed electrocoagulation of the entire surface of the right kidney, except for a 2-mm area of intact tissue around the hilum, followed 2 weeks later by left nephrectomy. At 6 weeks after nephrectomy, the mice were euthanized and skin samples were obtained. The degree of renal failure was evaluated by measuring blood urea nitrogen (BUN), serum creatinine, and hemoglobin concentrations on the day of euthanasia. We observed significantly lower hemoglobin (10.8 ± 0.3 vs. 13.3 ± 0.5 g/dL) and significantly higher BUN and serum creatinine levels in CKD mice relative to controls (61.5 ± 6.3 and 0.27 ± 0.05 vs. 19.9 ± 1.01 and 0.13 ± 0.04 mg/dL, respectively; Supplementary Figure S1). Skin samples from FVB mice subjected to a sham operation at the same age were used as controls. All experiments were approved by the Committee for the Care and Use of Laboratory Animals at Yonsei University College of Medicine.
Immunohistochemical staining
Full-thickness skin samples from the experimental mice and human subjects were obtained and embedded in paraffin. The samples were cut into sections with a thickness of 5 µm and incubated with rabbit polyclonal anti-PAR-2 antibody (ab180953; Abcam) at a dilution ratio of 1:100 for 2 h. Then, staining with secondary antibody (Real™ Envision™ HRP Rabbit/Mouse; DAKO, Carpinteria, California, USA) was performed, and the sections were counterstained with hematoxylin.
For renal histology, kidney sections were stained with Masson’s trichrome stain. The tubular injury score was scored semiquantitatively by an independent investigator who was blinded to the study design. Scoring was determined by assessing the cortex and corticomedullary junction as follows: 0, 0%; 1+, <25%; 2+, 25–50%; and 3+, >50% of tubules. Randomly selected fields (n = 10) from the cortex and corticomedullary junction in each kidney slide section were examined as described previously. 14
Statistical analysis
Data are presented as mean ± standard error of the mean. Statistical analyses were performed with Student’s t-test and one-way analysis of variance, as well as a post hoc Bonferroni test. All analyses were performed with SPSS software for Windows (V22.0; IBM Corporation, Armonk, New York, USA). In all analyses, p < 0.05 was taken to indicate statistical significance.
Results
Effects of IS on PAR-2 expression in NHEKs
NHEKs were stimulated with IS and PC at concentrations of 10, 50, 100, and 250 μg/mL. PAR-2 mRNA expression was evaluated at 12, 24, 48, and 72 h. RT-PCR revealed both dose-dependent and time-dependent upregulation of PAR-2 mRNA expression in IS-treated NHEKs. Higher concentrations of IS and longer incubation times, especially at 48 and 72 h, resulted in significantly elevated PAR-2 mRNA expression (1.78-fold at 250 μg/mL and 48 h, p < 0.001; 2.53-fold at 250 μg/mL and 72 h, p < 0.001) (Figure 1(a)). In addition, Western blotting analyses revealed 2.3-, 3.4-, and 4.8-fold increases in PAR-2 protein expression in NHEKs treated with IS at concentrations of 50, 100, and 250 μg/mL over an incubation period of 48 h (p < 0.001) (Figure 2(a)).

RT-PCR was performed to analyze PAR-2 mRNA expression. PAR-2 mRNA expression increased in a dose-dependent and time-dependent manner in NHEKs stimulated with (a) IS and (b) PC. Data are presented as mean ± SEM. *p < 0.05; **p < 0.01; ***p < 0.001. RT-PCR: real-time polymerase chain reaction; PAR-2: protease-activated receptor-2; NHEKs: normal human epidermal keratinocytes; IS: indoxyl sulfate; PC: p-cresol; SEM: standard error of the mean.
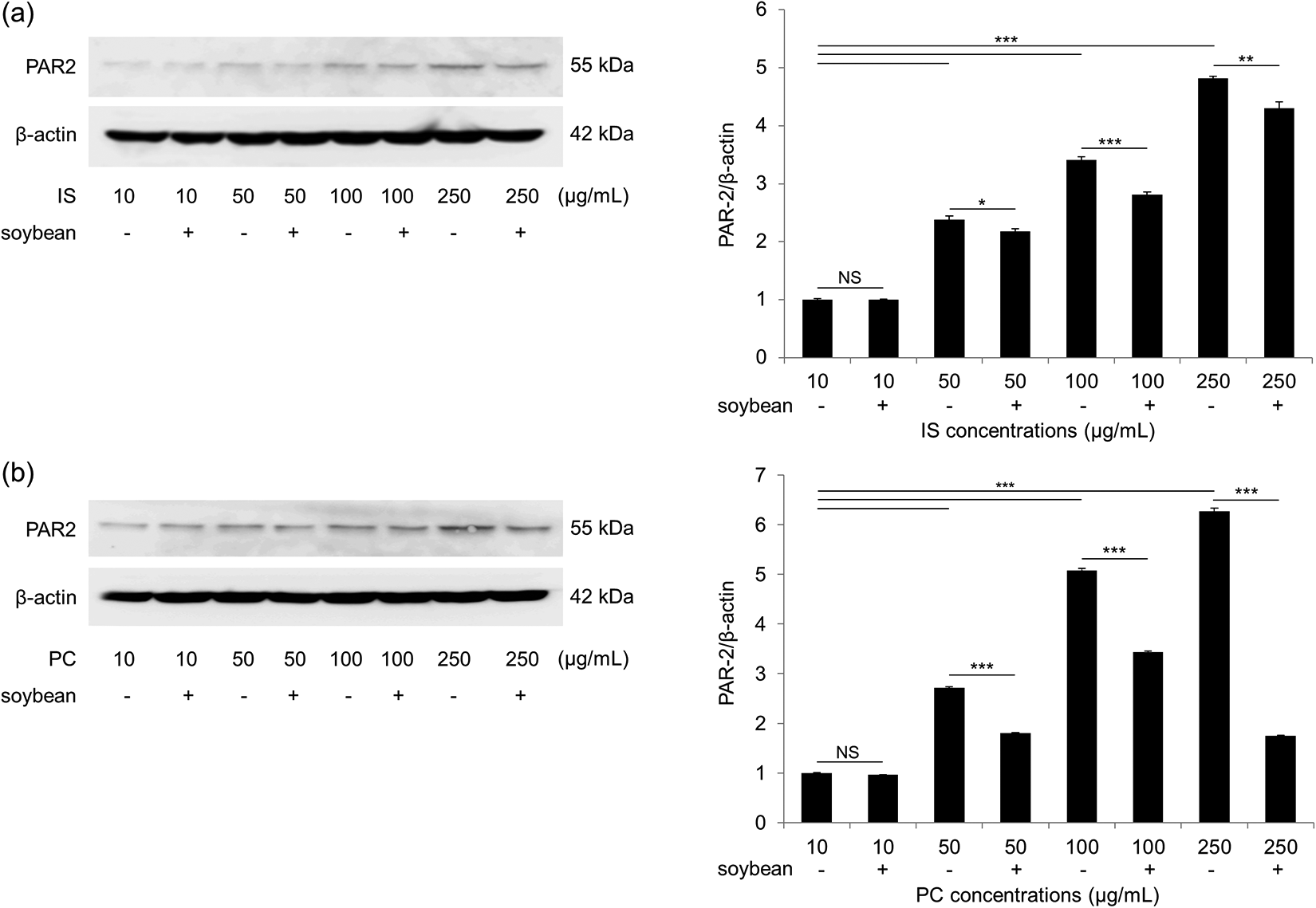
Western blotting for PAR-2 protein expression. Protein expression of PAR-2 was elevated in NHEKs stimulated with 50, 100, and 250 µg/mL of (a) IS and (b) PC for 48 h. Soybean trypsin inhibitor significantly inhibited PAR-2 protein expression. Data are presented as mean ± SEM. *p < 0.05; **p < 0.01; ***p < 0.001. NS: not significant; PAR-2: protease-activated receptor-2; NHEKs: normal human epidermal keratinocytes; IS: indoxyl sulfate; PC: p-cresol; SEM: standard error of the mean.
To evaluate the inhibitory effects of a protease inhibitor on uremic solute-induced PAR-2 expression, NHEKs in each experimental treatment group were additionally pretreated with soybean trypsin inhibitor over an incubation period of 48 h. Pretreatment of NHEKs with soybean trypsin inhibitor significantly inhibited PAR-2 protein expression (p < 0.05, p < 0.001, and p < 0.01, respectively) and mRNA expression (p < 0.01, p < 0.001, and p < 0.001, respectively) at IS concentrations of 50, 100, and 250 μg/mL (Figures 2(a) and 3(a)).

RT-PCR was performed to analyze PAR-2 mRNA expression after soybean trypsin inhibitor pretreatment. Pretreatment with soybean trypsin inhibitor significantly downregulated PAR-2 mRNA expression in NHEKs incubated with (a) IS and (b) PC for 48 h. Data are presented as mean ± SEM. *p < 0.05; **p < 0.01; ***p < 0.001. PBS: phosphate-buffered saline; ME: methanol; NS: not significant; RT-PCR: real-time polymerase chain reaction; PAR-2: protease-activated receptor-2; NHEKs: normal human epidermal keratinocytes; IS: indoxyl sulfate; PC: p-cresol; SEM: standard error of the mean.
Effects of PC on PAR-2 expression in NHEKs
As observed in IS-stimulated NHEKs, dose-dependent and time-dependent upregulation of PAR-2 mRNA expression was also recorded in NHEKs incubated with PC. Higher concentrations of PC, especially 50, 100, and 250 μg/mL, and longer incubation times, especially 48 and 72 h, led to significantly elevated PAR-2 mRNA expression (4.66-fold at 250 μg/mL and 48 h, p < 0.001; 5.09-fold at 250 μg/mL and 72 h, p < 0.001) (Figure 1(b)). In addition, Western blotting analyses showed 2.7-, 5.1-, and 6.3-fold increases in PAR-2 protein expression in NHEKs treated with PC at concentrations of 50, 100, and 250 μg/mL, respectively, after an incubation period of 48 h (p < 0.001) (Figure 2(b)). Pretreatment of NHEKs with soybean trypsin inhibitor also significantly suppressed PAR-2 protein and mRNA expression at PC concentrations of 50, 100, and 250 μg/mL (Figures 2(b) and 3(b)).
Effects of uremic serum on PAR-2 expression in NHEKs
To examine the effects of uremic serum on PAR-2 mRNA and protein expression, NHEKs were incubated with serum samples obtained from CKD patients with uremic pruritus and healthy controls for 12, 24, and 48 h. NHEKs treated with sera from CKD patients exhibited significant increases in PAR-2 mRNA at 48 h (CKD: 5.37 ± 0.08 vs. control: 2.46 ± 0.08, p < 0.001), but not at 12 and 24 h, compared to NHEKs treated with sera from healthy controls (Figure 4(a)). PAR-2 protein expression also increased significantly at 24 and 48 h in CKD sera-treated NHEKs, compared to those treated with sera from healthy controls (CKD: 1.80 ± 0.02 vs. control: 1.55 ± 0.02 at 24 h, p < 0.01; CKD: 2.08 ± 0.01 vs. control: 1.75 ± 0.01 at 48 h, p < 0.001) (Figure 4(b)). Pretreatment of NHEKs with soybean trypsin inhibitor significantly reduced both PAR-2 mRNA (p < 0.001) and protein expression (p < 0.01) after 48 h of incubation with healthy and CKD sera (Figure 5(a) and (b)).

RT-PCR and Western blotting were performed to analyze PAR-2 expression after serum incubation. The levels of PAR-2 (a) mRNA and (b) protein expression increased in NHEKs stimulated with 4% pooled sera from CKD patients with uremic pruritus compared with HC samples treated with 4% BSA, especially at 48 h. Data are presented as mean ± SEM. ANOVA was used for comparisons among groups. *p < 0.05; **p < 0.01; ***p < 0.001. NS: not significant; RT-PCR: real-time polymerase chain reaction; PAR-2: protease-activated receptor-2; NHEKs: normal human epidermal keratinocytes; CKD: chronic kidney disease; HC: healthy control; BSA: bovine serum albumin; SEM: standard error of the mean; ANOVA: analysis of variance.

RT-PCR and Western blotting were performed to analyze PAR-2 expression after soybean trypsin inhibitor pretreatment. Pretreatment with soybean trypsin inhibitor significantly reduced PAR-2 (a) mRNA and (b) protein expression in NHEKs after 48 h of incubation with sera from HCs and CKD patients. Data are presented as mean ± SEM. ANOVA was used for comparisons among groups. *p < 0.05; **p < 0.01; ***p < 0.001. NS: not significant; RT-PCR: real-time polymerase chain reaction; PAR-2: protease-activated receptor-2; NHEKs: normal human epidermal keratinocytes; CKD: chronic kidney disease; HC: healthy control; SEM: standard error of the mean; ANOVA: analysis of variance.
Effects of IS and PC on protease activity in NHEKs
To examine the effects of uremic serum on protease activity, NHEKs were stimulated with 100 μg/mL of IS or 50 μg/mL of PC for 12, 24, and 48 h. NHEKs treated with both IS and PC exhibited significant increases in protease activity at 12, 24, and 48 h (Figure 6(a) and (b)). These increases were considerably attenuated by pretreatment with soybean trypsin inhibitor (p < 0.01 to p < 0.001).

Protease activity after soybean trypsin inhibitor pretreatment. Pretreatment with soybean trypsin inhibitor significantly reduced protease activity in NHEKs incubated with (a) IS and (b) PC for 12, 24, and 48 h. Data are presented as mean ± SEM. *p < 0.05; **p < 0.01; ***p < 0.001. NS: not significant; NHEKs: normal human epidermal keratinocytes; IS: indoxyl sulfate; PC: p-cresol; SEM: standard error of the mean.
Cutaneous PAR-2 expression in CKD patients and CKD mice
Full-thickness skin samples from human CKD patients and from the experimental mice were stained to compare patterns of cutaneous PAR-2 expression. Skin samples from CKD patients exhibited marked increases in PAR-2 expression in the cytoplasm of epidermal cells compared to healthy control samples (Figure 7(a) and (b)). Notably, an increased granular pattern of PAR-2 expression was found throughout the entire epidermal layer. Meanwhile, just as in humans, skin samples from CKD mice exhibited upregulated expression of PAR-2 in the cytoplasm of epidermal cells compared to skin samples from control mice (Figure 7(c) and (d)).

Immunohistochemical staining for PAR-2 expression. Immunohistochemical staining for cutaneous PAR-2 expression in (a) CKD patients and (c) CKD male mice revealed marked increases in PAR-2 expression in the cytoplasm of epidermal cells, compared to that of HCs (b and d). Original magnification, ×400. PAR-2: protease-activated receptor-2; CKD: chronic kidney disease; HC: healthy control.
Discussion
Uremic solutes, which are retained in CKD patients, have deleterious effects on disease progression. 4,5 IS inhibits the proliferation and repair of endothelial cells and increases endothelial–leukocyte interactions. 4,15,16 In addition, IS and PC enhance epithelial-to-mesenchymal transition and the expression of cytokine and inflammatory genes in renal tubular cells. 17,18 In this study, we found that both IS and PC induced significant increases in PAR-2 expression in NHEKs in a dose-dependent manner. The stimulation of NHEKs with sera from CKD patients also generated increases in PAR-2 expression compared to cells treated with healthy control sera.
Proteases are regarded as hormone-like multifunctional signaling molecules under both physiological and pathophysiological conditions. 8,9 In the skin, trypsin-like serine proteases activate PAR-2, which is abundantly expressed in the epidermis and/or nerve fibers of patients with pruritic skin diseases, including atopic dermatitis and uremic pruritus. 11,12 A previous study reported significant increases in protease activity in human CKD skin, with the upper portion of the epidermis exhibiting significantly greater PAR-2 expression of higher intensity. 12 A uremic condition in CKD patients leads to upregulation of proteolytic activity and protein energy wasting that may result in increased PAR-2 expression in the skin in these patients. 12,19 How IS and PC regulate PAR-2 mRNA expression is, however, not fully understood, although several reports have suggested that IS and PC increase the expression of transcription factors. IS has been shown to upregulate the transcription factor GATA3 in human renal cell carcinoma cells, 20 and IS has been shown to activate NF-κB p65, followed by p53 expression, in renal proximal tubular cells. 21 Therefore, IS and PC could regulate PAR-2 gene expression by modulating transcription factors, although further studies are required to determine the precise pathways involved.
Cleavage and activation of PAR-2 are effectively inhibited by soybean-derived serine protease inhibitors. 22 In the present study, soybean trypsin inhibitor significantly inhibited PAR-2 mRNA and protein expression in NHEKs treated with IS, PC, and CKD sera. Although the serum samples from CKD patients and healthy controls contained many nonspecific inflammatory mediators that could have affected PAR-2 expression, CKD sera elicited a significantly greater upregulation of PAR-2 expression than did sera from healthy controls. Nevertheless, CKD sera-induced PAR-2 expression was effectively inhibited by soybean trypsin inhibitor. Accordingly, we suggest that IS, PC, and CKD sera may contribute to upregulating PAR-2 expression by directly or indirectly activating protease activity.
The understanding of uremic pruritus has progressed in several areas. Various mechanisms underlying uremic pruritus in CKD have been suggested, including uremic xerosis, secondary hyperparathyroidism, calcium phosphate crystals, uremic neuropathy, iron-deficiency anemia, and immune system alterations. 23 In addition, the regulation of abnormal itch sensations by the central nervous system has attracted attention, particularly the role of endogenous opioids in its treatment. Nalfurafine, a selective κ-opioid receptor agonist, has been introduced as a treatment option for uremic pruritus. 24,25 In addition, uremic pruritus has been linked to quality of life and survival. Several studies have documented relationships between uremic pruritus and quality of life, sleep quality, morbidity, and mortality. Pisoni et al. reported that patients with moderate to severe pruritus had poor sleep quality, depression, and lower mental and physical quality-of-life scores. 2,26,27 In addition, the authors showed that pruritus in hemodialysis patients was associated with higher mortality rates. However, treatments for uremic pruritus remain somewhat ineffective. Therefore, uremic pruritus should be regarded as an important health problem in CKD patients, and investigations of novel mechanisms underlying uremic pruritus may have significant implications for alleviating the symptoms of uremic pruritus in patients with CKD.
Currently, mouse models of uremia can be established with unilateral ureteral obstruction (UUO), 5/6 nephrectomy, adriamycin nephropathy, and coagulation. 13,28,29 UUO models are developed by unilateral ureter ligation, whereas 5/6 nephrectomy models are made by tying the upper and lower poles of the left kidney followed by right nephrectomy. 28,29 A CKD mouse model with adriamycin nephropathy can be developed by a single intravenous injection of adriamycin at a dose of 10 mg/kg. 28 These CKD mouse models, however, have several limitations; in UUO models, there is an aggressive progression of uremia, which may not fully reflect the human CKD condition, as well as compensation of renal function by the normal contralateral kidney. The 5/6 nephrectomy model can be strongly influenced by mouse strain background, affecting the severity of disease. In addition, mouse models developed with adriamycin nephropathy introduce adriamycin toxicity to other cells. 28 –30
In the present study, we developed our mouse model of uremia via a two-step procedure consisting of electrocoagulation of the entire surface of the right kidney and total left nephrectomy. We chose this model as it reportedly reflects both the decreased renal function and chronic complications in other organs associated with CKD, whereas the other three models, in which kidney dysfunction develops over a shorter period of 7–14 days, mainly focus on renal fibrosis and inflammation. 30 Accordingly, we believe that this mouse model may be more suitable for investigating uremic pruritus. Indeed, at 6 weeks after completing the two-step procedure, significant increases in PAR-2 expression were observed in the skin samples from the CKD mice, similar to those seen in skin specimens from human CKD patients.
The results of this study demonstrated that IS and PC uremic solutes significantly induce PAR-2 expression in NHEKs in a dose-dependent manner. In addition, we found that sera from CKD patients also stimulated increases in PAR-2 expression. Finally, we found that skin from human and mouse CKD subjects exhibit marked upregulation of PAR-2 expression throughout the epidermis, compared to their respective controls. Although the precise role of uremic solutes in cutaneous PAR-2 expression remains to be determined, our data suggest that uremic solutes either directly or indirectly affect PAR-2 and serine protease activity in skin from CKD subjects, potentially playing an important role in the pathogenesis of uremic pruritus.
Supplemental material
Supplemental Material, suppl_Figure_1 - Uremic solutes of indoxyl sulfate and p-cresol enhance protease-activated receptor-2 expression in vitro and in vivo in keratinocytes
Supplemental Material, suppl_Figure_1 for Uremic solutes of indoxyl sulfate and p-cresol enhance protease-activated receptor-2 expression in vitro and in vivo in keratinocytes by SJ Kim, X Zhang, SB Cho, CH Kim, HC Park and SJ Moon in Human & Experimental Toxicology
Footnotes
Author contributions
SJK and XZ contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a National Research Foundation of Korea grant funded by the Korea government (MSIP) [Grant No. 2014R1A1A1002439] and a research fund of Catholic Kwandong University International St. Mary’s Hospital [Grant No. CKURF-201601380001].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
