Abstract
Monoterpenes present in the essential oils exhibit anti-inflammatory properties. In this study, we investigated the preventive effect of alpha-pinene (AP), a monoterpene, against isoproterenol (ISO)-induced myocardial infarction and inflammation in Wistar rats. Male Wistar rats were pretreated with AP (50 mg/kg body weight (bw)) administration for 21 days and ISO (85 mg/kg bw) was administered subcutaneously for last two consecutive days (20th day and 21st day). We noticed that there was an increased activity of cardiac marker enzymes in ISO-treated rats. We also observed that elevated levels of lipid peroxidative indices decreased activities of antioxidant status in plasma, erythrocyte, and heart tissue in ISO-induced rats. Furthermore, ISO-treated rats showed an increase in the levels of inflammatory mediators like tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) in the serum. Besides, we confirmed the upregulated expression of TNF-α, IL-6, and nuclear factor kappa-light-chain-enhancer of activated B cells in ISO-induced rat heart tissue. Conversely, we found that AP pretreatment significantly decreased levels of cardiac markers like serum cardiac troponin T and cardiac troponin I, lipid peroxidative markers, and restored antioxidants status in ISO-treated rats. Besides, AP administration attenuated ISO-induced inflammatory marker expression. The present findings demonstrated that AP significantly protects the myocardium and exerts cardioprotective and anti-inflammatory effects in experimental rats.
Introduction
Cardiovascular disease (CVD) is the most significant cause of death worldwide. The incidence and mortality due to CVDs are alarmingly increasing despite the availability of advanced treatment options. 1 About 32.4 million myocardial infarctions (MIs) and strokes were reported worldwide every year. 2 The CVDs occur due to several pathological reasons, which have resulted in CVD-linked mortality. 3 The pathology for CVD includes atherosclerosis, coronary heart disease, congestive heart failure, and MI. 4 Several factors are responsible for the pathogenesis of MI, such as environmental, physical, chemical, and emotional stress. 5 Additionally, the intake of tobacco, excessive alcohol consumption, sedentary lifestyle, and obesity may also contribute substantially to MI. 6 The occurrence of MI can be observed by analyzing different cellular and molecular indices that include serum levels of troponin-1, cardiac tissue-specific marker enzymes, inflammatory marker expression, and oxidative damage indices. 7
There were several experimental protocols available to develop heart failure in animal models. 8 The isoproterenol hydrochloride (ISO) is a catecholamine and it is a nonselective β-adrenergic agonist, which induces oxidative damage-mediated MI in experimental animals. 9 Studies show inflammatory responses, oxidative stress, and development of cardiomyopathy during ISO administration. 10 More specifically, the administration of ISO induced myocardial impairment, multifunctional rate, coronary microcirculatory effects, hypoxia, and membrane permeability changes. 11 Oxidative damages have been well documented during ISO treatment, which has been linked with myocardial ischemic injury. 12 The ISO treatment induced free radicals and decreased the antioxidant defense system. 13 It has been well documented that ISO generates cardiac necrosis by redox imbalance, higher calcium overload, altered cellular metabolism, elevated cyclic adenosine monophosphate levels, altered electrolyte profile, intracellular acidosis, and altered membrane permeability. 14 Several clinical studies illustrate the role of free radicals in endothelial damage and MI. 15 Thus, the pathophysiological aspects of ISO-induced myocardial necrotic rat models are comparable with those developing in human beings. 16 Further, the rat model of ISO-mediated MI is a reliable experimental model for evaluating the efficacy of cardioprotective agents. 17
Inflammation plays an important role in the pathogenesis of MI. The activation of cytokine in the damaged myocardial tissue has been well documented. 18 Furthermore, MI effects in the upregulation of cytokines lead to tissue infiltration by the inflammatory cells. 19 The pro-inflammatory cytokines expedite MI, which is accompanying severe congestive heart disease. 20 The pro-inflammatory cytokines like tumor necrosis factor α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6) augment inflammatory reactions during MI. 21 The TNF-α triggers neutrophil migration into the ischemic region of infarcted myocardial tissue. 22 IL-6 is a pleiotropic cytokine involved in inflammatory responses to stress in the myocardial tissue. 19 MI-mediated adverse reactions are characterized by the activation of cellular signaling molecules like nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB). 23 NF-κB is a key regulator in the condition of inflammation and apoptosis. In MI, phosphorylation of NF-κB initiates the intracellular signaling cascade and finally induces pro-inflammatory cytokines, such as TNF-α, IL-6, IL-1β as well as other inflammation-related proteins that induce various pathophysiological changes. 24
The potential side effects of the currently available drugs for CVDs encourage the research to develop natural products-based nontoxic medicinal compounds as a source of drugs for CVD treatment. 25 Natural medicinal compounds are important nutritional supplements that provide potential health benefits in the prevention of CVD-linked mortality. 26 Terpenes are the major components of plant essential oils. 27 Monoterpenes present in the essential oils show antioxidant and anti-inflammatory properties. 28 Alpha-pinene (AP) is a major component in essential oils of pine trees that show significant anti-inflammatory property. 21 The AP shows anti-inflammatory property in several experimental models. Bae et al. reported that AP effectively attenuated cerulean-mediated pancreatic damage in experimental animals. 29 Pinheiro et al. showed the protective role of AP in gastric ulcer experimental models. 30 Furthermore, AP exhibits antiproliferative effect against hepatocarcinoma and melanoma. 31 Moreover, AP attenuates UVA radiation-mediated inflammation through modulation of matrix metalloproteinase expression in mouse skin. 32 However, the cardioprotective effect of AP has not yet been studied. Hence, we investigated the preventive effect of AP on ISO-induced cardiac markers, lipid peroxidation, antioxidant status, and cytokine levels in experimental animals.
Materials and methods
Experimental animals
Wistar albino rats of male, the weight of 160–180 g, were used for the experiments. All the rats were maintained under standard conditions in the clean air-conditioned room at a temperature 25 ± 2°C and 12-h:12-h dark and light cycles. Animals were fed with standard diets and drinking water was assessed to animals ad libitum. The experimental protocol was approved by the Institutional Animal Ethical Committee.
Chemicals
AP, ISO, phosphate-buffered saline, and bovine serum albumin (BSA) were obtained from Sigma Aldrich (St Louis, Missouri, USA). All the primary anti-TNF-α, IL-6, NF-κB, and secondary horseradish peroxidase (HRP)-conjugated antibodies were purchased from Cell signaling (Danvers, Massachusetts, USA). All the other chemicals of analytical grade used in the study were purchased from HiMEDIA (Mumbai, India).
Dose fixation study
MI was stimulated by subcutaneous (s.c.) injection of 85 mg/kg ISO to the experimental animals, as described earlier. 33 The first injection was given at 11 a.m. on the first day and the second injection at 11 a.m. on the next day.
A dose-dependent study was carried out to determine the effective dose of AP by evaluating serum marker enzyme activities in ISO-induced rats. AP was given at different doses (i.e. 25, 50, and 100 mg/kg body weight (bw) to different groups of animals. AP was administered orally once in a day for 7 days and ISO was given s.c. for the last two consecutive days. Among the three doses of 50 mg/kg bw, the dose was more effective than the other two doses. Therefore, 50 mg/kg bw of AP was used for further study.
Experimental design for MI studies
All the rats were classified into four experimental groups and each group contained six rats. Dimethyl sulfoxide (DMSO) (0.1%) was served as control rats (group I). The AP (50 mg/kg bw) in 0.1% DMSO was preadministered for 21 consecutive days (group II). Group III rats received ISO treatment at 85 mg/kg bw, s.c. on the 20th day and 21st day. Group IV rats received 50 mg/kg bw, orally for 21 days plus ISO (85 mg/kg bw, s.c. on the 20th day and 21st day.

After 24 h of the last ISO and/or AP treatment (22nd day), the rats were anesthetized using ketamine hydrochloride (24 mg/kg bw) and euthanized by cervical dislocation.
Serum preparation
Blood samples were collected in a dry test tube and allowed to coagulate at ambient temperature for 30 min. The serum was separated by centrifugation at 2000 r/min for 10 min. We measured the levels of creatine kinase (CK), creatine kinase-myocardial band (CK-MB), cardiac troponin T (cTnT), cardiac troponin I (cTnI), and inflammatory cytokines in the rat serum.
Plasma preparation
Blood samples collected in heparinized centrifuge tubes were centrifuged at 2000 r/min for 10 min and the plasma was separated by aspiration. We measured the lipid peroxidative markers and antioxidant status in the rat blood plasma and heart tissue homogenates.
Tissue homogenate preparations
Heart tissue (250 mg) was sliced into pieces and homogenized in the appropriate buffer in cold condition (pH 7.0) to give 10% homogenate (wt%/vol%). The homogenates were centrifuged at 1000 r/min for 10 min at 0°C in a cold centrifuge. The supernatant was separated and used for various biochemical estimations and Western blot analysis.
Erythrocyte preparation
After the separation of plasma, the buffy coat enriched in white cells was removed and the remaining erythrocytes were washed three times with physiological saline and made up to specific volume. A known volume of erythrocyte was lysed with hypotonic phosphate buffer at pH 7.4. The hemolysate was separated by centrifugation at 2500 r/min for 10 min and the supernatant was used for the estimation of enzymatic antioxidants.
Blood pressure measurement
Blood pressure and heart rate were measured on the initial (0 days) and final day (22nd day) of the experimental period (after treatment) by the tail-cuff method, as described previously. Briefly, the animals were kept in plastic retainers. Then, a pulse sensor was fixed with the tail of the rats. The blood pressure and heart rate values were measured using Model MK-2000. Data were reported as the average of sequential blood pressure measurements.
Activities of cardiac parameters
The CK-MB is an isoenzyme mainly present in the heart muscle tissue. The activity of CK-MB and CK was assayed according to the methods of Okinaka et al. 34 The cTnT and cTnI are the major regulatory markers that control cardiac actin and myosin interaction. The serum cTn T and I were measured using a commercial kit, respectively. The data were quantitatively calculated, as per the kit provided by the manufacturer (ELISA). 35,36
Lipid peroxidative markers in plasma and heart tissue
Isoproterenol produces superoxide anion and hydrogen peroxide upon oxidation at the cellular environment. This ISO-induced reactive oxygen species (ROS) eventually damages membrane lipids through oxidative damage mechanisms. Thiobarbituric acid reactive substances (TBARS) and lipid hydroperoxides (LHPs) are the oxidative stress markers elevated during ISO treatment. The plasma and heart TBARS levels were estimated by the method of Niehaus and Samuelson. 37 The reaction of malondialdehyde and TBA in the acetic acid was measured. Estimation of plasma and cardiac tissue LHPs was carried out by the method of Jiang et al. 38
Determinations of antioxidant status
Antioxidant enzymes play a major role in the redox homeostasis of tissues that scavenges ISO-induced free radicals. Superoxide dismutase (SOD) scavenges ROS by converting superoxide to hydrogen peroxide and molecular oxygen. The SOD activity was assayed by the method of Kakkar et al. 39 Catalase (CAT) degrades hydrogen peroxide into water and oxygen, thereby maintaining redox status in the cellular milieu. The CAT activity was assayed by the procedure of Sinha. 40 Glutathione peroxidase (GPx) catalyzes the reduction of hydrogen peroxide to water via the oxidation of reduced glutathione (GSH) into glutathione disulfide. The activity of GPx was assayed by the method of Rotruck et al. 41 The GSH content remaining after the reaction was measured. Estimation of GSH in plasma and the heart tissue was done by the method of Ellman. 42
Measurement of inflammatory cytokines
The levels of pro-inflammatory cytokines, that is, TNF-α and IL-6, were generally elevated during MI. The serum level of TNF-α and IL-6 was measured by ready to use commercial ELISA kit, as per the instructions given by the manufacturer (Cusabio Technology LLC, Wuhan, China). Briefly, the monoclonal antibody specific for TNF-α and IL-6 precoated microtiter plates was added with the experimental samples. A reaction of biotin-conjugated antibody specific for TNF-α and IL-6 with avidin-conjugated HRP was measured using a substrate solution. The developed color intensity was measured using a Tecan multimode reader (Männedorf, Switzerland).
Western blot analysis
The heart tissue was homogenized in radioimmunoprecipitation assay buffer containing phenyl methyl sulfonyl fluoride, as per the previous report. 9 The protein concentration was quantified by Lowry et al. method. 43 The protein samples stayed equally loaded into the gel (10% sodium dodecyl sulphate-polyacrylamide gel electrophoresis) and transferred onto polyvinylidene difluoride (PVDF) membrane. Then, nonspecific binding proteins were blocked with 5% BSA for 2 h. Further, the membrane was incubated using primary antibodies such as TNF-α, IL-6, and NF-κB overnight at 4°C. After finishing this reaction, the membranes were incubated with HRP-linked secondary antibodies for 2 h at 37°C. The membranes were washed three times with 10-min interval, and the bands were identified by enhanced chemiluminescence Western blotting substrate reagent.
Statistical analysis
The experimental results were expressed as the means ± standard deviation of six independent experiments (n = 6). The results were statistically analyzed by analysis of variance for the comparison of group mean values. Further, the mean values were compared by Duncan’s multiple range test (DMRT) using SPSS 17.0 software. The p-values ≤0.05 were considered as statistically significant.
Results
Dose fixation study
We observed increased activities of CK, CK-MB, and elevated levels of cTnT and cTnI in the serum of ISO-treated rats when compared to control rats. Conversely, AP administration prevented ISO-induced expression of cardiac marker enzymes in the rat serum. Among the three different concentrations (25, 50, and 100 mg/kg bw) of AP administration studied, we noticed that 50 mg/kg bw showed a higher cardioprotective effect than the other two doses (Table 1). Therefore, we selected 50 mg/kg bw of AP pretreatment for further MI experiments.
Effect of AP on CK, CK-MB, cTnT, and cTnI in the serum of control and ISO-induced rats.a

aValues are given as means ± SD (n = 6). Values not sharing a common marking superscript (a, b, c) are different significantly at p value ≤0.05. (DMRT).
DMRT: Duncan’s multiple range test; SD: standard deviation; ISO: isoproterenol; AP: alpha-pinene; CK: creatine kinase; cTnT: cardiac troponin T; cTnI: cardiac troponin I.
Effect of AP on ISO-induced hemodynamic measurements
ISO has been known to increase heart muscle contraction, thereby increasing heart rate and vasodilation in the experimental models. In this study, we noticed that the ISO treatment caused a significant (p < 0.05) decrease in systolic blood pressure, diastolic blood pressure, and increased heart rate when compared to control rats. Conversely, AP pretreatment restored the systolic blood pressure, diastolic blood pressure, and reduced heart rate in ISO-induced rats (Figure 1(a) and (b)).
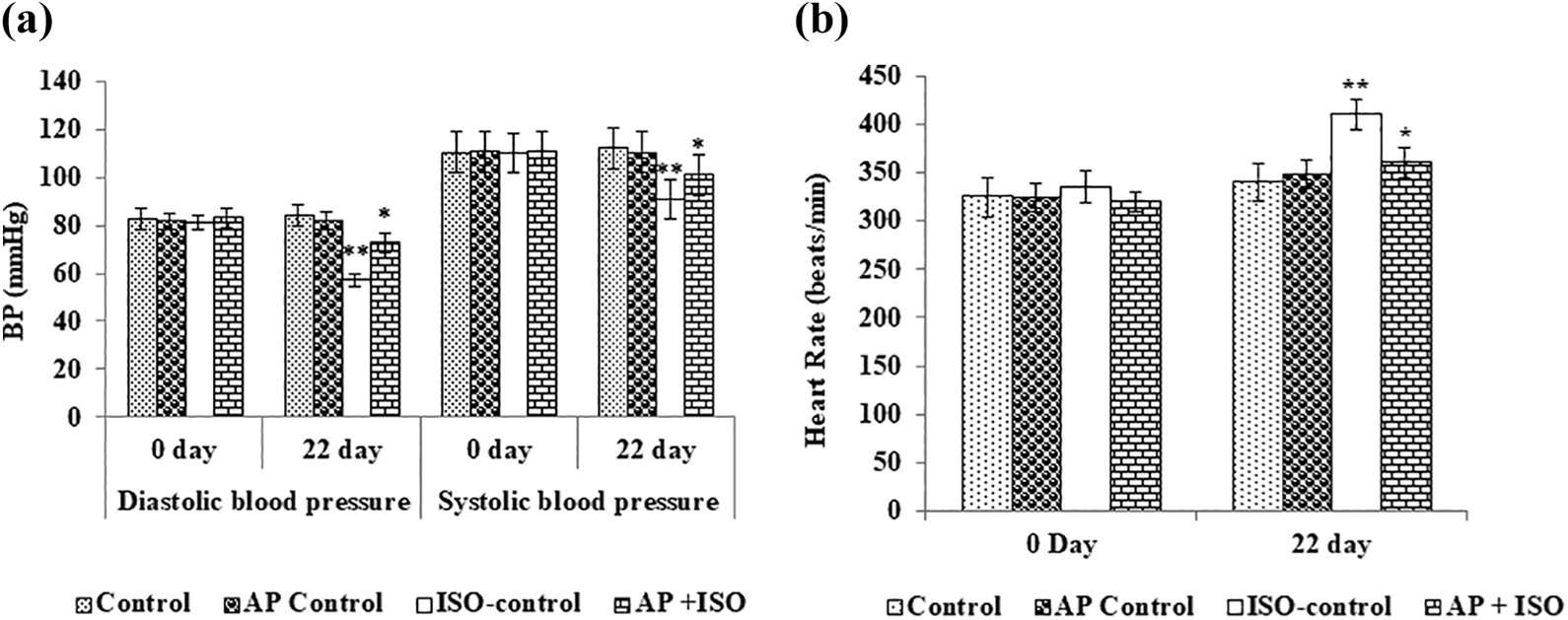
Effect of AP on ISO-induced blood pressure and heart rate measurement. (a) Effect of AP on ISO-induced blood pressure measurement. (b) Effect of AP on ISO-induced heart rate measurement. Data are given as means ± SD (n = 6). The data not sharing a common superscript are different significantly at p value ≤0.05 (DMRT). DMRT: Duncan’s multiple range test; SD: standard deviation; ISO: isoproterenol; AP: alpha-pinene.
Effect of AP on ISO-induced CK, CK-MB, cTnT, and cTnI levels in the serum
Cardiac specific enzymes/markers are released into the bloodstream when the cardiac tissue is infracted. In this study, rats treated with ISO caused significantly (p < 0.05) elevated activities of CK, CK-MB, and augmented levels of cTnT and cTnI when compared to control rats. Conversely, the AP pretreatment significantly prevented the ISO-mediated elevation of serum cardiac marker enzymes in the rat serum (Figure 2(a) and (b)).

(a, b) Effect of AP on CK, CK-MB, cTnT, and cTnI in the serum of control and ISO-treated rats. Data are given as means ± SD (n = 6). The data not sharing a common superscript (a, b, c) are different significantly at p value ≤0.05 (DMRT). DMRT: Duncan’s multiple range test; SD: standard deviation; ISO: isoproterenol; AP: alpha-pinene; CK: creatine kinase; cTnT: cardiac troponin T; cTnI: cardiac troponin I.
Effect of AP on ISO-induced lipid peroxidation and antioxidant status
ISO treatment caused an increase in the thiobarbituric acid reactive substance (TBARS)and lipid hydroperoxide (LOOH) levels in the plasma and heart tissue when compared to the control group (Table 2). AP administration significantly reduced the ISO-induced TBARS and LOOH formation. ISO-treated rats showed significantly (p < 0.05) reduced activities of enzymatic antioxidants like SOD, CAT, and GPx in erythrocytes and heart tissue and decreased levels of GSH when compared to the control group (Tables 3 and 4). However, AP treatment had significantly improved the antioxidant status as compared to ISO-treated rats. There were no significant differences observed in the control and AP alone treatment group.
Effect of AP on the levels of TBARS and LOOH in the plasma and heart of control and ISO-induced rats.a

a Values are given as means ± SD (n = 6). Values not sharing a common marking superscript (a, b, c) are different significantly at p value ≤0.05 (DMRT).
ISO: isoproterenol; AP: alpha-pinene; TBARS: Thiobarbituric acid reactive substances; DMRT: Duncan’s multiple range test.
Effect of AP on the activities of SOD, catalase, and GPx in erythrocytes and GSH in the plasma of control and ISO-induced rats.a

a Values are given as means ± SD (n = 6). Values not sharing a common marking superscript (a, b, c) are different significantly at p value ≤0.05 (DMRT).
ISO: isoproterenol; AP: alpha-pinene; SOD: superoxide dismutase; GSH: glutathione; GPx: glutathione peroxidase; SD: standard deviation; DMRT: Duncan’s multiple range test; U*: enzyme required to scavenge the chromogen formed by 50% in 1 min; U**: µmol of hydrogen peroxide decayed per minute. U@: µmol of GSH required per minute.
Effect of AP on the activities of SOD, catalase, GPx, and GSH in the heart of control and ISO-induced rats.a

a Values are given as means ± SD (n = 6). Values not sharing a common marking superscript (a, b, c) are different significantly at p value ≤0.05 (DMRT).
ISO: isoproterenol; AP: alpha-pinene; SOD: superoxide dismutase; GSH: glutathione; GPx: glutathione peroxidase; SD: standard deviation; DMRT: Duncan’s multiple range test; U*: enzyme required to scavenge the chromogen formed by 50% in 1 min; U**: µmol of hydrogen peroxide decayed per minute. U@: µmol of GSH required per minute.
Effect of AP on pro-inflammatory cytokines level in the serum
The serum level of TNF-α and IL-6 was significantly increased in ISO-treated rats when compared to control rats. Nevertheless, AP pretreatment significantly decreased the TNF-α and IL-6 levels in ISO-treated rats (Figure 3).

Effect of AP on pro-inflammatory cytokines level in the serum of control and ISO-treated rats. Data are given as means ± SD (n = 6). The data not sharing a common superscript (a, b, c) are different significantly at p value ≤0.05 (DMRT). DMRT: Duncan’s multiple range test; SD: standard deviation; ISO: isoproterenol; AP: alpha-pinene.
Effect of AP on ISO-induced inflammatory markers expression by Western blot analysis
The ISO administration significantly upregulated TNF-α expression along with elevated expressions of IL-6 and NF-κB as compared to control rats, whereas AP pretreatment significantly prevented ISO-induced overexpression inflammatory markers. No significant change was observed in normal control and AP alone treated group (Figure 4(a) and (b)).
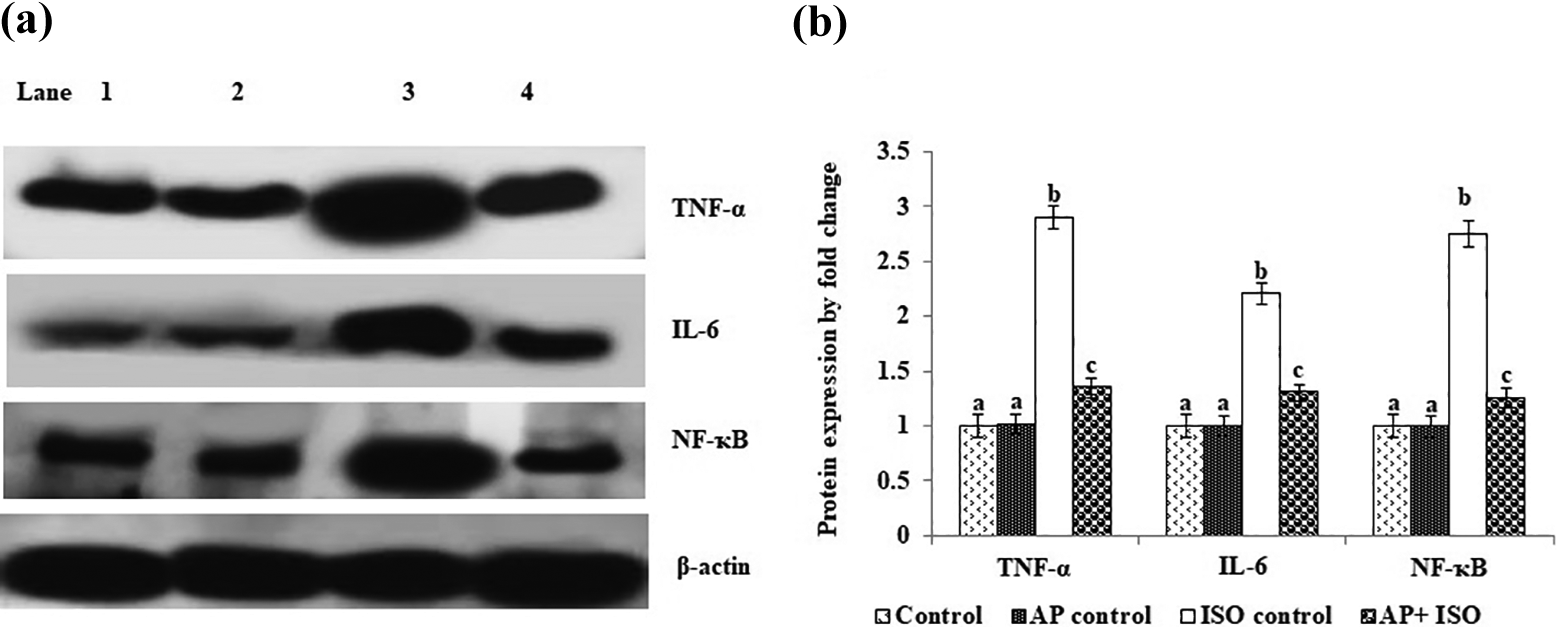
Effect of AP on TNF-α, IL-6, and NF-κB protein expressions in the heart tissue of control and ISO-induced rats. (a) TNF-α, IL-6, and NF-κB protein expressions by Western blot analysis. Lane 1: control; 2: AP control; 3: ISO control; 4: AP + ISO. (b) Band intensity scanned by the densitometer. The graph depicts quantitation of three independent experiments (means ± SD) with data normalized by the control group with TNF-α, IL-6, and NF-κB as unit 1. Values not sharing a common superscript (a, b, c) differ significantly at p ≤0.05 (DMRT). DMRT: Duncan’s multiple range test; SD: standard deviation; ISO: isoproterenol; AP: alpha-pinene; TNF: tumor necrosis factor; IL: interleukin; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cell.
Discussion
Scientific evidence shows that specific interventions might reduce the MI; the natural products-based medicinal compounds have been well documented to prevent oxidative damage-mediated myocardial damages. 44 We observed the protective effect of AP against ISO-mediated MI by measuring the cardiac markers, lipid peroxidative markers, and antioxidant status and by analyzing the inflammation markers. MI is mainly characterized by both systolic and diastolic dysfunction. 16 In this study, ISO treatment significantly increased systolic and diastolic blood pressure, increased heart rate, and vasodilatation. ISO is a sympathomimetic amine that is very much similar to adrenaline and acts on β-adrenergic receptors; it increased the cardiac output, thereby significantly increasing the heart rate in the experimental rats. 45 Conversely, AP administration prevented ISO-mediated systolic and diastolic blood pressure and heart rate. Monoterpenes have been reported to inhibit ISO-mediated oxidative stress and mitochondrial dysfunction. 46 Further, AP has been reported as an antioxidant and prevents UVA-mediated oxidative stress in skin cells. 47
Abnormally elevated levels of CK-MB are a specific indication of myocardial injury. During MI, the plasma levels of CK and CK-MB have been increased in serum within 3–6 h. 48 The cardiac-specific markers like troponins T and I are highly specific as well as sensitive markers for cardiac diseases and also the desired markers for the diagnosis of MI. 49 CTn is a constituent of myofibrillary cardiac tissue. The cTnI was considered to be a specific marker of MI. 50 Stabilization of cardiac muscle membrane injury has been considered as a novel approach to prevent ISO-mediated MI. 25 In this study, the ISO-treatment caused a rise in the CK and CK-MB levels and cTn T and I levels, which reflect extensive myocardial injury. In contrast, AP pretreatment prevented ISO-induced CK, CK-MB, and cTn T and I levels. Monoterpenes have been shown to stabilize membrane injury, thereby diminishing the infarct size and myocardial enzymes like CK and CK-MB, lactate dehydrogenase, and cTnT in acute MI of rats. 51
Lipid peroxidation is a dynamic pathogenic process in cardiac injury conditions. The effect of isoproterenol on lipid peroxidation in the myocardial tissue of mice has been well documented. 52 In this study, we noticed augmented levels of TBARS and LHPs in ISO-treated rats. The peroxidative damages were exerted via the formation of quinone intermediates of ISO that react with molecular oxygen to produce ROS that causes membrane lipid peroxidation. 14 We noticed that AP treatment significantly prevented ISO-mediated lipid peroxidation in myocardial tissue. As AP is a potent antioxidant, it may scavenge free radicals produced by ISO and protect the myocardium. Porres-Martínez et al. illustrated the preventive role of AP against hydrogen peroxide-mediated free-radical generations in PC12 cells. 53 The AP may donate hydrogen atoms to the free radicals generated during ISO treatment, thereby terminating free-radical chain reactions.
Mitochondrial impairment, oxidative damages associated with elevated lipid peroxidation, and depletion of endogenous antioxidant systems in the myocardial tissue are the other prominent hallmarks of cardiac ischemia. 54 The myocardial enzymatic antioxidant system works in a coordinated manner to maintain cellular redox balances. 55 This redox balance has been disturbed by ISO-mediated myocardial injury. The alteration in the redox balance during ISO treatment in myocardial tissue has been well documented. 56 In the present study, the s.c. injection of ISO caused a significant decrease in SOD, CAT, GPx, and GSH in the heart tissue. This could be due to the utilization of cellular enzymatic and nonenzymatic antioxidants for the scavenging of ISO-induced free radicals. However, AP pretreatment prevented ISO-induced loss of SOD, CAT, GPx, and GSH in the cardiac tissue. Because AP prevents the formation of free radicals, we observed restoration of antioxidant system in the ISO plus AP pretreated experimental group. Ali et al. 57 reported that AP has increased the antioxidant system in HCl/ethanol-mediated ulcers in rats. AP also increases the total antioxidant capacity in primary rat neurons without any alterations in N2a cells. 58 Karthikeyan et al. 47 reported that the AP prevents UVA-induced loss of antioxidant enzymes in epidermal keratinocytes.
Inflammatory reactions and signaling to play a major role in the CVD and varieties of synthetic drugs were used as the anti-inflammatory agents to diminish the myocardial damage. 59 Recent study reported that the excess of pro-inflammatory cytokines, TNF-α, and IL-6, is responsible for myocardial injury, which is mainly dependent on the NF-κB activation. 9 Generally, NF-κB is prevented from the activation of inhibitory kappa B (IκB) family. During MI conditions, IκB becomes phosphorylated and degraded by the IκB kinase enzymes. 60 Thus, IκB fails to control NF-κB, which further allows NF-κB activation and translocation from the cytosol to the nucleus and binds to the promoter sequence of target genes and causes transcription of pro-inflammatory cytokines, such as TNF-α and IL-6. 61 In the present study, the expression pattern of TNF-α, IL-6, and NF-κB was significantly increased in ISO-induced animals. Our results indicated that the pretreatment with AP significantly diminished TNF-α, IL-6, and NF-κB in the heart tissue when compared to control rats, suggesting that it has anti-inflammatory properties that protect the heart tissue. Monoterpenes from natural medicinal plants have been reported as promising preventive agents against inflammatory reactions and possess the ability to modulate the key signaling molecules involved in inflammation, such as pro- and anti-inflammatory cytokines. 62 We noticed that AP pretreatment significantly decreased TNF-α and IL-6 levels. Previously, AP and 1,8-cineole have been reported to inhibit TNF-α and IL-6 in experimental models. 63 Furthermore, AP exhibited anti-inflammatory activity through the suppression of NF-κB pathways in mouse model. 64
In conclusion, this study showed that AP pretreatment prevents ISO-mediated oxidative damage indices like lipid peroxidation, antioxidants loss, and cardiac markers expression in the heart tissue of experimental animals. Therefore, the cardioprotective activity of AP might be probably due to its antioxidant and anti-inflammatory properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
