Abstract
This study aimed to evaluate the synergistic effects of both vanillin (V) and chitosan nanoparticles (CNPs) in alleviating hepatotoxicity, oxidative injury, and genotoxicity induced by
Keywords
Introduction
Aging is related to the incidence of Alzheimer’s disease and many disturbances in vital organs; the new trend in treatment by combination of some compounds is considered and giving hope for the treatment of the several consequences of aging. 1
Oxygen-free radicals are reactive species that are called reactive oxygen species (ROS); they are generated permanently in the living cells as a portion of regular metabolism. The excessive production of ROS has many deleterious effects to the living cells and has the toxic impacts on cells involved in aging diseases pathogenesis.
DG is used to induce aging in animals for some pharmacological studies. There are several criteria which revealed that DG can cause changes related to aging including the increment of ROS and the decrement of the antioxidant enzyme capacities. 3
DG induced high lipid peroxidation, decreased the antioxidant enzyme activity, and thus reduced the duration of life. 4
Mice injected with DG for 6–10 weeks display the impairment of cumulative and progressive learning and memory abilities, elevated brain ROS generation, and progressive deterioration and damages of the neurogenesis in the hippocampus. 5 Accumulative evidences suggest that the chronic injection of DG induces neurological impairment, speeds up the metabolic rate, declines the antioxidant enzymes’ activity, induces oxidative stress and damage, and then speeds up the aging process. 6
Vanillin is a natural compound and has a wide range of beneficial pharmacological and biochemical capacities. Vanillin is the major component of vanilla; it is extremely used as a flavoring agent in foods and drugs. 5
It has been shown to induce several chemopreventive properties, including antioxidant activity and anemia treatment and decreasing mutagenic sequences, 7 anticarcinogenic effect, and inhibition of cell invasion. 8
Chitosan has attracted more attention due to its biomedical activity. It has a wide range of biological activities, such as antitumor agent, 9 anti-inflammatory capacities, 10 and free radical scavenging activities. 11
Chitosan is a polysaccharide composing of various amounts of glucosamine, which are linked by glycosidic bonds. 12 Chitosan nanoparticles (CNPs) display more superior actions as CNPs have multiple immune-ameliorative effect, anticancer, and antimicrobial activities. Additionally, nanoparticles possess a potent surface warp, as compared to the large particles; this produces more decay pressure with an analogous increment in saturation solubility. 13 The elevated saturation solubility enhances an increment in concentration between cells in the intestine and the mesenteric circulation beneath.
Chitosan is a polysaccharide that containing different amounts of (1-4)-glycosidic bonds. 14 Chitosan has attracted more attention as a biomedical compound, as it motivates a broad diversity of biological activities, such as antitumor activities, 13 immunostimulating effects, 15 anti-allergic effects, 16 hemostatic agent, 17 anticoagulant effects, 10 hypocholesterolemic effects, 11 anti-inflammatory activities, 18 and free radicals (ROS) scavenging activities. However, chitosan is insoluble in water and it has high molecular weight. Its applications are roughly with limits. Nanoparticle formulation supplies a sensible pharmaceutical principle and bases for promoting bioavailability orally and therapeutic effectiveness of chitosan particles. Nanoparticles of chitosan have more activities and have elevated immunity and anticancer activity than chitosan large particles. Additionally, nanoparticles possess a stronger surface area; this produces pressure with solubility increment. 19
Moreover, incorporation of bioactive compounds such as antioxidant agents and probiotics into the nanoparticle coatings represents an innovative concept. 20 Vanillin is widely used in food, beverage, and pharmaceutical industries due to its functional properties. 21
Vanillin is a phenolic compound exhibiting antioxidant and antimicrobial properties against yeasts, molds, and bacteria. Additionally, the inhibitory effect of vanillin on the growth of different types of bacteria in five fruit-based agar systems significantly is effective on the bacterial growth rate. 21
The role of vanillin in combination with CNPs against DG, which mimic oxidative stress series in the hepatic tissues in case of aging toxicity, has not been studied until date. Their effect on the hepatic tissues was estimated and evaluated on the bases of assessment of some hepatorenal biomarkers, redox status assays, histopathology, and ultrastructural changes. So, the purpose of the current study was to evaluate the synergistic effects of V and CNPs in alleviating ROS, genotoxicity, and hepatotoxicity produced during aging and to investigate the prospect advantageous impacts and the synergistic effect between V and CNPs against DG by using the rat aging model experiment.
Materials and methods
Chemicals
Pure chitosan (C) (C6nH11nNO4n) was purchased from Shanghai Macklin Biochemical Technology Co., Ltd. (China); vanillin (V) and DG were obtained from Sigma Aldrich chemicals, while the other chemical obtained from Jiangsu Dehe Biotechnology Co., Ltd (China) with a purity of 98%.
Preparation of CNPs
Twenty milligrams of C was added to 40 mL (2%) (v/v) of acetic acid. Twenty milliliters of (0.75 mg/mL; Na5P3O10) sodium tripolyphosphate was dropped wisely and quietly with continuous stirring. CNPs were stored in deionized water. Supernatant was isolated and CNPs were well dried. 22
Physicochemical characterization of CNPs
Particles size and their morphology
CNPs size and morphology were analyzed by using Nanotrac analyzer. CNPs were slashed into diverse sizes. CNPs’ particles size was found within the range of 45–70 nm with spherical black spots, and the nanoparticles’ morphology was organized with Malvern Instruments Ltd.
Zeta potential
Zeta potential was analyzed by using Malvern Analyser (Zeta sizer Ver. 7.01 Serial Number: MAL1082067, UK) by illuminating CNPs solution in a sample cell.
Size distribution
Particle size was measured by Malvern Analyser under the following conditions: refractive index: 1.330; cP: 0.8872s: 60 mm: 4.65 years; material absorption: 0.010 and 25°C.
Animal model
Seventy adult male Wistar rats (3 month age) were used in the current study. All the experiments were proceeded under the animal ethics of caring the experimental animals. All the experimental animals were healthy and weighing 170–180 g. The animals were allowed to free access to standard diet and water
Experimental design
The experimental animals were divided into seven groups as follows: first control group was administrated distilled water; second group was given DG at a dose of 100 mg/kg for 60 days 23 as previously reported that chronic administration of DG induces alterations that mimic induction of aging in animals 24 ; third and fourth groups were treated with CNPs (LD) and CNPs (HD), which are 140 mg/kg and 280 mg/kg, respectively, as previously reported by El-Denshary 25 ; fifth group was administrated V (100 mg/kg) 26 ; and sixth and seventh groups were administrated DG plus CNPs (LD) + V and DG plus CNPs (HD) + V, respectively. All the treated groups were treated intraperitoneally for 60 successive days. DG administration in intraperitoneal route will cross the blood–brain barrier and then make aging effect better than oral administration, as an ideal aging and Alzheimer’s disease rodent model capable of mimicking pathological alterations in aging. 26
Tissue homogenates preparation for measurement of redox state
Liver tissue portions (∼0.45 g) were used for the oxidative stress marker analysis. Hepatic tissues were perfused with buffer phosphate saline (50 mM, pH = 7.4) and then we added a protease inhibitor to protect the hepatic antioxidant enzymes which are very sensitive to the oxidation.
Oxidative and antioxidant biomarkers activity
Malondialdehyde (MDA) levels were evaluated according to Ohkawa et al. 27 Superoxide dismutase (SOD) activity was evaluated by Marklund and Marklund. 28 Catalase (CAT) was estimated by Aebi. 29 Glutathione (GSH) was evaluated by Couri and Abdel-Rahman. 30 Glutathione peroxidase (GPx) was evaluated by Hafeman et al. 31 Myeloperoxidase (MPO) was evaluated by Suzuki et al. 32 Xanthine oxidase was evaluated by Litwack et al. 33 Total thiols were assayed by Hu. 34
TNF-α and IL-6 activities in the liver homogenates
Cytokines levels of interleukin-6 (IL-6) and tumor necrosis factor alpha (TNF-α) were evaluated in the hepatic tissues homogenate by using enzyme-linked immunosorbent assay (ELISA) kit that wase obtained from Immuno-Biological Laboratories, Minneapolis, Minnesota. USA.
Lipid profile
Total cholesterol (TC) and triglycerides (TG) were estimated by Carr et al. 35 High-density lipoprotein (HDL-c) was estimated by Warnick et al. 36 Low-density lipoprotein (LDL-c) level was evaluated by Friedewald. 37 Protein levels were measured by Bradford. 38
Liver function biomarkers
Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) enzymes were evaluated according to Reitman and Frankel. 39 ALP was measured according to Young et al. 40 by using commercial kit “Biodiagnostic kits.” Lactate dehydrogenase (LDH) was determined by Vassault 41 by using “BioVision Colorimetric Assay Kit”, Milpitas, California, USA.
Inflammation marker (CRP) activity
C-reactive protein (CRP) levels were evaluated according to Wener et al. 42 by using SEA821-ELISA kit for CRP.
Mitochondrial function
Evaluation of SDH (complex II) activity
Metabolic viability-based assays are used for measuring the mitochondrial metabolic rate and reflect indirectly the number of viable cells. The mitochondrial succinate dehydrogenase (SDH) activity was evaluated also. The absorbance was measured using an ELISA kit at 570 nm (Tecan, Rainbow Thermo, Austria). 43
Mitochondrial ROS assay
The mitochondrial ROS generation was evaluated by using the dichlorodihydrofluorescein diacetate probe. An increase in fluorescence intensity indicates an increase in ROS generation due to exposure to inflammation of hepatotoxicity. 44
MMP (ΔΨ
m) assay
The mitochondria of the hepatic tissues were isolated from all groups. Briefly, the mitochondrial ROS generation was evaluated by using rhodamine 123 (Rh123) probe at a final concentration of 10 μM for 30 min at 30°C. An increase in fluorescence intensity indicates an increase in the collapse of mitochondria membrane potential (MMP). 45
Fluorescent detection of MMP
Animals after they were euthanized, hepatic tissues were washed thoroughly and rinsed with ice. They were gently blotted between the folds of a filter paper and weighed in an analytical balance. Homogenate (10%) was prepared in 0.05 M phosphate buffer (pH 7) by using a polytron homogenizer at 40°C according to manual instructions. Fluorescent kit cannot be measured by using a fluorometer microplate reader (Thermo Fisher Scientific Oy, Vantaa, Finland). MMP was expressed as JC-1 fluorescence ratio (J-aggregates:J-monomers).
Evaluation of swelling on mitochondria
Briefly, swelling of mitochondria in sizes of 10 and 100 nm (pure and impure) was measured by using an ELISA reader (Tecan, Rainbow Thermo) at 540 nm. A decrement in absorbance indicates an increment in mitochondria swelling. 45
Cytochrome c oxidase release assay
Evaluation of cytochrome c release that referred to mitochondrial volume was measured (Rat/Mouse cytochrome c Immunoassay kit, Minneapolis, MN, USA).
ATP content assay
The adenosine triphosphate (ATP) content was detected by using the luciferase enzyme. The intensity was evaluated by using Sirius tube luminometer (Berthold Detection System, Germany). ATP content was expressed as nmol/mg protein. 46
Histological examination and TEM evaluation
Liver portions were fixed in about “10% neutral buffered formalin” and then other histological processing were carried out as described previously by Gabe. 47
For ultrastructural examination, the hepatic tissues were fixed in 2.5% glutaraldehyde for about 48 h and then the other transmission electron microscopy (TEM) processing according to Weakley and Beginner. 48
Single-cell gel electrophoresis (comet assay)
Liver tissues were placed in a small Petri dish with an ice-cold solution (Ca2+and Mg2+and 10% dimethylsulphoxide and ethylenediaminetetraacetic acid). The cell viability was examined by using the comet images after the electrophoresis. 49 The comet assay was described previously by Collins and Dunsinka. 50
Statistical analysis
Data are expressed as “mean ± standard error.” Statistical analysis was performed by using analysis of variance. The statistical significance was set at
Results
TEM of CNPs
The micrograph showed the CNPs in the spherical form (Figure 1).

TEM images of biosynthesized CNPs (scale bar = 50 nm). TEM: transmission electron microscopy; CNP: chitosan nanoparticles.
Zeta potential measurement
It is an assessment technique of the surface charge of particles in the solution, which is directly related to the stability of the nanoparticles suspension. Figure 2 showed that the zeta potential measurement of CNPs was found to be −14.9 mV. This value revealed the high stability of the prepared CNPs.
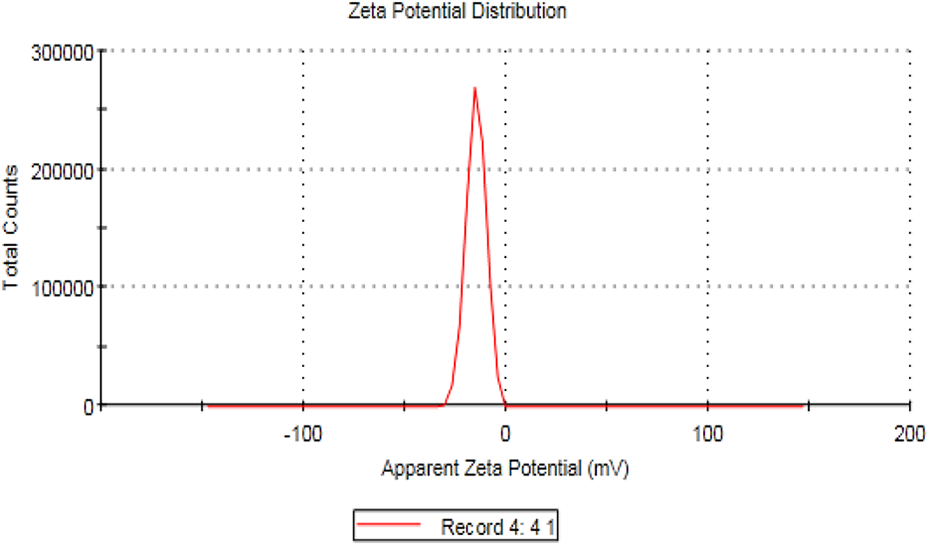
Zeta potential distribution of CNPs. CNP: chitosan nanoparticle.
Size distribution report by intensity
Figure 3 showed that the mean particle size of CNPs was found to be in the nano-range of the synthesized particles (ranged from 54 nm to 85 nm).

Size distribution of CNPs, Pdi = 0.9. CNP: chitosan nanoparticle; Pdi: polydispersity index.
Mortality rate and behavioral changes
It was noticed that rats treated with DG showed signs of dementia by hesitated motions and unconcentrated conscious actions.
During eating or drinking water, animals were not able to recognize the opening of drinking bottle with state of laziness almost the time, on the contrary to the rats treated with CNPs in two doses and combined groups. There was no mortality in all the treated groups.
Hepatic biomarkers and lipid profile
The ALT, AST, ALP, and LDH activities were significantly elevated in DG-treated group (Table 1), while these levels were significantly declined in the groups treated with DG in combination with either CNPs (LD) and (HD) or V and restored to about control levels except total proteins which were declined in the DG group and elevated in other treated groups with DG and CNPs (LD) and (HD) with V.
Changes in hepatic enzymes and biomarkers in male rats treated with DG, LD and HD CNPs, and/or V on male rats.a

ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; LDH: lactate dehydrogenase; DG:
a Values are expressed as means ± SE;
The rats treated with DG caused a significant increase in TG, TC, LDL-c, and very-low-density lipoprotein (vLDL-c) with declining HDL-c level as compared to the control group (Table 2). While these levels were equal to the levels of control group in CNPs (LD) and (HD) and V groups as well as declining these levels in groups treated with DG in combination with CNPs (LD) and CNPS (HD) and V with elevating of HDL-c levels. So, there was a synergistic effect between CNPs in two doses especially CNPs (HD) and V in improving the lipid profile picture and acting as hypocholesterolemic agents.
Changes in lipid profile in male rats treated with DG, LD and HD CNPs, and/or V on male rats.a
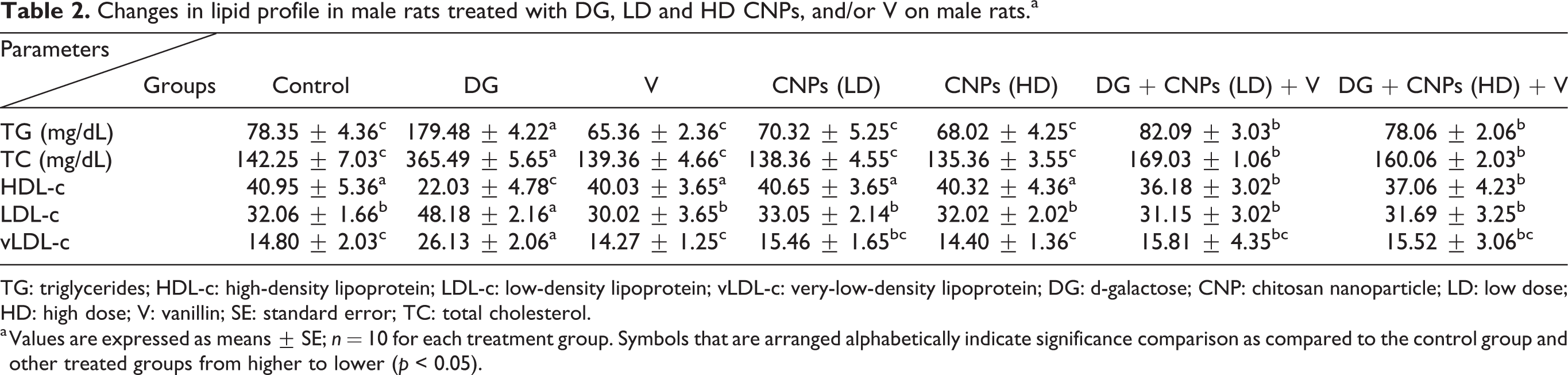
TG: triglycerides; HDL-c: high-density lipoprotein; LDL-c: low-density lipoprotein; vLDL-c: very-low-density lipoprotein; DG: d-galactose; CNP: chitosan nanoparticle; LD: low dose; HD: high dose; V: vanillin; SE: standard error; TC: total cholesterol.
a Values are expressed as means ± SE;
Tumor necrosis marker, IL-6, and CRP level
There were no changes in the levels of TNF-α and IL-6 in the rats that were treated with CNPs (either LD or HD) and V with DG compared to the control group (Table 3). The elevation of the inflammatory cytokines was observed in the DG group. The anti-inflammatory effect was noticed in the combined group treated with DG in combination with CNPs (LD) with V and CNPs (HD) with V, respectively.
Changes in serum TNF-α, IL-6, and CRP levels in male rats treated with DG, LD and HD CNPs, and/or V on male rats.a

TNF-α: tumor necrosis factor alpha; IL-6: interleukin-6; DG:
a Values are expressed as means ± SE;
Table 3 presents the effect of DG alone and in combination with CNPs (LD or HD) and V on CRP of hepatic tissue. The CRP levels were increased by 6.1-fold in the DG group as compared to the control group. The decrease of CRP in DG plus CNPs (LD) and V and DG plus CNPs (HD) and V as compared to its related group of DG alone.
Oxidative stress biomarkers
The SOD activity was declined significantly in DG-treated group (Table 4) as well as CAT and GPx activities which were also declined in DG-treated group and did not change in other treated groups with V and CNPs (LD) and (HD).
Effect of with DG, LH and HD CNPs, and/or V on antioxidant markers in liver homogenates in male rats.a

SOD: superoxide dismutase; MDA: malondialdehyde; GPx: Glutathione peroxidase; CAT: catalase; DG:
a Values are expressed as means ± SE;
There was a significant increment in MDA activity in the rats treated with DG. No critical changes were recorded in MPO levels of all treated groups except the DG-treated animals (Table 4). MPO activities were increased in DG-treated group as compared to the control group by 1.8-fold as well as in DG combined with V and/or CNPs (either LD or HD) as compared to its relative group of DG by 2.1- and 1.8-fold, respectively (Table 4).
GPx levels were declined significantly in DG-treated group by 64.76% as compared to control rats (Table 4). In both treatment, doses of CNPs and V with VDG. GPx was slightly significantly decreased at the lower dose and nonsignificantly decreased at the higher dose as compared to the control group.
In the DG group, the hepatic thiol activity declined by 2.1- fold as compared to the control group (Table 5). In the CNPs in two doses either LD or HD with V and combined with the DG group, the hepatic thiol activity decreased by 34.28 and 5.55%, respectively as compared to the control group. While MPO level was significantly elevated in DG-treated group while declined in the other groups treated with DG in combination either with CNPs (LD or HD) or with V (Table 5). So, there was a synergistic effect between CNPs in two doses especially CNPs (HD) and V and acting as antioxidant agents.
Changes in serum hepatic MPO antioxidant enzymes and thiol level in male rats treated with DG, LD and HD CNPs, and/or V on male rats.a
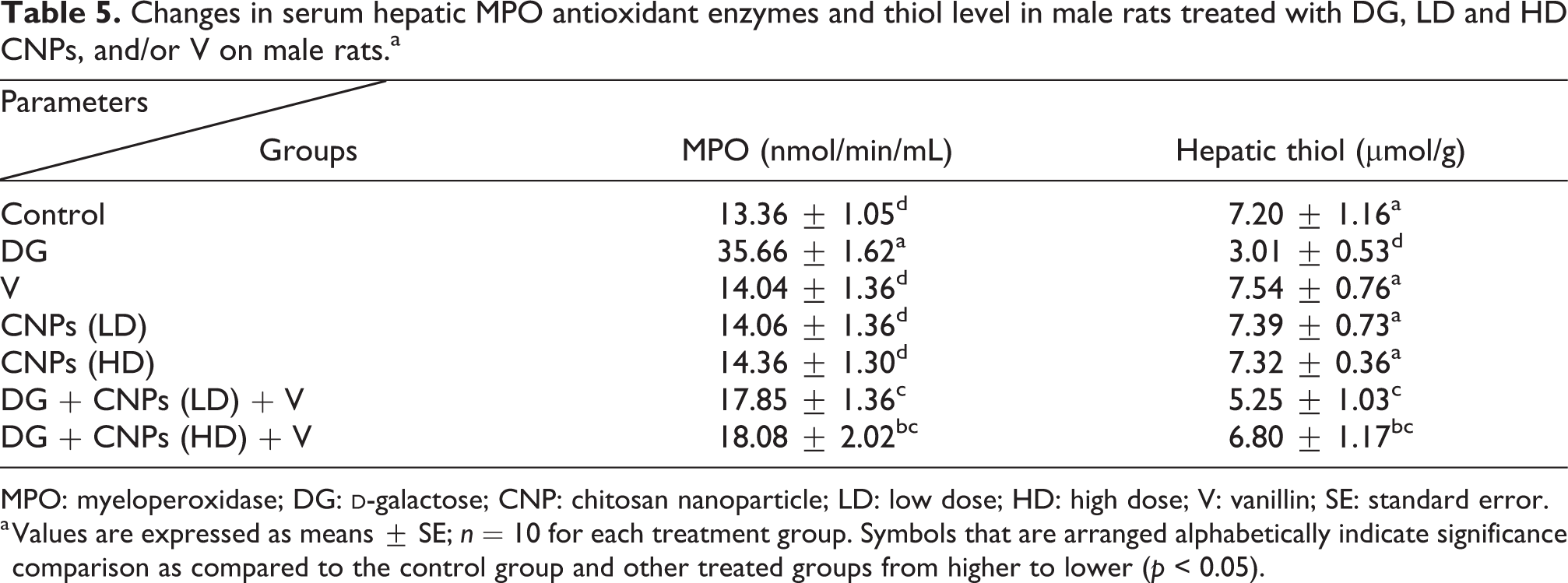
MPO: myeloperoxidase; DG:
a Values are expressed as means ± SE;
SDH, ROS, and MMP
The ROS increased significantly in the DG group by 3.5-fold as compared to the control group (Figure 4). The animals treated with CNPs in two doses (LD and HD) or with V or both of them lowered the ROS by 50.7, 56.9, and 74.8% as compared to DG-treated rats, respectively.

The efficacy of V and CNPs in two doses LD and HD against SDH and ROS in the rat model of aging induced by DG. ROS: reactive oxygen species; CNP: chitosan nanoparticle; DG:
The cell viable number detected by SDH percent as presented in Figure 4. The SDH was 93.4% in the control group and decreased significantly to 54% in DG-treated rats, while the groups treated with DG combined with CNPs (LD and/or HD) and V elevated the percent of SDH to about 82 and 87%, respectively.
The baseline value of ROS for normal healthy rats was 18 ± 1.5 as shown in Figure 4. The aging rats presented higher ROS content than controls for mitochondria (5.4-fold). The ROS generation decreased by 67.1% in CNPs (LD and HD). More reduction in ROS contents was observed in V and CNPs (LD) and CNPs (HD) and in combination with DG 71.6 and 73.4%, respectively.
On day 60, the effects of chitosan or/and vanillin on MMP (Δ

The efficacy of V and CNPs in two doses LD and HD against MMP in the rat model of aging induced by DG. CNP
Aging accelerated mitochondrial swelling as illustrated in Figure 6, where it was elevated as compared to control healthy rats. The swelling diminished for either CNPs either LD or HD either alone or in combination with V, respectively.

The efficacy of V and CNPs in two doses LD and HD against swelling mitochondria (mg/m3) and cytochrome C release (ng/mg protein) in the rat model of aging induced by DG. CNP
Aging rats release less ATP content as compared to the control rats (Figure 7). CNPs (LD and HD) treatment increased the ATP content as compared to DG animals. Similarly, CNPs (LD and HD) with V in combination with DG treatment elevated ATP content, separately. So, there was a synergistic effect between CNPs in two doses especially CNPs (HD) and V and acting as energy promoters and oxidative injury inhibitors.

The efficacy of V and CNPs in two doses LD and HD against ATP content in the rat model of aging induced by DG. CNP
TEM evaluation
Electron micrograph of liver sections (Figure 8): (a) control group showing the normal hepatocytes with normal condensed nucleus and normal-sized mitochondria and normal endoplasmic reticulum (ER; 5 µm). (b) DG-treated group showed pyknotic disintegrated nuclei with degradation of most hepatic structures with foamy cytoplasm and severe fatty change (5 µm). (c) to (e) V- and/or CNPs (LD and HD)-treated groups had normal appearance of hepatic structures with normal nucleus, normal mitochondria, and normal ER with few vacuoles in the cytoplasm of CNPs (5 µm). (f) and (g) DG group treated with thecombination of CNPs (LD) and V showed restoration of most hepatic tissues structure with the appearance of normal nuclei with regular boundaries with moderate-sized mitochondria and normal hepatic structures (5 µm).

Electron micrograph of liver section:
Histopathology evaluation
Photomicrographs of the hepatic tissues (Figure 9) of (a) control group: hepatic tissues were normal in appearance with normal central vein (CV) and hepatic lobule (×400). (b) DG-treated group showed markedly dilated CV lies at the center of the lobule surrounded by the hepatocytes. Pericentral zone shows necrotic hepatocytes with portocentral fibrosis (black-headed arrow; ×400). (c) to (e) Groups treated with either V or CNPs (LD and HD) showed normal hepatic structures with normal CV and normal hepatic lobules (×400). (f) and (g) DG group treated with either CNPs (LD or HD) or V showing the restoration of normal hepatic structures and very mild congested CV with the removal of fibrosis which appeared markedly in DG-treated group (×400). Histological activity index was assessed based on the degree of microscopic lesions in hepatic tissues as recorded in Table 6.

Photomicrographs of (a) control group: showing normal hepatic structures with normal CV (×400). (b)
HAI was assessed based on the degree of microscopic lesions in hepatic tissues as the effect of DG, LD and HD CNPs, and/or V separately or in combination in male rats.

HAI: histological activity index; −: no found; −−−+: found in 1–3 rats; ++−−: found in 4–6 rats; +++−: found in 6–7 rats; ++++: found in 7–8 rats at least; CNP: chitosan nanoparticle; V: vanillin; DG:
The protective effect of LD and HD CNPs and/or V separately or in combination against oxidative DNA damage in the liver (tail length, DNA percent, and tail moment) and apoptotic cell population (apoptosis percent) induced by DG.a
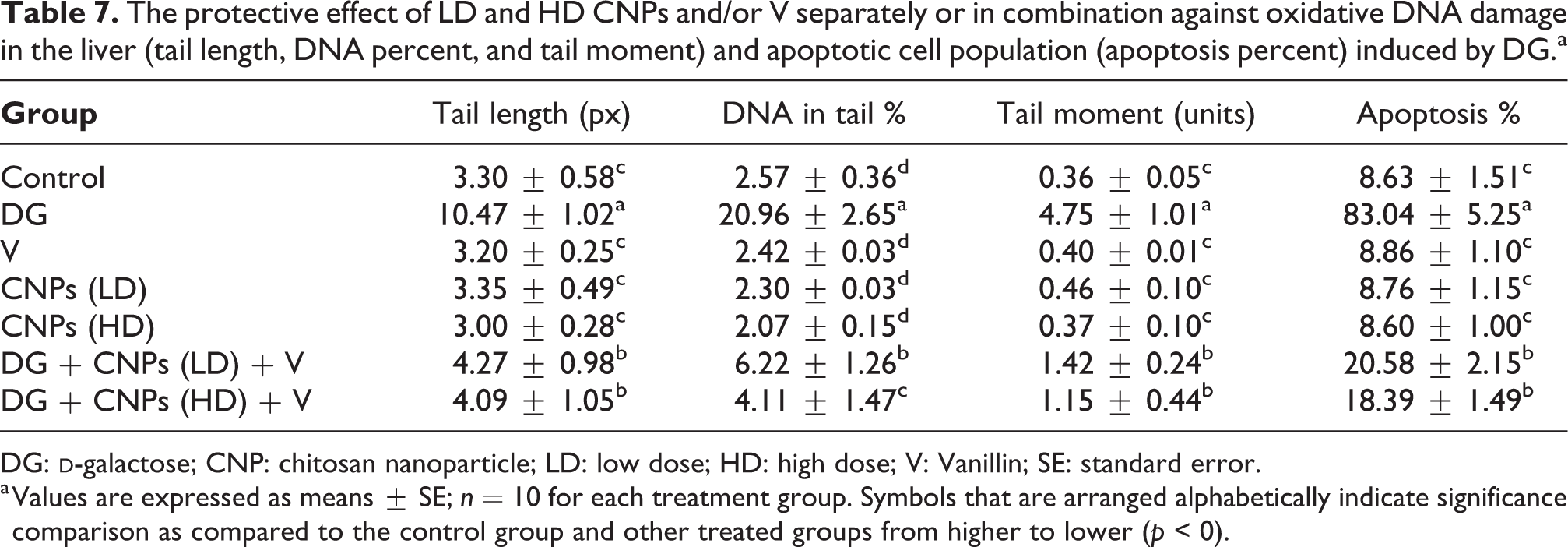
DG:
a Values are expressed as means ± SE;
Comet assay
Comet images of cells derived from the hepatic tissues of (a) control group showed normal rounded cell and intact nuclei without any appearance of tail. (b) DG group showed a higher degree of damages and injuries with the appearance of two apoptotic cells with a small head and large tail as a comet-shaped structure. (c) to (e) V- or CNPs (LD) and (HD)-treated groups showed more intact cells with undamaged DNA. (f) and (g) DG combined groups CNPs (LD and/or HD) with V showed high amelioration in the hepatic tissues as previously recorded by fewer tail length and percent of damaged DNA as shown in Figure 10 and the percentages of tail length and DNA damage were shown in Table 7.

Comet images of cells derived from the hepatic tissues of (a) control group which showed intact nuclei. (b) DG-treated group showing a high percent of apoptosis by tall shadow of nuclei. (c) to (e) Groups V and CNPs (LD and HD) showed more percent of intact nuclei without any comet. (f) and (g) Combined groups treated with DG in combination of CNPs (LD) with V and CNPs (HD) with V showed high percent intact nuclei with very percent of comet like the pinpoint. DG:
Mitochondrial membrane potential
The fluorescent detection of MMP showed high improvement on the hepatic tissues treated groups. (a) and (c) to (e) Treated groups with either V or CNPs (LD or HD) showing nonaccumulation of ROS in mitochondrial (50.0 µm). (b) DG-treated group showing high accumulation of ROS in the mitochondria, because ROS accumulation is a hallmark of oxidative cell death (50.0 µm). (f) and (g) Treated groups with DG in combination with either V or CNPs (LD) and/or (HD) showing high amelioration in mitochondrial potential with very low ROS accumulation (50.0 µm; Figure 11).

The fluorescent detection of MMP showed high improvement on the hepatic tissues treated groups. (a) and (c) to (e) Treated groups with either V or CNPs (LD or HD) showing nonaccumulation of ROS in mitochondrial (50.0 µm). (b) DG-treated group showing high accumulation of ROS in the mitochondria, because ROS accumulation is a hallmark of oxidative cell death (50.0 µm). (f) and (g) Treated groups with DG in combination with either V or CNPs (LD) and/or CNPs (HD) showing high amelioration in mitochondrial potential with very low ROS accumulation (50.0 µm). DG:
Discussion
The characterization of CNPs used in the current study against DG toxicity showed that CNPs have spherical shape with low molecular weight with high degree of stability. Zeta potential of CNPs is an assessment technique of the surface charge of particles in the solution and it is directly related to the stability of the nanoparticles suspension. When the zeta potential values are less than (more negative) −20 mV or greater than +20 mV, the particles are characterized by high stability; zeta potential of CNPs is −14.9 mV and thus indicates its stability. This resulted in facilitating the process of coupling with other molecules such as V and make high synergistic activity against aging hepatotoxicity.
The current in vivo study revealed the hepatoprotective effect of CNPs either alone or combined with V against the oxidative stress injury, hepatotoxicity, and genotoxicity that induced by DG in the rat model. As a solution for the problem of C poor water solubility, CNP provides a suitable pharmaceutical basis for promoting of the oral bioavailability and the therapeutic efficacy of C and other drugs that are poorly soluble in water. 19
Additionally, the nanoparticles possess a potent and stronger surface curvature; this may help in the production of more dissolution pressure with analogical increment in the saturation solubility. 51 The elevated saturation solubility aligns an increment in concentration gradient between the intestinal epithelial cells and the blood circulation. CNPs exhibit more activities than C due to their small size and proportion size effect. So, it is expected that CNPs have enhanced immune system, 52 anticancer activity, 53 antimutagenic activity, 54 and antimicrobial activity 55 than those of C.
The antioxidant capacity of CNPs has received more less attention. In the current study, treatment with DG and CNPs in two doses showed marked improvements in the hepatic biomarkers of toxicity, antioxidant enzymes, oxidative stress markers, genotoxicity alleviation of tumor markers, and inflammatory markers as well as the significant and marked improvement in the histological picture of the hepatic tissues. These findings are in harmony with Wen et al., 56 who reported a protective role of CNPs with a mean diameter of 83.66 nm against hydrogen peroxide-induced RAW-264.7 cell injury and damage via restoring the endogenous antioxidant enzyme activities of (SOD, GPx, and CAT) along with the raising of their gene expression.
In the current study, the liver has been selected in this study due to its more insightful to the oxidative stress as it contains the suitable concentration of antioxidant enzymes; it also needs high energy as well as contains the high cellular lipids and proteins levels. The damages in this vital organ generate excessive free radicals that can harm the proteins, lipids, as well as the other cellular contents as nucleic acids at the particle deposition location.
DG induced oxidative stress pathways in the brain and liver and this could be represented by elevated hepatic enzymes and marker of damages as shown in the present investigation where the levels of LDH, AST, ALT, and ALP were elevated with the elevation of lipid profile markers as TG, TC, LDL-c, and vLDL-c levels, elevating inflammatory marker CRP and tumor marker TNF-α. The use of DG-elicited histopathological and ultrastructural changes in the hepatic tissues and ROS excessive formation, which confirmed by declining the antioxidant enzymes activities of SOD, CAT, and GPx, declines the GPx and thiol levels as well as elevating marker of lipid peroxidation (MDA).
The CNPs hepatoprotective effect has been reported previously in some reports; for example, Al-Eisa
1
demonstrated the antioxidative effect of CNPs combined with lycopene on DG-induced hepatic damage and reported that CNPs have potent antioxidative effects, which decline the production of the free radicals and elevate antioxidant enzymes (CAT and SOD) activities during DG treatment. In the same concern, CNPs may stabilize the cellular membranes and may prevent a leakage of intracellular enzymes into the blood. Thus, the overall hepatoprotective effect of CNPs may be due to a counteraction of free radicals by its antioxidant nature and/or to its ability to inhibit lipid accumulation by its antilipidemic property.
57,58
Santhosh et al.
59
showed that cotreatment with chitosan may prevent
In the present study, treatment with CNPs succeeded by the tested two doses to improve the antioxidant capacities and decline the oxidative stress as indicated by the increment in the antioxidant enzymes (CAT, SOD, and GPx) and the decrement of MDA levels. Treatment with CNPs could significantly lower the high serum level of the hepatic enzymes (ALT, AST, and ALP) as well as decrement the increased levels of tumor marker (TNF-α). Moreover, CNPs succeeded to improve the histological changes that resulted in the hepatic tissues of DG-treated rats. Elevation of hepatic enzymes as ALT and AST enzymes reflects generalized damages and injury to the hepatocytes while the increment of ALP reflects cholestasis. 60
In another study, they reported also that increment of ALT and AST reflects hepatocyte damages and the increment of ALP refers to cholestasis, 61 which is in complete agreement with the current obtained results. The elevated levels of ALT, AST, and ALP in the current study in DG-treated group indicated the incidence of severe hepatic damages and cellular injuries.
Ragab et al. 62 demonstrated that orally administered DG elevated MDA and diminished the GSH levels in brain tissue. The increment in MDA level in the hepatic tissues showed that the DG group proved the increment in lipid peroxide levels resulting in hepatic tissue damage and mitochondrial dysfunction due to the excessive free radicals formation and thus injuring the antioxidant capacity system. These findings confirmed the histopathological, ultrastructural, and mitochondrial potential membrane activity alterations that are found in DG-treated group. These results are consistent with Al-Eisa, 1 who confirmed that DG induced sever hepatic congestion and dilation of CV with the appearance of pyknotic nuclei in both TEM and comet assay investigation.
CRP is a protein made by the liver. CRP levels in the blood increase when there is a condition causing inflammation somewhere in the body. A CRP test measures the amount of CRP in the blood to detect inflammation due to acute conditions or to monitor the severe disease in chronic conditions. 63 In the current study, CRP was significantly elevated in DG-treated group as compared to the normal control group and was declined in the groups treated with CNPs (LD or HD) combined with V.
DG is a reducing sugar and can be converted into either aldose or hydroperoxide, resulting in anion superoxide and ROS production, such as H2O2 and O. 2 radicals. 64 This finding explained the high TC, TG, and LDL-c levels of DG-treated group with declining of antioxidant enzyme activities (SOD, CAT, and GPx) with elevating the final marker of lipid peroxidation (MDA).
Ahangarpour et al. 65 reported that systemic administration of DG induced significant elevation in TG, LDL-c, and v-LDL-c, which is greatly supported the current finding of the present study and confirming the damaging effect of DG on lipid profile biomarkers.
Ahangarpour et al. 65 reported that regular DG administration triggers ROS formation in mitochondria and promotes the formation of advanced glycation end (AGE) products. Over-expressed AGE activates nuclear factor kappa B and other inflammatory mediators that may be associated with the metabolic disorders, aging, and age-related disorders and diseases, especially affecting the liver. ROS, SDH, MMP, and swelling mitochondria were severely increased due to the administration of DG with declining of ATP contents and this confirmed the ameliorative role of CNPs (LD and HD) in combination with V in retrieving most of these parameters and reducing the triggering of ROS accumulation.
There were no available data in the literature about the effect of V in combination with CNPs (LD and HD) on the liver. The mixture has elevated the activities of SOD, CAT, and GPx and of the hepatic tissues than that the effect of DG alone. Also, this mixture has the ability to adjust MPO and thiol levels than DG.
Nanoparticle formulation of chitosan supplied a suitable pharmaceutical basis for ameliorating of the therapeutic efficiency and oral availability of chitosan. Additionally, nanoparticles own a potent and stronger surface and this triggers more dissolution pressure. 19 Al-Eissa 1 found that CNPs have ameliorated antioxidant capacities of the treated rats as reduced the marker of lipid peroxidation (MDA) and improved the hepatic function enzymes and the hepatic structures of treated animals, which are all confirmed in the current study.
The chitosan hepatoprotective effects may be due to ROS and free radicals formation avoidance by its antioxidant capacities and its ability to inhibit lipids accumulation by its anti-lipidmic characters. 66 Santhosh et al. 59 demonstrated that chitosan coadministration may inhibit the hepatotoxicity elicited by drugs in male rats.
Abd-Elhakeem et al. 67 found that CNPs succeeded greatly to afford significant ameliorating effects in all the biochemical parameters of the hepatic tissues and they concluded that CNPs induce high protection against hepatotoxicity and it is considered as a promising candidate as a drug in the hepatic diseases’ treatments, which is greatly consistent with the obtained results.
Moreover, Virginia et al. 68 found that quercetin was encapsulated in chitosan/alginate nanoparticles revealed a good safety profile. These results suggested that the encapsulation of chitosan/alginate nanoformulations might represent an effective therapeutic approach against oxidative stress-induced liver injury as proved in the current result study. This opens the gate to our new treatment strategy by using CNPs and V, which showed high synergistic action against the hepatic injury especially HD of CNPs with V which showed great improvements in all hepatic enzymes, lipid profile, declining tumor marker, inflammatory marker, declining DNA damage, elevating antioxidant enzymes, and declining marker of lipid peroxidation in addition to improvement of hepatic histological structures.
V is a compound that is widely used in foods, beverages, cosmetics, and drugs and it has been reported to exhibit multifunctional effects such as antimutagenic, antiangiogenetic, and antianalgesic effects. However, Tai et al. 69 proved the antioxidant capacities of vanillin by different methods.
The high level of vanillin intake from foods and beverages appears to exert some effects on human health. 69 They proved that vanillin showed stronger activity than did ascorbic acid and Trolox in the scavenging assay. Thus, the oral administration of vanillin increased the antioxidant activity. Thus, the antioxidant activity of vanillin might be more beneficial than has been thought for daily health care.
The antioxidant, antigenotoxicity, and anti-inflammatory effects of vanillin are considered as the important forces in the protection against the hepatic damage and fibrosis. Makni et al. 70 investigated the protective effects of vanillin against “CCl4” that induced hepatotoxicity in male rats. Treatment with V prior to the administration of CCl4 markedly prevented the decrement of protein synthesis and the increment in ALT, and AST levels. Additionally, V inhibited hepatic marker of lipid peroxidation (MDA) and alleviated CCl4-mediated decrement of antioxidant enzyme CAT and SOD activities and GSH in the hepatic tissues. Additionally, V significantly alleviated the expression levels of cytokines as TNF-α, IL-1β, and IL-6 and alleviated CCl4-induced hepatic alterations and necrosis, as indicated by liver histopathology. These findings suggest that the antioxidant and anti-inflammatory effects of V against DG-induced hepatic damage may involve its ability to block DG-generated free radicals.
The results of the current study coincide with previous studies showing also that V effectively inhibits the expression of the inflammatory cytokines, including TNF-α, IL-6, and aminotransferase hepatic enzymes, subsequently inhibiting the downregulating mediators of inflammation and its effects is very important for attenuating the hepatic damage and liver fibrosis. 71
Additionally, Saad et al. 72 demonstrated that vanillin treatment alleviated the renal toxicity of cadmium and restored the biochemical parameters to near control values. Also, vanillin proved to possess a stronger scavenging radical activity than vitamin C as well as its capacity to protect genes expression and histopathological changes and the finding of the current study supported all these findings and explain the great role of vanillin as potent antioxidant agent, mitigate the hepatic damage effect of DG, restore most of the parameters to normal levels, give high synergistic effect when combining with CNPs, and prevent the accumulation of ROS as appeared clearly in the results of the current study.
The previous finding supports the highly synergistic effect between CNPs and V in scavenging free radicals, declining MMP, elevating antioxidant enzymes, declining marker of lipid peroxidation (MDA), declining tumor and inflammatory markers, declining hepatic genotoxicity, and improving hepatic cellular structures, as clarified by histopathological investigation due to the high antioxidant and anti-inflammatory of both compounds and presence of chemical integration between them that cause this synergism especially the size of CNPs and its surface, which facilitate this integration and this high mobility and achieve this potent antioxidant, antigenotoxic, and anti-inflammatory actions.
Conclusion
In conclusion, DG suspiciously affects the hepatic structure of the rats as well as to its effects on biomarkers of the antioxidant enzymes activities (SOD, CAT, MPO, thiol levels, and GPx) and MMP with their related parameters such as SDH percent, ROS, ATP, MMP, and mitochondrial swelling level. DG caused hepatic degeneration supported by histopathological, ultrastructural, and comet assay alteration. The results revealed the mechanisms through which stress may lead to cellular oxidative status in the liver by DG. Therefore, CNPs were an effective agent in the drug delivery in the hepatic diseases medications and act as a carrier for V and make synergism that achieved the high antioxidant potency. V in combination with CNPs either LD or HD was proved to be a benefit in decreasing the toxicity of DG and its consequence of aging especially the combination between CNPs (HD) and V with DG. Their synergistic ameliorated effects could be related to their abilities to scavenge the free radicals accumulation and alleviate oxidative stress greatly.
Supplemental material
Supplemental Material, graphical_abstract.jpg_DG - Synergistic antioxidant capacities of vanillin and chitosan nanoparticles against reactive oxygen species, hepatotoxicity, and genotoxicity induced by aging in male Wistar rats
Supplemental Material, graphical_abstract.jpg_DG for Synergistic antioxidant capacities of vanillin and chitosan nanoparticles against reactive oxygen species, hepatotoxicity, and genotoxicity induced by aging in male Wistar rats by NM Al-Baqami and RZ Hamza in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding of this publication was achieved by the Deanship of Scientific Research, King Abdulaziz University, Jeddah [project no. J: 10-247-1440]. The research is also funded by Taif University Researcher supporting project number (TURSP-2020/21), Taif University, Taif, Saudi Arabia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
