Abstract
Arsenic is known to cause damage to the body’s immune system by inducing epigenetic changes. However, the molecular mechanism of this damage remains elusive. Here, we report that arsenic disrupts the morphology of lymphocytes, decreases cell viability, and results in abnormal proportions of T lymphocyte subsets. Moreover, our results revealed that arsenic can reduce global acetylation of histone H4 at K16 (H4K16 ac) in lymphocytes via decreasing the level of males absent on the first but upregulates mRNA and protein levels of the forkhead/winged-helix box P3 (Foxp3) gene by increasing the acetylation of histone H4 at K16 (H4K16) at the promoter of Foxp3. Finally, arsenic-induced dysfunction of regulatory T cells (Tregs) could be ameliorated by trichostatin A. Our research indicates that arsenic-induced immunosuppressive effect in human lymphocytes may be related to the acetylation of H4K16 at the promoter of Foxp3 and that histone deacetylase inhibitors may play a role in the prevention and treatment of immune injury caused by arsenic.
Introduction
Arsenic, an environmental toxicant widely found in soil, water, and air, 1 has been identified as a human carcinogen by the International Agency for Research on Cancer. 2 The main route of human arsenic exposure is through arsenic-contaminated water, food, and air. When arsenic intake exceeds arsenic excretion, it will accumulate in the human body, which eventually leads to arsenic poisoning. 3,4 At present, more than 200 million persons worldwide might be exposed to drinking water in which the level of arsenic exceeds a safe limit, 5,6 and approximately 60 million people in China are under threat of arsenic-contaminated groundwater. 7 Coal-burning arsenic poisoning occurs primarily in the Guizhou and Shaanxi Provinces of China, where burning coal with a high arsenic content in unventilated, indoor stoves is the major resource of energy for heating and cooking. 4 A study has demonstrated that chronic exposure to arsenic is detrimental to human health and arsenic exposure has even been linked to cancers. 8 Moreover, a large number of animal experiments and epidemiological data demonstrate that chronic exposure to arsenic can damage the immune system and ultimately lead to immune dysfunction. 9 –12
There is increasing evidence from animal studies, indicating a correlation between exposure to arsenic and decreased spleen and thymus indices, the production of immune cells, and the impaired development of immune organs. 13,14 Chronic arsenic exposure also significantly lowers the percentage of CD4+ T cell subsets in human peripheral blood and the secretion of interleukin-2, leading to impaired immune system function. 15,16 A study also showed that exposure to arsenic during pregnancy is associated with dysfunction of the immune system both at birth and later in life. 17 These studies suggest that arsenic exposure can cause lasting and irreversible damage to the immune system and its function. Nevertheless, the molecular mechanisms that underlie these unfavorable effects of arsenic on the immune system remain enigmatic.
Tregs are a subset of CD4+ T cells that play roles in the maintenance of self-tolerance and homeostasis. 18,19 The immunosuppressive activity of Tregs can be attributed to different mechanisms, including the secretion of cytokines and/or cell–cell contact inhibition or the induction of cell apoptosis and death through the perforin-granzyme B (PRF-GZB) pathway. 20,21 Forkhead/winged-helix box P3 (Foxp3), a member of the forkhead/winged-helix family, is a pivotal transcription factor required for the development and function of Tregs. 22 As indicated in the literature, the activity of Foxp3 is regulated by histone acetylation. 23 Acetylation and deacetylation are catalyzed by histone acetyltransferases (HATs) and histone deacetylases (HDACs), respectively. The dynamic balance between HATs and HDACs controls the structure of chromatin and the expression of genes. Histone acetylation usually occurs on lysine residues at the tails of histones H3 and H4, and the N-terminal tail of histone H4 contains four acetylated lysine sites (K5, K8, K12, and K16). Among these acetylated sites, H4K16 ac, the first histone modification discovered, plays a vital role in gene transcription. 24 MOF, also known as KAT8 or MYST1, is the main HAT of H4K16 ac and specifically involved in the modification of H4K16 ac. 25 A study demonstrated that the level of H4K16 ac is highly correlated with the deficiency or overexpression of MOF and that the loss of MOF in animals decreased the level of H4K16 ac, leading to cell cycle arrest and chromosome distortion. 26 Foxp3-mediated gene activation is related to H4K16 ac, which recruits MOF to the binding site of H4K16 and causes its acetylation. 27
Trichostatin A (TSA) is a histone deacetylase inhibitor (HDACI). By inhibiting HDAC activity, TSA promotes histone acetylation, thereby causing chromosomal remodeling, which in turn regulates the expression of specific genes. Recently, increasing evidence suggests that TSA enhances the acetylation of Foxp3, which plays a decisive role in the development and function of Tregs. 28,29 Therefore, TSA was used to intervene in human lymphocytes exposed to sodium arsenite (NaAsO2) to find an effective intervention measure to regulate changes in immune homeostasis and immune function due to arsenic poisoning.
Materials and methods
Lymphocyte isolation and culture
Peripheral blood samples from healthy young volunteers were collected in 5-mL anticoagulant tubes, and Ficoll gradient centrifugation was then used to isolate peripheral blood mononuclear cells (PBMCs). After separation, the isolated PBMCs were moved to a culture dish in Roswell Park Memorial Institute (RPMI)-1640 medium (Gibco-Invitrogen, California, USA) containing 4% fetal bovine serum. Subsequently, the culture dish was placed in an incubator containing 5% CO2 at 37°C for 24 h. Then, lymphocytes were purified due to their differential adherence times. After incubation for 24 h, the upper cell suspension contained only purified lymphocytes.
MTT assay
Cell viability was assessed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay according to the manufacturer’s instructions. The purified lymphocytes were counted with the trypan blue exclusion method, seeded in 96-well plates (2 × 106 cells/well) for 24 h, and then treated with different concentrations of NaAsO2 (0, 5, 10, 20, 30, and 40 μmol/L) for 12, 24, and 48 h. After that, 10 µL of 5 mg/mL MTT was added to each well, and the plates were incubated in the dark for 4 h at 37°C. Subsequently, 100 µL of formazan-dissolving solution was added to each well, and the plates were placed in an incubator and incubated continuously at 37°C for approximately 4 h until all the purple crystals had dissolved. Then, a microplate spectrophotometer (Bio-Rad Laboratories, Inc., Hercules, California, USA) was used to detect the absorbance at 570 nm (A 570). The experiment was repeated three times.
Hematoxylin and eosin staining
The infected cells were added evenly to slides and dried naturally. Cells were fixed in 95% ethanol for 15 min and stained with hematoxylin for 30 s, following which the dye was washed with tap water. Subsequently, the cells were stained with eosin for 2 min and washed with tap water. An ethanol gradient (70%, 90%, and 95%) was used to dehydrate cells for 30 s for each solution. Then, the cells were washed twice with 100% ethanol for 1 min/wash to dehydrate the cells. Xylene was then used to clear the cells for 15 min. Finally, the slides were mounted and observed under a light microscope.
Chromatin immunoprecipitation
A chromatin immunoprecipitation (ChIP) assay was conducted using an EZ-ChIP kit (Millipore, Upstate, USA). Briefly, the cells were collected by centrifugation at 2500 ×g for 10 min at 4°C. Cytomembranes were dissociated with Ez-Zyme lysis buffer supplemented with protease inhibitor cocktail II. Then, the samples were centrifuged at 2500 ×g for 10 min at 4°C, and the supernatants were discarded. The pellets (nuclei) were lysed with Ez-Zyme digestion buffer supplemented with protease inhibitor cocktail II to yield chromatic fragments, followed by incubation with protein A/G agarose at 4°C. Protein A/G agarose–antibody/chromatin complexes were eluted with ChIP elution buffer. Protein/DNA complexes were dissociated with 5 M NaCl at 65°C for 4 h and then incubated with RNase A at 37°C for 30 min and ethylenediaminetetraacetic acid -Tris plus proteinase K at 45°C for 2 h. Subsequently, a CFX96 real-time polymerase chain reaction (PCR) detection system (Bio-Rad Laboratories Inc.) was used to perform quantitative PCR. The primers used for Foxp3 promoter loci were as follows: 5′-TCCTCCCTGCCCAACTACAA-3′ (forward) and 5′-CGGCTAACAATCAGGGGCAT-3′ (reverse). The displayed results are relative to those of the input DNA.
Quantitative real-time reverse transcription-PCR
Total RNA was extracted from lymphocytes using TRIzol (Invitrogen, Carlsbad, California, USA), and complementary DNA (cDNA) was synthesized according to the manufacturer’s instructions. Then, using cDNA as a template, quantitative reverse transcription (RT)-PCR was carried out with Foxp3- and MOF-specific primers. The reactions were carried out as follows: 95°C for 30 s and 40 cycles of 95°C for 5 s, and 60°C for 30 s. The primer sequences are displayed in Table 1. GAPDH was used as an internal control, and the relative expression level of each target gene was calculated as 2− ΔΔ Ct , where ΔCt = Ct target − Ct GAPDH and ΔΔCt = ΔCt treatment − ΔCt control. Quantitative RT-PCR experiments were performed in triplicate.
Sequences of the primers used.

Western blotting analysis
After treatment with NaAsO2, the cells were moved to a clear tube and lysed with RIPA lysis buffer. Then, the protein concentration was measured using the bicinchoninic acid (BCA) assay. Proteins were separated by 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). After electrophoresis, the proteins were transferred onto a labeled polyvinylidene difluoride (PVDF) membrane at 300 mA for 1 h. Then, the membrane was blocked in tris-buffered saline with tween (TBST) containing 5% nonfat milk and incubated with primary antibodies overnight at 4°C. Subsequently, the membrane was washed with TBST three times (5 min/wash). Next, the membrane was transferred to a box containing secondary antibodies and incubated on a shaker for 2 h. Finally, the membrane was developed with an ECL kit (Millipore).
Analysis of lymphocyte subsets using flow cytometry
After exposure, the cells were moved to 1.5 mL tubes and collected by centrifugation. Then, cold phosphate-buffered saline containing 2% fetal bovine serum was used to wash the cells twice. The cells were incubated with Fc Block solution for 30 min at room temperature. Then, the cells were incubated with anti-BB515-CD4 antibody and anti-APC-CD25 antibody (Becton Dickinson and Company, New Jersey, USA) at 4°C for 30 min in the dark. The cytomembranes were destroyed by incubation with fix/perm buffer at 4°C for 45 min. Subsequently, the cells were stained with PE-Foxp3 antibody at 4°C for 45 min. Finally, the cells were analyzed by flow cytometry (Becton Dickinson Company).
Enzyme-linked immunosorbent assay
The culture supernatants were collected after centrifugation, and the concentrations of cytotoxic T lymphocyte-associated antigen 4 (CTLA-4), transforming growth factor β (TGF-β), PRF, and GZB were then measured according to the instructions of an enzyme-linked immunosorbent assay (ELISA) kit (Anoric Biotechnology Co. Ltd. Tianjin, China). Subsequently, the absorbance was measured at 450 nm (A 450). Finally, sample concentrations were obtained according to a standard curve.
TSA intervention
According to the immunosuppressive effects observed in lymphocytes exposed to NaAsO2, a dose of 20 μmol/L NaAsO2 was chosen as treatment for the exposure group in the TSA intervention test. The dose of TSA chosen was 50 nmol/L. After treatment, the protein expression of Foxp3 and H4K16 ac was detected by Western blotting, the level of H4K16 ac in the promoter region of the Foxp3 gene was detected by ChIP, the proportion of Tregs among CD4+ T cells was detected by flow cytometry, and the expression levels of cytokines were detected by ELISA.
Statistical analysis
Statistical analyses were conducted with SPSS 17.0 software. Data are presented as the mean ± standard deviation. Quantitative data from multiple groups were compared by one-way analysis of variance, and the least significant difference (LSD) and Dunnett tests (equal variance) were used for further pairwise comparisons. The p value <0.05 indicated statistical significance.
Results
Effects of arsenic exposure on cell viability
To explore the effects of arsenic exposure on lymphocytes’ viability, MTT assay was used to measure cell viability that was treated with different concentrations of arsenic for 12, 24, and 48 h. As shown in Figure 1(a), compared with the control group, cell viability increased in the 5 μmol/L NaAsO2-treated group but gradually decreased with the increase of exposure concentration and time, and the differences were statistically significant (p < 0.05). Since the insignificant changes in cell viability at 12 h and low cell viability at 48 h, 24 h of exposure was selected for subsequent study. However, cell viability was relatively low in the 40 μmol/L NaAsO2-treated group, so 5, 10, 20, and 30 μmol/L were chosen as arsenic exposure doses for the present study.
Arsenic induces the changes of the morphology in lymphocytes
Changes in the morphology of the lymphocytes were observed by hematoxylin staining (Figure 1(b) and (c)). After exposure to the indicated doses of NaAsO2, lymphocytes in the control group exhibited a round shape, intact membrane, and dark purple nuclei. However, among lymphocytes in the NaAsO2-treated groups (≥5 μmol/L NaAsO2), the morphology of the lymphocytes was irregular, and the membrane was ruptured. In particular, a mass of lymphocytes appeared as vacuoles in the 30 μmol/L NaAsO2-treated group.

Changes of cell viability and morphology of lymphocytes exposed to arsenic. (a) Cells were treated with different concentrations of NaAsO2 for 12, 24, and 48 h, and cell-viability rates were determined using the MTT assay. The results were represented as mean ± SD. (b, c) The morphological changes of lymphocytes after exposure to different doses of NaAsO2 for 24 h. SD: standard deviation; NaAsO2: sodium arsenite.
Arsenic decreased the global H4K16 ac modification of lymphocytes by inhibiting the activity of MOF
To explore whether arsenic exposure is correlated with global H4K16 ac modification of the lymphocyte genome, we compared the levels of H4K16 ac after treatment with different concentrations of NaAsO2. As shown in Figure 2(a), the levels of H4K16 ac were substantially reduced as the concentration of NaAsO2 increased.
Since the expression levels of MOF are crucial for the H4K16 ac modifications, we next investigated the mRNA and protein expression levels of MOF. As the exposure dose increased, the protein levels of MOF decreased (Figure 2(c)). However, the mRNA expression of MOF was not noticeably changed (Figure 2(b)), which suggested that arsenic may affect the stability of MOF protein and make it degradation, so as to reduce the modification level of H4K16 ac.
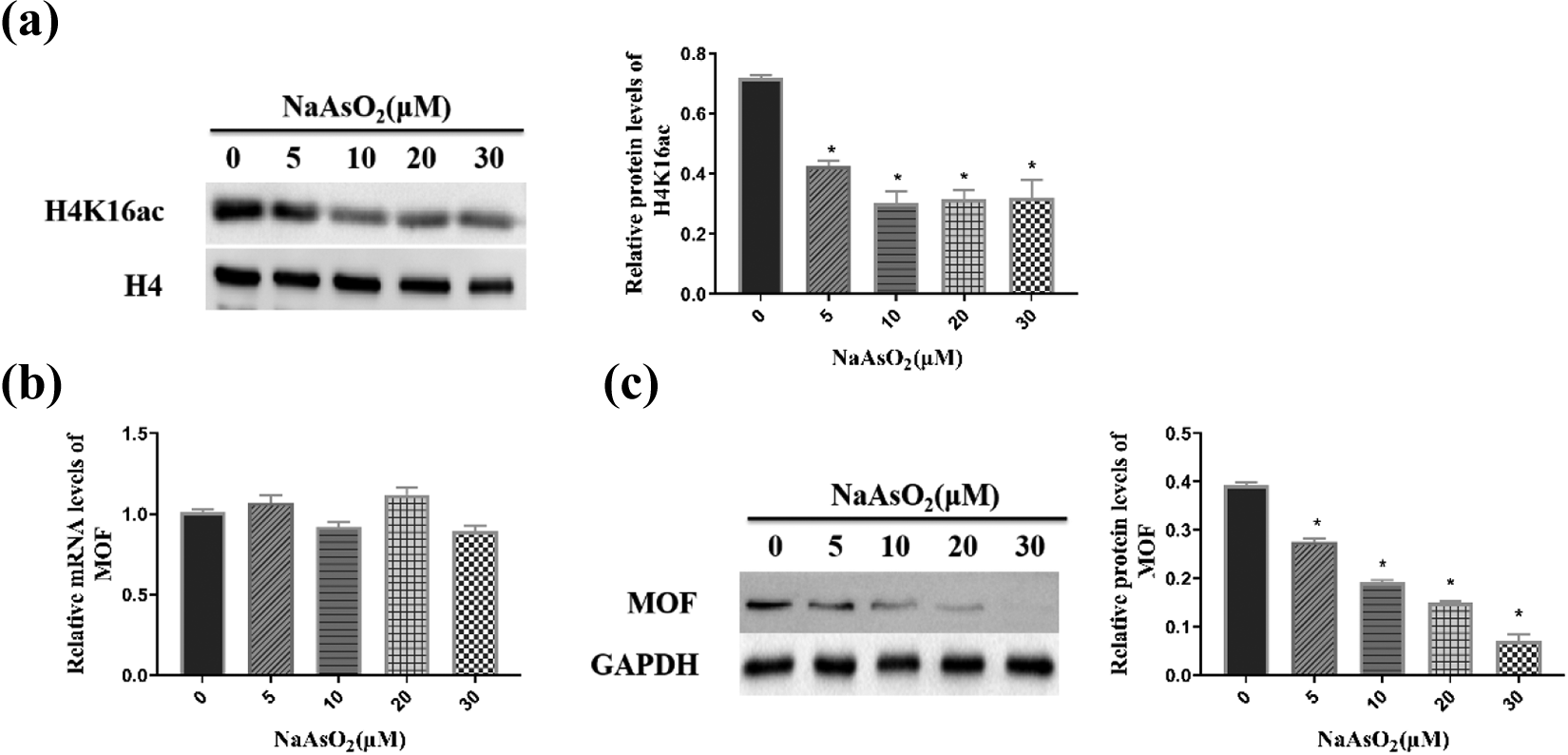
H4K16 ac modification in response to arsenic exposure. (a, c) Protein levels of H4K16 ac and MOF were tested by Western blotting. (b) The mRNA level of MOF was measured by RT-PCR. “*”indicates a significant change with respect to the control at p < 0.05. RT-PCR: reverse transcription polymerase chain reaction.
Arsenic increased H4K16 ac accumulation in the promoter region of the Foxp3 gene
To investigate the effect of arsenic exposure on H4K16 ac at the promoter region of the Foxp3 gene in lymphocytes, the enrichment of H4K16 ac at the promoter region of Foxp3 was detected by ChIP. As shown in Figure 3(a), the level of H4K16 ac modification at the promoter region of the Foxp3 gene in cells treated with 10–30 μmol/L arsenic was obviously augmented compared to that in the control group (p < 0.05). IgG, which served as a negative control, showed low levels of enrichment, indicating nonspecific binding to the promoter region of the Foxp3 gene.
Simultaneously, we tested the mRNA and protein expression of the Foxp3 gene after lymphocytes were exposed to NaAsO2. As exhibited in Figure 3(b) and (c), with increasing dose, the mRNA and protein expression levels were significantly upregulated in comparison to the control group (p < 0.05).

Effects of NaAsO2 on H4K16 ac enrichment in the promoter region of the Foxp3 gene and mRNA and protein expression of the Foxp3 gene. (a) ChIP analysis showed a distinct increase in H4K16 ac at the promoter region of the Foxp3 gene. (b) Foxp3 mRNA levels were measured by RT-PCR. (c) Protein levels of the Foxp3 gene were tested by Western blotting. “*” indicates a significant change with respect to the control at p < 0.05. RT-PCR: reverse transcription polymerase chain reaction; NaAsO2: sodium arsenite; Foxp3: forkhead/winged-helix box P3.
Arsenic altered the proportion of Tregs and secretion of cytokines
Previous studies have shown that arsenic may affect the immune function of lymphocytes by changing the proportion of Tregs. Therefore, we treated human lymphocytes with arsenic and detected the proportion of Tregs. As demonstrated in Figure 4(a) and (b), with increasing NaAsO2 concentrations, the proportion of Tregs was obviously elevated in comparison to the control group (p < 0.05).
Cytokines are essential to maintain the function of Tregs. Normally, an imbalance between pro- and anti-inflammatory cytokines will cause immune damage. Figure 4(c) shows that the levels of CTLA-4, PRF, TGF-β, and GZB were strongly enhanced following treatment with NaAsO2 (p < 0.05).

Effects of NaAsO2 on Tregs and secretion of cytokines. Tregs are the main effector of arsenic-induced immunosuppression. (a, b) Flow cytometry was used to test the proportion of Tregs. (c) ELISA was used to detect the expression of CTLA-4, TGF-β, PRF, and GZB. ‘*’ indicates a significant change with respect to the control at p < 0.05. CTLA-4: cytotoxic T lymphocyte-associated antigen 4; TGF-β: transforming growth factor β; NaAsO2: sodium arsenite; ELISA: enzyme-linked immunosorbent assay.
TSA regulated global levels of the H4K16 ac modification in lymphocytes but not H4K16 ac at the promoter region of the Foxp3 gene
TSA is an HDACI that can specifically inhibit the activity of HDAC and promote histone acetylation, thus participating in the transcriptional regulation of many genes. Therefore, we next assessed whether TSA can reverse changes in the expression of Foxp3 induced by arsenic through the regulation of global H4K16 ac or H4K16 ac of the Foxp3 gene promoter. Our results showed that global H4K16 ac was higher in the arsenic + TSA group than in the arsenic exposure group (Figure 5(a), p < 0.05). However, there was no difference in H4K16 ac of the Foxp3 gene promoter (Figure 5(b)). As shown in Figure 5(c), the protein level of Foxp3 was reduced in the arsenic + TSA group compared to the arsenic group (p < 0.05), suggesting that TSA intervention can decrease the expression level of Foxp3 by regulating global H4K16 ac modification.

Effects of TSA intervention on arsenic-induced Foxp3 expression. After TSA intervention, (a) the level of H4K16 ac increased and (b) H4K16 ac enrichment at the Foxp3 promoter was not significantly changed. (c) The protein expression of Foxp3 was reduced. *p < 0.05. Foxp3: forkhead/winged-helix box P3; TSA: trichostatin A.
TSA reduced the proportion of Tregs and secretion of cytokines
The effects of TSA on the proportion of Treg cells and secretion of cytokines were tested. Figure 6(a) and (b) shows that the proportion of Tregs was reduced in the arsenic + TSA group in comparison to the arsenic group (p < 0.05). As shown in Figure 6(c), the levels of CTLA-4, TGF-β, PRF, and GZB were higher in the arsenic + TSA group than in the control group (p < 0.05). Moreover, the levels of CTLA-4, TGF-β, PRF, and GZB were decreased in the arsenic + TSA group compared with the arsenic group (p < 0.05).

Effects of TSA intervention on the proportion of Tregs and secretion of cytokines. After TSA intervention, (a,b) the proportion of Tregs and secretion of cytokines (c) were reduced. *p < 0.05. TSA: trichostatin A.
Discussion
Lymphocytes, the basic components of the immune system are integral to maintaining immune homeostasis. 30 A study showed that 31 arsenic can cause damage to the immune system, but the underlying mechanism of this damage is poorly understood. In the present study, lymphocytes were treated with NaAsO2 to further explore the mechanism by which arsenic induces immune system dysfunction. As shown by HE staining, cells became swollen with irregular or ruptured membranes or shrunken nuclei after exposure to arsenic. The results of the MTT assay also showed a visible decrease in cell viability. The above results indicate that arsenic exposure may induce the changes of normal structure of lymphocytes and result in the decreases of lymphocytes viability rates.
Previous studies confirmed that arsenic can cause lymphocyte subset abnormalities, and abnormalities in Tregs are related to the occurrence of autoimmune diseases. 32,33 Foxp3 is a key regulator of immunosuppression in Tregs. 34,35 Mutations in the Foxp3 gene cause the defective development of Tregs, resulting in autoimmune diseases and immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome. 36 It has also been reported that the loss of Foxp3 expression reduced the immunosuppression of Tregs. 37 Our study reveals an increase in the mRNA and protein expression levels of the Foxp3 gene upon arsenic exposure. Furthermore, the results of flow cytometry showed that the proportion of Tregs in CD4+ T cells increased. Meanwhile, the levels of CTLA-4, TGF-β, PRF, and GZB secretion were also enhanced. CTLA-4, an important antigen on the surface of Tregs, competes with CD28 for B7 and then restrains T cell activation via blocking the CD28/B7 pathway. Additionally, CTLA-4 can promote secretion of the inhibitory cytokine TGF-β, which can enhance the conversion of naive T cells to Tregs, thereby enhancing their immunosuppressive function. 38 PRF and GZB are the main effectors of activated cytotoxic T lymphocytes and natural killer cells, which can induce target cell and CD4+ T and CD8+ T cells apoptosis and death to avoid an excessive immune response. 39 We found that the expression of Foxp3 was increased by arsenic, which in turn promoted the differentiation and development of Tregs, and that the latter secreted cytokines to suppress the activation of T cells. These results suggest that the arsenic-induced lymphocyte immunosuppressive effect occurs through abnormal expression of the Foxp3 gene. We then asked how the expression of the Foxp3 gene is regulated.
Recent studies have found that histone acetylation, which acts as an epigenetic code, is crucial to gene transcriptional activation. Evidence suggests that H4K16 ac modification levels are associated with gene expression and that abnormal levels of this modification mediate a variety of pathological processes. 40,41 The primary enzyme that acetylates histone H4K16 is MOF. Li et al. 42 found that knocking out MOF in mice led to a global reduction of H4K16 acetylation. In addition, there are studies showed that arsenic might reduce the expression of H4K16 by inhibiting the activity of MOF. 43,44 In our study, after the expression of MOF decreased, the expression of H4K16 also reduced, which is consistent with the above reports. We then wondered whether hyperacetylation of H4K16 at the promoter of the Foxp3 gene is the main cause of the upregulation of Foxp3 gene expression.
Hyperacetylated H4K16 has been found at the promoter sites of the Foxp3 gene and is highly associated with high Foxp3 expression. 27 To address this question, we detected the enrichment of H4K16 ac at the Foxp3 promoter region in arsenic-treated lymphocytes using ChIP-PCR. The level of H4K16 ac at the Foxp3 promoter region was increased after exposure to arsenic. Taken together, these data indicate that upon arsenic exposure, the increased expression of Foxp3 at both the mRNA and protein levels is regulated by hyperacetylation of histone H4K16 at the promoter region of the Foxp3 gene.
Histone acetylation is a dynamic and reversible process, and a study showed that deacetylase inhibitors can selectively regulate the transcript levels of genes involved in pro-inflammatory responses. 45 Liu et al. 46 found that TSA could downregulate expression of the Foxp3 gene in the mouse spleen and decrease the proportion of Tregs among CD4+ T cells. In our study, after TSA intervention, the level of global H4K16 ac modification, but not H4K16 ac at the promoter region of the Foxp3 gene, was increased compared with that in the arsenic-treated group. Moreover, both the proportion of Tregs among CD4+ T cells and the expression of cytokines were shown to be decreased. The above results suggest that TSA can increase the tolerance of lymphocytes to arsenic toxicity through upregulating the global H4K16 ac level and inhibiting the immunosuppressive function of Tregs; TSA thus plays a role in antagonizing the immunosuppressive effect induced by arsenic in lymphocytes.
Conclusions
In summary, our study demonstrates that arsenic-induced immunosuppressive damage in human lymphocytes is exacerbated through the hyperacetylation of H4K16 at the Foxp3 promoter, which thus upregulates the immunosuppressive effect of Tregs. Furthermore, the immunosuppressive effect of arsenic is reversible with TSA treatment, suggesting a role for TSA in the prevention and treatment of immune injury caused by arsenic poisoning.
Footnotes
Author contributions
The authors JC and JJ contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the key program of the NSFC [81430077] and the First-Class Discipline Construction Project in GuizhouProvince, Public Health and Preventive Medicine (No. 2017[85]).
