Abstract
The present study investigates the possible ameliorative effects of diallyl trisulfide (DATS) against arsenic (As)-induced hepatotoxicity and oxidative stress in rats. The four experimental groups evaluated include: (1) vehicle control; (2) As (5 mg/kg/day); (3) DATS (80 mg/kg/day) + As; and (4) DATS. Induction of As in rats caused severe hepatotoxicity as evidenced by an elevation of serum aspartate aminotransferase and alanine aminotransferase activities and increased total bilirubin concentration, indicating hepatic function abnormalities. Histopathological examination revealed various structural changes in the liver, characterized by hepatocyte degeneration/necrosis, congestion, sinusoidal dilatation, vacuolation, and inflammatory cell infiltration. The significant decrease in reduced glutathione content, catalase, superoxide dismutase, glutathione peroxidase, and glutathione reductase activities and the significant increase in lipid peroxidation (thiobarbituric acid reactive substance) and protein oxidation (protein carbonyl) contents indicated that As-induced hepatotoxicity was mediated through oxidative stress. As intoxication also elevated the levels of Cas-3 and nitric oxide and increased the expression of nuclear factor-κB p65 in the liver. In contrast, DATS pretreatment significantly improved As-induced serum biochemical, immunohistochemical, and histopathological alterations reflecting hepatic dysfunction. These results may contribute to a better understanding of the hepatoprotective role of DATS, emphasizing the influence of this garlic trisulfide in the diet for human health, possibly preventing the hepatic injury associated with As intoxication, presumably due to its ability to inhibit lipid peroxidation, protein oxidation, and restoration of antioxidant status.
1. Introduction
Arsenic (As) is a common environmental pollutant that threatens the life of many people, especially in developing countries of the world. It is widely spread in nature in many forms, and its compound is used extensively as herbicides, insecticides, rodenticides, food preservatives, and drugs. 1 Humans may be predominantly exposed to As through contaminated drinking water, while inhalation and skin absorption can contribute to minor entry routes. 2 Besides, the sources of As-contaminated drinking water, use of As-containing pesticides, herbicides, and rodenticides are also potential origins of As toxicity. 3 An early event in As-induced carcinogenesis is the molecular alterations in both humans and animals which manifest dose-dependent chromosomal breaks and alterations. Several reports have implicated that the oxidative stress is the major causator of As-induced cytotoxicity and genotoxicity. 1
In fact, the liver has been reported to be a critical target of As in humans and As exposure is associated with the development of hepatocellular carcinomas as well as other toxic lesions.
4
The liver is not also unused from inorganic As toxicity. As is well absorbed from the gastrointestinal tract and first reaches the liver. Arsenate is reduced to arsenite in the liver. Because the liver is rich in glutathione (GSH), it is a major site of As detoxification, either from GSH acting as an antioxidant or by GSH-As conjugation for cellular efflux and biliary excretion. The liver is also the major site of As methylation, which is catalyzed by arsenic methyltransferase using
A greater number of synthetic drugs, including As-specific antidotes have been tested to reduce hepatic damage or necrosis and to inhibit liver fibrogenesis. However, many of them met with serious side effects. 7 The most effective and promising treatment for As poisoning is the chelation therapy. Chelators, such as British anti-Lewisite, sodium 2,3-dimercaptopropane-1-sulfonate, and meso 2,3-dimercaptosuccinic acid, also result in a number of undesirable complications. 8 Therefore, the current research trend is to exploit the therapeutic potential of phytochemical antioxidants from medicinal plants to combat the As-induced oxidative stress-mediated hepatotoxicity.
It has been suggested that the number of sulfur atoms may play a determining factor in the biological activities of garlic organosulfur compounds. 9 Structure and function relationship studies demonstrate that the polysulfide diallyl trisulfide (DATS) is more potent than the other two organosulfur compounds in the modulation of the antioxidant and phase II enzymes and effectively lessens carbon tetrachloride-induced liver injury. 10 The organosulfur compounds effectively modulate the antioxidant system in rat liver and red blood cells, where DATS was found to be the most effective allyl compound compared to other organosulfurs. 10
Several earlier reports have claimed that DATS, a free radical scavenger, 11 can protect bioconstituents from free radical damage which otherwise can lead to oxidative damage. A recent report from our laboratory also claimed that DATS protect the rat liver mitochondria from As-induced toxicity mainly because of its antioxidant activity. 12 Hence, in view of its potential benefits, the present study has been designed to investigate whether DATS pretreatment exerts any protective influence on hepatic oxidative stress, inflammation, apoptosis, and DNA damage in hepatocytes of rats intoxicated with As.
Materials and methods
Chemicals
DATS was purchased from Lukang Cisen Pharmaceutical Co., Ltd (Shangdong, China). As (sodium arsenate-Na3AsO4) was obtained from Sigma Chemical Co. (St Louis, Missouri, USA). Serum hepatic marker enzyme diagnostic kits were obtained from Sigma Diagnostics (I) Pvt. Ltd (Baroda, Gujarat, India). Reagent kits were obtained from Span Diagnostics (Mumbai, Maharashtra, India). Anti-nuclear factor-κB (NF-κB) p65 antibody (1:100) was obtained from Biolegend (San Diego, California, USA).
Animals
Male albino rats weighing 170–190 g were used in this study. They were maintained in an environmentally controlled animal house (temperature 24 ± 2°C) with a 12-h light/12-h dark schedule and free access to deionized drinking water. The animal treatment and protocol employed were approved by the Institutional Animal Ethics Committee, Annamalai University (registration number: 885/2012/CPCSEA), Annamalainagar, Tamil Nadu, Chennai.
Experimental design
In the present study, As was administered as Na3AsO4 intragastrically intubation at a dose of 5 mg/kg body weight (BW)/day for 4 weeks, which was one-eighth of the oral median lethal dose values in rats.
11
A pilot study was conducted with three different doses of DATS (20, 40, and 80 mg/kg) to determine the dose-dependent effect on As-treated rats. After 4 weeks of the experiment, it was observed that DATS pretreatment at the doses of 20, 40, and 80 mg/kg BW significantly (
For experimental analyses, 24 rats were randomly selected and divided into 4 groups consisting of 6 rats in each group. The groups include: group I-control rats were orally administered with normal saline and corn oil for 28 days; group II-rats were orally administered with As as Na3AsO4 (5 mg/kg.BW) in normal saline for 28 days; group III-rats were orally preadministered with DATS (80 mg/kg BW) 90 min before As as Na3AsO4 (5 mg/kg.BW) intoxication; and group IV- rats were orally administered with DATS alone (80 mg/kg BW). The animals from all the groups were provided with a pellet foodstuff from the Amrut laboratory animal feed, Pune, Maharashtra, India for feeding and water ad libitum.
At the end of the experimental period, the rats in each group were fasted overnight, and then the animals were killed by decapitation. Blood samples were collected in tubes for the separation of serum. Liver tissue was surgically removed, washed with cold physiological saline, cleared off adherent lipids, and immediately transferred to ice-cold containers. Liver tissue (250 mg) was sliced into pieces and homogenized in appropriate buffer in cold condition (pH 7.0) to give 20% homogenate (w/v). The homogenate was centrifuged at 1000 r/min for 10 min at 0°C in cold centrifuge. The supernatant was separated and used for the assay of various biochemical estimations. For comet assay, liver was cut into small pieces and washed with Hank’s balanced salt solution. The individual cell suspensions were obtained by enzymatic digestion with collagenase at 37°C. After filtration on 100 and 40 mm mesh successively, the resulting cell suspensions were centrifuged for 10 min at 3000 r/min. The cell pellet was then suspended in phosphate-buffered saline and used for the estimation of DNA damage. Urine was collected for 24 h (8 a.m to 8 a.m) according to the standard laboratory protocol as described by Chanda et al.. 13
Estimation of serum hepatic marker enzymes, bilirubin assay, and proinflammatory cytokines
The activities of serum aspartate aminotransferase (AST; E.C.2.6.1.1), alanine aminotransferase (ALT; E.C. 2.6.1.2), alkaline phosphatase (ALP; E.C.3.1.3.1), lactate dehydrogenase (LDH, E.C.3.1.3.1), and total bilirubin were assayed using commercially available diagnostic kits (Sigma Diagnostics (I) Pvt. Ltd). γ-Glutamyl transferase (GGT; E.C. 2.3.2.2) activity was determined by the method described by Rosalki et al.
14
using γ-glutamyl-
Lipid peroxidation and oxidative stress marker assays
Lipid peroxidation in liver was estimated spectrophotometrically by measuring thiobarbituric acid reactive substances (TBARS) and lipid hydroperoxides (LOOH) by the method of Niehaus and Samuelsson 16 and Jiang et al., 17 respectively. Protein carbonyl (PC) content was determined by the method described by Levine et al. 18 The levels of conjugated dienes (CDs) were assessed by the method of Rao and Racknagel. 19
Determination of nonenzymatic antioxidants
Reduced GSH was determined by the method of Ellman. 20 Total sulfhydryl (TSH) groups were measured by the method of Ellman. 20 Vitamin C concentration was measured as previously reported. 21 Vitamin E (α-tocopherol) was estimated by the method of Desai. 22
Determination of antioxidant enzymes
Superoxide dismutase (SOD) activity was determined by the method of Kakkar et al..
23
The activity of catalase (CAT) was determined by the method of Sinha.
24
Glutathione peroxidase (GPx) activity was estimated by the method of Rotruck et al..
25
Glutathione
Intracellular ATP determination from isolated hepatocytes
Hepatocytes were isolated from rat liver following the method of Sarkar and Sil 30 with some modifications and were used for analysis of intracellular adenosine triphosphate (ATP) as described by Kalbheim and Koch. 31
Hepatic membrane-bound enzymes’ ATPase estimation
The sediment after centrifugation was resuspended in ice-cold tris(hydroxymethyl)aminomethane (Tris)–hydrochloric acid (HCl) buffer (0.1 M) pH 7.4. This was used for the estimations of membrane-bound enzymes and protein content. The membrane-bound enzymes such as sodium ion (Na+)/potassium ion (K+)-ATPase, calcium ion (Ca2+)-ATPase and magnesium ion (Mg2+)-ATPase activities were assayed by estimating the amount of phosphorous liberated from the incubation mixture containing tissue homogenate, ATP, and the respective chloride salt of the electrolytes. 32 –34 Total protein content was estimated by the method described by Lowry et al. 29
Determination of blood ALAD level
The activity of blood δ-aminolevulinic acid dehydratase (ALAD) was assayed according to the procedure followed by Berlin and Schaller. 35 The values are expressed as nanomoles per minute per milliliter.
Liver nitrite assay
Nitric oxide (NO) decomposes rapidly in aerated solution to form stable nitrite/nitrate products. Therefore, nitrite accumulation was estimated by the Griess reaction 36 and was used as an index of NO production. The concentration of nitrite (in milli molar) in the sample was calculated from a sodium nitrite curve. 37
Determination of As concentration
For determination of As in blood, liver, and urine, 1 ml of blood, 1 g of tissue, and 1 ml of urine were digested with nitric acid in microwave oven. After digestion, As was continuously preconcentrated and determined by flame atomic absorption spectrophotometry. A Perkin-Elmer 5000 atomic absorption spectrometer (Waltham, Massachusetts, USA) furnished with an As hollow cathode lamp (lamp current 4 mA) was used to determine the As concentration. The instrument was set at 228.8 nm with a slit width of 0.5 nm. The acetylene flow rate was 2.0 l/min, and an airflow rate of 17.0 l/min was employed to ensure an oxidizing flame. As estimation is based on the concentration of the element which will produce a signal/noise ratio of 3. Thus, the detection limit considers both the signal amplitude and the baseline noise and is the lowest concentration that can be clearly differentiated from zero. The specific limit of detection (i.e. actual concentration limit of detection for As analysis) is about 2 mg/g tissue.
Comet assay
Hepatocytes isolated from control and experimental groups were processed for alkaline comet assay as described previously. 38 The slides were immersed in lysis buffer for 1 h at 4°C and equilibrated in alkaline solution for 20 min, followed by electrophoresis at 18 V, 300 mA (Sub-Cell GT system with Power Pac basic power supply, Bio-Rad Laboratories Inc., Hercules, California, USA). After electrophoresis, the slides were neutralized and stained by ethidium bromide. The images were captured using a fluorescence microscope (Eclipse TS100, Nikon Instruments Inc., Melville, New York, USA). Fifty images per slide were analyzed for tail length (TL) and olive tail moment (OTM) using image analyzer CASP software version 1.2.2.
Estimation of caspase 3 activity
Liver tissues were placed in 0.15 M potassium chloride and homogenized using Teflon glass homogenizer. The resulting homogenates were centrifuged at 3000 r/min for 15 min, and the supernatant fractions were used for caspase 3 activity measurement. Caspase 3 activity was estimated at 96-well plates according to the manufacturer’s protocol by measuring enzymatic cleavage of the Florigenic substrate Ac-DEVD-AMC (R&D System). Then, 50 μl of homogenate (200 μg of total protein) was added to 50 μl of the assay buffer followed by 5 μl of caspase 3 colorimetric substrate (DEVD-pNA) and incubated at 37°C for 2 h. Absorbance was measured at 405 nm using a microplate reader (BMG Lab Technologies, Chicago, Illinois, USA). Experiments were performed in duplicate. The caspase 3 activity was expressed as nanomole per milligram protein.
Immunohistochemistry of NF-kB p65
To examine the protective effects of DATS on markers of inflammation in the liver, NF-κB p65 expression in the liver was assessed by immunohistochemical staining. Liver sections on polylysine-coated slides obtained were fixed in neutral-buffered formalin, embedded in paraffin, and were treated for NF-kB p65 antibody for immunohistochemical analysis. The procedures were processed according to the manufacturer’s protocol recommended for the NF-kB p65 immunohistochemistry with slight modifications. Following deparaffinization and rehydration, sections were irradiated in 0.1 mol/l sodium citrate buffer (pH 6.0) in a microwave oven (medium low temperature) for 20 min. Then, the sections were exposed to 3% H2O2 for 10 min to bleach endogenous peroxidases, followed by rinsing 3 times in Tris buffer (pH 7.4) for 10 min. Sections were selectively incubated under humid conditions using an anti-NF-kB p65 antibody (1:100; Biolegend) overnight at 4°C. The next day, slides were washed three times in Tris buffer for 10 min each. The specificity of the antibody was tested by omission of the primary antibody and a positive control of rat tonsil tissue. After washing in Tris buffer (pH 7.4), tissues were visualized with 3,3′-diaminobenzidine and counterstained with hematoxylin. Finally, the sections were dehydrated in xylene, mounted with a mixture of distyrene, a plasticizer, and xylene (DPX), and cover slipped. Slides prepared for this case was examined using light microscopy. Positive and negative controls were conducted in parallel with NF-kB p65-stained sections. Staining of sections with commercially available antibodies served as the positive control, and for the negative controls tissue sections were stained with the omission of the primary antibody.
Liver TAC assay
Antioxidant power of liver homogenate was determined by measuring their ability to reduce ferric (Fe3+) to ferrous (Fe2+) ion established and named as fluorescence recovery after photobleaching (FRAP) test and described previously. 39 Briefly, in this test, the medium is exposed to Fe3+ and the antioxidants present in medium start to produce Fe2+ as an antioxidant activity. The reagent included 300 mmol/l acetate buffer, pH 3.6 and 16 ml acetic acid/l of buffer solution, 10 mmol/l 2,4,6-tris(2-pyridyl)-1,3,5-triazine (TPTZ) in 40 mmol/l HCl, and 20 mmol/l ferric chloride hexahydrate (FeCl3 6H2O). Working FRAP reagent was prepared as required by mixing 25 ml acetate buffer, 2.5 ml TPTZ solution, and 2.5 ml FeCl3 6H2O solution. Then, 10 μl of sample diluted in water was added to 300 ml freshly prepared reagent warmed at 37°C. The complex between Fe2+ and TPTZ gives a blue color with absorbance at 593 nm.
Histopathological studies
The livers were removed and embedded in a paraffin block after being kept in Bouin fixative. Sections of 4–5 mm thick were prepared from the blocks. These sections were stained with hematoxylin and eosin (H&E) to observe the general structure and viewed with the Olympus light microscope (Japan). To evaluate the degree of hepatic injury, 10 slides made from each treatment group were examined and assigned for severity of changes using scores on a scale of (−) = none, (+) = mild, (++) = moderate, and (+++) = severe by two blinded histologists semiquantitatively.
Electron microscopy
Electron microscopic liver specimens were prefixed in 3% glutaraldehyde solution in 0.1 M phosphate buffer for 1.5 h at 4°C. Following this, they were washed in 0.15 M phosphate buffer (pH 7.2) and postfixed in 2% osmium tetroxide solution in 10 mM sodium phosphate buffer and left overnight. Dehydration was performed in acetone, and inclusion was done in the epoxy embedding resin Epon 812. The blocks were cut with an ultramicrotome type LKB at 70 nm thickness. The sections were doubly contrasted with solutions of uranyl acetate and lead citrate and analyzed with a Tecnai 12 Biotwin transmission electron microscope (FEI, Hillsboro, Oregon, USA).
Statistical analysis
All the data were expressed as mean ± SD of a number of experiments (
Results
The effect of DATS on morphological changes induced by As intoxication
Table 1 shows the effect of As and DATS on the BW gain (%), organ BW ratio (liver), water intake, and food intake in control and experimental rats. A significantly (
Effect of DATS on animal body weight, weight gain, organ–body weight ratio, water intake, and food intake in As-treated rats.a

As: arsenic; DATS: diallyl trisulfide; DMRT: Duncan’s multiple range test.
aValues are given as mean ± SD from six rats in each group. Values with different superscript letters (b–d) in the same column differ significantly at
Effect on DATS on As induced changes in serum hepatic marker enzymes and proinflammatory cytokines
Table 2 depicts significantly (
Effect of DATS on As-induced changes in serum hepatic biomarkers.a

As: arsenic; DATS: diallyl trisulfide; DMRT: Duncan’s multiple range test; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; LDH: lactate dehydrogenase; GGT: γ-glutamyl transferase; TNF-α: tumor necrosis factor α; IL-6: interleukin 6.
aValues are given as mean ± SD from six rats in each group. Values with different superscript letters (b–d) in the same column differ significantly at
Effect of DATS on As induced changes in hepatic oxidative stress markers
Table 3 illustrates the changes in the levels of hepatic lipid peroxidation, hydroperoxides (HP), PC content, and CD in control and experimental rats. The levels of TBARS, LOOH, PC content, and CDs were significantly increased (
Effect of DATS on arsenic induced changes in hepatic oxidative stress markers.a

As: arsenic; DATS: diallyl trisulfide; DMRT: Duncan’s multiple range test; TBARS: thiobarbituric acid reactive substances; LOOH: lipid hydroperoxides; CD: conjugated dienes; PC: protein carbonyl.
aValues are given as mean ± SD from six rats in each group. Values with different superscript letters (b–e) in the same column differ significantly at
Effect of DATS on As-induced changes in nonenzymatic antioxidant activity of rat liver
Table 4 demonstrates the alterations in the levels of nonenzymatic antioxidants in the liver tissues of control and experimental rats. A significant decrease (
Effect of DATS on As-induced changes in nonenzymatic antioxidant activity of rat liver.a
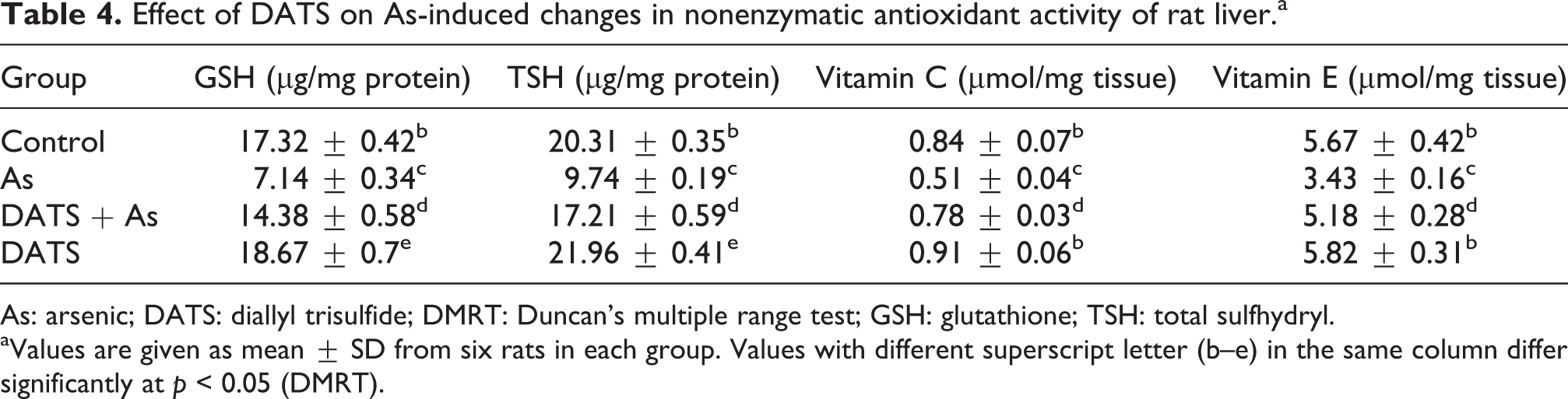
As: arsenic; DATS: diallyl trisulfide; DMRT: Duncan’s multiple range test; GSH: glutathione; TSH: total sulfhydryl.
aValues are given as mean ± SD from six rats in each group. Values with different superscript letter (b–e) in the same column differ significantly at
Effect of DATS on As-induced changes in antioxidant enzyme activity of rat liver
The activities of enzymatic antioxidants namely SOD, CAT, GPx, GST, and GSH-metabolizing enzymes (GR and G6PD) significantly (
Effect of DATS on As-induced changes in antioxidant enzyme activity of rat liver.a

As: arsenic; DATS: diallyl trisulfide; DMRT: Duncan’s multiple range test; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; GR: glutathione reductase; GST: glutathione
aValues are given as mean ± SD from six rats in each group. Values with different superscript letter (b–e) in the same column differ significantly at
Effect of DATS on the cellular ATP level in hepatocytes of As-treated rats liver
Figure 1 depicts the cellular ATP level in hepatocytes, where the level of intracellular ATP significantly lowered in As-treated rat when compared with the control rats. However, pretreatment of DATS in As-treated rats could prevent this As-induced lowering of ATP level when compared with As alone-treated rats, and it shows restoring ability. DATS alone treatment caused a significant (

Effect of DATS on the cellular ATP level in hepatocytes of control and experimental rats. Values are represented as mean ± SD (
Effect of DATS on the activitities of hepatic ATPases in the liver of As-treated rats
Figure 2 shows the activities of hepatic total ATPase, Na+/K+ -ATPase, Mg2+-ATPase, and Ca2+-ATPase in the liver of control and experimental rats. The activities of these membrane-bound ATPases in the liver tissue of As-treated rats were significantly (

(a to d) Effect of DATS on the levels of membrane-bound ATPase enzymes in the liver of control and experimental rats. The effect of DATS on the levels of (a) total ATPase, (b) Na+/K+-ATPase, (c) Ca2+-ATPase, and (d) Mg2+-ATPase in the liver of control and experimental rats. Values are represented as mean ± SD (
Effect of DATS on As induced changes in the level of blood ALAD
Figure 3 shows the level of blood ALAD in control and experimental rats. As-intoxicated rats showed a significant (
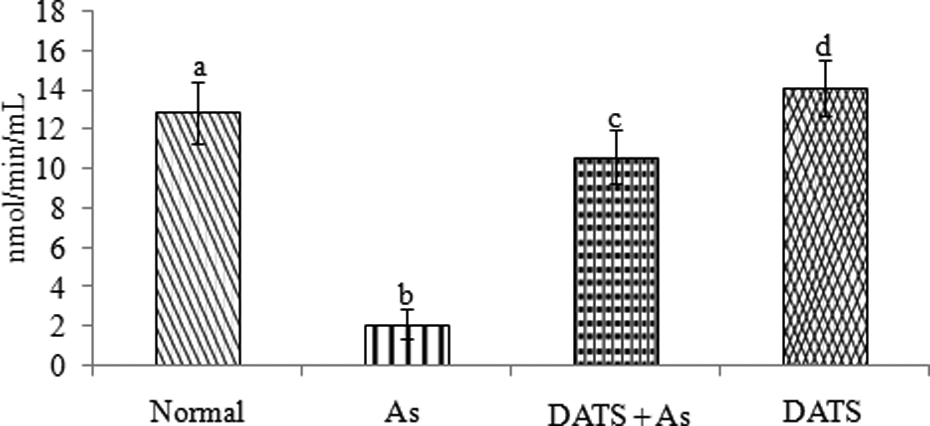
Effect of DATS on the level of blood ALAD in control and experimental rats. Values are represented as mean ± SD (
Effect of DATS on NO production in the liver of the As-intoxicated rats
Figure 4 depicts significantly (

Effect of DATS on liver NO production in control and experimental rats. Values are represented as mean ± SD (
Effect of DATS on As concentration in blood, liver, and urine of the As-intoxicated rats
Figure 5 shows the As concentration in blood, liver, and urine of control and experimental rats. The exposure of As resulted a significant increase (

(a) Concentration of arsenic in blood of control and experimental rats; (b) concentration of arsenic in liver of control and experimental rats; (c) concentration of arsenic in urine of control and experimental rats. Values are represented as mean ± SD (
Effect of DATS on average tail length of comets in the hepatocytes of As-treated rats
The data in Table 6 shows the effect of DATS on average tail length of comets in the hepatocytes of control and experimental rats. A significant (
Effect of DATS on average tail length of comets assay in liver tissue of As-treated rats.a
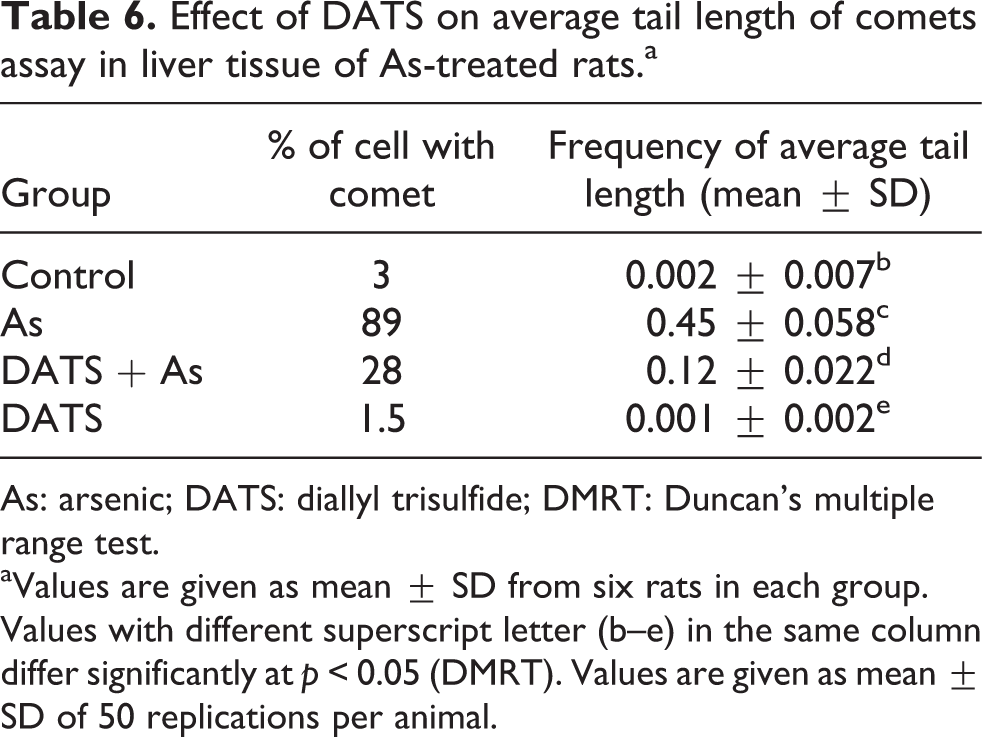
As: arsenic; DATS: diallyl trisulfide; DMRT: Duncan’s multiple range test.
aValues are given as mean ± SD from six rats in each group. Values with different superscript letter (b–e) in the same column differ significantly at
Effect on DATS on As-induced DNA damages in rat
As-induced alterations in the comet assay parameters of control and experimental rats is illustrated in Figure 6. A significant (

Effect of DATS on DNA damage in the hepatocytes of control and experimental rats. (a) % DNA in tail; (b) tail length (in micrometers); (c) olive tail moment (arbitrary unit). Values are represented as mean ± SD (
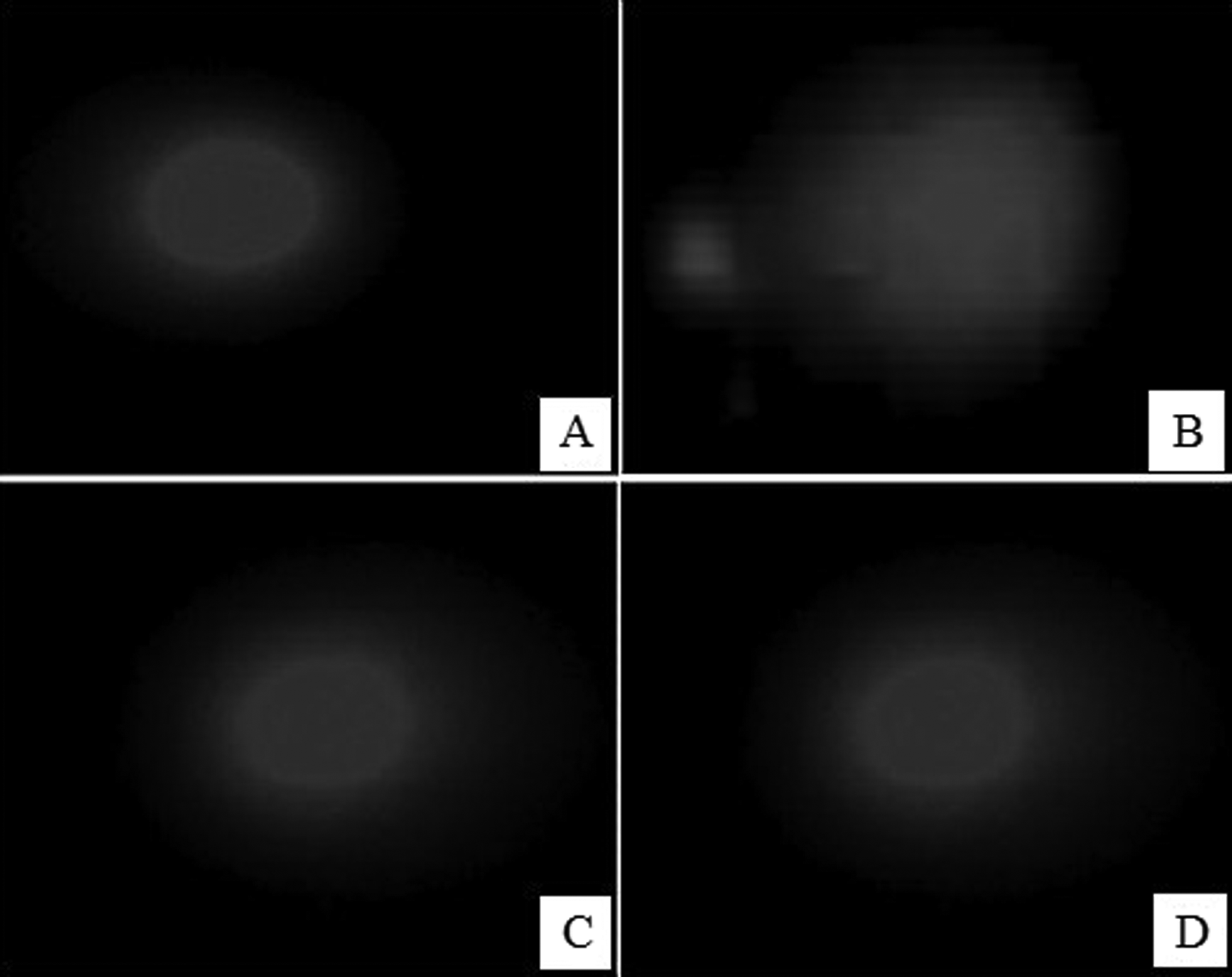
Representative photomicrographs of comets stained with ethidium bromide at ×200 magnification showing the DNA migration pattern in hepatocytes. (a) Control rats show no DNA migration. (b) Arsenic-treated rats show extensive DNA migration. (c) DATS-preadministered arsenic-intoxicated rats show minimal DNA migration. (d) DATS alone-administered rats show no DNA migration. DATS: diallyl trisulfide.
Effect of DATS on hepatic caspase 3 activity in As intoxication
A significant increase in hepatic caspase 3 activity was observed in As-treated rats as compared to control rats. DATS preadministration resulted in a significant reduction in caspase 3 level in As-treated rats in comparison with As alone-exposed rats (Figure 8). DATS administration to normal rats produced no change in renal caspase 3 activity.

Effect of DATS preadministration on hepatic caspase 3 activity in As-induced hepatatotoxicity in rats. Values are represented as mean ± SD (
Effect of DATS on As-induced expression of NF-kB p65 in rat liver
The expression of NF-kB p65 protein is given in Figure 9. The immunohistochemical study revealed that there was no staining of NF-kB p65 observed in the control rats, while As-treated rats show intense and positive staining as shown by arrows in As-treated rats when compared with the control group. There was moderate staining of NF-kB p65 in DATS-pretreated rats showing negligible NF-kB p65 immunostaining. There was no significant NF-kB p65 immunostaining of liver in DATS alone-treated rats.

Representative photomicrographs of NF-κB p65 protein expression (×400). (a) In control group (a′), NF-κB p65 protein immunostaining showed no staining; in As-treated rats (b′), NF-κB p65 protein immunostaining shows intense and positive stain nucleus as revealed by arrows; in DATS pretreated As intoxicated rat liver (c′), almost negligible NF-κB p65 immunostaining was observed; and in rats treated with DATS alone (d′), NF-κB p65 immunostaining shows very weak and diffused staining of hepatocytes. (b) Expression level of NF-κB p65. Values are represented as mean ± SD (
Effect of DATS on liver TAC in As intoxication
As depicted in Figure 10, liver total antioxidant capacity (TAC) in As-intoxicated rat significantly (
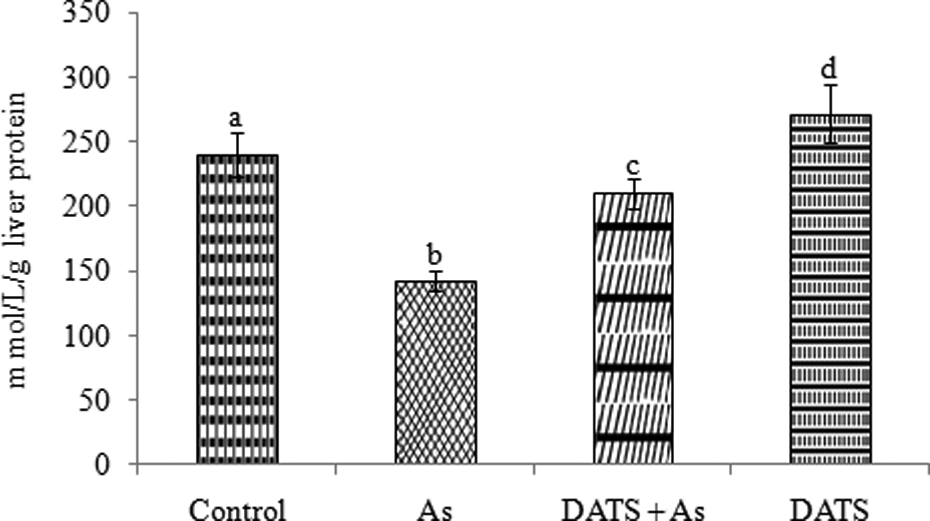
Effect of DATS preadministration in As-intoxicated rats liver TAC. Values are represented as mean ± SD (
Effect of DATS on As-induced pathological changes in the liver of control and experimental rats
Table 7 illustrates the histopathological scores of the liver of control and experimental rats. In control and DATS alone-treated rats, there were no indication of histopathological grades when compared with As alone-treated rats. As alone-treated rats showed severe grade in portal inflammation, necrosis, and sinusoidal dilation with moderate grade for inflammatory cell infiltration and hepatocellular vacuolization. Significantly after preadministration of DATS to As-exposed rats show mild grades when compared with As alone-treated rats.
Histopathological scores in the liver of control and experimental rats.a

aPathological scores: (−) = none; (+) = mild; (++) = moderate; (+++) = severe.
Effect of DATS on histopathological studies of As-treated rat liver
In Figure 11, the histopathological studies showed that As administration induces severe pathological changes in the liver (400×). The liver of control rats (Figure 11(a)) and DATS-treated rats (Figure 11(d)) showed a normal histoarchitectural pattern of liver. As exposure resulted in histological alterations in the liver, and it involved infiltration of inflammatory cells, hepatocyte vacuolation and necrosis, and dilatation of interhepatocyte spaces (Figure 11(b)). DATS preadministration before As exposure (Figure 11(c)) reduced the infiltration of inflammatory cells and vacuolation of hepatocytes.
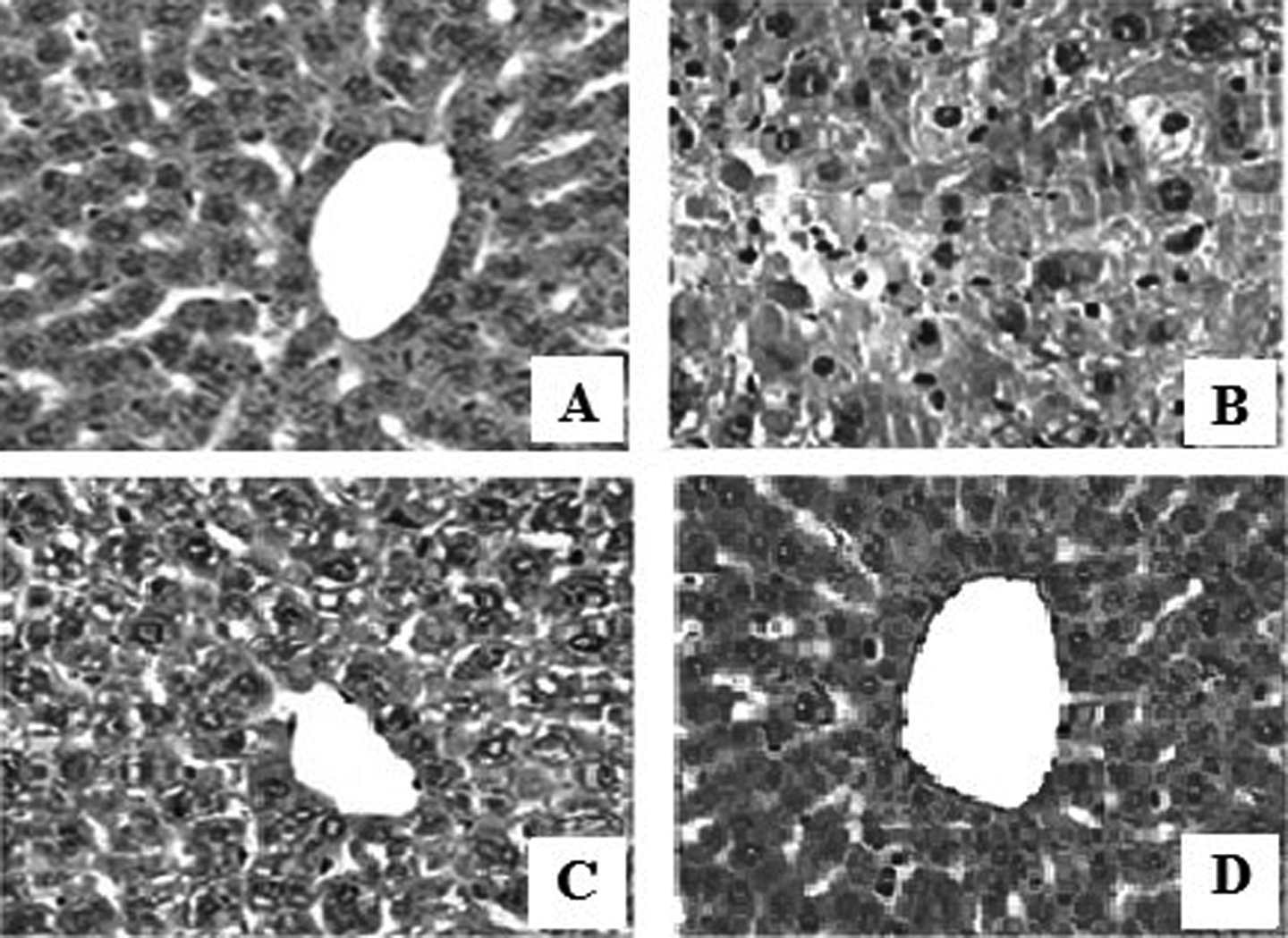
(a) Normal liver histology in control rats (hematoxylin–eosin; ×400); (b) infiltration of inflammatory cells, hepatocyte vacuolation, and necrosis and dilatation of interhepatocyte spaces in As-treated rats (hematoxylin–eosin; ×400); (c) DATS pretreatment in As-intoxicated rats revealed significant improvement in liver damage with reduced infiltration of inflammatory cells and vacuolation of hepatocytes (hematoxylin–eosin; ×400); and (d) normal liver histology in DATS alone-treated group (hematoxylin–eosin; ×400). As: arsenic; DATS: diallyl trisulfide.
The effect of DATS on electron microscopic examination of rat liver intoxicated by As
Figure 12 reveals the electron microscopic examination of the control and treated livers. The hepatocytes of control group have normal structures with few fat droplets with well-distributed cytoplasm. The nucleus was normal with other organelles (Figure 12(a)). In As-treated group, hepatocytes showed altered nucleus, mitochondria, rough endoplasmic reticulum, and Golgi complex. Also, flattened nuclei, flattened microvilli with unevenly distributed glycogen and a number of lipid droplets were observed (Figure 12(b) and (c)). As and DATS pretreated group showed hepatocytes with free ribosome, regular-shaped nuclei, and dense chromatin. In DATS alone-treated group hepatocytes with normal mitochondria, rough endoplasmic reticulum, and Golgi complex were observed. Hepatocytes with large nucleus and evenly distributed chromatin were also seen (Figure 12(d)).

Electron micrographs of the control and treated livers. (a) control group hepatocytes have normal structures with few fat droplets with well-distributed cytoplasm. The nucleus was normal with other organelles (×6000). (b) As-treated group hepatocytes showing altered nucleus, mitochondria, rough endoplasmic reticulum, and Golgi complex. Flattened nuclei, flattened microvilli with unevenly distributed glycogen and a number of lipid droplets were observed (×6000). (c) As and DATS-treated group shows hepatocytes along with free ribosome, regular-shaped nuclei, and dense chromatin were observed (×6000). (d) DATS alone-treated group shows hepatocytes with normal mitochondria, rough endoplasmic reticulum, and Golgi complex. Hepatocytes with large nucleus, evenly distributed chromatin were also seen (×6000). As: arsenic; DATS: diallyl trisulfide.
Discussion
The protective role of DATS against As-induced hepatotoxicity in rats were revealed with the outcome of the present study. The results of the present study showed that the pretreatment of DATS prior to As intoxication exerted a marked amelioration of As-induced hepatotoxicity as evidenced by the significant restoration of altered BWs, liver BW ratio, serum, and hepatic biochemical parameters toward normalization. The DATS pretreatment before As intoxication significantly recuperated the perturbed hepatic parameters entailing its antioxidant role. Figure 13 shows a summary of As-induced oxidative liver injury and its prevention by DATS.

Graphical abstract showing a summary of As-induced oxidative liver injury and its prevention by DATS. As: arsenic; DATS: diallyl trisulfide.
Reduction in BW is used as an indicator for the deterioration of rat general health status. It has been reported that As could induce toxicological effects and biochemical dysfunctions representing serious health hazards. The findings from the present study indicate that As exposure has reduced BW, liver BW ratio, food intake, and water intake leading to the deterioration of general health of rats. These findings were in line with the reports of Yousef et al., 40 who reported that As exposure have significantly induced the disturbances of the BW, liver BW ratio, food intake, and water intake in rats. While the pretreatment of DATS potentially attenuate all these morphological changes observed in As-intoxicated rats.
The liver is the major target organ for As-induced hepatotoxicity, because metabolism of As involved only in the liver and the leakage of hepatic marker enzymes commonly used as an index of hepatocellular damage. 38 In the present study, As intoxication caused a significant increase in the levels of AST, ALT, ALP, LDH, and GGT which reflects the damage of hepatocytes. The elevated levels of serum hepatic marker enzymes and bilirubin of the rats exposed to As indicate the loss of functional integrity of hepatocellular membrane and their increased permeability leading to leakage of hepatic enzymes into the blood stream and abnormal liver function. Pretreatment of DATS before As exposure significantly normalized the levels of hepatic marker enzymes and bilirubin. This could be due to its and anti-lipoperoxidative action on maintaining membrane integrity thereby restricting the leakage of these hepatic markers into circulation.
Increased levels of lipid peroxidation markers such as TBARS, HP, and CD along with protein carbonylation content in the liver tissue of As-treated rats clearly signify the commencement of hepatic oxidative stress. The increased level of lipid peroxides and PC contents in As-induced rats might be due to free radical-mediated membrane damage. 41 Preadministration of DATS resulted in a significant reduction of TBARS, LOOH, CD, and PC in the liver of As-treated rats. This might be due to the free radical scavenging and antioxidant ability of DATS through conjugation with GSH leading to the control of oxidative damage and tissue protection. Furthermore, the orientation of –S– groups in DATS facilitates the elimination of As, possibly by coordinating with several sulfur atoms at a time forming a complex by acting as a multidentate ligands. 42
In the present study, the level of nonenzymatic antioxidants namely GSH, TSH, and vitamins C and E significantly decreased in the liver of As-treated rats when compared with the normal rats, which strengthen the onset of As-induced hepatic oxidative stress. Depleted levels of these nonenzymatic antioxidants represent an excess free radical production, which may be due to the binding of As with various sulfhydryls that exist in the cell. 43 Interestingly, restoration of these nonenzymatic antioxidants has been observed in DATS-pretreated rats clearly indicate the protective nature of DATS against As-induced thiol depletion and oxidative stress. DATS has already been reported to enhance the level of intracellular thiols and the activity of GPx, GR, and GST in liver. 9,42 The direct free radical scavenging ability of DATS is mainly attributed to its trisulfur moieties present in it and thereby decreasing the As-induced ROS-mediated lipid peroxidation, which could reduce the utilization of nonenzymatic antioxidants and consequently leading to the improvement of vitamins C and E and total sulfydryl levels in liver.
In the present study, it has been observed that As-intoxicated rats significantly reduced the activities of all the antioxidant enzymes, namely SOD, CAT, GPx, GST, GR, and G6PD. As-induced ROS and subsequent depletion of antioxidant defenses can result in the disruption of antioxidant/prooxidant equilibrium in liver.
44
Surprisingly pretreatment with DATS markedly increased the activities of these antioxidant enzymes. This may be due to the antioxidant and metal-chelating properties of DATS. It has been reported that DATS has the ability to increase the activity of phase II enzymes quinone reductase and GST, activities of the hepatic antioxidant enzymes (SOD, CAT, and GR) and the mitochondrial antioxidant enzymes (SOD, GR, and GPx) and thereby reduced the oxidative stress. DATS has already been reported as a potent inducer of phase II enzymes via activating the Nrf2
Our data show a depleted level of ATP in hepatocytes of As-treated rats. The ATP level of hepatocytes in As-treated rats decreased mainly due to the arsenate ability to block the enzymatic activity in mitochondria and its capability to replace phosphorus, so it combines with adenosine diphosphate to replacing phosphate and subsequent formation of an unstable arsenate ester bond that is rapidly hydrolysate. As a result, though oxidation is occurring, not only production of ATP through phosphorylation is hampered and source of energy in cell reduces continually but also it disturbs the electron transfer of inorganic phosphorus with ATP. 46 Prior administration of DATS in As intoxicated rats significantly elevated the level of ATP in hepatocytes. This may be due to the ability of DATS to form a complex with As and thus preventing the As-induced inactivation of mitochondrial enzymes.
In the present study, the levels of membrane-bound total ATPases, Na+/K+-ATPase, Ca2+-ATPase, and Mg2+-ATPase were significantly decreased in As-treated rats. Decreased activity of Na+/K+-ATPase could be due to the enhanced lipid peroxidation by free radicals on As induction, since Na+/K+-ATPase is an “SH” group containing enzyme and is lipid dependent. Decreased activity of Na+/K+-ATPase can lead to a decrease in sodium efflux, thereby altering membrane permeability. The disruption of membrane permeability or fragmentation of the membrane leads to the leakage of Ca2+ ions into cells thereby potentiating irreversible cell destruction. The Ca2+ overload medicated by As also decreased the Ca2+-ATPase activity in cell membranes. It is generally accepted that due to the high affinity for SH groups, As binds keenly to various enzyme proteins and inactivates them. Mg2+-ATPase activity is involved in other energy-requiring process in the cell and its activity is sensitive to lipid peroxidation. Pretreatment with DATS in As-intoxicated rats significantly reduced the peroxidation of membrane lipids in the hepatocytes and restored the activity of membrane-bound enzymes through its ability to defend the SH groups from the oxidative damage and thus stabilized the hepatocyte membrane. 11
ALAD is a sulfhydryl-containing enzyme involved in the biosynthesis of tetrapyroles (such as heme), the prosthetic group of various tissue proteins (such as hemoglobin), and its marked inhibition in the blood by As in the present study was evidently due to the binding of As with sulfhydryls, as As has a high affinity for SH groups. ALAD inactivation can also lead to overproduction of δ-aminolevulinic acid that can cause an overproduction of ROS which, in part, could explain the As-induced oxidative stress. 47 The results of the present study revealed that DATS pretreatment significantly reduced the ALAD activity toward near-normal level. Such beneficial effect of DATS might be due to its direct scavenging of ROS and the ability to inhibit the thiol group oxidation. 42 DATS, on the other hand, forms complexes with As, possibly due to its ability to coordinate with several sulfur atoms at a time and enhances its excretion via activating the phase II antioxidant system. 45
In our study, administration of As results to cause a significant increase in NO production of liver and this might be one of the reasons for the aggravated tissue damage and inflammatory changes in the liver of As-intoxicated rats. These findings were in accordance with the report of Bera et al. 48 In addition, during As exposure overproduction of ROS can occur, resulting in an oxidative stress, which is another risk factor for vascular dysfunction and was related to the inhibition of endothelial NO synthase (NOS) that ultimately leads to excessive NO production. 49 In our study, DATS-preadministered As-intoxicated rats showed a decreased level of NO in the liver, which might be due to its free radical scavenging and antioxidative activity. 46
In the present study, compared with As-treated rats, we observed an increase in urinary excretion of As in DATS-preadministered rats with a concomitant decrease in load of As in both hepatic tissue and blood samples. Our results are in full agreement with the reports of Pari and Murugavel 50 and suggest that an augmented detoxifying process of inorganic As in hepatic tissue and blood by DATS. The chelating efficacy of DATS possibly contributed in reducing As load in liver tissue and facilitated the excretion of As through urine.
Alkaline single-cell gel electrophoresis is a rapid, sensitive, and versatile method for the quantification of DNA damage in the individual cells. In the present study, the percentage of cells with comets, frequency of average TL, and the levels of % DNA in tail, TL, and tail movement were significantly increased in the hepatocytes of As-treated rats. Earlier reports showed that As induced generation of lipid peroxidation products, in particular, malondialdehyde and 4-hydroxynonenal have been identified to bind with DNA and damage them. 51 DNA damage significantly reduced in the hepatocytes of DATS-treated rats. Preadministration of DATS in As-treated rats were found to decrease in the percentage of cells with comets, the frequency of average TL and effective result in ameliorating As-induced DNA damage, suggesting efficacy of DATS on ROS-mediated lipid peroxidation and oxidative DNA damage induced by As intoxication. 11
To analyze the intensity of apoptotic changes and DNA damage, we performed comet assay by alkaline single-cell gel electrophoresis. An increased DNA damaging effect suggesting DNA fragmentation was observed in As-treated group confirming that apoptotic changes had entailed damage even at cytogenetic level. Caspases, which are cysteine proteases, cleave numerous intracellular target molecules, resulting in cell shrinkage, chromatic condensation, and DNA fragmentation. In this study, As-treated rats showed a marked increase in caspase 3 activity in hepatic tissues, suggesting that As-induced apoptotic changes in the liver of rat is caspase dependent. These apoptotic-related molecular changes were effectively attenuated in the DATS preadministered rats, suggesting the possible protective role of DATS against As-induced molecular changes in the progression of hepatic damage.
Elevated ROS levels were known to be an important step in the activation of proinflammatory cytokines and NF-kB signaling pathway. It was reported that As intoxication increased the generation of ROS by activating the expression of nicotinamide adenine dinucleotide phosphate oxidases in liver. In addition, it has been demonstrated that increased NO production is implicated in As-induced cytotoxicity and oxidative damage. 52 Moreover, the increased expressions of NF-kB p65 with subsequent release of proinflammatory mediators like TNF-α and IL-6 are the major events responsible for inflammation. 53 In this study, As intoxication significantly elevated the levels of proinflammatory cytokines TNF-α and IL-6 in the serum and increased the expression of hepatic NF-kB p65 clearly suggesting As-induced hepatic inflammation. The present results revealed that DATS pretreatment significantly suppressed lipid peroxidation, restored the antioxidant defense mechanisms, attenuated the overproduction of TNF-α and IL-6, and reduced the expression of NF-kB p65 in the liver of rats exposed to As. The hepatoprotective effect of DATS can be attributed to its ability to inhibit the activation of the NF-kB signaling pathway which promotes the transcription of TNF-α, inducible NOS, and COX-2 genes. However, this point warrants further investigations.
The histopathological scores of the liver of As alone-treated rats showed severe grade in portal inflammation, necrosis, and sinusoidal dilation with moderate grade for inflammatory cell infiltration and hepatocellular vacuolization. The histopathological observation in As-treated rats showed alterations in the liver histomorphology involving infiltration of inflammatory cells, hepatocyte vacuolization, necrosis, portal inflammation, and dilatation of sinusoidal spaces. This could be due to the increased formation of lipid peroxides, PCs, and associated DNA damages in the liver, and it is well correlated with the biochemical alterations provoked by As. Preadministration of DATS shows histopathological scores of mild grades and curtailed the histological alterations caused by As. This might be due to the antioxidant and chelating effect of DATS. The chelation of As by DATS significantly reduced the oxidative threat that might lead to a restoration of normal histological architecture of the liver tissue.
Transmission electron microscopy studies also support the histological observations showing the dysfunction and deterioration of As-induced hepatic ultrastructure. The electron microscopy of As-treated liver has revealed the altered subcellular organelles like mitochondria, rough endoplasmic reticulum, Golgi complex, and nucleus. Flattened nuclei, flattened microvilli with unevenly distributed glycogen and a number of lipid droplets were also observed in As-intoxicated rat liver. However, preadministration of DATS to As-treated rats significantly ameliorated the ultrastructural alterations in the liver tissue confirming its protective role in arsenicosis. There are no such reports available on the protective nature of DATS on the hepatic ultrastructural changes during experimental arsenicosis. Hence, this investigation should be considered as an innovative assessment to prove the hepatoprotective action of DATS in rats exposed to As.
To conclude, our findings clearly demonstrate that DATS has a hepatorotective effect against As-induced biochemical and pathological changes and further warrants the need for molecular studies to elucidate the mechanisms underlying the protective effects of DATS. On the other hand, its widespread use as dietary supplement should be encouraged to ward off As-associated oxidative stress because of its antioxidant and metal-chelating properties. Pharmacological agents like DATS, which is capable of scavenging free radicals, maintaining antioxidant homeostasis, and/or inhibiting LOOH, protein oxidation, and thereby protecting hepatocytes from oxidative injuries, may provide useful therapeutic potentials for the prevention or treatment for the As-induced degenerative changes in tissues.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was supported by the University Grants Commission and major portion of the project research was funded with a grant by the Government of India (F. No: 41-171/2012 (SR) dated 09/07/2012).
