Abstract
There is a major concern that exposure to titanium dioxide (TiO2) nanoparticles (NPs) can have degrading effects on human health as well as mammary gland because of the increased use in numerous sorts of nanotech-based health care and food merchandise. Also, there is a scarcity in NP toxicity studies on the mammary gland; therefore, the aim of the present study was to compare toxicity caused by nano- and bulk-phase TiO2 particles on the human mammary gland in vitro. In comparison to bulk-TiO2 particles, nano-TiO2 cause a significant (
Keywords
Introduction
Nanotechnology is the science of using nanomaterials (NMs) or nanoparticles (NPs) for the benefit and innovation in medical and health research. 1 NPs, natural or manufactured, are generally considered to have either of its dimensions within a size range of 1–100 nm. 2 The recent advancement in nanotechnology has enabled the use of NPs in physics, chemistry, biology, electronics, antimicrobials, and agriculture. 3 Titanium dioxide NPs (TiO2-NPs or nano-TiO2) are one of the highly studied, enormously produced, and widely used NPs. Besides the popularity of metal nano-TiO2 for its high insolubility, thermal and flammable stability, and cross-metal non-reactivity, 4 nano-TiO2 have also been on top of priority list for numerous applications in plastics, rubber, paper, cosmetics, paints, medicines, and pharmaceuticals. 5 Nano-TiO2 is commonly used in various products because of their distinct properties, for example, ultraviolet radiation absorption, photocatalytic properties, or higher specific surface connected with catalytic properties. 6
Owing to their mega-production and wide use, the safety and toxicity concerns have equally become important for nano-TiO2. They behave differently, in comparison to bulk-TiO2, due to their small size and large surface area that increase their reactivity and penetration into human system 5 . It has been shown through various in vitro and in vivo studies that nano-TiO2 entered the bloodstream after gastrointestinal absorption and damaged the liver, spleen, kidney, lungs, brain, and reproductive organs. 7 –9 The nanotoxicity or damage occurs to cells and organs has been attributed, mainly because of generation of reactive oxygen species (ROS), 9 cytotoxicity, 10 genotoxicity, 11 and lipid peroxidation. 12 However, there is a scarcity in the reports of in vitro TiO2-NPs toxicity in mammary gland cell models (like MCF-7 cells). A recent and only report 12 suggested the cytotoxicity and oxidative stress induced by Zn-doped TiO2-NPs, and more such studies are required to assess, comprehensively, the effects of TiO2-NPs on mammary cells.
Besides the scarce reports on toxic effects of nano-TiO2 on mammary cells, no data could be retrieved on comparative assessment of bulk- and nano-TiO2 with same cells. The bulk (or micro) particles should also be tested for their toxicity, as suggested in a pioneer report by Lee et al. 13 when long exposure of pigment-grade TiO2 was seen to induce lung tumors in rats. Until that, the metal TiO2 (bulk or micro forms) had always been considered safe and used to be experimented as a negative control dust in a number of in vitro and in vivo toxicological studies. 4 Interestingly, in vitro mechanistic studies (especially for adverse outcomes) have been suggested to be deemed imperative for generation of primary data, which would pave the way for future in vivo trials. 5,14
Considering the abovementioned facts and gaps in knowledge, the comparative preclinical data of TiO2 (bulk and nano) toxicity was generated through experiments on morphology, cytotoxicity, genotoxicity, and apoptosis of human mammary epithelial cells (MCF-7 cells). Importantly, this study may be helpful in establishing an in vitro nanotoxicity model system for human mammary gland.
Materials and methods
Chemicals and biological materials
Nano-TiO2 (titanium (IV) oxide, nanopowder, <100 nm particle size, Cat. No. 634662), Bulk-TiO2 (titanium (IV) oxide, powder, particle size −325 mesh, Cat. No. 248576), Annexin V-fluorescein isothiocyanate (FITC) were from Sigma-Aldrich (St. Louis, MO, USA). Dimethylsulfoxide, ethanol, and acetic acid were from Hi-Media (Maharashtra, India). Fetal bovine serum (FBS) was from Gibco (Thermo Fisher Scientific, Waltham, MA, USA). Dulbecco's Modified Essential Medium and Ham's F-12 Medium (DMEM/F12), with
Culturing and maintenance of cells
Human mammary epithelial cells (MCF-7 cells) were cultured as monolayers in FBS (10%) and antibiotics (penicillin and streptomycin) supplemented DMEM/F12 at 37°C under a humidified atmosphere with 5% CO2 as suggested previously. After attaining 70–80% confluency, cells were detached using trypsin-EDTA solution (0.25% trypsin and1 mM EDTA) in sterile phosphate-buffered saline (PBS) (Ca2+ and Mg2+ free), counted in a Neubauer hemocytometer method and subcultured.
Preparation of nano-TiO2 stock solution
Nano-TiO2 and bulk-TiO2 stock (1 mg/mL) suspensions were prepared in PBS and sonication was performed using a probe sonicator (Branson Sonifier, B-12, Danbury, Connecticut, USA) at specified conditions (15 min at 4°C on ice), then different dilutions were made to expose human mammary epithelial cells with NPs.
Characterization of nano-TiO2 particles
The characterization of the nano-TiO2 was performed by dynamic light scattering (DLS) (Malvern Zetasizer-Nano-ZS, MAL1101853, UK) and by scanning electron microscopy (SEM) (Carl Zeiss Microscope, EVO18-15-34, Germany) following previously standardized lab protocols. 15
Evaluation of morphological changes in mammary epithelial cells
Morphological evaluation was done according to previously standardized protocol. 16 The human mammary epithelial cells were exposed to different concentrations (1–100 µg/mL) of bulk-TiO2 and nano-TiO2 for 12 h. After completion of the exposure period, the cells were observed by phase contrast inverted microscope (Olympus-CK40, Japan), and representative images were captured at 200× magnification. Morphological changes in mammary epithelial cells induced by bulk-TiO2 and nano-TiO2 were observed, recorded, and compared.
Interaction of bulk and NPs with mammary epithelial cells
Human mammary epithelial cells (MCF-7) were grown for 24 h on adherent cover-slips inserted in six-well plates. The cells were treated with nano-TiO2 and bulk-TiO2 concentration of 50 µg/mL for 12 h and morphological change was analyzed under a SEM as suggested earlier. 17 The adhered cells on cover-slips were later fixed with glutaraldehyde (2.5% in sodium cacodylate buffer, pH 7.4) for 2 h at 4°C, washed thrice with the same buffer for 5 min, kept at 4°C for 1.5 h after dropping one drop of osmium tetraoxide, sequentially dehydrated through a series of alcohol concentrations (25%, 30%, 50%, 85%, 95%, and 100%), and visualized under SEM after gold coating (Carl Zeiss Microscope, EVO18-15-34).
WST assay for cell proliferation and cytotoxicity
Water-soluble tetrazolium (WST) assay was carried out using the WST-1 Cell Proliferation Assay Kit (Cayman Chemicals, Item-10008883, Michigan, USA). Human mammary epithelial cells (MCF-7) were seeded (2 × 104 cells/well) in 96-well tissue culture plate and allowed to grow up to 24 h. Cells were then incubated with nano-TiO2 and bulk particles (1–100 μg/mL) for 6, 12, 24, and 48 h. Post incubation, the cell viability was calculated on the basis of color developed due to WST reagent. Color development was measured by reading absorbance spectrophotometrically at 570 nm in a microplate reader (Microscan (MS5605A) Electronic Co-operation of India Ltd, Maharashtra, India). Absorbance values of blank were subtracted from the measured values. The cell viability showed as a percent of the control value (cells grown in medium without exposure of the particles).
ROS generation by dichlorodihydrofluorescein diacetate assay
The cytotoxicity on the basis of ROS generation was done as per previously published protocol with slight modifications. 18 Cells were grown in adherent black 96-well plates for 24 h followed by treatment with different concentrations (1–100 μg/mL) of nano-TiO2 and bulk-TiO2 for 3, 6, 12, and 24 h. On completion of treatment, cells were washed with PBS and incubated with 40 µM dichlorodihydrofluorescein diacetate assay (DCFH-DA) for 30 min. Subsequently washed with PBS, lysed with NaOH, and measured the fluorescence (TECAN Infinite® 200 Pro, Microplate Reader, Switzerland) at excitation and emission wavelengths of 485 nm and 530 nm, respectively. For the visual identification of ROS generation, the similarly treated cells were washed with PBS and then loaded with 40 µM DCFH-DA for 30 min. The fluorescence in cells was visualized using a fluorescence microscope (Olympus-BX51, Japan).
Genotoxicity estimation by comet assay
The experimental procedure was conducted following the previously published protocol. 19 Briefly, the ROS-based DNA damage was analyzed by single cell gel electrophoresis. Mammary epithelial cells were grown to confluence for 24 h after seeding and treated with different concentrations (1–100 μg/mL) of bulk- and nano-TiO2 particles for 12 h. Later, cells were washed with PBS, lysed, electrophoresis (Bio-Rad, Mini Sub-Cell GT, USA) was performed for further staining with ethidium bromide. Further, the level of damage was analyzed by ImageJ 1.x software (ImageJ 1.x-1.52p, NIH, USA) on the basis of area and percentage tail DNA for valid comets only.
Apoptosis detection by Annexin V-FITC/propidium iodide staining
Detection of apoptosis with Annexin V and propidium iodide (PI) was conducted using an apoptosis detection kit (APOAF, 1001720658, Sigma, St. Louis, Missouri, USA) and following previously standardized lab protocol. 16 The mammary epithelial cells were first cultured on four-chamber slides and exposed to various concentrations of bulk-TiO2 and nano-TiO2 (1–100 μg/mL) for 12 h. Treated cells were fixed with paraformaldehyde (4%) for 30 min, washed in PBS, and incubated in Annexin V-FITC (5 μL) for 30 min in the dark, for further addition of PI solution (1 mg/mL) to the samples followed by incubation at room temperature for 10 min in the dark. The cells were observed with a fluorescence microscope (Olympus-BX51) with filters set at excitation/emission spectra 485/535 nm (FITC) and excitation/emission spectra 435/617 (PI). The intensity of green fluorescence is directly proportional to the degree of apoptosis and was determined using ImageJ 1.x software.
Statistical analysis
Statistical analyses were performed using the Graph Pad Prism software (Graph Pad Software Inc., San Diego, California, USA). Data for all the experiments were presented as the mean ± standard error of the mean and were analyzed by two-way analysis of variance. A minimum of three independent experiments were performed for each experimental condition tested and samples were drawn in triplicates. The value of
Results
Characterization of TiO2 NPs
The average hydrodynamic size of nano-TiO2 was measured using DLS and found to be 107.3 nm (Figure 1(a)). NP’s size was also confirmed by particle size analyzer tool of ImageJ 1.x software using SEM images which showed the average particle size of nano-TiO2 was 56.08 nm (Figure 1(b)).

Characterization of NPs. The results of DLS and particle size analysis using SEM. (a) The measurements of DLS show the average hydrodynamic size of nano-TiO2 (107.3 nm) and (b) the estimation of the average particle size of nano-TiO2 (56.08 nm) from SEM images by particle size analysis tool of ImageJ 1.x software. DLS: dynamic light scattering; SEM: scanning electron microscopy; NP: nanoparticle; TiO2: titanium dioxide.
Morphological changes in mammary epithelial cells
We found that there was comparatively more decrease in growth of mammary epithelial cells (MCF-7) upon exposure to nano-TiO2 (Figure 2(f) to (j)) than bulk-TiO2 (Figure 2(a) to (e)) in a dose-dependent manner as confirmed by the decrease in cell confluency (density of cells in the culture flask). It was observed through direct microscopy that MCF-7 cells in the control group (Figure 2(a) and (f)) were elongated and well spreaded, while the cells treated with nano-TiO2 were roundish (Figure 2(f), (i), and (j)), especially at higher doses (i.e. 50 and 100 µg/mL). Although dose response ratio was observed at lower doses of both compounds but at highest dose toxicity was shown in a non-linear fashion 20,21 (Figure 2).
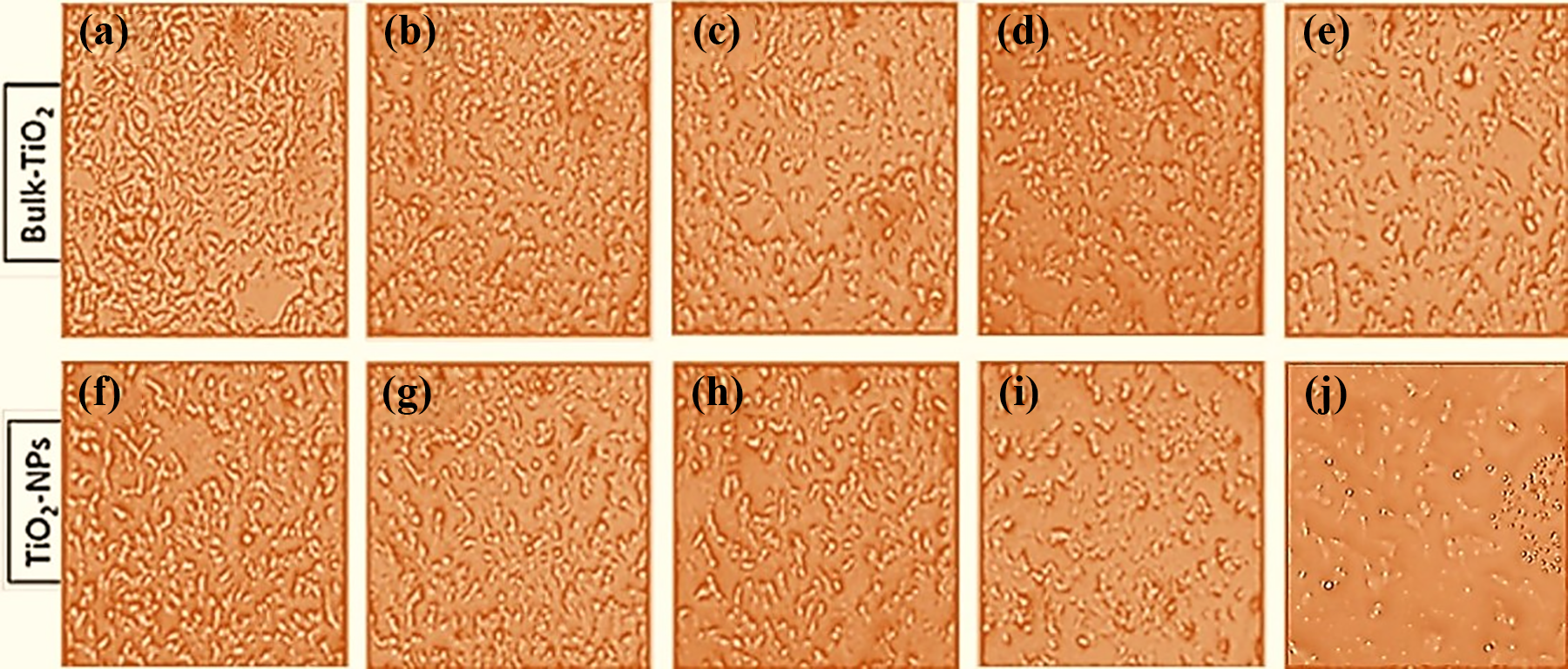
Morphological changes in MCF-7 cells. Representative images showing effects of bulk-TiO2 (a to e) and nano-TiO2 (f to j) on the morphology and growth of MCF-7 cells in a dose-dependent manner. Results obtained after treatment to MCF-7 cells with concentration of 1–100 µg/mL for 12 h. Different treatment groups of bulk-TiO2 and nano-TiO2 have been shown in figure as (a) and (f): control (untreated); (b) and (g): 1 µg/mL; (c) and (h): 10 µg/mL; (d) and (i): 50 µg/mL; and (e) and (j): 100 µg/mL. Images shown in figure were captured at ×100 magnification. TiO2: titanium dioxide.
Interaction of bulk-TiO2 and nano-TiO2 with mammary epithelial cells
SEM analysis showed the mammary epithelial cells were healthy, flat, and elongated in the control group (Figure 3(a)), while rounded or oval-shaped cells in the treated groups, that is, dose of 50 μg/mL (Figure 3(b) and (c)). Nano-TiO2 particles caused more shrinkage in the size of mammary epithelial cells in comparison to bulk particles, as can be seen clearly in SEM images (Figure 3(b) and (c)). Moreover, nano-TiO2 particles were observed to be evenly distributed and interacting with mammary epithelial cells (Figure 3(b)) whereas the bulk-TiO2 particles aggregated in clumps over and near to the cells (Figure 3(c)).

SEM images showing morphological changes induced by nano-TiO2 and bulk-TiO2 in the MCF-7 cells at 12 h. Different treatment groups were (a) control (untreated) MCF-7 cells, (b) MCF-7 cells treated with a dose of 50 μg/mL of nano-TiO2, and (c) MCF-7 cells treated with a dose of 50 μg/mL of bulk-TiO2. SEM images showing the interaction of (b) nano-TiO2 and (c) bulk-TiO2 over the surface of MCF-7 cells. Images were captured at 10 µm scale bar. SEM: scanning electron microscopy; TiO2: titanium dioxide.
Cell proliferation and cytotoxicity
Overall, the WST profiling showed that nano-TiO2 caused a comparatively higher cytotoxic response in the human mammary epithelial cells than bulk-TiO2 in a time- and dose-dependent manner (Figure 4(a) to (d)). The significant differences (

Cytotoxicity measurement by WST assay. Figure showing the percentage cell viability of MCF-7 cells treated with nano-TiO2 and bulk-TiO2. Percentage viabilities at (a) 6 h, (b) 12 h, (c) 24 h, and (d) 48 h after exposure with concentration of 1–100 µg/mL. Cells not exposed to bulk-TiO2 or nano-TiO2 served as a control. Statistical significance was evaluated by two-way ANOVA and data represented are mean ± SE of three identical experiments made in three replicates. *
Cytotoxicity by ROS generation
The cytotoxic effects of nano-TiO2 and bulk-TiO2 were assayed by measuring the fluorescence intensity of DCF (green color), which is the oxidized product of DCFH-DA dye. DCF-fluorescence represents ROS levels in cells. In case of nano-TiO2, the ROS generated in the human mammary epithelial cells was significantly higher (
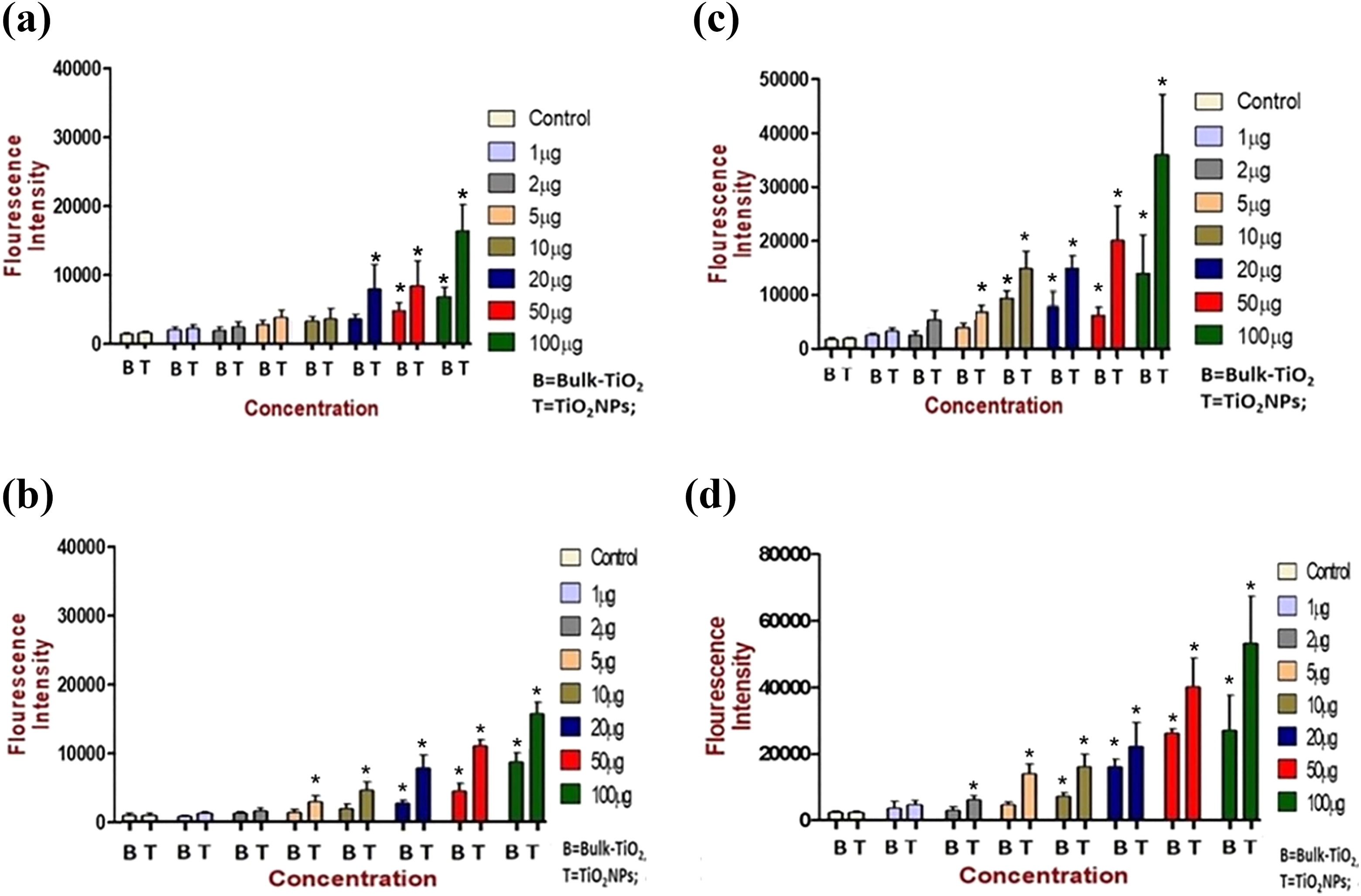
ROS measurement by DCF-DA assay. ROS mediated cytotoxic effects imposed by nano-TiO2 and bulk-TiO2 on intracellular ROS levels in MCF-7 cells. Measurements were done by DCF-DA method at (a) 3 h, (b) 6 h, (c) 12 h, and (d) 24 h of exposure with bulk-TiO2 and nano-TiO2 to MCF-7 cells with concentrations ranging from control untreated to 50 μg/mL. The intracellular ROS generation detected using a fluorescence plate reader. The green fluorescence signal indicated the level of intracellular ROS. Statistical significance was evaluated by two-way ANOVA and data represented are mean ± SE of three identical experiments made in three replicate. *
Fluorescence microscopy also exhibited the same pattern of ROS generation as an increased intensity of green color in treated human mammary epithelial cells, but comparatively higher in case of nano-TiO2 (Figure 6(f) to (j)) than in bulk-TiO2 (Figure 6(a) to (e)). The effect was dose-dependent and higher ROS generation was found in the dose of 10 μg/mL (Figure 6(h)), 50 μg/mL (Figure 6(i)), and 100 μg/mL (Figure 6(j)).

DCF-DA fluorescence images of MCF-7 cells. Images showing effects of bulk-TiO2 (a to e) and nano-TiO2 (f to j) on intracellular ROS generation in MCF-7 cells and detected using fluorescence microscopy. MCF-7 cells showing DCF fluorescence as ROS in images were taken after treatment of nano-TiO2 and bulk-TiO2 to MCF-7 cells with a concentration of 1–100 µg/mL for 12 h. Treatment groups shown in figure were control (a and f), 1 µg/mL (b and g), 10 µg/mL (c and h), 50 µg/mL (d and i), and 100 µg/mL (e and j). Images shown in figure were captured at ×100 magnification. DCF-DA: dichlorodihydrofluorescein diacetate; ROS: reactive oxygen species; TiO2: titanium dioxide.
Genotoxicity by comet assay
The results of fluorescence microscopy of the comet assay depicted a visual difference between comets of control and treated samples was observed in the DNA of human mammary epithelial cells (Figure 7(a)). Differences in the comets from control and tested concentration of 100 µg/mL in both bulk-TiO2 and nano-TiO2 have been shown via the enlarged images (Figure 7(b)). However, the lowest tested concentration (1 µg/mL) did not show any considerable dissimilarity in comets among control and treated samples.

Typical comet images from MCF-7 cells after 12 h of treatment with nano-TiO2 and bulk-TiO2 representing the level of DNA damage. Comets were stained with EtBr. (a) Comets image from control, 1, 10, and 100 µg/mL of bulk-TiO2- and nano-TiO2-treated groups. Images shown in figure were captured at ×100 magnification. (b) Enlarged images of comets analyzed by ImageJ 1.x software shown by green arrow from control, bulk-TiO2 (100 µg/mL), and nano-TiO2 (100 µg/mL) treatment groups. (c) Graph showing difference in the total area of comet DNA from MCF-7 cells. (d) Graph representing % DNA in the tail as representative of % DNA damage in MCF-7 cells. Statistical significance was evaluated by two-way ANOVA and data represented are mean ± SE of three identical experiments made in three replicate.
The ImageJ 1.x-based quantification for both treatments (nano-TiO2 and bulk-TiO2) also showed the highly significant (
Apoptosis and necrosis detection by Annexin V-FITC
As per Annexin V-FITC staining, an increased level of apoptosis (green) and necrosis (red) in the human mammary epithelial cells was observed upon exposure of bulk- and nano-TiO2 at the concentrations of 5–100 µg/mL (Figure 8(a) and (b)). We found that the apoptotic potential of nano-TiO2 to the mammary epithelial cells was high in comparison to bulk-TiO2.

Fluorescence microscopic images of MCF-7 cells stained with Annexin V-FITC (green) and PI (red). Images shown in the figure were captured at ×100 magnification. (a) MCF-7 cells treated with bulk-TiO2 for 12 h and (b) MCF-7 cells treated with nano-TiO2 for 12 h (green fluorescence showing apoptosis and red showing necrosis in MCF-7 cells after being exposed to various concentrations of NPs ranging from 5 μg/mL to 200 μg/mL). (c) Rate of necrosis in MCF-7 cells at 12 h and (d) rate of apoptosis in MCF-7 cells at 12 h. Rate of apoptosis and necrosis were determined using fluorescence intensities of PI and Annexin V-FITC. Statistical significance was evaluated by two-way ANOVA followed by Tukey test for comparison of multiple groups and data represented are mean ± SE of three identical experiments made in three replicate. The dissimilar subscript alphabets (a, b, c, etc.) represent statistically significant differences (
The rate of necrosis in the human mammary epithelial cells increased significantly at 50, 100, and 200 µg/mL concentrations of nano-TiO2 in comparison to bulk-TiO2 (Figure 8(c)). Similarly, the rates of apoptosis increased significantly at 10, 50, 100, and 200 µg/mL concentrations of nano-TiO2 in comparison to bulk TiO2 (Figure 8(d)). However, no difference in the rate of apoptosis or necrosis was observed in the control and 5 µg/mL dose groups of both nano-TiO2 and bulk-TiO2.
Discussion
Reports on the toxicities of engineered and naturally occurring NPs are quickly expanding. However, scarcity in the reports of toxicity of nano-TiO2 and bulk-TiO2 on the mammary gland has been seen, hence some related combinations of other NPs and human cells have also been discussed. Before exploiting the beneficial effects of NPs in diversified model frameworks, like cell lines and animal models, their toxicity and biocompatibility in these models should be tested. 5
Our study demonstrated the morphological changes in the human mammary epithelial cells (MCF-7) after treatment of both nano-TiO2 and bulk-TiO2. The phenomenon of change in cell morphology such as compactness and roundness after such treatments has also been reported previously, 7 and suggested to be happened due to an increase in ROS-mediated damage to the membranes of cell organelles. 7,22 Interestingly, NPs of the Cu metal and Cu-Zn alloy were reported to be highly membrane damaging even at significantly lower doses (20 µg/mL), in contrast to our dose (50 µg/mL) of nano-TiO2. 23 Charged particles, for example, cationic QD-NPs, commonly incite transient poration of the biomembranes, which may result in cytotoxic impacts. 24
Microculture tetrazolium assays, including color based tests like WST/MTT/MTS, are a few of different existing measures for determining cell proliferation and cytotoxicity. These assays are based on the reduction of tetrazolium salt to a soluble purple formazan by trans-plasma membrane electron transport from nicotinamide adenine dinucleotide or NADH via an electron-mediated transport across the plasma membrane of dividing cells. 25 We found that a dose (100 μg/mL) of nano-TiO2 in comparison to bulk-TiO2 was more cytotoxic to mammary epithelial cells as confirmed by a decrease in percentage cell viability of mammary epithelial cells. Similarly, earlier studies also showed cytotoxicity upon exposure to nano-TiO2 in the mammary epithelial cells. 26 MnO2 NPs were also reported to cause a reduction in cell viability and membrane damage in both mammary epithelial (MCF-7) and fibrosarcoma (HT1080) cells in a dose-dependent manner. 27
An increase in oxidative stress (ROS) in the mammary epithelial cells upon exposure to nano-TiO2 was recorded, but the results were less pronounced when cells were exposed to bulk-TiO2. Also, elevation of ROS levels was time and dose dependent, that is, 3–24 h and 1–100 µg/mL, respectively. At 24 h, nano-TiO2 elevated ROS level and the corresponding cytotoxicity was shown even at very low concentrations of NPs, but the bulk-TiO2 required comparatively much higher concentration for the same effect. Our results are in agreement with the results of earlier studies which indicate that NPs induced cytotoxicity through the generation of ROS in other cell types. 10,9,22
The property of nano-TiO2 to induce the production of ROS from the water and oxygen is a result of its nano size. 5,7 Excessive generation of ROS is conceivably dangerous to cells on account of their capacity to oxidize a scope of biomolecules that are engaged with fluctuated parts of cell signaling and in addition in the defense mechanism of the immune system. 6
We found that nano-TiO2 at the concentration of 10 and 100 µg/mL induce a high degree of DNA damage in MCF-7 cells upon a dose-dependent exposure, however, the DNA damage was significantly less in case of bulk TiO2. DNA damage implemented by only nano-TiO2 suggested that small size, equal distribution, and generation of ROS were the countable factors behind the cellular damage in cell organelles, suggesting the DNA strand breaks as one of the plausible mechanisms of cytotoxicity. 22,28
Other studies have also reported DNA strand breaks after nano-TiO2 treatment incorporate the likelihood of lipid peroxidation in biomembranes, DNA harm as strand breakage or oxidized nucleotides, oxidation of amino acids and protein synergist focuses in the wake of collaborating with cells. 29,30 Earlier in vivo studies on rats reported that a dose of 5 mg/kg body weight of TiO2 NPs (21 nm) caused genotoxicity in bone marrow cells. 31 Our results extended knowledge of the previous in vitro genotoxicity studies with the comet assays in other human cells where the toxicity of nano-TiO2 was shown. 5
Apoptosis is recognized as a distinct mode of programmed cell death for the elimination of cells with irreparable genetic damage. On the same line, our results of increased DNA damage through Comet assay (Figure 7) were verified by getting increased apoptosis levels in nano-TiO2-treated cells (Figure 8). Similar results of simultaneous enhancement in DNA damage and apoptosis have been reported previously. 5
The process of apoptosis takes place by means of autophagy inside the cells with deterioration of mature proteins and disposal of organelles like mitochondria. 32,33 Disturbance of mitochondrial function assumes a key part in the commencement of apoptosis; in the same way our WST assay resulted in the disturbed mitochondrial function and the resultant enhancement in apoptosis was observed. We reported a dose-dependent increase in apoptosis upon exposure to both test compounds, but the results were more pronounced in case of nano-TiO2 than bulk-TiO2. An increase in surface reactivity causes production of ROS which leads to damage to cell organelles and damage to DNA as strand breaks can be the main reason for the induction of apoptotic pathways. 34
Similarly, induction of apoptosis in MCF-7 and HT1080 cells was observed upon exposure to MnO2 NPs and it was reported that these NPs alter the regulation of apoptotic genes (p53, bax&bcl-2), cause cell cycle arrest, and decrease mitochondrial membrane potential. 27 Earlier studies found that NPs trigger autophagy at low concentrations and it was evidenced by morphological and biochemical hallmarks such as autophagolysosomes. 35 Distinctive sorts of nanomaterials have been reported to increase autophagy in numerous cell types and thus lead to cell death by means of apoptosis or necrosis. 2,36 The physicochemical properties (like size, surface area, chemical reactivity, melting point, fluorescence, electrical conductivity, and magnetic permeability) of NPs have unanimously been reported to be changed in comparison to their bulk forms and such changes may be responsible for the higher toxicity of TiO2 NPs observed in the present work. 37,38
Considering toxicity implemented by nano-TiO2 in the human mammary epithelial cells, the dose–response ratio of nano-TiO2 on mammary gland and milk synthesizing potential can further be investigated.
Conclusions
We investigated the difference in the toxicity caused by nano- and bulk-TiO2 particles in the human mammary gland cells in vitro. The comparatively harmful effects (decreased viabilities, increased ROS production, and DNA strand breaks) of nano-TiO2 in MCF-7 cells indicated for a need of regulatory framework to ensure their judicial use in health care. However, few experiments were also displaying marginally lower toxicity with bulk-TiO2 and raised the scope of more comprehensive testing. Moreover, in vitro toxicity experiments have to be replicated in in vivo systems, with toxicokinetics of NPs in various organs. Present experimental model may serve as a reference for future research works, where similar experiments could be planned for TiO2 or other nanomaterials. This information may also prove its importance among nanotechnology industries, which can modulate the NPs designs for better biocompatibility of NPs. Overall, we can conclude that the toxicity response caused by nano-TiO2 on the human mammary gland cells was comparatively higher than the bulk phase of particles.
Footnotes
Acknowledgments
The authors thank Dr Nisha Bara, State Forensic Science Laboratory, Ranchi, Jharkhand, India, for providing her expertise and help in this study. The authors also thank Ms Priyanka, Department of Biochemical Engineering and Biotechnology, IIT Delhi, India, for help in manuscript preparation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Indian Council of Agriculture Research (ICAR)-National Dairy Research Institute (NDRI), Karnal, India, which provided laboratory facilities and funding for the research work, and the Indian Council of Medical Research (ICMR), New Delhi, India, which provided a monthly stipend in the form of Junior Research Fellowship and an annual contingency grant to the research fellow (Grant No. 3/1/3/JRF-2013/HRD-18(32355)).
