Abstract
Garlic contains diallylsulfide (DAS) and other structurally related compounds that are widely believed to be active agents in preventing cancer. This study shows the effect of DAS (a phenolic antioxidant used in foods, cosmetics, and pharmaceutical products
Keywords
Introduction
Oxidative damage apparently increases with age and thus may overwhelm the natural repair systems in the elderly. It is induced by many chemical compounds that contribute to toxicity and carcinogenicity after a prolonged and sustained exposure. It is also known that that oxidative free radical stress is an important contributor to the aging process and many age-related diseases, the focus must be on reducing oxidative stress in vivo. Henceforth, diet rich in phytochemicals with antioxidant properties or dietary supplements of antioxidants is the best way to neutralize free radical-mediated oxidative stress in the elderly. 1
Garlic has been found to contain a large number of potent bioactive compounds with anticancer properties, largely allylsulfide derivatives. Garlic derivatives have been found to influence an increasing number of molecular mechanisms in carcinogenesis, including DNA adduct formation, scavenging of free radicals, mutagenesis, cell proliferation and differentiation, and angiogenesis. 2 It has also been reported that cancer can be prevented by organosulfur compounds from garlic and onion. 3,4 Garlic-derived organosulfur compounds such as diallylsulfide (DAS), diallyldisulfide, and diallyltrisulfide provide significant protection against carcinogenesis. Thus, specific and predetermined manipulations of the diet may represent a promising noninvasive approach to minimize cancer. A number of pharmacological effects of garlic constituents have also been reported, which provided sufficient evidences that garlic along with its organosulfur components possess pleiotropic beneficial health effects including antioxidative, antimutagenic, and antitumorigenic properties. 5 –9
The majority of the diseases associated with old age are initiated by free radicals. Previous studies have shown that intake of garlic by humans may either prevent or decrease the incidence of major chronic diseases associated with old age such as atherosclerosis, stroke, cancer, immune disorders, cerebral aging, arthritis, and cataract formation. Garlic has strong antioxidant properties, and epidemiological studies support the fact that diets rich in antioxidants may prevent some of the above-mentioned chronic diseases. It may also promote general health by preventing premature aging of the skin and increases glutathione (GSH) and energy levels. Thus, garlic can be beneficial if taken by the elderly in preventing or reducing chronic diseases associated with old age. 10
Garlic or its constituents are known to suppress hepatic damage and tumor promotion by scavenging hydroxyl radicals and augmenting the antioxidant enzyme activities. 11 It has been shown in earlier studies that ferric nitrilotriacetate (Fe-NTA) is a hepatic as well as a renal tumor promoter and acts by depleting antioxidant armory of the tissues. 12,13 Age-dependent renal accumulation of 4-hydroxy-2-nonrenal-modified proteins following parenteral administration of Fe-NTA and intranuclear iron deposition in hepatocytes has also been shown. 14,15
Some of the studies have reported that oxidative stress, toxicity, and hyperproloiferative response mediated by Fe-NTA in rats were attenuated after animals were pretreated with garlic oil. 16 Since DAS is known to be a major component of garlic, possessing some antitumor activity, 17 –19 the present study was undertaken to study the suppressing effect of DAS on Fe-NTA-mediated hepatic toxicity in rats.
Materials and methods
Preparation of Fe-NTA
The Fe-NTA solution was prepared by the method described by Awai et al. 20
Animals and treatment
Male albino rats of Wistar strain weighing 125–150 g were used in this study and were housed in an air-conditioned room and had free access to pellet diet and water ad libitum. All animal studies were approved by Animal Care and Use Committee.
Study design
For various sets of biological studies, different groups of animals were used. A total of 30 rats were used for studying the effect of DAS (Sigma Aldrich, St Louis, Missouri, USA) on Fe-NTA-mediated induction of oxidative stress.
For studying the effect of prophylactic treatment of DAS on Fe-NTA-mediated induction of hepatic toxicity, 30 male rats were taken, which were divided into 5 groups of 6 rats each. Group I received intraperitoneal (i.p.) injection of 10 ml kg−1 body weight (bw) saline and group II received an oral treatment of higher dose of DAS (200 mg kg−1 ml−1 corn oil) and both served as negative control. Group III received only 1.0 ml kg−1 corn oil for a period of 1 week through the gavage. Groups IV and V received DAS at a dose level of 100 mg and 200 mg kg−1 ml−1 corn oil, respectively, for a period of 1 week administered through the gavage. Twenty four hours after the last treatment of DAS or corn oil, the animals of groups III, IV, and V received an i.p. injection of Fe-NTA (9 mg Fe kg−1 bw). All the animals were killed 3 h after the treatment of Fe-NTA or saline by cervical dislocation. Just before the killing, blood samples from retro-orbital sinus of these animals were collected for the estimation of serum transaminases (alanine transaminase (ALT) and aspartate aminotransferase (AST)) and lactate dehydrogenase (LDH). Livers were collected and processed for the estimation of antioxidants including GSH, glutathione reductase (GR), and glutathione
Preparation of PMS and microsomes
Livers were quickly removed, perfused immediately with ice-cold saline (0.85% w/v sodium chloride) and homogenized in chilled phosphate buffer (0.1 M, pH 7.4) that contained potassium chloride (KCl; 1.17% w/v) using a homogenizer. The homogenate was filtered through a muslin cloth and was centrifuged at 800
Lipid peroxidation
The assay for hepatic microsomal lipid peroxidation was done following the method of Wright et al.
21
The reaction mixture, in a total volume of 1.0 ml, contained 0.58 ml phosphate buffer (0.1 M, pH 7.4), 0.2 ml microsome (10% w/v), 0.2 ml ascorbic acid (100 mM), and 0.02 ml ferric chloride (100 mM). The reaction mixture was incubated at 37°C in a shaking water bath for 1 h. The reaction was stopped by the addition of 1.0 ml trichloroacetic acid (TCA; 10% w/v). Following addition of 1.0 ml thiobarbituric acid (TBA; 0.67% w/v), all tubes were placed in a boiling water bath for a period of 20 min. At the end, the tubes were shifted to an ice-bath and centrifuged at 2500
Estimation of serum transaminases (ALT and AST)
In a water bath, 0.5 ml of 2 mM α-ketoglutatarate was incubated for 5 min at 37°C. Serum (0.1 ml) was then added, and the volume was adjusted to 1 ml with sodium phosphate buffer. The reaction mixture was incubated at 37°C for 20 min. The reaction was terminated with an equal volume of TCA (10% w/v), stored in ice, and then centrifuged at 4000
Estimation of LDH activity
The reaction mixture containing 2.7 ml of phosphate buffer (0.1 ml, pH 7.4), 0.01 ml serum, and 0.1 ml of reduced nicotinamide adenine dinucleotide was incubated for 20 min. To this, 0.1 ml of pyruvate (2.5 mg ml−1) was added. The absorbance was read for 5 min at an interval of 30 s. The activity was expressed as milli international units. 23
Estimation of GSH
Reduced GSH in liver was assayed by the method followed by Jollow et al.
24
An aliquot of 1.0 ml of PMS (10% w/v) was precipitated with 1.0 ml of sulfo salicylic acid (4% w/v). The samples were kept at 4°C for at least 1 h and then subjected to centrifugation at 1200
GR activity
GR activity was assayed by the method of Carlberg and Mannervik as 25 modified by Mohandas et al. 26 The assay system consisted of 1.65 ml phosphate buffer (0.1 M, pH 7.6), 0.1 ml ethylenediaminetetraacetic acid (0.5 mM), 0.05 ml oxidized GSH (1 mM), 0.1 ml nicotinamide adenine dinucleotide phosphate (NADPH; 0.1 mM), and 0.1 ml of PMS (10% w/v) in a total volume of 2.0 ml. The enzyme activity was quantitated at 25°C by measuring the disappearance of NADPH at 340 nm and was calculated as nanomoles of NADPH oxidized per minute per milligram protein using a molar extinction coefficient of 6.223103 M cm−1.
GST activity
The reaction mixture consisted of 1.425 ml phosphate buffer (0.1 M, pH 6.5), 0.2 ml reduced GSH (1 mM), 0.025 ml 1-chloro-2,4-dinitrobenzene (CDNB; 1 mM), and 0.3 ml and 0.05 ml of PMS (10% w/v), respectively, in a total volume of 2.0 ml. The changes in absorbance were recorded at 340 nm and enzyme activity was calculated as nanomole CDNB conjugate formed per minute per milligram protein using a molar extinction coefficient of 9.63103 M cm−1. 27
H2O2 assay
Hydrogen peroxide (H2O2) was assessed by H2O2-mediated horseradish peroxidase-dependent oxidation of phenol red by the method of Pick and Keisari.
28
Microsomes, 2.0 ml (10% w/v) of each, were suspended in 1.0 ml of solution containing phenol red (0.28 nM), horseradish peroxidase (8.5 U), dextrose (5.5 nM), and phosphate buffer (0.05 M, pH 7.0) and were incubated at 37°C for 60 min. The reaction was stopped by the addition of 0.01 ml NaOH (10 N) and then centrifuged at 800
Determination of protein carbonyl content
Protein oxidation was measured by an estimation of carbonyl groups with slight modifications using DNPH reagent.
29
Then, 0.1 ml of serum was treated with 0.5 ml of 10 mM DNPH, dissolved in 2 M hydrochloric acid (HCl) as a sample or with 0.5 ml of 2 M HCl as a control blank. The reaction mixtures were allowed to stand for 1 h at room temperature and in dark, with stirring at 15 min intervals. To this, 0.5 ml of ice-cold 20% TCA was added, and the sample was left on ice for 15 min. The tubes were then centrifuged at 11,000 r min−1 for 5 min to obtain the protein pellet. The precipitated proteins were subsequently washed 3 times with 1 ml of ethanol–ethyl acetate (1:1) to remove unreacted DNPH and lipid remnants. Each washing step was followed by centrifugation at 3000 r min−1 for 7 min. The final protein pellet was dissolved in 0.25 ml of 6 M guanidine hydrochloride and incubated at 37°C for 10 min. The carbonyl content was calculated from peak absorption (360 nm) using an absorption coefficient (
Statistical analysis
Values were expressed as means ± SEM. The level of significance between different groups is based on Dunnett’s
Results
Effect of DAS on Fe-NTA-mediated increase in the activities of serum transaminases (ALT and AST) and LDH
The effect of pretreatment of DAS on Fe-NTA-mediated increase in the activities of serum transaminases (ALT and AST) and LDH is shown in Table 1. Fe-NTA administration led to about 2.5-fold increase in the values of both ALT and AST, respectively, and to about 3.2-fold increase in the activity of LDH. The effect was dependent on the dose of DAS administered and a significant reduction in the values of LDH, ALT, and AST was observed in rats receiving a pretreatment of DAS. As the DAS dose increases, a reduction the fold levels for AST, ALT, and LDH to 1.3–1.56 (
Modulatory effect of DAS on Fe-NTA-induced levels of serum transaminases (ALT and AST), and LDH.a

DAS: diallyl sulfide; Fe-NTA: ferric nitrilotriacetate; ALT: alanine transaminase; AST: aspartate aminotransferase; LDH: lactate hydrogenase; D1: 100 mg kg−1 ml−1 DAS/animal/day; D2: 200 mg kg−1 ml−1 DAS/animal/day.
aData represent mean ± SE of six animals. Dose regimen and treatment protocol are described in the text. Saline-treated group served as control for group III. Fe-NTA-treated group served as control for groups IV and V.
b
c
Effect of DAS on Fe-NTA-induced changes in antioxidants
The effect of pretreatment with DAS on Fe-NTA-mediated depletion in hepatic antioxidants is shown in Figure 1. There was decrease in the level of GSH, GR, and GST to the level of 60–80% on treatment with Fe-NTA compared with the control. Pretreatment with DAS resulted in a significant recovery of these enzymes. At a lower dose of DAS, enzymatic activity recovery was observed to 75%, whereas at higher dose recovery was up to 92% as compared to Fe-NTA-treated group.

Modulatory effect of DAS on Fe-NTA-induced hepatic levels of GSH, the activities of GSH metabolizing enzymes and antioxidant enzymes. (I) Saline, (II) DAS (D2), (III) Fe-NTA, (IV) DAS (D1) + Fe-NTA, (V) DAS (D2) + Fe-NTA. Data represent mean ± SE of six animals. Saline-treated group served as control for Fe-NTA group III. Fe-NTA-treated group served as control for groups IV and V. Dose regimen and treatment protocol are described in the text. *
Effect of DAS on Fe-NTA-induced lipid peroxidation and H2O2 generation
Fe-NTA treatment resulted in increase of microsomal lipid peroxidation to 1.5-fold and H2O2 generation was increased to about 2.3-folds. This increase of Fe-NTA-mediated lipid peroxidation and H2O2 generation was reduced by DAS in a dose-dependent manner. There was a significant reduction in both the doses of DAS studied. At a lower dose, DAS decreased lipid peroxidation to about 76% compared with the Fe-NTA-treated group, but at the higher dose of 200 mg kg−1 bw, the level of lipid peroxidation reached almost the value of control. H2O2 generation was also significantly reduced to 78.6% (
Modulatory effect of DAS on Fe-NTA-induced hepatic microsomal lipid peroxidation and generation of H2O2.a
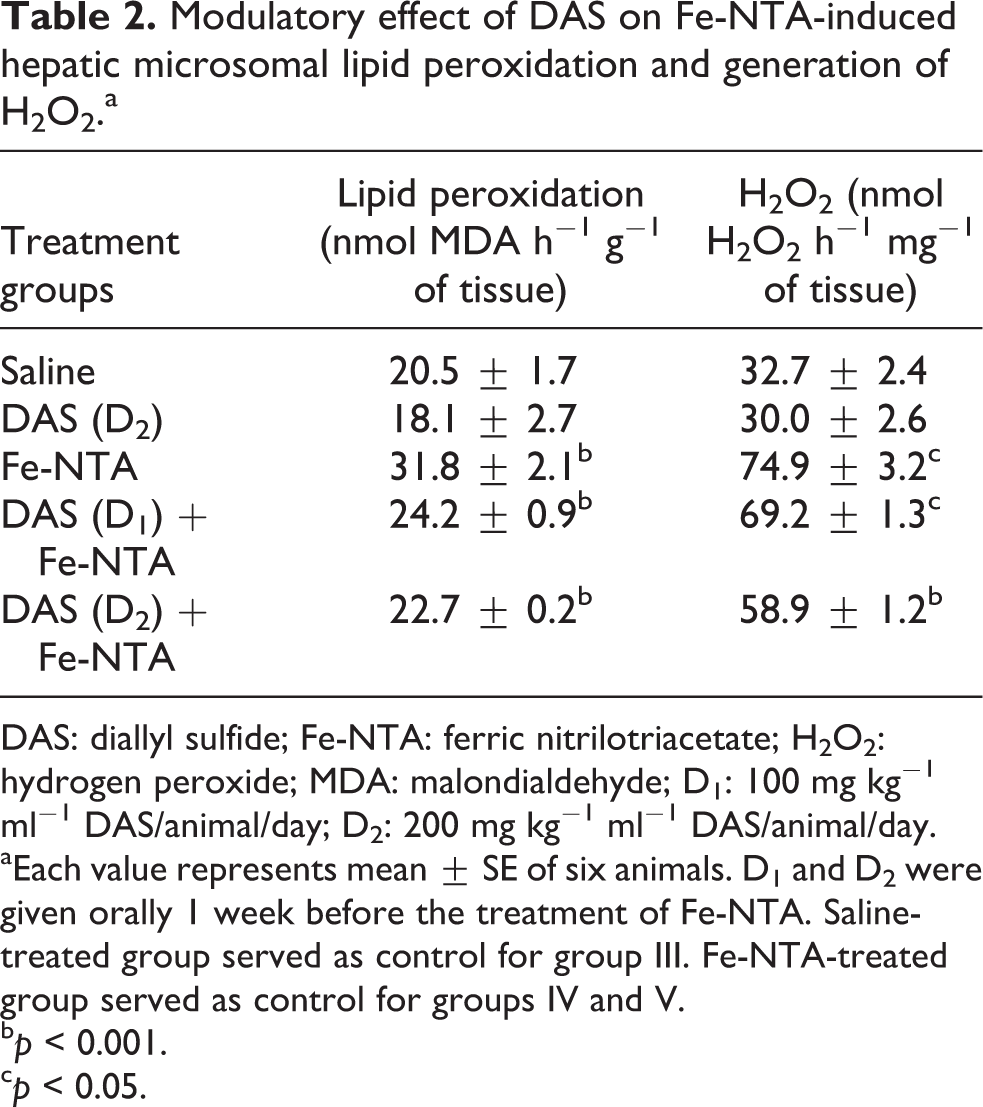
DAS: diallyl sulfide; Fe-NTA: ferric nitrilotriacetate; H2O2: hydrogen peroxide; MDA: malondialdehyde; D1: 100 mg kg−1 ml−1 DAS/animal/day; D2: 200 mg kg−1 ml−1 DAS/animal/day.
aEach value represents mean ± SE of six animals. D1 and D2 were given orally 1 week before the treatment of Fe-NTA. Saline-treated group served as control for group III. Fe-NTA-treated group served as control for groups IV and V.
b
c
Effect of pretreatment of rats with DAS on Fe-NTA-mediated induction of protein carbonyl
Fe-NTA (alone)-treatment results in a maximum of 2.65-fold increase in the DNPH incorporation into cystosolic proteins in rat livers. This increase in the incorporation of DNPH was reversed significantly in groups receiving a pretreatment with DAS as compared to Fe-NTA-treated group. The decrease in the incorporation of DNPH into cytosolic protein was dependent on the dose of DAS used. There was significant decrease (
Modulatory effect of DAS on Fe-NTA-mediated induction of protein carbonyl.a

DAS: diallyl sulfide; Fe-NTA: ferric nitrilotriacetate; D1: 100 mg kg−1 ml−1 DAS/animal/day; D2: 200 mg kg−1 ml−1 DAS/animal/day.
aEach value represents mean ± SE of six animals. D1 and D2 were given orally 1 week before the treatment of Fe-NTA. Saline-treated group served as control for group III. Fe-NTA-treated group served as control for groups IV and V.
b
c
Discussion
A number of agents particularly those that are part of our diet afford protection against the onset of various diseases including cancer. Many of their activities have been related to their abilities to enhance the carcinogenic metabolizing enzymes or bind with them, thus decreasing their effective critical concentration and acting as antioxidants to counteract the increased amount of oxidants generated by toxicants. Dietary intake of natural antioxidants has also been suggested to be useful against oxidant-mediated toxicity. 30 –33
It is observed that the toxic manifestations of Fe-NTA gradually increase with the increasing age of animals. A dose of Fe-NTA, which produces almost 100% mortality in aged rats, causes 70% mortality in adults, 30% in pups, 20% in litters, and less than 10% in neonates. 14 In this study, i.p. injection of Fe-NTA to rats induced alterations in both serum and tissue biomarkers of hepatotoxicity. These alterations included significant increases in the serum levels of ALT and AST enzymes and also significant increases of lipid peroxidation by-product MDA and the activity of GSH metabolizing enzymes (GR and GST) with a decrease of reduced GSH content in rat livers. It is known that Fe-NTA is a potent hepatotoxic chemical agent used for the induction of hepatotoxicity in laboratory animal models. The hepatotoxic effect of Fe-NTA in the current study was similar to that demonstrated in previous studies. 4,12,31
DAS, a major component of garlic, diminished Fe-NTA-mediated hepatic oxidative stress, toxicity, and, thereby, suppressed hyperproliferative response. DAS possesses strong antioxidant properties and has earlier been shown possessing anticancer and antimicrobial properties. 34 –40 Our earlier studies have shown Fe-NTA-induced oxidative stress and renal damage in rats. 41 Since liver is a major metabolic organ in the body, it was important to understand the function of DAS on Fe-NTA-induced hepatic damage. The results in this study clearly indicate that pretreatment of animals with DAS attenuated the Fe-NTA-mediated hepatic toxicity and thereby suppresses hyperproliferation. Earlier, some studies showed protective effect of DAS in gentamicin-induced oxidative stress preserving the activity of the antioxidant enzymes in kidney and preventing nitrosamine tumorigenesis. 42,43
It has also been shown earlier that Fe-NTA induces oxidative stress in liver by depleting the levels of decreasing antioxidant enzyme activities with the concomitant increase in the production of lipid peroxides and H2O2. 4,44 The involvement of oxidative stress in Fe-NTA-treated animals is also evident from the present study where an increase in the formation of hepatic protein carbonyl, an indication of oxidation of cellular protein as a result of increased generation of oxidant was observed. The protective effect of DAS may be due to its scavenging action on the hydroxyl radicals generated in response to Fe-NTA and secondly augmenting the activities of GSH and antioxidant enzymes.
DAS acts as an antioxidant, which is evident from its protection against Fe-NTA-induced hepatic damage and generation of oxidants by Fe-NTA. A decrease in the level of serum transaminases in DAS-pretreated groups also suggest that prophylaxis of DAS is effective in improving hepatic function of Fe-NTA-treated group. DAS abrogated the Fe-NTA-induced hepatic lipid peroxidation and reduced enhanced generation of H2O2 and formation of hepatic protein carbonyl, showing its strong role in reducing Fe-NTA-mediated toxicity in this study. The exact mechanism by which DAS suppresses Fe-NTA-mediated hepatic toxicity and hyperproliferation is however not known.
Conclusion
DAS may be a useful antioxidant agent for the prevention of Fe-NTA-induced hepatic damage in rats. This study suggests that dietary intake of DAS or garlic with food formulations may be beneficial for patients who suffer from liver diseases due to oxidative stress. In summary, the present data show that in vivo antioxidant effect of DAS may be involved, at least in part, in its protective effect in rats with Fe-NTA-induced hepatotoxicity.
Footnotes
Acknowledgment
This research project is supported by Research Center of the Center for Female Scientific and Medical Colleges, Deanship of Scientific Research, King Saud University.
Conflict of interest
The authors declared no conflicts of interest.
